Abstract
Neuropsychological impairment is a core component of major depression, yet its relationship to clinical state is unclear. The aims of the present review were to determine which neuropsychological domains and tasks were most sensitive to improvement in clinical state in major depression and to highlight the methodological issues in such research. Studies that included a baseline and at least one follow-up neuropsychological testing session in adults with major depression were identifed using MEDLINE, Web of Science and ScienceDirect databases. Thirty studies were included in the review. Findings in younger adult populations suggested that improvement in mood was most strongly related to improved verbal memory and verbal fluency, while measures of executive functioning and attention tended to remain impaired across treatment. In late-life major depression, improved psychomotor speed was most closely related to treatment response, but there was much inconsistency between study findings, which may be due to methodological issues. In major depression, particular neuropsychological domains are more strongly related to clinical state than others. The findings from the present review suggest that the domains most sensitive to clinical state are verbal learning and memory, verbal fluency and psychomotor speed. In contrast, measures of attention and executive functioning perhaps represent more trait-like markers of major depression. With further methodologically sound research, the changes in neuropsychological function associated with treatment response may provide a means of evaluating different treatment strategies in major depression.
Among the many disabling features of major depressive disorder (MDD) is neuropsychological dysfunction, which is increasingly becoming recognized as a core component of the disorder [1]. A fundamental question regarding this neuropsychological dysfunction is whether it is part of the underlying and stable neurobiological vulnerability to MDD (i.e. a trait) or whether it occurs only during episodes of depressed mood (i.e. state). Alternatively, it is possible that functions subserved by certain areas of the brain may be more sensitive to clinical state, while other functions are less changeable but more clearly trait-related.
Longitudinal description of the pattern of neuropsychological impairment in MDD during episodes, and during and following treatment may help to answer these fundamental questions. In addition, longitudinal assessment would be useful for the following reasons. First, it may further elucidate changes in brain function in MDD both during episodes and following recovery. Second, measuring changes in neuropsychological function may be helpful in assessing the effects of various treatments including both pharmacological and psychological. Measuring neuropsychological changes could provide a more objective marker of treatment response and may show change or differentiation between treatments at an earlier time point than traditional depression rating scales. The ability to measure response or non-response to treatment at an early stage could potentially also enable clinicians to either increase dosing or alter treatment in order to determine the best treatment for the patient as quickly as possible.
The aims of the present review were therefore as follows: (i) to examine change in neuropsychological function in MDD and its relationship to symptomatology and to further examine which domains of function appear to be sensitive to improvement in clinical state; (ii) to examine which neuropsychological domains or tests would be most likely to be useful in determining treatment response in MDD; and (iii) to examine methodological issues in studies examining change in neuropsychological function over time in MDD.
We reviewed all available literature in which neuropsychological testing and clinical measures of severity of MDD at baseline and after a specified amount of time after treatment, were conducted.
Methods
Search strategy
Up to 1 May 2009, electronic database searches were carried out for relevant papers, utilizing MEDLINE and Web of Science. In the initial search, the terms: ‘major depressive disorder’ or ‘major depression’ or ‘depression’ and ‘neuropsychological impairment’ or ‘neuropsychological function’ or ‘cognitive impairment’ or ‘cognitive function’ were used. To enable the inclusion of more recent articles, ScienceDirect was also searched using the aforementioned keywords in the ‘abstract, title, keywords’ field. We also checked the reference lists of all the relevant papers and the Web of Science was used to review the articles that had cited the relevant articles found using these search strategies.
Inclusion criteria
We selected only those articles involving assessment of neuropsychological function and depression severity at least twice during treatment of a major depressive episode (unipolar or bipolar) in younger adult or late-life samples.
Exclusion criteria
Reasons for exclusion were: (i) use of a depressed sample with comorbid major medical, neurological, or endocrinological conditions (the number of studies in this criterion is unknown because many studies of disorders such as dementia or Parkinson's disease involve comorbid depression); (ii) inclusion of patients who were being treated with electroconvulsive therapy (ECT; a review of all papers conducting follow-up neuropsychological testing in ECT patients has previously been published [2]); (iii) lack of comparison of change in neuropsycho-logical function between test occasions (n = 3); or (iv) lack of presentation of results for described neuropsychological testing procedures (n = 2). For the latter exclusion condition, the authors were contacted via email and asked to send their neuropsychological data, but no replies were received. All studies were limited to English-language publications.
Results
Study characteristics
Thirty studies met the inclusion criteria (Table 1). Of these studies, 18 included healthy controls but two of these studies only tested the controls at baseline [3,4]. The analyses conducted in these studies were able to be categorized under five main headings (some studies included more than one type of analysis), as follows.
(1) Those that measured overall change in mood rating scales and overall change in neuropsychological function, but did not report the direct relationship between these two measures [5–9]. In the Majer et al. and Reppermund et al. studies, patients had been classed as remitters versus nonremitters (and responders vs non-responders), but this information was used only for baseline neuropsychological comparisons and predictions [6,7].
(2) Those in which all or nearly all patients were deemed to be remitters (or responders) at follow up. For these studies, the change in neuropsychological function over time was presented and, in some cases, compared with healthy controls [10–16].
(3) Those in which only the neuropsychological performance of the remitters (or responders) was presented and, in some cases, compared with healthy controls and/or the depressed group as a whole [3,17–21].
(4) Those that categorized patients into remitters (or responders) and nonremitters (or nonresponders) and compared the change in neuropsychological function between the two groups over time [3,4,21–28].
(5) Those that correlated change in mood rating scales with change in neuropsychological performance over time [3,18,21,29–32].
The latter two types of studies are more informative than the others when examining the association between treatment response and changes in neuropsychological function. Thus, more of an emphasis will be placed on these studies in the review. The results from the review (Table 1) will be discussed in detail in the subsequent sections, followed by a discussion of the methodological inconsistencies between these studies.
Reviewed studies
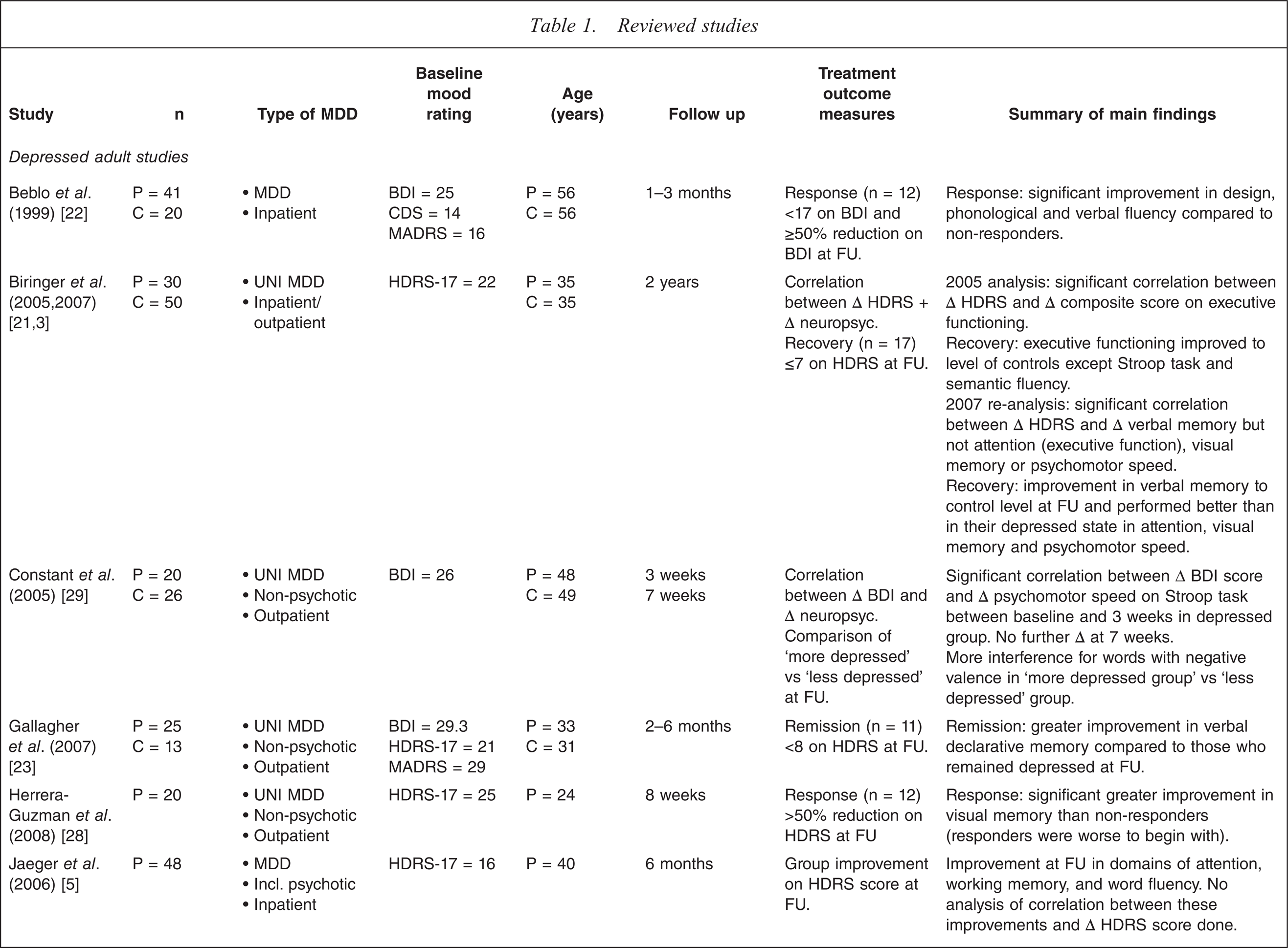
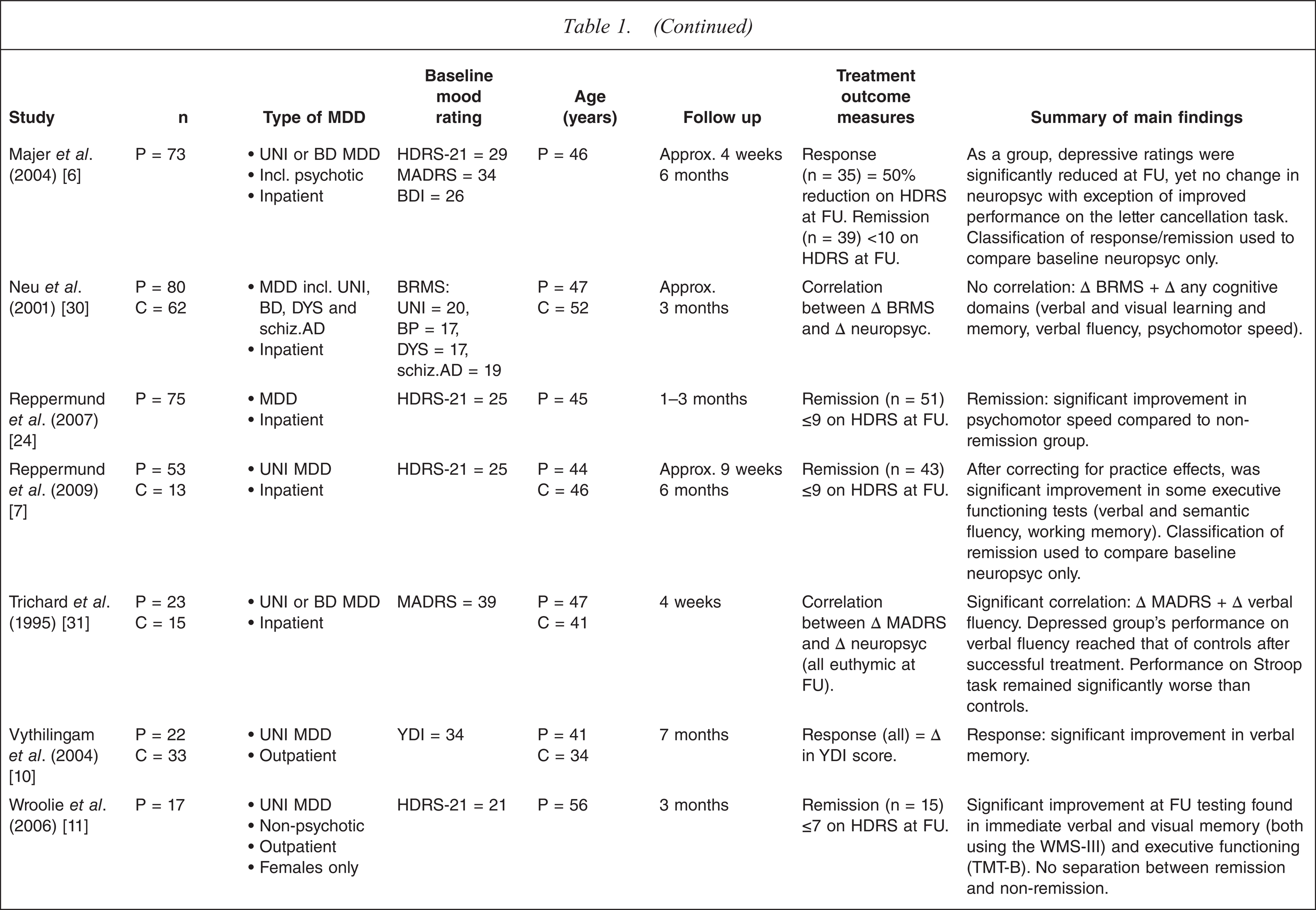
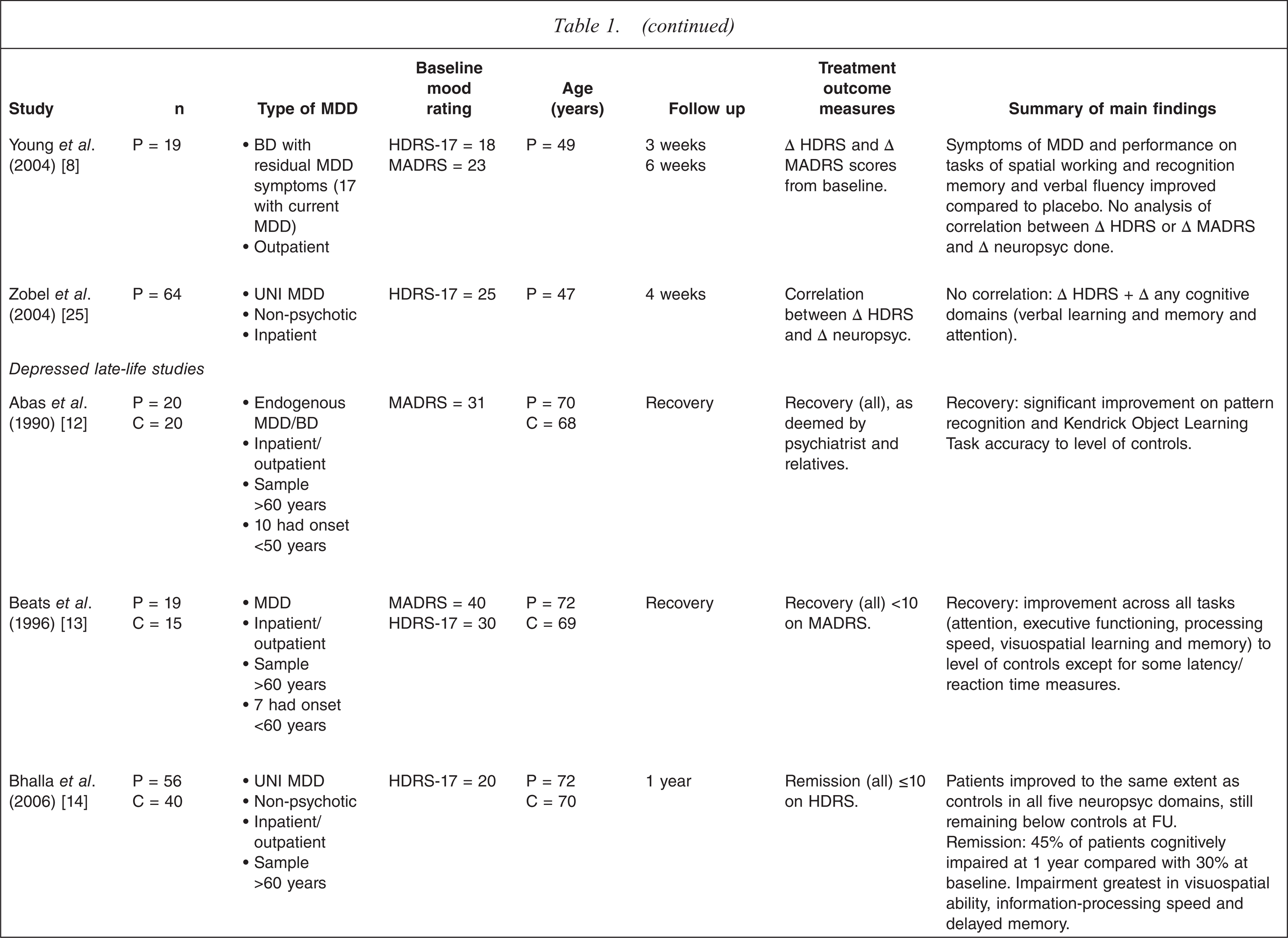
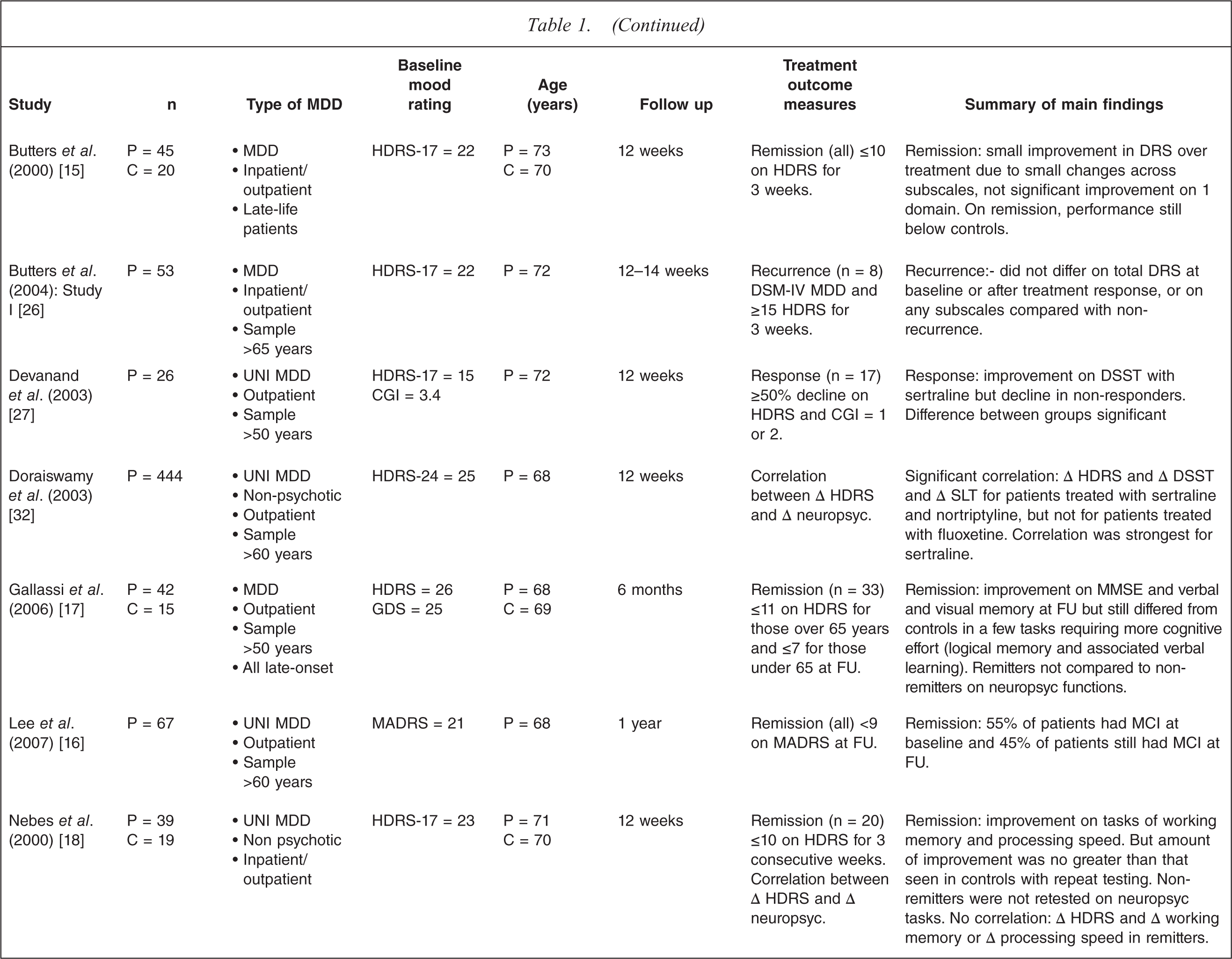
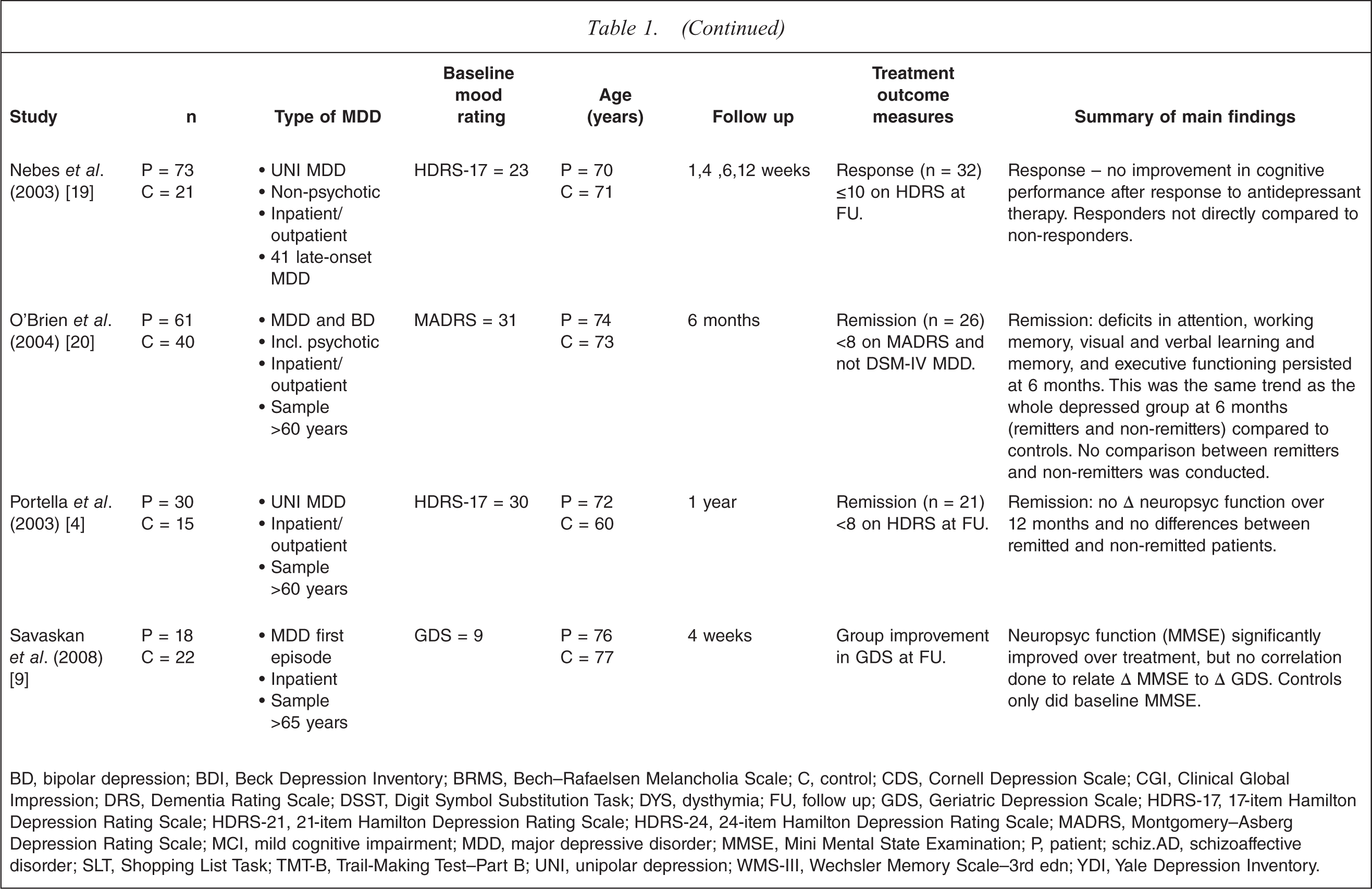
BD, bipolar depression; BDI, Beck Depression Inventory; BRMS, Bech–Rafaelsen Melancholia Scale; C, control; CDS, Cornell Depression Scale; CGI, Clinical Global Impression; DRS, Dementia Rating Scale; DSST, Digit Symbol Substitution Task; DYS, dysthymia; FU, follow up; GDS, Geriatric Depression Scale; HDRS-17, 17-item Hamilton Depression Rating Scale; HDRS-21, 21-item Hamilton Depression Rating Scale; HDRS-24, 24-item Hamilton Depression Rating Scale; MADRS, Montgomery–Asberg Depression Rating Scale; MCI, mild cognitive impairment; MDD, major depressive disorder; MMSE, Mini Mental State Examination; P, patient; schiz. AD, schizoaffective disorder; SLT, Shopping List Task; TMT–B, Trail-Making Test–Part B; UNI, unipolar depression; WMS-III, Wechsler Memory Scale–3rd edn; YDI, Yale Depression Inventory.
Neuropsychological changes during treatment of MDD
To make the results clearer, we have separated the study findings into four main neuropsychological domains: executive functioning/attention, verbal memory, non-verbal memory, and psychomotor speed.
Sustained attention and executive functioning
There are inconsistencies in the literature concerning the categorization of attention and executive functioning. Furthermore, the tasks used to measure these functions have considerable overlap [33]. We have, therefore, combined findings from both of these neuropsycho-logical domains. It should also be noted that although working memory is sometimes classed under the umbrella of ‘memory functions’, we have included it in this section too.
Short-term studies suggest that various measures of attention and executive functioning remain impaired during treatment of and recovery from MDD. In a short-term study in a severely depressed sample, Trichard et al. found no correlation between improvement in MDD and performance on the Stroop task at 4 weeks [31]. After 2–6 months in a moderately depressed sample, Gallagher et al. found no differences in executive functioning/attention changes between responders and non-responders (using spatial working memory and the Tower of London tasks) [23]. Jaeger et al., in a sample including patients with psychotic depression, found improvement in composite executive function and composite attention scores at 6 months, but did not separate responders and non-responders in their analysis [5]. A very much longer-term follow-up study found a significant correlation between improvement in MDD and change in a composite score of executive functioning at 2 years [21]. When re-analysing the results from that study with a thorough operationalization procedure, however, the correlation between improvement in composite executive functioning/ attention and mood did not remain. The recovered depressed patients performed better in attention than in their depressed states, but still not to the level of healthy controls [3].
Overall, the studies that found relatively stable impairment in executive functioning/attention tasks conducted the follow-up neuropsycho-logical batteries at ≤6 months. The second study by Biringer et al. [3] involved follow up at 2 years and found that although tasks of attention significantly improved with recovery from MDD, performance never completely returned to normal. This conclusion is supported by studies examining fully recovered or euthymic patients previously diagnosed with MDD who show persistent impairment on tasks of executive functioning/attention [34], even when performance on other neuropsycho-logical tasks is comparable to healthy controls [35,36].
Twin studies offer the ability to determine whether genetic liability to MDD is associated with neuropsychological impairment. Christensen et al. found that broad executive functioning impairment was present before the onset of MDD in healthy twins discordant for unipolar MDD [37], suggesting that those at high risk of MDD have reduced executive ability; the extent of this impairment may of course increase during episodes of MDD.
Two measures within the domain of executive functioning for which there is evidence of an association with changes in depressive symptoms are verbal and design fluency. That is, the ease with which a person can produce words or drawings given a certain set of rules. Verbal fluency tasks require patients to produce as many words as possible starting with a certain letter (phonological) or belonging to a particular category (semantic) in either 60 or 90 s. Design fluency, in contrast, involves producing as many different drawings or symbols within the same amount of time. Mildly and severely depressed patients have shown similar trends across treatment in verbal fluency tasks, with performance normalizing with successful treatment of MDD [7,8,22,31]. Two particularly strong studies directly related clinical state to change in verbal fluency. Among a battery of many neuropsychological measures, Beblo et al. found that improvement in both verbal and design fluency was the only neuropsychological measure that was significantly different between responders and non-responders [22]. Furthermore, in their severely depressed inpatient sample, Trichard et al. demonstrated a significant correlation between improvement in mood and improvement in verbal fluency, while the Stroop task remained impaired, as already described [31].
In summary, except for tests of fluency, executive functioning/ attention has generally been shown to remain impaired during and following treatment compared with healthy controls but shows slight improvement if given enough time. The diverse nature of executive functioning tasks, however, highlights the need to classify executive functioning measures into subcategories in order to understand the precise brain mechanisms controlling each task.
Evidence of executive functioning/attention improvement in late-life MDD is sparse. Many studies have been unsuccessful in finding a relationship between changes in executive functioning/attention and MDD recovery. This includes a robust study by Butters et al. in which patients whose MDD had not recurred at 12–14 weeks showed no improvement in executive functioning [26]. It appears that most studies find executive functioning/attention throughout late-life MDD episodes to be relatively insensitive to response to various pharmacological treatments over 3 months [15,27], 6 months [20] and 12 months [14]. In fact, to our knowledge, the only study that has found significant improvement in executive functioning/attention following recovery from depression, among improvement across a broad range of neuropsychological tasks, was that by Beats et al. [13].
Verbal learning and memory
Despite a relatively small number of studies, a relationship between improvement in verbal learning and memory (VLM) and improvement in depressive symptoms is a fairly consistent finding. In the Biringer et al. 2 year follow-up study, improvement in VLM was found to be significantly correlated with improvement in depressive symptoms [3]. Several studies with follow-up testing between 2 months and 2 years after baseline have also shown VLM to improve significantly more in those who achieve remission or response to treatment compared with those who do not [3,10,23]. These changes occurred within the context of a comprehensive neuropsychological battery in which all other tests, including measures of executive functioning/attention and psychomotor speed, were unrelated to clinical response. The studies had diverse samples of patients that differed in terms of hospitalization, severity, MDD type, and number of episodes of MDD. All patients, however, were diagnosed with unipolar MDD. Measures of non-VLM (NVLM) in two of these studies also failed to relate to clinical state [3,23]. Consistent with these findings, cross-sectional studies in patients who have fully recovered from MDD, or are in their euthymic phase, have found that VLM is unimpaired compared with healthy controls [35,36].
The Beblo et al. study stands out among the others because no association between VLM and MDD recovery was shown [22]. Performance on VLM tasks remained impaired at the follow up, which varied from 1 to 3 months after baseline. A major difference between the Beblo et al. study and those already described above involved the mean age of the depressed samples: being 33 years [23], 36 years [3], 41 years [10], and 56 years [22].
Two studies in late-life MDD also suggest that VLM performance is sensitive to clinical state. In a large study Doraiswamy et al. demonstrated a significant correlation between improvement in clinical state and improvement on the Shopping List Task (also known as the Buschke-Fuld Selective Reminding Test) after 12 weeks of treatment [32]. Gallassi et al. showed improvement in VLM in remitters, but in VLM tasks that required more cognitive effort (logical memory and associated verbal learning) these patients still differed from controls [17]. In addition, remitters were not directly compared to nonremitters in that study.
A number of studies have shown persistence of VLM impairment in late-life MDD populations [4,15,19,20,27], possibly relating to prolonged treatment responses in older populations [38]. VLM also appears to be more severely impaired in late-life MDD patients than younger patients [39]. O’Brien et al. found an association between impaired memory (a composite of verbal and visual memory) and hippocampal volume at 6 months, suggesting that in late-life MDD structural changes may be related to persisting neuropsychological impairment [20]. A very large follow-up study in MDD that did not compare neuropsycho-logical function between tests, however, examined predictors of neuropsychological dysfunction at baseline and at a follow-up session several weeks later following clinical response. At follow up, length of previous depressive history was a significant predictor of memory function (paragraph learning) [40]. This may be related to structural changes, such as reduced hippocampal volume, in MDD [41].
Non-verbal learning and memory
Of the studies investigating NVLM throughout an MDD episode, four studies found differences in performance with changes in MDD severity. In a study of patients with bipolar disorder and depressive symptoms (some were subsyndromal for MDD), spatial recognition memory showed significant improvement after treatment with mifepri-stone [8]. Wroolie et al., in a sample of depressed female subjects, also found an improvement in immediate visual memory after treatment with escitalopram [11]. These two studies, however, only showed group improvements in depressive symptoms and group improvements in NVLM, the relationship between the two was not examined. Vythilingam et al. found that performance on the visual selective reminding test improved in treatment responders (all patients were classified as treatment responders) even though, compared with control participants, the depressed outpatients were not impaired on this task to begin with [10]. In a study of treatment with bupropion, Herrera-Guzman et al. found that responders improved to a greater extent that non-responders in NVLM after 8 weeks [28], but this was the smallest sample in the review (responders = 12, non-responders = 8), making the findings somewhat preliminary. Larger neuropsychological studies that have directly related change in NVLM to change in depressive symptomatology have not produced any significant findings [3,22,23].
In two late-life MDD studies that tested specifically for NVLM changes as opposed to VLM, performance on some but not all visuospatial memory tasks nor malized to the level of controls on recovery from MDD [12,13]. As well as the improvements in VLM discussed in the previous section, Gallassi et al. found similar improvements in visual memory in remitters yet, as stated earlier, remitters were not compared with nonremitters [17].
Psychomotor speed
Studies generally suggest that psychomotor speed is unrelated to MDD severity or clinical state in younger adult populations. Several different measures of psychomotor speed have failed to show sensitivity to change in clinical state including the Stroop task – reaction time [31], the Phasic Alertness Task – reaction time [22], the Digit Symbol Substitution Task (DSST) [23], Trail-Making A [10], and the California Computerized Assessment Package, which was developed as a specialized package to measure psychomotor speed [3]. Constant et al., however, found a significant correlation between improvement in psychomotor speed after 3 weeks of sertraline treatment and MDD severity [29] but at 7 weeks, after further improvement in mood, there was still significant psychomotor slowing compared with controls and no correlation with clinical improvement. Another study showing sensitivity of psychomotor speed to treatment response, among various other neuropsychological tasks, was conducted by Reppermund et al. [24] using the Zahlen–Verbindungs-Test in inpatients. In that study remitters showed significant improvement in reaction time compared with nonremitters.
Overall there are some data suggesting that psychomotor speed is sensitive to clinical state in the younger depressed population. Clinically marked psychomotor retardation is observed in adult depressed patients and improves with successful treatment. It may be that findings from patients with clear psychomotor retardation are washed out when means are analysed in groups of relatively less severely depressed patients.
In late-life depressed samples it is more likely that baseline impairment in psychomotor speed will be observed [42,43]. Although Beats et al. found a broad range of improvement in many neuropsychological domains on recovery from various pharmacological treatments, measures of reaction times were the exception and remained impaired [13]. Conversely, in their sample of late-life depressed patients, Devanand et al. found a specific improvement in psychomotor speed in responders compared with non-responders using the DSST [27]. Interestingly, non-responders to sertraline actually worsened from baseline to follow up. Doraiswamy et al. also found evidence of psychomotor sensitivity to sertraline treatment response [32]. In their more severely depressed sample, improvement on the DSST significantly correlated with improvement of mood. Overall, the data are more consistent in suggesting a relationship between clinical response and improved psychomotor function in late-life MDD, as might be expected from the greater incidence of psychomotor retardation in this age-group.
Methodological issues of reviewed studies
Matched controls
A major problem with several studies was the lack of a matched healthy control group. A control group is necessary when there are repeated neuropsychological testing sessions to examine the extent to which improvements in performance are related to symptom change, as opposed to practice effects from repeated administration. Even if parallel forms are used, improvement can still occur due to enhanced learning strategies. This issue can be controlled for only if controls perform equivalent testing sessions to the patient group.
The control group should also be appropriately matched to the patients, bearing in mind that many demographic factors may affect neuropsychological function. These factors include gender [44], age and education [45], and phase of menstrual cycle [46,47]. In addition, non-psychiatric related exclusion criteria (such as pregnancy, previous head injury, or serious medical conditions) should be the same for patients and controls.
Medications
The type of medication that patients and controls are taking is important, particularly in studies recruiting patients with milder forms of MDD whose impairment may be more subtle [48]. Research suggests that neuropsychological impairment in MDD is not solely due to the effects of antidepressant medication [5,49], because studies in unmedicated depressed patients have shown significant differences in neuropsychological function compared with controls [50]. Some classes of antidepressants, however, are more likely to cause neuropsychological dysfunction, for instance, tricyclic antidepressants [51,52].
Categorization of patients by treatment outcome
A common way of analysing the relationship between change in depressive symptomatology and change in neuropsychological function was to compare remitters with nonremitters, or responders with non-responders, at the end of the treatment period. The definition of remission, however, varies between studies, as does the mood rating scale used. Furthermore, separating patients into remitters or nonremitters could result in individuals who score close to the cut-off being misclassified [3]. In our opinion, measurement of improvement in depressive symptomatology and improvement of neuropsychological function on a continuum is preferable because this captures more of the variance in the measures.
Time to follow up
The time between baseline and follow-up testing sessions varied greatly between studies. Many of the studies involving neuropsycho-logical testing were drug trials, and hence had 6–12 weeks between the testing sessions. The importance of timing is crucial because not all variables that remain the same over the follow-up sessions should automatically be classed as trait markers of MDD. It may be that these variables simply take longer to return to normal than those classed as state markers.
Subtype of MDD
The issue of clinical factors such as severity of MDD, inpatient status, presence of melancholia, comorbid anxiety and personality disorders, and number of episodes of depression should, if possible, be taken into account and at least reported [48]. Certain aspects of these factors may affect both the profile of neuropsychological dysfunction in MDD and also the speed and completeness of response to treatment.
It is also likely that subtypes of MDD, such as bipolar or psychotic depression, could have different neuropsychological profiles during treatment. Table 1 lists studies in which bipolar or psychotically depressed patients were included, but in some studies this information was not provided. Only one study specifically examined bipolar depression [8], while remaining studies that included bipolar depressed patients did not separate the analysis into subtypes, meaning that it cannot be determined whether there are different neuropsychological changes over time between unipolar and bipolar depression. Regarding psychotic depression, it is known that this subtype is associated with a more widespread neuropsychological impairment [53], but we found no studies looking specifically at changes in neuropsychological performance during treatment in psychotic depression. Again, many studies in the review included depressed patients with psychotic features, but did not conduct analyses to compare neuropsychological changes in patients with non-psychotic features.
Age
Studies have suggested that the neuropsychological impairment associated with late-life MDD is more severe than in younger depressed patients [54], particularly in the domains of psychomotor speed [42,43] and in verbal memory [39]. An issue in late-life studies, however, is that there may be an important difference between early- and late-onset MDD. Most late-life studies in the present review include a mixture of depressed patients with early- and late-onset MDD. Because of the possible different biological origin between early- and late-onset MDD [55], this should be taken into account when examining neuropsychological function in this group. Another important methodological factor is the follow-up interval. Patients with late-life MDD may take longer to respond to treatment than younger patients with MDD [38], perhaps resulting in changes in neuropsychological function taking longer. Long follow-up intervals, however, could be confounded by age-related decline and by progression of a number of patients to dementia. This issue can of course be controlled for by using an age-matched control group.
Discussion
After an extensive literature search, we identified 30 studies eligible for inclusion in this review. In adult MDD samples, verbal measures of learning and memory and fluency produced the most robust evidence of sensitivity to clinical state, with improvement in these tasks being significantly related to improvement in mood in a number of comprehensive studies. Why verbal measures appeared to be the most sensitive is unclear, but it may be in part due to the relative ease of administering verbal compared to non-verbal neuropsychological tasks. Pure measures of psychomotor speed in the adult MDD samples showed some support of an association with improvement in clinical state. It should be noted that a major component of verbal fluency, and various other timed neuropsychological measures, is psychomotor speed. Measures of executive functioning and attention tended to remain impaired across treatment, suggesting a more trait-like feature of these domains.
In late-life MDD the neuropsychological domain that demonstrated the clearest association with treatment response was psychomotor speed. Two studies that supported this view (using correlation and responders vs non-responders as treatment outcomes) produced significant evidence of improvement in psychomotor speed with treatment response using the DSST [33], a pure measure of psychomotor speed. In contrast, verbal memory and attention/executive functioning appeared to remain impaired over time.
Given the findings from younger adult and late-life MDD samples, we recommend that a neuropsychological testing battery including specific measures of VLM, verbal fluency, and psychomotor speed be used in future studies aiming to examine the relationship between mood and neuropsychological functioning in MDD. Typical measures of VLM used in this review were word list-learning tasks such as the Rey Auditory–Verbal Learning Task, the California Verbal Learning Task, and the verbal subtests of the Wechsler Memory Scale–Revised [33]. Inclusion of delayed recall and recognition components allows for a more detailed examination of VLM, but in severely depressed samples the original versions of these tasks are likely to be poorly tolerated by some patients and subject to floor effects. Thus, shortening or modifying of the tasks may be necessary. The Controlled Oral Word Association Task was the most common measure of phonological and semantic (verbal) fluency, while design fluency was measured with the Five-point Test [33]. The DSST produced strong findings in the late-life MDD samples in the present review, and hence is our recommendation for a pure psychomotor measure. In late-life MDD studies it is important to have longer-term follow up to allow for the possibility of prolonged treatment response.
This review also highlighted the variability of findings in neuropsychological studies of MDD and it was proposed that methodological issues were likely to be major contributors to these inconsistencies. We suggest that studies examining neuropsychological functioning across treatment of MDD should include matched healthy controls (matched for age, gender, IQ and menstrual cycle), take into account the medication status of patients and the subtype of MDD they are diagnosed with, and directly relate treatment response to changes in neuropsychological function. We believe that correlation of changes in mood rating scale scores with changes in neuropsychological test scores is the optimal analysis, because it eliminates the issue of categorical misclassification into responders or remitters.
Future directions
The current review has presented evidence from studies using traditional neuropsychological tasks. Increasing attention, however, has been given to emotionally valenced neuropsychological tasks in MDD, with one reviewed article finding evidence of sensitivity to clinical state using the emotional Stroop test [29]. Facial emotion recognition tasks may also be useful objective markers of treatment response. It has been found that depressed individuals are more likely to classify ambiguous faces as negative [56], consistent with psychological theories of MDD that emphasize the role of negative biases in information processing [57]. Furthermore, studies have begun to clarify the relationship between facial emotion recognition impairments and specific brain structures and neural pathways involved in emotion perception [58]. Harmer provided preliminary evidence of negative biases in facial emotion recognition in MDD norma lizing with successful treatment [59], supporting a review that presented indirect support for facial expression perception as an objective outcome measure for treatment studies in MDD [60]. Given the promising initial findings, we believe that the inclusion of a facial emotion recognition task [61] could provide useful insights into neurobiological changes occurring during the treatment of MDD.
In addition to focusing our attention on alternative measures that may indicate treatment response, it is also important to continue to search for reasons why neuro –psychological function is impaired in MDD. A possibility is that severe or multiple episodes of MDD may result in changes in neurobiology, giving rise to permanently impaired neuropsychological function. Thus, long-term description of the effects of multiple depressive episodes on neuropsychological function would be of great interest.
Conclusion
The present review suggests that certain tests of neuropsychological function may be useful indicators of treatment response in MDD. Being able to objectively examine changes in brain function during treatment may add to assessment of treatment response and potentially aid clinicians in optimizing treatment. It may also add to assessment of treatment efficacy in trials of treatments for depression. Before this can occur, however, many more methodologically sound, large-sample studies are required to clarify the neuropsychological domains and tests most sensitive to clinical state in MDD.
