Abstract
Patients with chronic daily headache with medication overuse are difficult to treat, especially when the doses of analgesia are substantial. We have previously shown that intravenous lignocaine (lidocaine) infusion is useful in maintaining pain control while the offending analgesic agent is withdrawn in these patients. The published data on long-term efficacy of this treatment is limited. We undertook a retrospective survey of 71 consecutive patients admitted for lignocaine infusion (mean 8.7 days) for treatment of chronic daily headache, with substantial analgesic abuse. Ninety percent of patients had a history of migraine headaches. In 80% of patients codeine was the predominant agent implicated in the analgesic rebound headaches (mean 1053 mg/week) and 24% used ergotamine-containing medications (mean 16 mg/week). Thirty-one percent frequently used injected narcotics. At completion 90% reported that their daily headache was absent or improved, and the analgesic agent was withdrawn successfully in 97%. At six month follow-up, 70% of patients reported that their daily headache was absent or improved and 72% of patients remained free of the offending analgesic agent. Intravenous lignocaine is a useful treatment in the management of chronic daily headache with substantial medication overuse. The benefits of the program last for at least six months.
Introduction
Chronic daily headache (CDH) is often associated with medication overuse. There are four major diagnostic categories of CDH: transformed migraine (TM), chronic tension-type headache (CTTH), new daily persistent headache (NDPH) and hemicrania continua. These headaches last for greater than four hours, and occur on 15 or more days per month. CDH affects 4–5% of the population (1–3) and sufferers represent 35% of the patients seen in subspecialty headache practices (4). The most troublesome cases of CDH, requiring specialist treatment, usually evolve from episodic migraine (72% of cases) or less commonly from episodic tension-type headache (20% of cases) (5). Less often CDH occurs abruptly as NDPH. The transformation from an episodic to a daily pattern is associated with overuse of analgesics, opioids, ergotamine or triptans in up to 80% of cases (6). The syndrome is characterized by the regular, dependable and predictable development of a headache within hours of the waning therapeutic effect of the last dose of medication, creating the refractory headache cycle. Headache patients are more prone to be caught in this cycle than patients suffering from other chronic pain (7).
CDH associated with medication overuse is a difficult form of headache to manage. The basis for treatment is withdrawal of the offending medication in order to break the cycle of recurrent but ineffective analgesic intake (8–11). Combinations of outpatient and inpatient withdrawal programs have been suggested using symptomatic treatment, dietary changes, migraine prophylaxis, biofeedback and behavioural therapy. A number of pharmacological agents have been suggested to limit the symptoms of withdrawal and improve the likelihood of reversion back to episodic headache. These include sumatriptan (12), valproate (13), tricyclic antidepressants (14, 15), selective serotonin reuptake inhibitors (16, 17), methadone (18), corticosteroids (19), dihydroergotamine (20, 21), histamine (22) and lignocaine (23). In most reported series, the success rate of medication withdrawal is greater than 75% at six months, with at least moderate improvement of headache in greater than 50% of patients (Table 1). However most studies include few of the most difficult patients with high dose codeine or narcotic overuse, and some even excluded them (19).
Drug-induced headache: long-term follow-up

Inpatient trials are shown in Bold. % codeine, percentage of patients using codeine; dose mg/ week, dose of codeine per week; % narcotics, percentage of patients using narcotic medications; % ergots, percentage of patients using ergotamine based medications; 50% decrease, percentage of patients whose headache frequency had reduced by at least 50% at study end; % better, percentage of patients whose headaches had improved on treatment; IV DHE, intravenous dihydroergotamine; S/C DHE, subcutaneous dihydroergotamine; n/a, not available.
The patient population in our headache clinic frequently overuses codeine. Tablets of codeine 8 or 10 mg in combination with paracetamol or aspirin are available in Australia without medical prescription and tablets of codeine 30 mg in combination with paracetamol 500 mg, with or without doxylamine, are frequently prescribed by general practitioners for headache treatment. Many of these patients require inpatient withdrawal therapy. We have previously shown that intravenous lignocaine infusion given for severe CDH is safe (24). Lignocaine is known as lidocaine in North America. The present study was undertaken to assess the efficacy of this program in patients who have CDH with substantial medication overuse.
Methods
Study design
This study is a retrospective survey of patients admitted to hospital for lignocaine infusion for treatment of CDH with associated medication overuse. Seventy-one consecutive patients who were referred to our headache clinic, and admitted to one of three hospitals for treatment, were included. Two hospitals were large suburban private hospitals and the other was a large public teaching hospital in Melbourne, Australia. Admission for treatment was considered only in those patients who had failed outpatient withdrawal, or were considered to be at risk of substantial symptoms of withdrawal due to the dose of analgesics being used. Patients with significant cardiac disease, epileptic seizures, heart block, and allergic reactions to lignocaine were excluded. Results were obtained from case notes of all patients, and via structured interview in clinic or by telephone. Data were collected at discharge from hospital, one month and six months later. Patients were asked to compare their headaches to the time prior to medication withdrawal. They were asked if their daily headaches were absent (<7 days per fortnight), improved (>7 days per fortnight, less severe), or not improved. These two-week data were averaged over the month to most accurately reflect the quality and frequency of headache at the time of the interview. Data regarding medication use in the two weeks prior to interview were also obtained from the patient.
Patients
Seventy-one patients, 62 women and 9 men, aged 16–74 years (mean 44 years) were surveyed (Table 2). All presented with headaches that fulfilled the criteria for CDH with medication overuse (25). The mean duration of CDH prior to treatment was 66 months (range 3–480 months). Ninety percent of patients had transformed migraine, 4% had chronic tension-type headache and 6% had new daily persistent headache. The mean number of headache days per month was 29 (range 22–30 days, except for one patient) and headache medications were used on a mean of 29 days per month (again 20–30 days except for the same single patient who took pethidine 200 mg IM 8 times a month). Sixty-two percent of patients used multiple agents. Codeine was implicated in 80% of cases (mean 1053 mg/week), other opioid preparations in 47% (parenteral in 31%), triptans in 17% (mean 3.7 doses/week) and ergotamine containing medications in 24% (mean 16 mg/week).
Patient characteristics

Patient management
We have previously shown that the treatment of CDH with lignocaine infusion is safe (24) and the protocol previously reported was used for this series of patients (Table 3). All patients were informed of the course of treatment and the importance of complete withdrawal of medications implicated in their CDH. On admission these medications were abruptly stopped and lignocaine infusion was started at 2 mg/minute. Rescue medications used included nonsteroidal anti-inflammatory (NSAID) agents and paracetamol. Triptans were used only in those patients in whom they were not implicated in the CDH, and who had previously suffered episodic migraine. Inpatient psychiatric assessment was offered to all 40 (62%) patients who were treated in the public hospital. Twenty-four hour bedside cardiac monitoring was performed throughout the admission in the public hospital, and continuous monitoring by telemetry was undertaken for at least the first two days in the private hospitals. Except for the method of cardiac monitoring all other aspects of care in public and private hospitals were equivalent. The infusion was maintained on the ward for at least seven days and no longer than 14 days. Prophylactic medication for migraine was usually commenced prior to discharge. Patients were given a strategy for headache management to avoid medication overuse, and were followed-up in the headache clinic at least one month and six months after discharge.
Lignocaine infusion protocol

Results
Treatment was generally well tolerated, with 66 (93%) patients completing the full protocol. In five (7%) patients treatment was stopped before seven days at the patients’ request. Four patients discharged themselves before day five because of perceived lack of response. The fifth was transferred to a psychiatric institution following an episode of acute psychosis that had started prior to admission.
Fifteen (21%) patients suffered mild side-effects from the treatment. Six (8%) had painful IV sites, five (7%) had IV site infections requiring oral antibiotics. Two patients required peripherally inserted central catheters because of poor intravenous access. One patient taking antihypertensive medications had symptomatic hypotension. The infusion was stopped for two hours, and oral hypotensive agents were withheld. One patient with a past history of supraventricular tachycardia (SVT) had an episode of SVT during the infusion. One patient complained of oscillopsia, one of hot flushes and two patients had diarrhoea. In no patients was treatment aborted because of side-effects.
The mean length of stay in hospital was 8.7 days (range 3–17), and the mean duration of lignocaine infusion was 8 days (range 3–14). Narcotic withdrawal symptoms including mild to moderate agitation, insomnia and self limiting diarrhoea were encountered frequently. Benzodiazepines were occasionally used to manage patients with moderately severe symptoms. Rescue medications were used to treat rebound headache in 80% of cases. Non-steroidal anti-inflammatory drugs (NSAIDs) were used as acute headache treatment in 70% of cases, oral triptans in 18% and other medications in 13%. Migraine prophylaxis was commenced in 45% patients.
Ninety seven percent of patients who completed the protocol had successfully withdrawn from the implicated medication, and 90% noticed an improvement in their headaches (Fig. 1). The discharge plan for managing episodic migraine included NSAIDs for 65% of patients and triptans for 51%; 65% were discharged on migraine prophylaxis, 51% on antidepressants and 23% on sedatives (Table 4).
Headache frequency and medication use before and after lignocaine infusion
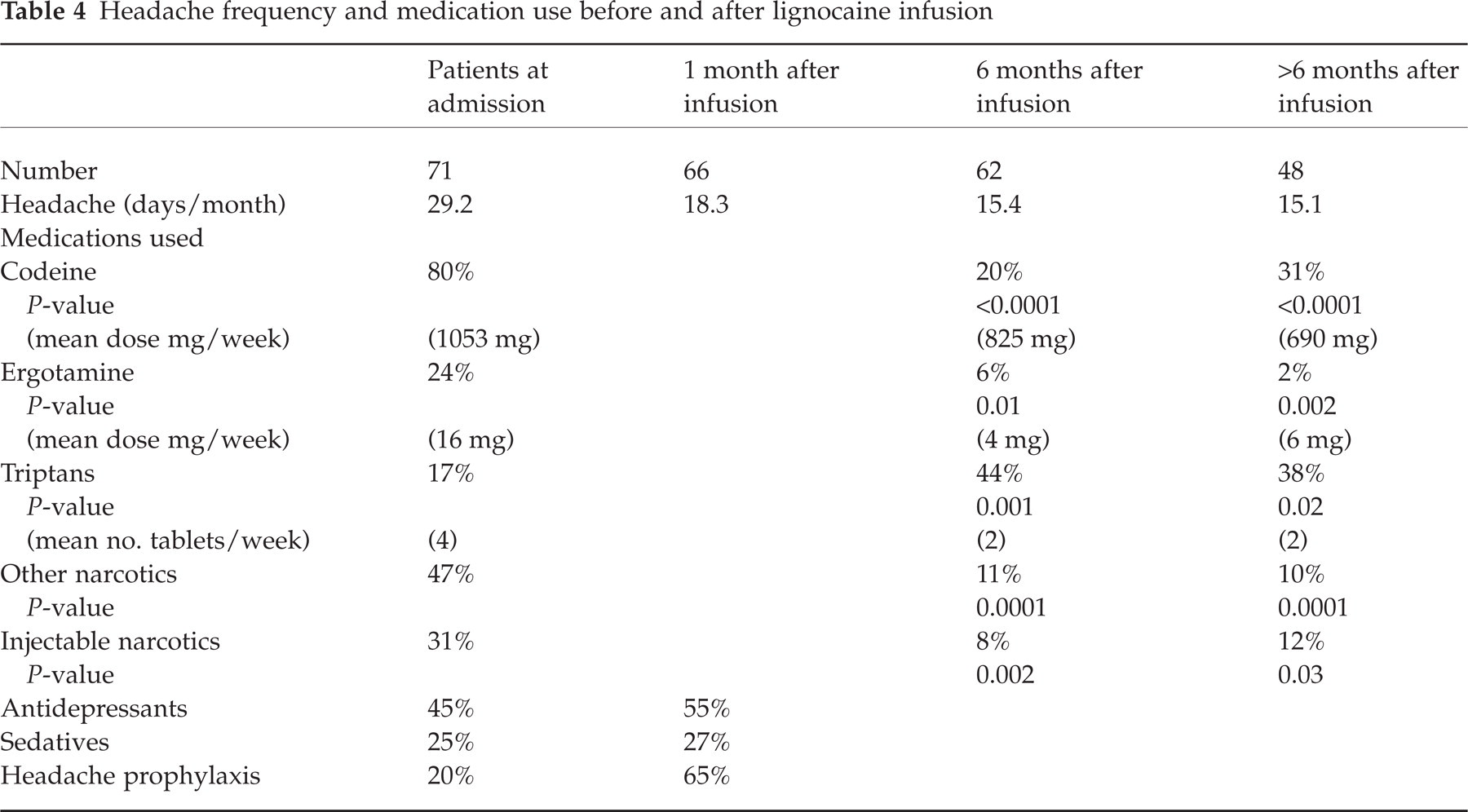

Headache outcome.
One month after discharge 88% of patients remained free of the offending medication. The daily headache improved in 76% of patients. The mean number of headache days per month had fallen from 29.2 to 18.3 days.
Six months after discharge 71% of patients remained free of the offending medication. The daily headache was absent in 51% of patients, and a further 20% of patients rated their daily headache as improved. The mean number of headache days had decreased to 15.4 days per month. The number of patients using codeine based medications decreased by 76% (from 57 to 12, P < 0.001). Of those who were still using codeine based medications the mean weekly dose decreased by 22%, to 825 mg (median 420 mg; interquartile range 300–1120 mg) (Table 4). The number of patients using ergotamine based medications decreased by 71% (from 15 to 4, P= 0.01). Of those who were still using ergotamine based medications the mean weekly dose decreased by 77%, to 4 mg (Table 4).
Forty-eight patients have been surveyed beyond 6 months, a mean of 16 months (range 8–34 months) after leaving hospital. Of these patients 58% remained free of the offending medication, a decrease of 16% from interview at six months. However the mean weekly dose of analgesic and the number of headache days per month were less than had been the case at 6 months. The daily headache was absent or improved in the same number of patients as at six months (Fig. 1).
In the group of patients with transformed migraine, the results were better than those observed for other headache types. At most recent review daily headache was absent in 63% of patients, a mean of 15 months after treatment, compared with 17% in patients with other headache types. The number of patients free from the offending medication at that time was substantially different between the two groups (65% and 33%, respectively).
We assessed whether reversion to use of the offending medication was associated with poorer control of daily headache. At six-month review, 29% of patients were again using the medication implicated in their CDH. Of this group of patients 72% said that their headache was not improved, and only 6% had no CDH (i.e. had headaches less than 15 days per month) (Fig. 2). By contrast, in the group who remained free from the offending agent only 13% had no improvement, and 69% had no CDH (Fig. 3).

Headache outcome: patients on medication at 6 months.
Headache outcome: patients off medication at 6 months.
In the six months following admission several patients were offered other therapies as part of their on-going management. Two were treated with buprenorphine for opiate withdrawal following a return to medication overuse, one was successfully prescribed an angiotensin converting enzyme inhibitor by her local doctor for migraine prophylaxis and five started antidepressant medication.
Discussion
Chronic daily headache with medication overuse is most effectively treated by the complete discontinuation of the offending medication. This strategy may be thwarted by substantial and intolerable symptoms of withdrawal (26) and the coexistence of psychiatric conditions often adds to the complexity of the situation (11). The results of this study suggest that CDH with substantial medication overuse, including the overuse of opioids such as codeine in high doses, can be successfully treated using lignocaine infusion as part of a comprehensive inpatient withdrawal program. After six months CDH was absent in 51%, and the mean frequency of headache was reduced from 29.2 to 15.4 days per month. Total medication use was reduced and continued to decline during six-month follow up. Patients with TM tended to do better than those with CTTH or NPDH with medication overuse, but small numbers in the latter two groups did not allow statistical analysis. A reduction in headache frequency and intensity persists for more than one year after treatment.
Treatment of chronic daily headaches with medication overuse
Lignocaine infusion for the treatment of CDH with medication overuse is offered to a minority of patients in our headache clinic. Published inpatient withdrawal programs have generally been reserved for those who have failed outpatient therapy, or are at risk of significant side-effects of withdrawal (12, 20, 27–29). A recent review of prophylactic pharmacological treatments for chronic daily headache (30) recommends antidepressants, either tricyclics or SSRIs, as first line therapy. If these prove ineffective antiepileptics are recommended as add-on therapy. Third line therapy includes muscle relaxants, if pericranial muscle involvement is part of the CDH syndrome, and antianxiety agents, if anxiety is a coexistent factor. Short-term parenteral therapy is recommended when outpatient therapy fails.
The degree of analgesic and ergotamine overuse in the current study is substantially more than in other studies (Table 1). Codeine doses and the amount of opioid medication set this population apart from those in previous reports. Studies of the long-term efficacy of parenteral dihydroergotamine (DHE) (20, 21) and histamine (22), in patients with lesser degrees of analgesic overuse, have demonstrated overall positive treatment effects. A reduction in headache frequency to less than 15 days per month is reported in around two-thirds of these patients. Global assessment of long-term improvement was documented in around three-quarters (Table 1). The current study achieved similar reductions in headache frequency and quality over at least six months.
Side-effects were seen in 21% of patients during the lignocaine infusion. They were mainly trivial, and none resulted in the termination of treatment. This compares favourably to the frequency of side-effects seen in patients receiving IV DHE, where 50% of patients suffered from side-effects, mainly nausea, diarrhoea and vomiting, resulting in a small number of treatment failures (20, 21).
In this study exposure to all classes of analgesics except triptans decreased in the first six months following inpatient treatment (Fig. 4). To decrease the risk of returning to a pattern of analgesic abuse patients were encouraged to avoid even one-off doses of the medication implicated in their CDH. Coincident with this significant reduction of exposure to analgesics is a reduction of mean headache days per month (Table 4). The increased exposure of patients to triptans over the first six months reflects the usual treatment of those who previously suffered from episodic migraine. It is our practice to use triptans in these patients when they were not implicated in the development of CDH.

Medication use.
The natural history of withdrawal headaches has recently been reported (26). The mean duration of withdrawal headache in those who overuse analgesic medications is 9.5 days. It is somewhat less in those who overuse ergots (6.7 days) and triptans (4.1 days). The duration of infusion in our protocol should therefore be sufficient to treat most patients. A briefer infusion may not adequately treat those who overuse analgesics. In our study 84% of patients overused codeine containing medications and the mean length of stay was 8.7 days (range 4–13).
Long-term outcomes
The long-term effect of lignocaine infusion for treatment of CDH cannot be interpreted in isolation. Admission to hospital and infusion are part of a comprehensive, ongoing treatment program that includes psychiatric assessment and outpatient follow-up. Avoidance of medication overuse remains the cornerstone of long-term treatment, and is reflected in the results of this study. Despite a 97% success rate of inpatient withdrawal of medications in those completing the protocol, 29% of patients had reverted to using the medication implicated in their CDH at six-month review. This group of patients was much less likely to be free of daily headache than those who remained free from the offending agent (Figs 2, 3).
Of the patients who were followed for more than six months there was no significant change in headache frequency or quality (Fig. 1), however, there was a trend for increased use of medications implicated in their CDH. Only 58% of patients remained free of the offending medication, compared to 71% at six months. The increase in medication use was mainly in the group who were off the agent at six month follow-up (Figs 2, 3). These figures include any use of the medication, even one-off doses. The mean weekly dose of all medications used continued to decrease over that time (Table 4), suggesting that one-off doses contributed to the increased percentage of patients using offending agents. This implies a more rational approach to medication use and suggests that limited reversion to use of the offending agent after 6 months may be less dangerous than reversion before 6 months. Despite this, we encourage our patients to remain off these agents indefinitely.
Lignocaine infusion as a treatment for CDH
Only few previous studies have investigated the effect of lignocaine infusion for treatment of CDH. It has been shown to have, at best, a modest effect when used briefly in the treatment of acute migraine (31). One limited study found that lignocaine infusion over a two day period was effective (23). We have previously used prolonged lignocaine infusion for treatment of patients with severe CDH associated with the use of high doses of codeine and ergotamine containing medications (24) because we have found outpatient withdrawal protocols ineffective in this patient population. Lignocaine is frequently used for the treatment of peripheral neuropathic pain (32).
The pathophysiology of CDH associated with medication overuse is incompletely understood (33). Central sensitization (34), cutaneous allodynia and intracranial hypersensitivity (35) are likely to play a role. Some of these effects may be mediated by sodium channel-related ectopic discharges in spinothalamic pathways (36). There is mounting evidence demonstrating abnormal cellular adaptation in the serotonin-pain control system in patients with CDH who overuse medications (37, 38). Animal models of craniovascular nociception have been used to investigate possible mechanisms of lignocaine's effect on this pain (23, 39). They suggest that lignocaine may act to interrupt part of the nociceptive pathway, but is unlikely to act at the central generator of the disorder (23). It is speculated that prolonged lignocaine infusion in CDH acts to reset this central sensitization, by modulation of peripheral nociceptor dysfunction and/or intracranial hypersensitivity, through effects on sodium channels. This is likely to be particularly useful in patients who also have abnormal cellular adaptation in opiate-dependent nociceptor pathways due to chronic opioid use (40).
Summary
We believe that prolonged intravenous infusion of lignocaine is a practical means of treating the particularly difficult group of patients with CDH associated with abuse of large quantities of analgesics, including codeine. It seems to have a better side-effect profile than other parenteral therapies such as DHE and requires a similar length of stay in hospital. As part of an integrated treatment program, including long-term follow up and psychiatric assessment, long-term improvement in headache frequency and intensity is seen past six months. Improvement is most likely if the medications implicated in CDH are avoided completely.
Footnotes
Acknowledgements
We thank the patients who participated in this survey and those who referred them. We also thank other members of the neurology unit at the Alfred Hospital who assisted in the care of these patients, and Dr David Lowenstern who performed the psychiatric assessments.
