Abstract
Study Design
Prospective study.
Objective
The aim of this study was to evaluate the clinical and radiologic results of using free vascularized fibular graft (FVFG) for anterior reconstruction of the cervical spine following with varying levels of corpectomy.
Methods
Ten patients underwent anterior cervical reconstruction using an FVFG after cervical corpectomy augmented with internal instrumentation. All patients were evaluated neurologically according to the Japanese Orthopaedic Association (JOA) and modified JOA scoring systems and the Nurick grading system. The neurologic recovery rate was determined, and the clinical outcome was assessed based on three factors: neck pain, dependence on pain medication, and ability to return to work. The fusion status and maintenance of lordotic correction by the strut graft were determined by measuring the lordosis angle and fused segment height (FSH).
Results
All patients achieved successful fusion. The mean follow-up period was 35.2 months (range, 28 to 44 months). Graft union occurred at a mean of 3.5 months. The mean loss of lordotic correction was 0.95 degrees, and the mean change in FSH was <1 mm. The neurologic recovery rate was excellent in four patients, good in five, and fair in one. All patients achieved satisfactory clinical outcome. No neurologic injuries occurred during the operations.
Conclusion
The use of FVFG is a valuable and effective technique in anterior cervical reconstruction for complex disorders.
Keywords
Introduction
Cervical spondylotic myelopathy (CSM) results in neurologic impairment secondary to degeneration of the structures in the cervical region of the spinal column causing narrowing of the spinal canal. CSM is the most common cause of spinal cord dysfunction worldwide. 1 Traumatic spinal cord injury (SCI) leads to varying degrees of motor and/or sensory deficits and paralysis. 2 The annual incidence of acute and chronic SCI in the United States is more than 10,000, resulting in 720 cases per million persons enduring permanent disability each year. The economic impact of SCI is estimated to be more than $4 billion annually. 3 Although CSM and SCI are the major causes of spinal cord compression, other causes include neoplasm and infection. 4 , 5
Anterior spinal cord compressive lesions of the cervical spine are ideally addressed via an anterior approach. The procedure involves the removal of the vertebral body (corpectomy) along with the intervening disk, placement of a graft into the resulting gap, and fixation using a locking anterior cervical plate. Anterior cervical corpectomy is the most direct and thorough surgical approach for anterior decompression when the spinal cord compression is located directly behind a vertebral body. Indications for this procedure include degenerative disorders, 6 neoplasm, 4 trauma, 7 infectious diseases, 5 or correction of degenerative or iatrogenic kyphosis. 8 The procedure has demonstrated good clinical results in terms of neurologic outcome and success in arthrodesis and stabilization. 9
Most reports in the literature indicate an increase in the nonunion rate when fusion is attempted at more than one level, and occasionally this failure of fusion is associated with neurologic aggravation. 10 , 11 Although a long iliac bone graft should provide a satisfactory opportunity for fusion, its inherent strength is not sufficient to resist graft fracture and segmental collapse. Therefore, a fibular graft is used to circumvent graft failure due to compressive loading across multiple segments. However, the fibular graft needs over a year to incorporate, which may require long-term postoperative immobilization even with halo orthoses. 12 As cell viability is maintained in the vascularized bone graft, primary bone healing occurs at the fibula strut–vertebral body interface in contrast to creeping substitution into the scaffold of necrotic nonvascularized bone graft. Therefore, extensive bone remodeling, including revascularization, resorption, and production of new bone, does not occur as in nonvascularized bone grafts and so structural viability and strength need to be maintained throughout the healing process in the vascularized bone grafts. 13 , 14 Also, the resorption of the nonvascularized bone graft during this process leaves the bone so weak that the graft may fracture even after being incorporated. 15
We hypothesized that the free vascularized fibular graft (FVFG) could resolve the mechanical and biological problems associated with nonvascularized grafts. To our knowledge, an FVFG has only been used in anterior cervical reconstruction after corpectomy in a few cases. The purpose of our study was to evaluate the clinical and radiographic results of using an FVFG in the anterior reconstruction of the cervical spine following varying levels of corpectomy.
Patients and Methods
Following approval of the local institutional review board, 10 patients underwent anterior cervical reconstruction using an FVFG after corpectomy between January 2008 and April 2010. Each patient provided informed consent to participate in the study. The patients comprised 7 men and 3 women with a mean age of 56 years (range, 45 to 65 years) at the time of surgery. The following inclusion criteria were applied: (1) clinical evidence of CSM, (2) motor and/or sensory deficits determined by clinical examination and confirmed by neurophysiologic studies, and (3) minimum of two levels of cervical disk affection documented by magnetic resonance imaging (MRI). The surgical indications and affected cervical levels are shown in Table 1. The mean duration from the onset of symptoms to the time of clinical evaluation was 6 months, ranging from 4 to 12 months. The most common presenting symptom was gait dysfunction in 6 patients. Two patients walk with someone else's help or with the aid of a frame, and 1 patient with gait deterioration reported multiple episodes of falls; the remaining 3 patients were chair-bound. The other 4 patients presented with deterioration of hand function. Six patients were smokers. Six patients had electrocardiogram ischemic changes, and 5 patients had hypertension.
Patient demographics and preoperative data
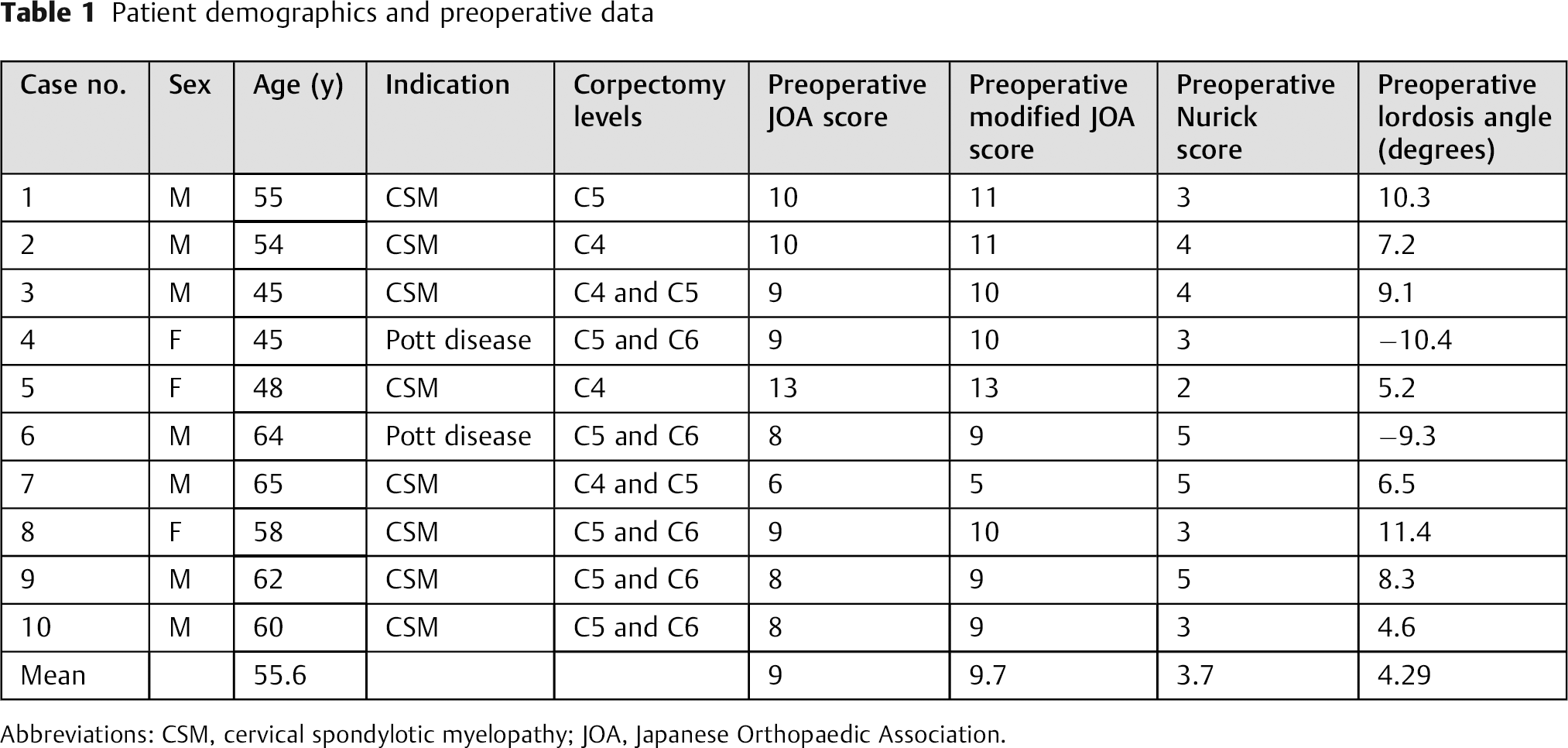
Abbreviations: CSM, cervical spondylotic myelopathy; JOA, Japanese Orthopaedic Association.
Anteroposterior and lateral radiographs of the cervical spine were obtained and the lordosis angle was measured. MRI revealed T2-weighted signal abnormalities within the spinal cord parenchyma in all patients. To standardize each patient's neurologic assessment, the Japanese Orthopaedic Association (JOA), modified JOA, and Nurick scores were calculated. The mean preoperative JOA, modified JOA, and Nurick scores were 9, 9.7, and 3.7, respectively.
Surgical Technique
Two surgical teams operated simultaneously. We used the Smith-Robinson anterior cervical approach for corpectomy (Fig. 1) and a lateral approach to harvest the fibular graft. 11 The skin was incised vertically to facilitate the dissection of the recipient vessels and graft placement. After incising the skin and platysma muscle, the dissection was continued carefully to prepare a superficial recipient vein (branch of the external jugular veins). Superficial veins are better than deep venae comitantes because they have larger diameters, thicker walls, and better drainage. Furthermore, anastomosis is much easier because it is carried superficially in a wide field. The pretracheal fascia was sharply incised medially to the carotid sheath. Based on the levels affected, either the superior thyroid artery or lingual artery was dissected gently and prepared as a recipient vessel. The prepared recipient vessel was well protected throughout the remainder of the procedure. We performed diskectomy followed by corpectomy at the desired levels until removal of the posterior longitudinal ligament, exposure of the dura, and complete decompression of the spinal cord were achieved.
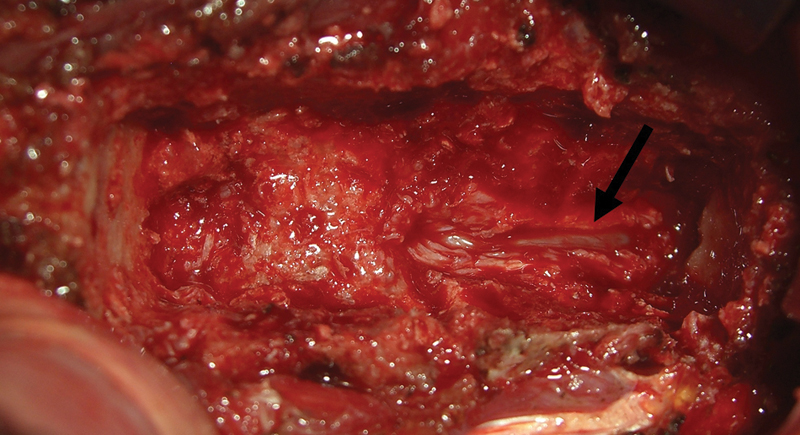
Intraoperative view of the corpectomy defect with the black arrow pointing to the site of division of the posterior longitudinal ligament.
We used the superior thyroid artery in eight patients and the lingual artery in two as the recipient artery, and the venae comitantes or a branch of the external jugular veins as the recipient vein. The FVFG remained attached to its vascular pedicle until the moment of actual transfer. The length of the free fibular graft was accurately measured according to the size of the cervical defect (Fig. 2). We cut the fibula using an electric reciprocating saw while protecting the vascular pedicle at all times. The fibular graft was positioned with the vascular pedicle ending in the cephalic direction and directed to the side of the incision (right or left). The most lateral parts of the vertebral body were not removed during corpectomy to protect the vertebral artery. At the site of exit of the vascular pedicle, the most lateral part of the vertebral body was completely removed to provide passage for the vascular pedicle without compression.
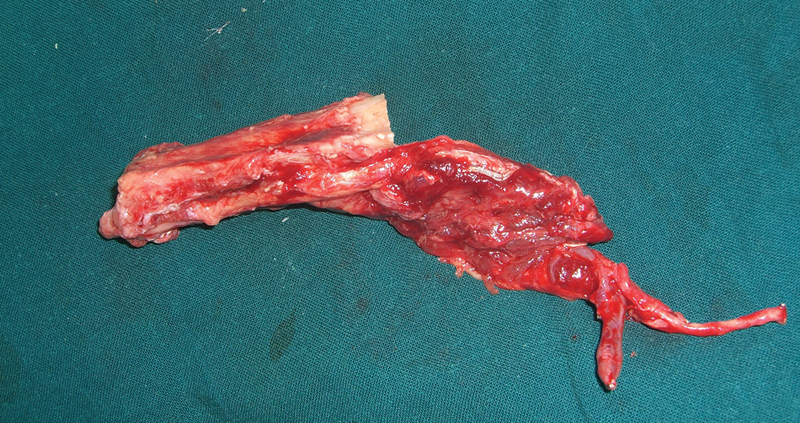
Harvested free fibular graft after being shaped to match the defect size.
A locked anterior cervical plate was applied prior to performing vascular anastomoses to protect the vascular pedicle. The plate was carefully fixed to avoid injury to the pedicle and recipient vessels. Then, end-to-end arterial and venous anastomoses were performed using an operation microscope. Postoperatively, the neck was immobilized in a rigid collar with chin support all day for 2 weeks and then during the daytime only for another 4 weeks.
All patients were clinically evaluated at discharge, monthly for 6 months, at the end of the first year, and annually thereafter according to the clinical evaluation score that was based on three factors 16 : (1) neck pain: no pain = 0, mild pain and the patient generally able to perform daily activities = 1, moderate pain that is tolerable with some limitations on daily activities = 2, severe pain with serious limitations on daily activities = 3; (2) pain medication: none = 0, non-narcotic medication = 1, and narcotic medication = 2; (3) ability to return to work: full return = 0, modified work duties = 1, unable to return to work = 2. The total score (0 to 7) was interpreted as follow: 0 to 1 = excellent (satisfactory), 2 to 3 = good (satisfactory), 4 to 5 = fair (unsatisfactory), 6 to 7 = poor (unsatisfactory). Neurologic evaluation was done according to the JOA, modified JOA, and Nurick scores at the same clinical follow-up visits. The neurologic recovery rate was determined using the formula described by Hirabayashi et al, 17 in which neurologic recovery rate = [(postoperative JOA score − preoperative JOA score)/(full score − preoperative JOA score)] × 100 (%). Surgical outcome was further quantified by categorizing the recovery rate into five groups previously designated by Matsuda et al 18 : excellent, 75 to 100%; good, 50 to 74%; fair, 25 to 49%; unchanged, 0 to 24%; and worse, <0%.
The anteroposterior, lateral, and flexion–extension lateral radiographs of all the patients were evaluated by an independent and experienced radiologist. The status of fusion and maintenance of lordotic correction by the strut graft were determined by measuring the lordosis angle and fused segment height (FSH) at discharge and final follow-up. 19 Radiographic fusion was defined as an absence of motion across the operated level on lateral flexion–extension radiographs and an absence of lucent lines or presence of bridging bony trabeculae at the fibula strut–vertebral body interface (Fig. 3). 16 , 19 , 20 The lordosis angle was determined from the tangent of the posterior body line between the C2 and C7. The FSH was assessed by measuring the distance between the anterior–superior angle of adjacent vertebrae above and the anterior–inferior angle of adjacent vertebrae below (height of the anterior border) and measurement of the distance between the posterior–superior angle of adjacent vertebrae above and the posterior–inferior angle of adjacent vertebrae below (height of the posterior border). The FSH was calculated as the mean value of height of the anterior border and height of the posterior border (Fig. 4). 19 Computed tomography was used to confirm the diagnosis of fusion. Gadolinium-enhanced MRI was performed in all patients 2 weeks postoperatively to confirm the perfusion and vascularity of the fibular graft. 21 We used Wilcoxon signed rank test for statistical analysis of the difference between the preoperative and postoperative neurologic scores with the level of significance set at P < 0.05.
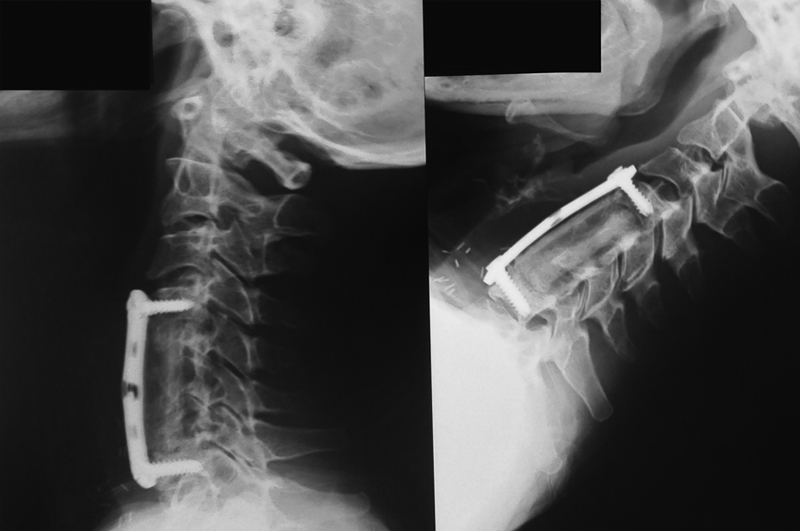
Plain X-ray lateral extension and flexion views showing solid fusion extending from C4 to C7.
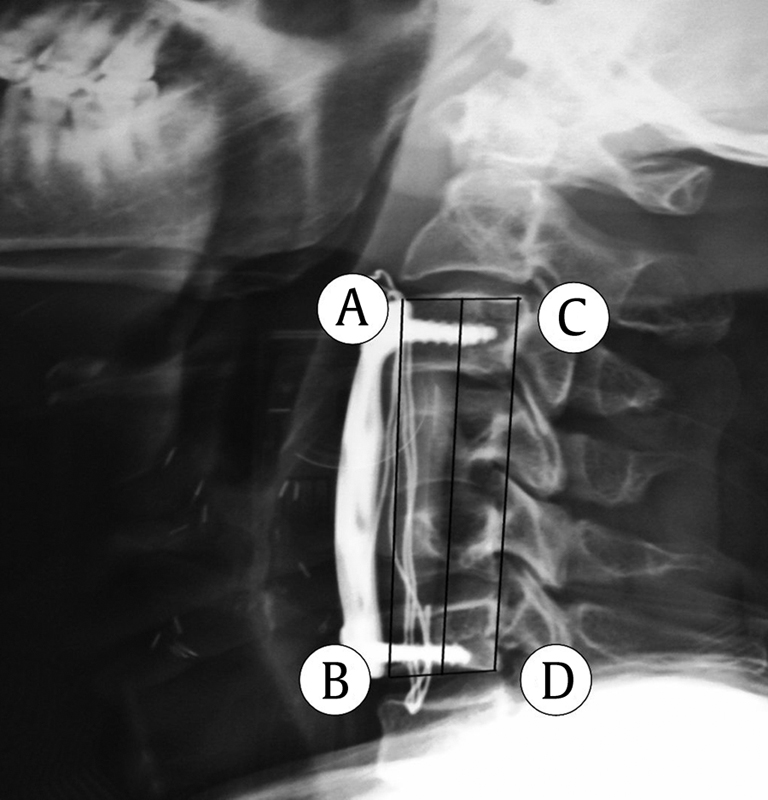
Measurement of the fused segment height: calculated as the mean of AB (height of the anterior border) and CD (height of the posterior border).
Results
The mean operating time was 6 hours (range, 4.5 to 8 hours), and the mean hospital stay was 7.5 days (range, 5 to 10 days). Uniform enhancement of the FVFG with good vascularity was seen in all patients with gadolinium-enhanced MRI. All patients achieved successful fusion at a mean of 3.5 months (range, 2.5 to 5 months; Fig. 5). The mean follow-up period was 35.2 months (range, 28 to 44 months).
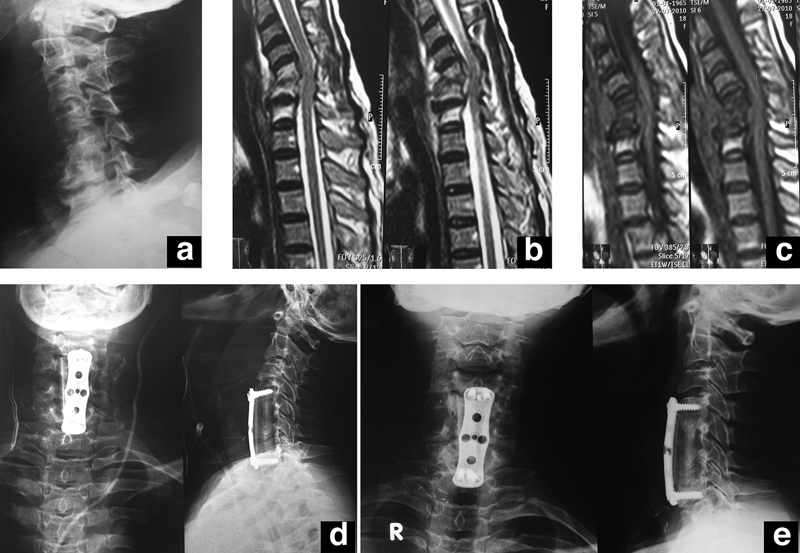
(a) Preoperative X-ray; (b, c) preoperative magnetic resonance imaging (T2 and T1); (d) immediately postoperative X-rays; (e) final follow up X-rays showing solid fusion of the graft.
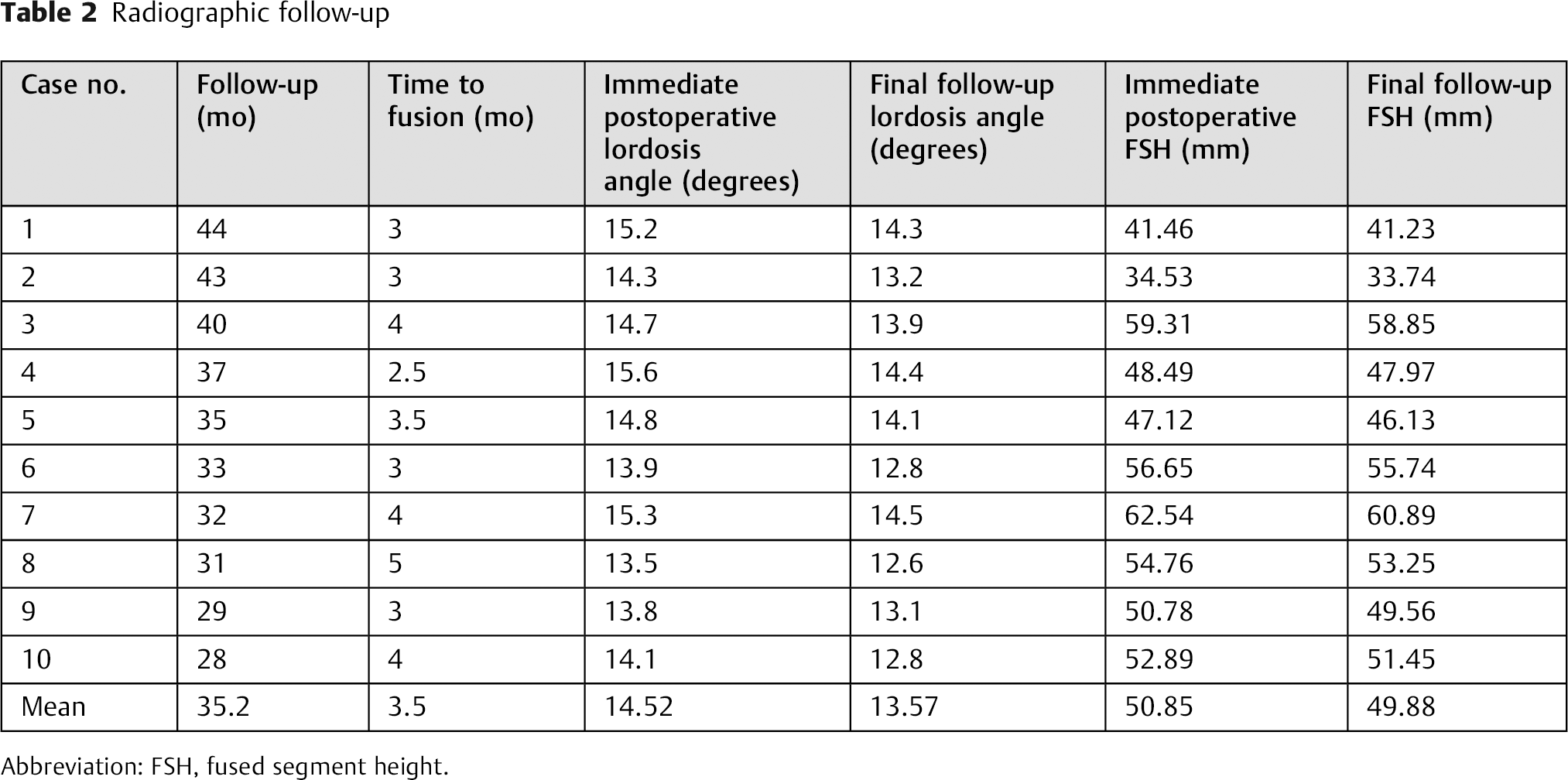
The mean lordosis angle was 4.29 degrees (range, −10.4 to 11.4 degrees) preoperatively, 14.52 degrees (range, 13.5 to 15.6 degrees) immediately postoperatively, and 13.57 degrees (range, 12.6 to 14.5 degrees) at the final follow-up visit. No significant differences were observed between the immediate postoperative and final follow-up lordosis angles (Table 2). The mean loss of lordotic correction was 0.95 degrees (range, 0.7 to 1.3 degrees). The incidence of significant loss of lordosis (>3.176 degrees) was 0%, and no patient developed cervical kyphosis. 22
Radiographic follow-up
Abbreviation: FSH, fused segment height.
Subsidence was determined as the change in FSH, with a change of ≥3 mm defined as significant subsidence. 20 In our study, the mean change in FSH was <1 mm (0.982 mm; range, 0.23 to 1.65 mm).
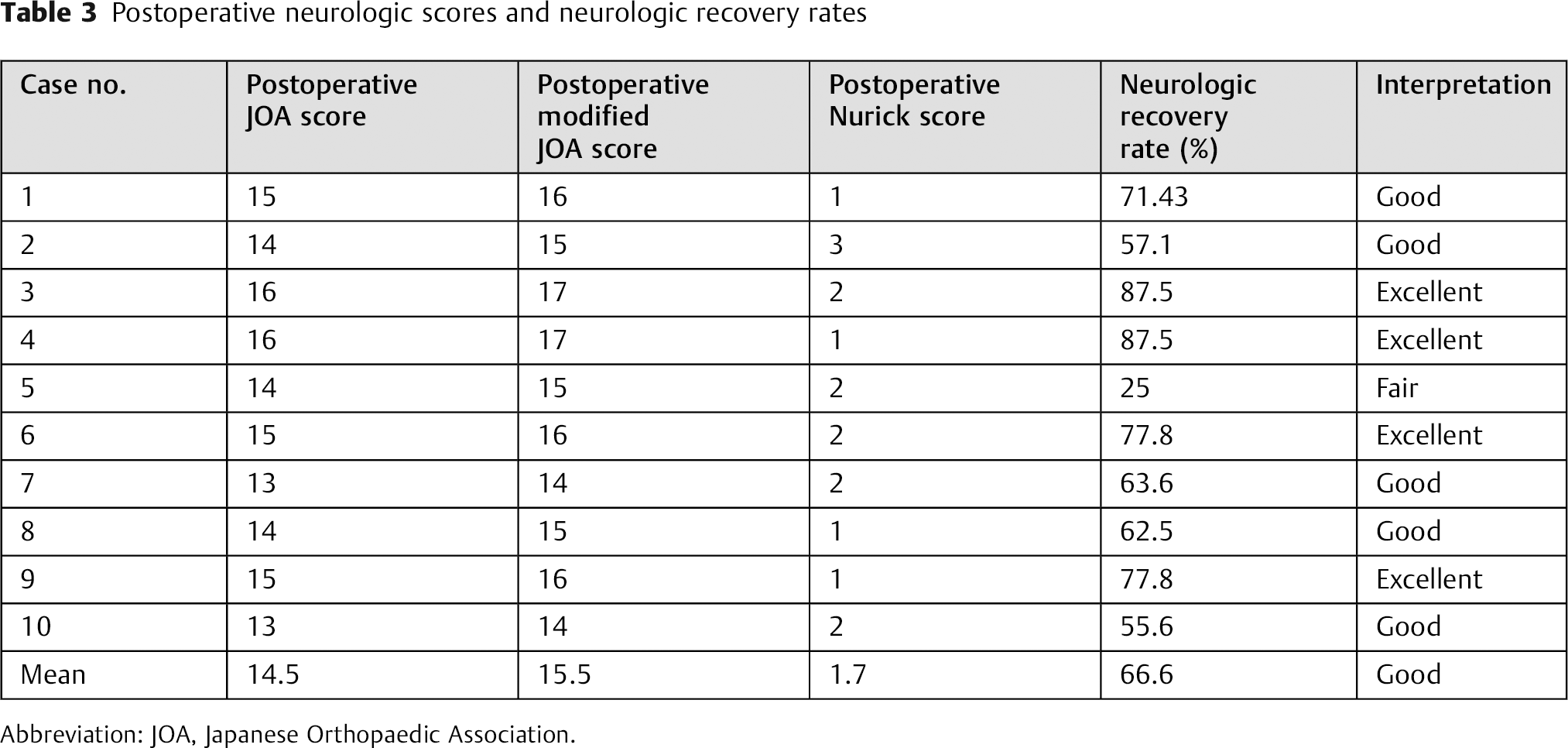
Three patients showed excellent clinical outcome, and the remaining 7 had good clinical outcome. After surgery, the neurologic status improved in all cases (Fig. 6), and no neurologic deterioration was observed in any of the cases. The mean postoperative JOA, modified JOA, and Nurick scores were 14.5, 15.5, and 1.7, respectively. Statistical analysis of the differences between the preoperative and postoperative JOA, modified JOA, and Nurick scores was performed using Wilcoxon signed rank test and was found to be statistically significant (P = 0.004, 0.005, and 0.007, respectively). The neurologic recovery rate was excellent in 4 patients, good in 5, and fair in 1 (Table 3). No neurologic injuries or other complications occurred during the intervention. Early complications included serous discharge from the cervical wound in 1 patient, which closed after 2 weeks of antibiotic administration. Late complications included pullout of the locked plate's lower screws, despite successful fusion, in 1 patient at 44 months and adjacent segment degeneration of the C5–C6 in another patient at 36 months. Graft dislodgment or pseudarthrosis was not observed. Delayed wound healing with superficial wound infection was observed in one patient, which was controlled with antibiotic therapy. Another patient complained of residual donor site pain for 4 months after the intervention. Otherwise, there was no neurologic or functional impairment.
Postoperative neurologic scores and neurologic recovery rates
Abbreviation: JOA, Japanese Orthopaedic Association.
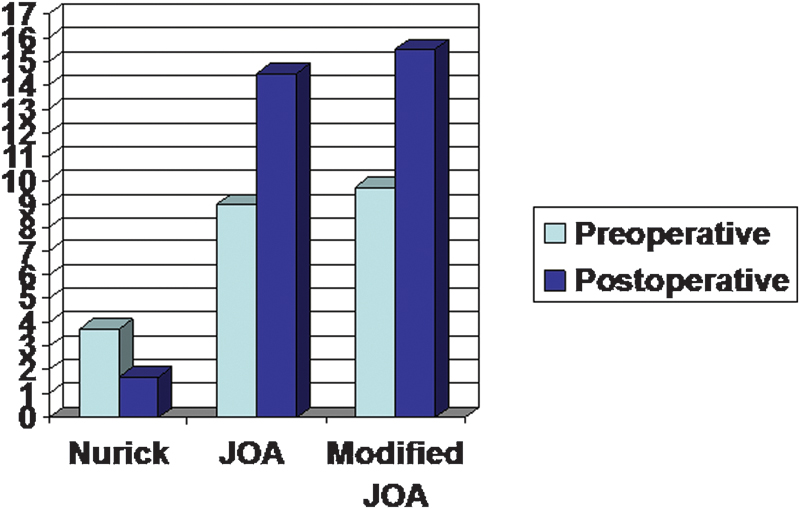
Preoperative and postoperative mean of neurologic scores. Abbreviation: JOA, Japanese Orthopaedic Association.
Discussion
The advantages of vascularized over nonvascularized bone grafts include rapid consolidation, resistance to infection, hypertrophic reaction guided by mechanical load, and tolerance of therapeutic levels of radiation. 23 When successfully revascularized, free vascularized bone grafts maintain their cellular viability as a result of the preserved endosteal and periosteal vascular blood flow. Therefore, revascularized bone grafts actively participate in bony union and provide fast and reliable periosteal growth and healing, comparable with fracture healing under the normal physiologic state. 24
In most cases of anterior cervical reconstruction, substantial volume and/or length of bone are needed, and high compression strength is desired. The fibula is superior in axial strength; is easily accessible in the prone, supine, and lateral positions; it has low donor site morbidity; and it can be easily harvested simultaneously with spinal surgery. 25
Two main problems are encountered after strut graft reconstruction following cervical corpectomy: pseudarthrosis and graft complications such as dislocation or fatigue fracture. The literature shows that the use of autogenous nonvascularized fibular grafts or fibular allografts is associated with significant pseudarthrosis or graft displacement when used for anterior cervical reconstruction following corpectomy. Fernyhough et al performed a retrospective study of 126 multilevel corpectomy cases for spondylosis to evaluate the fusion rates using autograft and allograft fibula strut graft for reconstruction. 26 The nonunion rate was high in both groups (27% of the autograft group and 41% of the allograft group) and was found to be correlated with increasing numbers of motion segments fused. Whitecloud and LaRocca reported 26 patients with fibula strut graft reconstruction of the cervical spine. 12 In the 26 cases, there were 3 graft dislocations and 2 graft fractures. Since then, other reports have documented dislocation/fracture rates for bone struts of 0, 27 , 28 2.4, 26 3.8, 29 6.5, 30 6.9, 31 7.1, 32 7.8, 33 , 8.7, 16 9.1, 34 10.2, 35 10.8, 36 11.1, 37 13.6, 38 15.0, 39 21.4, 40 26.3, 12 30.0, 41 50.0, 42 and 71.4%. 43
Pseudarthrosis, which means failure to achieve successful fusion at the strut graft–vertebral body interface with the development of a false joint, remains a serious complication with bony struts, particularly those extending over three or more disk levels, and contributes to recurrent pain with neurologic deterioration and patient disability. It might be associated with hardware failure, graft displacement, or migration that can lead to spinal cord compression. 33 Pseudarthrosis increases in the presence of several risk factors, including smoking, 44 increased number of levels fused, and medical comorbidities such as advanced age, obesity, osteoporosis, chronic steroid use, malnutrition, and chronic illnesses. The choice of the graft material, surgical technique, and use of instrumentation affect the rate of successful fusion. 45 It is quite difficult to determine pseudarthrosis rates from the literature because allografted and autografted cases are often mixed together or because autogenous bone grafts obtained by corpectomy are used in conjunction with fibular strut allograft in the same patients. 16 Where possible to gather data about the fusion for strut grafts, pseudarthrosis rates for autografts (two-level corpectomy) were 12, 35 14.3, 40 14.8, 38 21.4, 32 24, 46 27, 27 and 30%. 41 The rates for allografts were 13.4, 47 21.21, 37 29, 46 41, 26 and 41%, 27 and the pseudarthrosis rate for mixed series of allografts and autografts was 13.0%. 16
Our study showed solid fusion in all patients in a relatively short time (mean, 3.5 months) compared with a nonvascularized fibular graft, which takes longer to incorporate. 12 This short fusion time directly prevented certain complications, such as graft dislocation, dislodgment, and fracture. Furthermore, the rapid incorporation of the FVFG and solid fusion resulted in maintenance of lordotic correction by the strut graft, demonstrated by an insignificant loss of lordosis (mean, 0.95 degrees) and FSH (mean, 0.982 mm) and the fact that no patient developed cervical kyphosis during the follow-up. Accordingly, one might consider using the vascularized fibular graft reconstruction particularly in cases of corpectomy extending over an increased number of disk levels with an elevated risk of pseudarthrosis. Also, due to its resistance to infection, it would be advantageous to replace the destroyed vertebral body in cases of either pyogenic or tuberculous cervical spondylodiskitis. One may consider it after tumor resection due to its tolerance to therapeutic levels of radiation. It also might also be considered in patients with associated comorbidities such as diabetes, osteoporosis, thyroid dysfunction, and rheumatoid arthritis and in patients treated with oral steroids who are more likely to develop pseudarthrosis.
We believe that the achievement of rapid and reliable fusion was also reflected in the neurologic recovery rate, which was excellent in 4 patients, good in 5, and fair in 1. We think that the lower screw pullout that occurred in 1 patient might be due to the short period of immobilization, and the adjacent segment degeneration that developed at the caudal disk space of the fused C3–C5 in the third month in 1 patient might be related to the short distance between the tip of the plate and the adjacent disk (<5 mm) and the slight increase in lordotic curvature of the plate.
There have been reports of successful multilevel anterior cervical fusion using an FVFG. 48 Although the number of patients in our study was small, to the best of our knowledge it represents the largest series of patients treated using FVFGs in anterior cervical reconstruction that included both degenerative and infection cases.
The use of anterior instrumentation is intended to help prevent graft dislodgment, promote union, and maintain sagittal alignment. Routine angiographic evaluation of the vasculature in every patient undergoing free fibula flap surgery is probably unnecessary and expensive and does not provide benefits that outweigh the risks. 49 Therefore, no preoperative angiographic imaging was obtained in any of the patients in this study.
The main limitation of our study is the small number of patients. Moreover, the use of FVFG in anterior cervical reconstruction involves a complex spinal surgery, with wide surgical exposure and long procedures with extensive blood loss. Thus, these patients are susceptible to hypothermia and poor peripheral circulation, with an increased risk of deep venous thrombosis. 21 , 50 However, these complications did not occur in our study.
Conclusion
Our findings show that use of an FVFG can achieve reliable and successful fusion with both clinical and neurologic improvement; therefore, we believe it is a valuable and effective technique in anterior cervical reconstruction.
Disclosures
Ahmad Ibraheem Addosooki, none
Mohamed Alam-Eldin, none
Mohamed El-Sayed Abdel-Wanis, none
Mohamed Abdelhamid Ali Yousef, none
Paolo Dionigi, none
Mohamed Omar Kenawey, none
