Abstract
In order to evaluate a possible association between migraine and idiopathic narcolepsy, we performed a multicentre case-control study on the comorbidity of narcolepsy and different headaches. In total, 96 patients with idiopathic narcolepsy were enrolled. The migraine frequency in the patients and in the control group was 21.9% and 19.8%, respectively (P = 0.722). The migraine features did not differ significantly between both groups. However, headache fulfilling the criteria for tension-type headache was significantly more often reported by narcolepsy patients than by the control group (60.3% vs. 40.7%, P = 0.006). We conclude that there is no association between migraine and narcolepsy but that patients with narcolepsy show more unspecific headache, probably due to sleep disturbances.
Introduction
Narcolepsy is a disease with disturbances of the sleep-wake cycle and a genetic predisposition. The main characteristics are overwhelming episodes of sleep, cataplexy, hypnagogic hallucinations, sleep paralysis, excessive daytime sleepiness, and disturbed nocturnal sleep. Narcolepsy has an estimated prevalence in Western countries of 20–60 per 100 000 varying in the different populations (1). The clinical manifestations of narcolepsy can be explained by two principles: The inability to stay awake or asleep for a defined time period and the dissociation of REM sleep leading to cataplexy, sleep paralysis, and hypnagogic hallucinations.
Dahmen et al. (2) published an observational study showing that 81% of patients with narcolepsy suffer from different types of headache as diagnosed by the criteria of the International Headache Society (IHS) (3) and that 64% of the women and 35% of the men fulfilled the criteria for migraine. They concluded that there is a significant association between migraine and narcolepsy. Mayer et al. (4) found an increased frequency of unspecific headache in narcolepsy. However, except a case report on the occurrence of narcolepsy in a patient with episodic cluster headache (5), no other reports of the association between headache and narcolepsy, to the best of our knowledge, have been published to date.
In contrast, the frequency of HLA-DR2 antigen, which is highly associated with narcolepsy in Caucasians with cataplexy (2), was shown to be significantly decreased in migraine with aura but not in migraine without aura (6). This was not true for the HLA-DQB1∗0602 which has an even stronger association with narcolepsy.
The amazing number of patients with migraine among patients suffering from narcolepsy in the study by Dahmen et al. (2) in contrast to the reported decreased frequency of HLA-DR2 among migraine patients lead to initiate a multicentre case-control study in order to elucidate the epidemiological association between migraine and narcolepsy. Eight German centres collaborating in a study group of the German Migraine and Headache Society (DMKG) collected data of patients with narcolepsy using the International Classification of Sleep Disorders (ICSD) who were additionally interviewed with respect to migraine and headache in general using the criteria of the IHS.
Methods
Consecutive patients with idiopathic narcolepsy were recruited during a one year period from sleep laboratories approved by the German Society for Sleep Medicine. They were interviewed after providing informed consent in different university headache outpatient clinics in Germany (listed in the appendix). All patients included in this study had to fulfil the criteria of the ICSD for narcolepsy (7) with overwhelming daytime sleep episodes as a mandatory symptom. All patients were Caucasian since ethnicity plays a major role in the diagnosis of narcolepsy, at least in the HLA phenotype (8). The patients were examined neurologically and received electroencephalography and brain imaging in order to exclude symptomatic forms of narcolepsy. They were interviewed by investigators running a headache outpatient clinic and thus experienced in the diagnosis of headache. For the diagnosis of migraine all criteria of the IHS necessary for the diagnosis of migraine had to be fulfilled (3). The control group was matched by age (±1 year) and sex in a one to one fashion. It consisted of relatives of patients waiting for an appointment in the paediatric, orthopaedic and surgical outpatient clinic and of patients with traumatic bone fractures. We calculated the rate of migraine in our samples as the life time prevalence.
We evaluated the clinical and demographic data including migraine frequency and duration, migraine family history, other types of headache, duration of narcolepsy and narcolepsy family history. The family history of migraine or of narcolepsy was regarded positive if at least one first degree relative suffered from the same condition. For the diagnosis of narcolepsy, we registered the sleep history and performed polysomnography and multiple sleep latency test (MSLT). The polysomnography was analysed with respect to early onset REM. The MSLT was positive if the mean sleep latency (stage I) was five minutes or less and at least two sleep-onset REM periods occurred during five naps. HLA DR2 was measured by routine laboratory testing in a venous blood sample.
Statistical analysis was performed by nonparametric tests. We used the Mann–Whitney-U-test for quantitative and the χ2 test (Fisher's exact test for subgroups smaller than five) for qualitative data. Odds ratio (OR) and 95% confidence interval (CI) were calculated as well. Significance level was set at P= 0.05.
Results
A total sample of 96 patients with idiopathic narcolepsy according to the criteria of the ICSD and 96 healthy controls were enrolled in the study. The demographic and clinical data of our samples are presented in Table 1. There was a nearly similar rate of migraine in the patient (n = 21) and in the control group (n = 19) with no difference in sex between the migraine subjects in both groups (57% and 63%, respectively). The OR for migraine as a risk factor for narcolepsy was 1.13 with a 95% CI of 0.56–2.27. The migraine features did not differ significantly between migraineurs with and without narcolepsy. There was a significantly higher rate of other headache types in the narcolepsy group as compared to the control group (OR = 2.23; CI = 1.38–3.61). These other headaches fulfilled the criteria for tension-type headache in all but two cases. However, a symptomatic cause of the headache (i.e. sleep disturbance) could not be ruled out. One patient in either group suffered from cervicogenic headache.
Demographic and clinical data of patients with idiopathic narcolepsy and migraine.
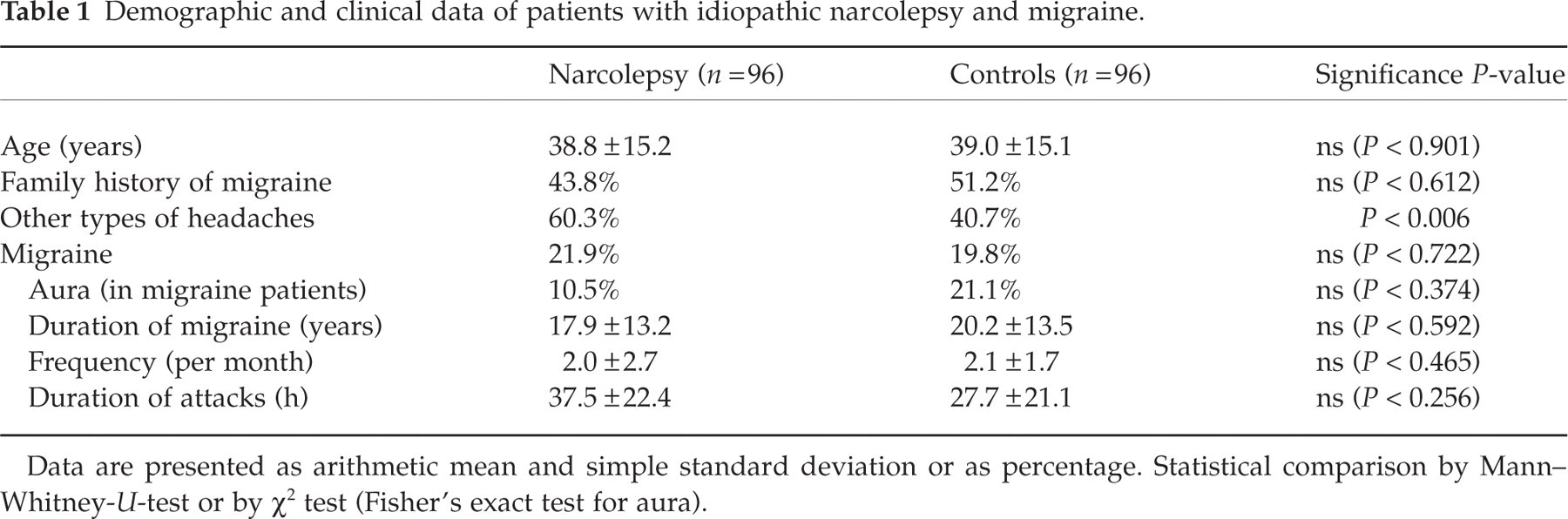
Data are presented as arithmetic mean and simple standard deviation or as percentage. Statistical comparison by Mann–Whitney-U-test or by χ2 test (Fisher's exact test for aura).
In Table 2, the clinical data of the narcolepsy patients are shown. The mean age at onset of narcolepsy was about 25 years with a range from 10 to 55 years. Overwhelming sleep episodes was an inclusion criterion and, thus, was positive in all patients. There were no significant differences in the features and symptoms of narcolepsy between those patients with narcolepsy and migraine and those patients with narcolepsy and no migraine. However, all diagnostic criteria for narcolepsy were more present in migraine patients than in patients without migraine.
Clinical data of patients with idiopathic narcolepsy given for the total group and for the subgroups with and without migraine

Data are presented as arithmetic mean with simple standard deviation or as percentage. No statistically significant differences between either subgroups. OR, odds ratio; CI, 95% confidence interval; na, not applicable.
Discussion
The most important finding in our sample is that migraine is not significantly associated with narcolepsy. We thus cannot confirm the results previously reported in the literature that the prevalence of migraine is increased in narcolepsy as compared to the data from different epidemiological studies (2). It is difficult to explain the contradictive results of the two studies. We performed a case-control study and did not compare our data to prevalence data published in the literature previously in order to calculate odds ratios. The IHS and the ICSD criteria for migraine and narcolepsy, respectively, were strictly applied. All headache diagnoses were made by experienced clinicians and researchers in the field of headache. One reason for the contradictive results might be, probably only in part, that the patients in the previous study (2) all suffered from cataplexy whereas we only had about 80% patients with cataplexy.
Interestingly, we did find a significant difference between patients with narcolepsy and our control group regarding the occurrence of unspecific headache. There was a significantly increased rate of headache in narcoleptic patients which was nearly exclusively tension-type headache. However, it is very difficult to classify this type of headache according to the IHS criteria. Although the criteria for tension-type headache were fulfilled by these patients, we cannot exclude the possibility of a symptomatic headache due to sleep disturbances. This would mean to classify the headache in group 10.1–10.3 of the IHS classification system (3). An increased frequency of unspecific headache has been also reported recently in another case series (4).
Patients with sleep disturbances, in particular patients with sleep apnea syndrome, show a higher rate of daytime headache which shows very similar pattern to tension-type headache (9–12). Narcolepsy is associated with other sleep-related disorders such as periodic limb movements and sleep apnea resulting in a disturbed and often disrupted nocturnal sleep (1, 13). This might be an explanation why unspecific daytime headache, fulfilling the criteria for tension-type headache, was reported more frequently in our sample of patients with narcolepsy. Another explanation might be that narcolepsy as a disorder of brainstem functions (1) does also affect antinociceptive brainstem functions resulting in more frequent unspecific headache. A third reason for the observed higher frequency of headache could be the intake of different drugs for the treatment of narcolepsy. Some of these drugs, in particular amphetamines, methylphenidate, ephedrine, and MAO inhibitors, show unspecific headache as a side-effect (1). However, we did not control for drug therapy in our sample.
The narcolepsy patients in our study show the typical symptoms and signs of their disease as reported in the literature in a very typical distribution (1). However, the frequency of all symptoms is higher in our sample as compared to the mean frequency in other studies (e.g. cataplexy 80% in our study vs. 70% in the literature; hypnagogic hallucinations 50% vs. 30%; sleep paralysis 45% vs. 25%). Probably, we applied more strictly the criteria to establish the diagnosis of narcolepsy.
A significant association between migraine and narcolepsy should result in a higher frequency of HLA-DR2 antigen in migraine patients. This has not been confirmed in a recent study (6). In contrast, a decreased frequency of HLA-DR2 antigen (but not of HLA-DQB1∗0602) was described exclusively for migraine with aura. This is in concordance with our finding that migraine with aura was less frequent (although not significantly) in narcolepsy patients than in the control group.
The migraine features did not differ significantly between the narcolepsy patients and the control group. This also supports our observation that migraine and narcolepsy are not associated. Vice versa, the narcolepsy features were not significantly different between the patients with and without migraine suggesting as well that there is no link between migraine and narcolepsy.
In conclusion, we could not detect any significant association between narcolepsy and migraine. This finding suggests that there are differences in the genetic background of both conditions. However, headache fulfilling the criteria for tension-type headache can be observed significantly more often in narcolepsy. This is possibly due to associated sleep disturbances with subsequent nightly hypoxic episodes causing a similar symptomatology as tension-type headache.
Footnotes
Appendix
Members of the DMKG Study Group who contributed to this study:
Stefan Evers MD, (University of Münster, Germany);
Stefanie Förderreuther MD, (University of Munich, Germany);
Gudrun Goßrau MD, (University of Dresden, Germany);
Karsten Henkel MD, (University of Ulm, Germany);
Martin Marziniak MD, (University of Würzburg, Germany);
Arne May MD, (University of Regensburg, Germany);
Saskia Kaube, (University of Regensburg);
Alexandra Rahmann MD, (University of Münster, Germany);
Gabriele Sixt MD, (Department of Neurology, University of Innsbruck, Austria);
Michael Teepker MD, (University of Marburg, Germany).
