Abstract
Previously we have reported an increased prevalence of migraine in narcoleptic patients. Because of the theoretical and clinical implications of this finding we recruited an independent new study sample of 100 patients with proven narcolepsy and conducted a structured 26-item interview based on the international diagnostic criteria for headache disorders, the Kiel Headache Questionnaire. Narcolepsy symptoms were measured by means of the Stanford Centre for Narcolepsy Sleep Inventory. Migraine prevalence was twofold to fourfold increased in the narcoleptic patients and amounted to 44.4% in women and 28.3% in men. The onset of narcolepsy symptoms was 12.3 ± 11.4 years before the onset of migraine symptoms. The results might be regarded as indicative of a common pathophysiological pathway relevant to both of the two disorders.
Introduction
Narcolepsy is a disorder of the sleep–wake cycle with a prevalence of approximately 0.05% that differs between different populations (1). Pathophysiologically, rapid eye movement (REM) dysregulation is thought to play a crucial role (1). The main symptoms are excessive daytime somnolence, overwhelming episodes of sleep, disturbed nocturnal sleep, hypnagogic hallucinations, sleep paralysis and cataplexy (1). Based on a clinical study with 68 narcoleptic patients we have previously reported an increased frequency of migraine in narcoleptic patients (2). The starting point of this first investigation was the clinical observation that roughly half of the patients being referred to our sleep laboratory for the confirmation of the diagnosis of idiopathic narcolepsy suffered from migraine headaches. Previously, only a more general relation between sleep and migraine attacks had been recognized (3, 4). This relation has been described in various ways: sleep modulates the course of migraine attacks (5), migraine attacks can be precipitated by excessive sleep as well as by sleep deprivation (5), and some patients intentionally sleep to terminate migraine attacks (6). In patients diagnosed with migraine non-discriminative polysomnographic alterations have been described (7). Finally, individual migraine attacks are accompanied and preceded by typical alterations of the sleep pattern in some patients (8).
Because of the clinical and theoretical implications of an increased prevalence of migraine in narcoleptic patients we recruited 100 additional patients with proven narcolepsy to confirm the suspected association of both conditions in an independent study population. In addition, we systematically explored the potential relations of narcolepsy symptoms, the body mass index (BMI) and HLA status with the occurrence of migraine in the affected patients.
Methods
Patients
One hundred patients suffering from narcolepsy were studied. All patients fulfilled diagnostic criteria according to the Diagnostic and Statistical Manual of the American Psychiatry Association, fourth edition (DSM IV) (9), and the International Classification of Sleep Disease (ICSD) of the American Sleep Disorders Association. All patients reported cataplectic attacks. HLA-DR2 status was determined in all patients according to a protocol described elsewhere (10).
Exclusion criteria were acute depression, schizophrenia, alcohol addiction and anxiety disorders. One patient whose narcoleptic symptomatology had started after a severe head trauma was also excluded.
The patients were either from the hospitals' sleep ambulance or were recruited with the help of a narcolepsy patient organization. This study is part of a broader effort to study narcolepsy that was approved by the local ethical committee. Patients gave written informed consent. A description of the participating patients is given in Table 1.
Patients' characteristics

Examination and questionnaire
On the day of examination a structured headache interview, a structured sleep questionnaire and a semi-structured general clinical interview were performed in all participants. The headache interview was the Kiel Headache Questionnaire (11). It consists of 26 questions and allows us to discriminate different types of headaches based on the criteria of the IHS (International Headache Society) (12) and to assess the severity and duration of symptoms. It does not distinguish between migraine with and without aura.
For the structured assessment of sleep-related symptoms, all patients filled out the Stanford Centre for Narcolepsy Sleep Inventory, a 146-item questionnaire organized in nine sections. Sections I and II report demographic information, section III includes the Epworth Sleepiness scale (13), section IV covers sleeping habits, sections V-VIII contain items on cataplexy, hallucinations, sleep paralysis and automatic behaviour, and section IX offers an opportunity to the patient to give remarks on items not already mentioned in the questionnaire. This questionnaire is frequently used in narcolepsy studies, its cataplexy part has recently been validated in 983 sleep-disorder patients (14).
The semi-structured clinical interview included symptoms and diagnostic criteria checklists for narcolepsy and for migraine, as well as for general medical conditions. Special consideration was given to the medication status of the patients, because many drugs used in the treatment of narcolepsy are known to exert effects on headache and migraine symptomatology.
Results
Out of the 100 patients examined 37 (37%) were suffering from migraine (all IHS criteria fulfilled). Twenty-four of these patients were women, representing 44.4% of the 54 women who took part in the study. Thirteen patients were male, representing 28.3% of the 46 male patients (Fig. 1).

Lifetime prevalence of migraine in narcoleptic patients.
The narcoleptic patients reported 49.2 ± 56.4 (mean±SD) headache days per year, with the women exhibiting 43.2 ± 50.4 and the men 62.4 ± 64.8 headache days per year. The duration (mean±SD) of migraine was 17.6 ± 14.8 years (17.1 ± 15.5 years for women and 18.6 ± 13.3 years for men). The symptomatology included all typical migraine symptoms, including phonophobia (88%), photophobia (77%), vomiting (49%) and nausea (66%). Symptoms worsened upon motoric activity (88%). Ninety-one per cent of the patients reported that the migraine symptomatology was restricting their daily life activities.
Because most of the patients were taking prescription drugs, some with the potential to cause headaches, the medication was compared in both groups.
Twelve (30%) of the 37 migraineurs were taking antidepressants, with six patients (16%) taking selective serotonin reuptake inhibitors (SSRIs), compared with 18 (30%) of the 63 non-migraineurs, including three patients on SSRIs (5%). When the 30 patients taking antidepressants were excluded from the study, the resulting migraine prevalence was 36% (men 31%, women 39%). When only the patients taking SSRIs were excluded, the prevalence was 34% (men 24%, women 42%).
Twenty-one (56%) of the migraineurs were taking stimulatory drugs including modafinil, with seven (19%) patients taking modafinil, in comparison with 29 (46%) of the non-migraineurs, who included nine patients (14%) on modafinil. When all 50 patients taking psychostimulants were excluded from the study, the resulting migraine prevalence was 32% (men 32%, women 32%). When only the patients on modafinil were excluded the prevalence was 36% (men 31%, women 40%).
Next, it was interesting to explore whether or not there is a relationship between onset of narcolepsy and onset of migraine and whether the patients suffering from migraine and narcolepsy differ with regard to narcolepsy symptoms from the patients not being afflicted by migraine.
The age of narcolepsy symptom onset was 20.9 ± 10.5 years for all narcolepsy patients, 18.8 ± 9.3 years for migraineurs and 22.3 ± 10.9 years for non-migraineurs (P=0.11, t-test). The age of cataplexy onset was 26.0 ± 11.5 years for all narcolepsy patients (migraineurs 22.8 ± 9.9, non-migraineurs 27.9 ± 12.2, P=0.04, t-test). The mean age of migraine onset was 30.6 ± 14.8 years; nine patients (31%) developed migraine after the age of 40. From the 29 patients who had been able to recall symptom onsets for both conditions, 25 patients developed narcolepsy symptoms prior to migraine symptoms (P < 0001, paired t-test). From the 27 patients who were able to specifically recall the onset of cataplexies and the onset of migraine, 24 patients developed cataplexies prior to the onset of migraine (P=0.003, paired t-test). The mean time-span between narcolepsy onset and migraine onset was 12.3 ± 11.4 years for all patients and 4.0 ± 3.2 years for the four migraine-first patients (Fig. 2).
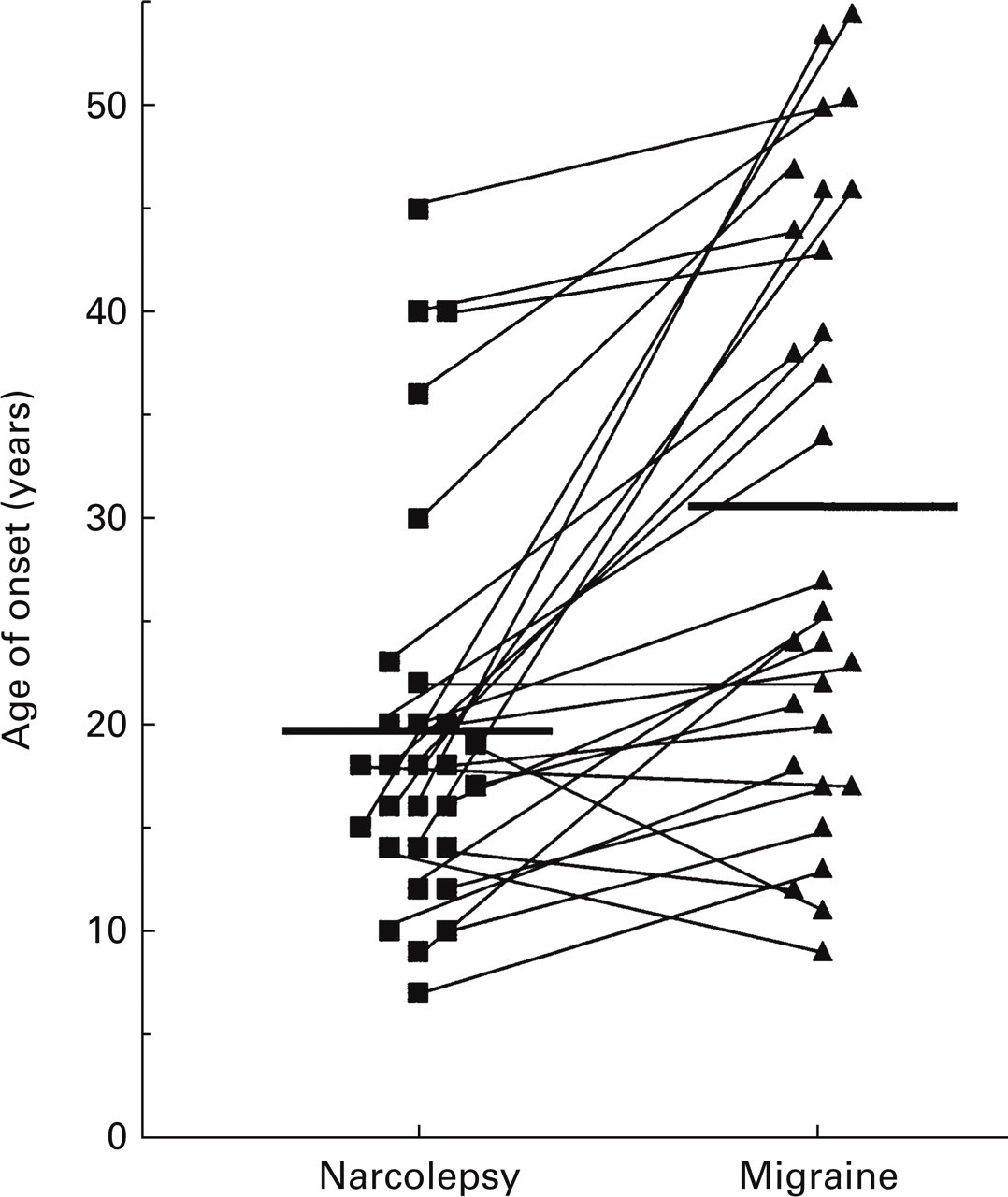
Time relationship between onset of first narcolepsy symptoms and first migraine pain attack. In most patients migraine occurred after the onset of narcolepsy (P ≤ 0.0001; paired t-test).
Because of the time relationship between narcolepsy and migraine we sought to investigate whether or not specific symptoms of narcolepsy might be associated with an increased incidence of migraine. For this analysis we dichotomized patients into those with severe daytime somnolence (as measured in items 15, 18 and 20 of the sleep inventory, corresponding to items 3, 6 and 8 of the Epworth sleepiness scale; answers 3 or 4) and those with moderate daytime somnolence (answers 0 or 1 in the items mentioned above), further in those with frequent (> 1/week) and infrequent (< 1/week) cataplexies. For the symptoms sleep paralysis, sleep-related hallucinations and automatic behaviour, patients were dichotomized according to the presence or absence of the respective symptom. In addition, we looked at BMI and at the HLA-DR2 phenotype (positive/negative). With daytime somnolence, automatic behaviour, BMI and HLA-DR2 status no obvious pattern emerged with regard to migraine prevalences. However, for the REM-associated symptoms, that is cataplexies, sleep paralysis and sleep-related hallucinations, the prevalence of migraine was numerically higher in the patients with the respective symptom present (sleep paralysis, hallucination) or more pronounced (cataplexies). This relation, however, was not significant for any single symptom (chi-square test) and because the different observations cannot be considered independent, not statistically significant on the whole (Fig. 3).
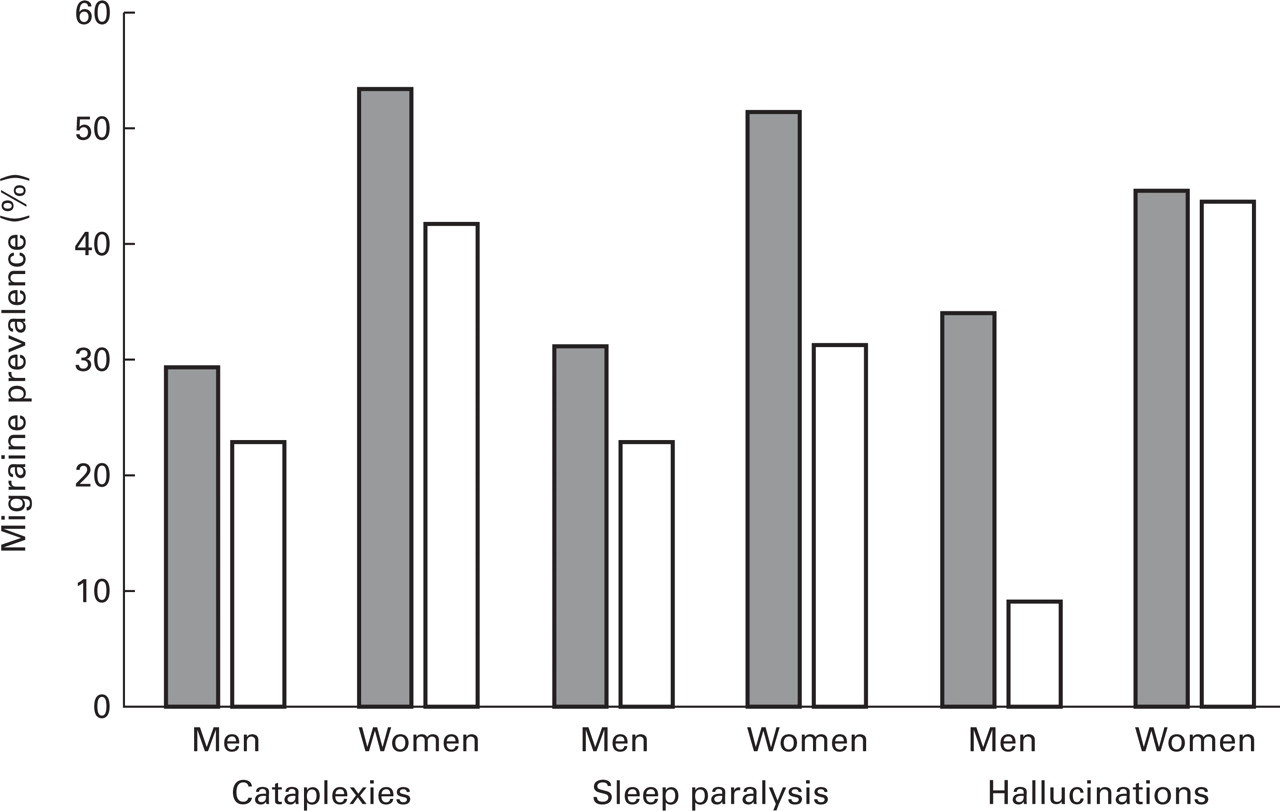
Relation between lifetime migraine prevalence of narcoleptic patients and narcolepsy symptoms. Grey bars indicate cataplexies of more than one per week or a history of sleep paralysis or sleep-related hallucinations, respectively. White bars indicate cataplexies of no more than one per week or a negative history of sleep paralysis or hallucinations, respectively.
Discussion
Almost half of the narcoleptic women and roughly a quarter of the narcoleptic men, and thus roughly 40% of the narcoleptic study population, suffered from migraine. In contrast, the lifetime prevalence of migraine in the general population as measured according to IHS criteria has been reported to be in the range of 16–25% for women and 7–8% in men (15, 16), although prevalence data obtained according to other criteria vary considerably and have been reported in the range of 1.7–46.3% for men and 3.2–57.1 in women (17). Göbel et al. (18) measured the migraine prevalence in 4061 persons aged 18 and older selected from an initial panel of 30 000 representative German households with the Kiel headache questionnaire that we also used in this study. Here, the lifetime prevalence for migraine meeting all IHS criteria was 15% in women and 7% in men. Thus, narcoleptic patients, women and men, appear to have a two- to fourfold increased risk of also suffering from migraine. These results confirm our previous study of 68 narcoleptic patients, that also had shown similarly increased migraine rates (women 64.4%, men 34.8%) (2). The increased prevalence of migraine was not due to pharmacological narcolepsy therapies, as the migraine prevalence remained elevated even when all patients on antidepressants or stimulants were excluded.
One might ask whether the study population was biased towards migraineurs, as they might be more inclined to come forward for clinical examination and to register in patient's organizations. Such a bias cannot be ruled out completely because our patients were not drawn from a representative population sample. Also, because narcolepsy is generally under-diagnosed, clinical samples might differ from the affected patients as a whole (19). In any case, the increased prevalence of migraine might not be simply attributable to the distortion of sleep observed in narcoleptic patients because distortions of sleep in snorers, patients suffering from sleep apnoea or brought upon by sleep deprivation have repeatedly been shown to be accompanied by an increased risk of various headaches but not by a specific increase in migraineous symptoms (20–23).
The second line of evidence arguing in favour of the specificity of the relation between narcolepsy and migraine is the time relationship between the ages of onset of both disorders. On average first migraine symptoms occurred 12.3 years after the first narcolepsy symptom. This is all the more remarkable as many of the migraine patients became migraineous rather late in life when typically the risk of still developing migraine becomes very small. For example, in the representative population survey of Göbel (11), only 5% of the migraineurs had developed symptoms after the age of 35 and only a ‘few exceptions’ after the age of 40. In the narcolepsy patients the corresponding numbers were 41% and 31%. Thus, narcolepsy appears to put people at risk for migraine who otherwise would have outlived this risk. Moreover, narcoleptic patients with migraine displayed a tendency towards an earlier onset of narcolepsy symptoms.
The increased migraine prevalence observed in our current as well as in the previous narcoleptic study population might indicate common pathophysiological pathways relevant to both disorders. However, no clear-cut relation was established between the severity of narcolepsy symptoms and the migraine lifetime prevalence. Nevertheless, for all REM-associated symptoms (cataplexies, sleep paralysis and hypnagogic or hypnopompic hallucinations) the prevalence of migraine was numerically highest when the respective symptom was present or more pronounced. Although not statistically significant, this observation might point towards a relation between REM distortion and migraine rather than a relation between vigilance regulation and migraine. This interpretation is also in accordance with some of the potential links between narcolepsy and migraine.
The neuroanatomical base of both disorders may involve the brainstem. Lesions in the periaqueductal grey and the dorsal raphe nuclei have been reported to induce migraine symptoms (24). Furthermore, regional blood flow studies have shown that brainstem regions corresponding to the predominantly serotonergic dorsal raphe nuclei and the predominantly noradrenergic locus ceruleus are activated during acute migraineous pain attacks (25). Cells in these regions play a critical role in REM sleep (26), a regulatory disturbance of which is thought to underlie narcolepsy/cataplexy. Most cells of these regions show a similar discharge pattern during the sleep–wake cycle, with the lowest discharge rate during REM sleep (27, 28). REM sleep itself has been linked to migraine and it has been reported that patients awakening to migraine attacks do so frequently from the REM phase of sleep (29). Furthermore, REM sleep time might be altered in migraine patients (7).
In addition, the serotonergic system might provide a link between the two disorders. So-called ponto-geniculo-occipital spikes, an electrophysiological feature of REM sleep, can be inhibited by serotonergic mechanisms (30). Several serotonergic drugs, including antidepressant selective serotonin reuptake inhibitors (SSRIs), are known to modulate REM sleep and also to affect migraine states. Clinically, anti-depressive medication sometimes has to be discontinued because of an increased frequency of migraine attacks (31). In some patients, however, SSRI antidepressants may reduce the incidence of migraine attacks (32, 33).
More recently, dysbalances in the hypothalamic orexinergic systems and a loss of orexinergic cells have been proposed to play a causative role in idiopathic narcolepsy (34). To our knowledge data on the integrity of the orexinergic system in migraine patients have not been reported so far. However, data derived from cultured spinal cord neurones and orexin expression data suggest that in addition to possible roles in endocrine regulation, the descending orexinergic fibre system may play a role in modulation of sensory input, particularly in regions related to pain perception and autonomic tone (35).
In summary, the study confirms our previous observation of an association of migraine with idiopathic narcolepsy in both women and men. The confirmation of this association in an independent large study sample may serve to stimulate aetiologically orientated research in either of the two conditions. Clinically, we recommend specifically inquiring about headache symptoms in narcoleptic patients.
Footnotes
Acknowledgement
This article contains parts of the doctoral thesis of S.W., B.U. and M.K. The authors wish to thank Mrs Lydia Michaelis for the skillful assistance in the preparation of the manuscript.
