Abstract
Cognitive processing as measured by event-related potentials (ERP) in patients suffering from the explosive subtype of headache associated with sexual activity (HSA type 2) was investigated. Visual ERP were measured in 24 patients with HSA type 2 outside the headache period. The differences of the first and the second part of measurement were evaluated separately to determine the amount of cognitive habituation. Twenty-four sex- and age-matched healthy subjects and 24 patients with migraine without aura served as controls. A missing increase of P3 latency during the second part of the measurement was found in 79% of patients with HSA type 2 and in 75% with migraine, but only in 17% of the healthy controls (P < 0.001). The P3 amplitude was increased during the second part in 71% of patients with HSA type 2 and in 79% with migraine, but only in 33% of the healthy controls (P = 0.02). Mean P3 latency was decreased and mean P3 amplitude was increased during the second part of the measurement in HSA type 2 and in migraine but not in the healthy control group. Patients with HSA type 2 have a loss of cognitive habituation as measured by ERP. This specific information processing is very similar to that in migraine observed in previous studies.
Introduction
Headaches associated with sexual activity (HSA) have been recognized since the time of Hippocrates (1). Kriz described 51 patients with neurological complications occurring during sexual intercourse (2). Intense headaches were the most frequent symptoms the patients complained of. The first systematic investigations of HSA were published in the 1970s (3–5). In their landmark papers, Paulson and Klawans (5) and Lance (6) distinguished three different types of HSA which are regarded as idiopathic.
The International Headache Society (IHS) classifies HSA as an idiopathic headache under ‘miscellaneous headaches unassociated with structural lesions’ (diagnosis 4.6.1–4.6.3) and differentiates three subtypes (7). Type 1 is a dull ache in the head and neck which intensifies as sexual excitement increases. Type 3 is a postural headache resembling the one caused by low cerebrospinal fluid pressure. This headache develops after coitus. Type 2, also called the explosive subtype, is the most frequent type, accounting for 69% of the first 70 published cases of HSA (8). Its intensity and sudden onset make the exclusion of a subarachnoid haemorrhage necessary (9).
Although much data are available on epidemiology (8), co-morbidity (10, 11), natural course (12, 13), and therapy (14) of HSA, there are no experimental studies on the pathogenesis of HSA. The present pathophysiological concepts of HSA are still based on the hypotheses of the discoverers of this headache type (5, 6).
Event-related potentials (ERP) are a useful tool to investigate cognitive processing in primary headache disorders. Migraine patients are characterized by potentiation instead of habituation of stimulation-evoked cortical responses. This abnormality is not found in other primary headache disorders such as tension-type headache or cluster headache, and may play a pivotal role in migraine pathogenesis (15–17). Since there is a significant association between HSA type 2 and other primary headache disorders, we performed a study on visual ERP in patients with HSA type 2 to investigate the involvement of cognitive processing in the pathophysiology of this headache disorder. Data were compared with a healthy control group, as well as with a group of patients suffering from migraine without aura (MO).
Methods
Subjects
We enrolled 25 consecutive patients (19 men, six women) with the diagnosis of HSA type 2 (explosive type) according to the criteria of the IHS (7). Mean age was 41.7 ± 11.3 years, mean duration of the disease was 3.4 ± 6.2 years (range 0–20 years). Sixteen patients were investigated after their first attack or first series of attacks of HSA type 2. In all patients with the first HSA attack, symptomatic headache was excluded by a computed tomography (CT) or magnetic resonance imaging (MRI) scan of the brain and by a lumbar puncture. Optionally, an additional CT or MRI angiography of the brain was performed. Patients’ histories were taken, and coexisting primary headaches were diagnosed according to the IHS criteria (7). A possible family history of migraine was investigated. Twenty-two patients were examined in a headache-free period at least 24 h after the end of the headache attack, one during the headache phase of HSA, and two both during and after the headache phase.
The data of the 24 headache-free HSA patients (19 men, five women) were compared with the data of two control groups. Control group 1 consisted of 24 healthy subjects. This control group was matched for sex (19 men, five women) and age (mean age 41.7 ± 11.3 years). The control subjects had no history of any primary or secondary headache. In control group 2, we enrolled 24 patients suffering from MO according to the criteria of the IHS (7). The migraine group was also matched for sex (19 men, five women) and age (41.4 ± 11.8 years). Mean duration of migraine was 12.6 ± 9.2 years. The examination was performed at least 72 h after the last migraine attack.
The occurrence of the next headache episode was controlled for by a telephone call 48 h after the examination. No patient included in the study suffered from a HSA or migraine attack between the examination and the telephone call. The patients with MO had no history of any other headache. No continuous drug intake and no acute drug intake on the day of examination or the day before were allowed for patients and controls.
ERP measurement
ERP were always measured at the same time of day in a dark air-conditioned room by a method published previously (16). To summarize, the subjects were seated in a comfortable chair and asked to watch a 30 × 30-cm screen. They were instructed to press a button whenever a red light occurred on the screen and to ignore a white light. ERP were evoked by an oddball paradigm using 15% red flashes of light (6 candela) and 85% white flashes of light (17 candela). A total of 400 red and white flashes of light were presented in random order. The duration of a single flash was 100 ms, with an interval of 1800 ms. EEG was recorded by an amplifier using Ag/AgCl surface electrodes placed according to the International 10–20 System at centroparietal (Pz) and linked to the right mastoid as the reference electrode. Eye movement was controlled by electro-oculography to exclude EEG periods with eye movement artefacts from the subsequent averaging process. A high-frequency filter was set at 70 Hz and a low-frequency filter at 0.1 Hz. The EEG was stored digitally. EEG periods of 300 ms before and 1100 ms after stimulus onset were averaged separately for the red and the white flashes of light.
The components of the ERP after the red (= target) stimulus were evaluated. Latencies of the endogenous P2, N2, and P3 components, the amplitude of the P3 component (compared with prestimulus baseline), and the mean choice reaction time (i.e. onset of pressing the button) were determined according to international recommendations (18). The endogenous components represent the information processing such as short-term memory and stimulus evaluation. The analysis of the ERP components was performed by a physician who was blinded to the diagnosis. Latencies and amplitudes were evaluated considering the whole measurement (60 target stimuli).
In a subanalysis, EEG periods were averaged and ERP components and the reaction time were evaluated separately for the first and the second half of the measurement, here termed ‘first trial’ and ‘second trial’ (30 target stimuli per trial), to determine the amount of cognitive habituation (16). An increase of the P3 latency and a decrease of the P3 amplitude during the second trial were termed ‘habituation’, no change or even a decrease of the P3 latency and an increase of the P3 amplitude were termed ‘loss of habituation’.
Statistical analysis
The data are presented as arithmetic means with 1 SD or as percentage. Non-parametric tests for statistical comparisons were used. The data of the HSA group and the control groups were compared by the Kruskal–Wallis analysis. Comparison between values of the first trial and the second trial was performed by the Wilcoxon test. The frequency of a ‘loss of habituation’ in the HSA and the control groups was compared with the χ2 test. Latencies were related to the age (all groups) and to the duration of the disease (HSA, MO) by regression analysis (Spearman rank coefficient). The level of significance was set at P < 0.05.
Results
Nine patients with HSA type 2 suffered from this disorder alone, 10 had one other primary headache, four had two, and two had three other headaches. Other primary headaches were diagnosed as episodic tension-type headache (IHS diagnosis 2.1, n = 9), MO (diagnosis 2.1, n = 6), benign exertional headache (diagnosis 4.5, n = 6), chronic tension-type headache (diagnosis 2.2, n = 2), and idiopathic stabbing headache (diagnosis 4.1, n = 1). Out of the six HSA patients suffering from MO, five also had a family history of migraine.
The mean latencies and amplitudes of the ERP components and the mean reaction time in pressing the button in the HSA and the control groups are presented in Table 1. Statistical analysis revealed no significant differences between the groups. There was a tendency to an increased P3 amplitude (P = 0.07) and a decreased mean choice reaction time (P = 0.06) in migraine patients. P3 latency was significantly correlated with age (r = 0.5, P = 0.001), but not with the duration of the disease in the HSA and MO groups (r = 0.04).
Total latencies and amplitudes of event-related potential (ERP) components at the centroparietal electrode and mean reaction time (arithmetic mean and simple standard deviation)

Statistical comparison between the groups by Kruskal–Wallis analysis (for further details see text). NS, Not significant.
The latencies of the ERP components, the P3 amplitude, and the reaction time were evaluated separately for the first half of the measurement (first trial) and the second half (second trial). P2 latencies, N2 latencies, and reaction times revealed only small differences not reaching a statistically significant level. The mean P3 latency was significantly increased during the second trial in the control group (P = 0.001, Fig. 1), and there was a tendency to a reduced P3 amplitude (P = 0.06, Fig. 2). In contrast, the mean P3 latency was significantly decreased during the second trial in the HSA group analysed during a headache-free period (P = 0.02, Fig. 1), and the P3 amplitude was significantly increased (P = 0.02, Fig. 2). In the MO group, there was a tendency towards a decreased P3 latency in the second trial (P = 0.06, Fig. 1), and the P3 amplitude was significantly increased (P = 0.002, Fig. 2). The mean values of the P3 latencies and amplitudes in the first and second trials are presented in Figs 1 and 2. Typical computer printouts of an ERP in a patient suffering from HSA type 2 and of an ERP in a healthy subject are presented in Fig. 3.

P3 latency in ms of the first trial (▪) and the second trial (□) in healthy controls, migraine without aura (MO), and headache associated with sexual activity (HSA) type 2. Statistical significance between both trials (Wilcoxon test) is marked: ∗P < 0.05.

P3 amplitude in µV of the first trial (▪) and the second trial (□) in healthy controls, migraine without aura (MO), and headache associated with sexual activity (HSA) type 2. Statistical significance between both trials (Wilcoxon test) is marked: ∗P < 0.05.
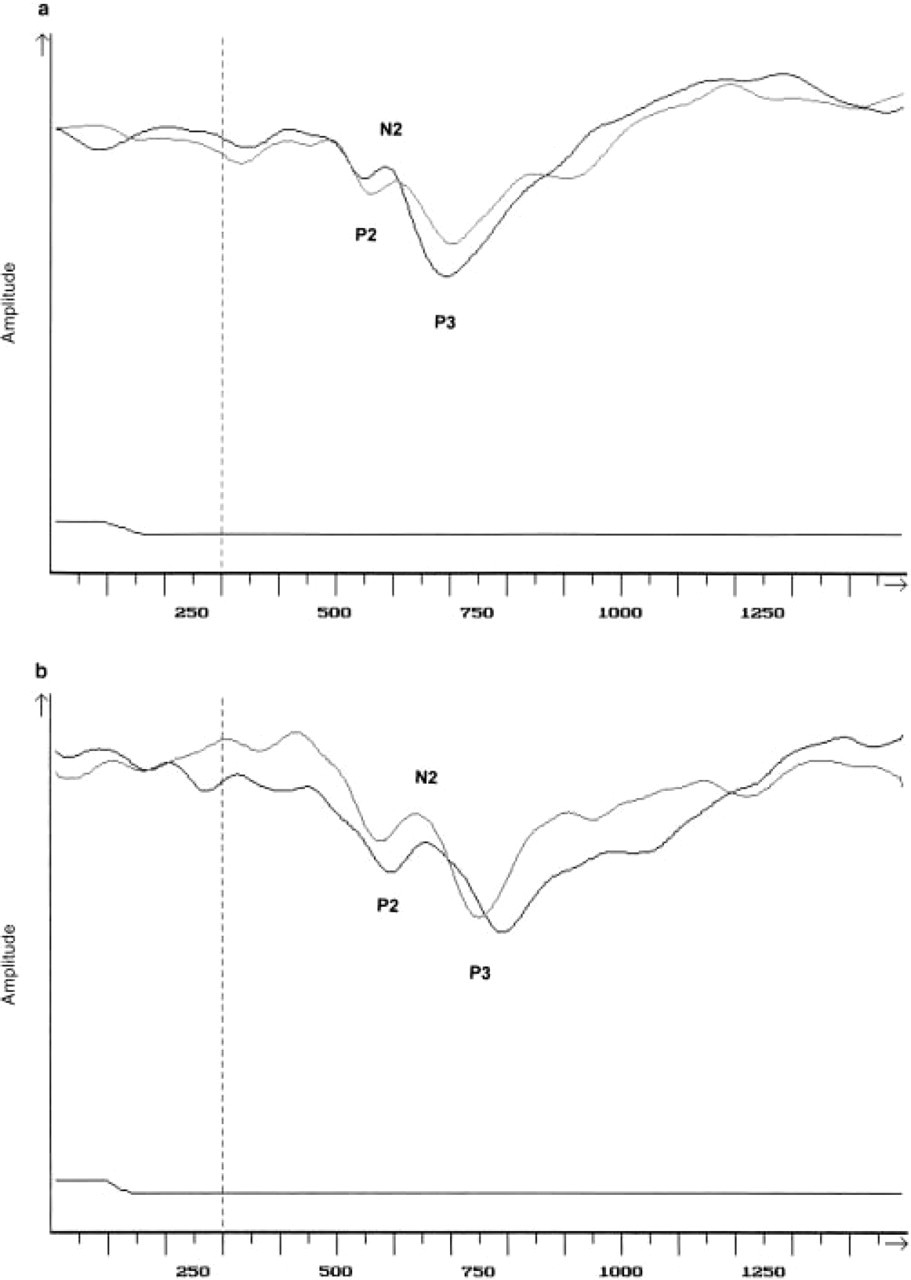
a. Typical computer printout of event-related potentials (ERP) in a patient suffering from headache associated with sexual activity (HSA) type 2. b. Typical computer printout of ERP in a healthy control. Thin lines, ERP of the first trial; thick lines, ERP of the second trial. Note that the P3 component of the second trial is faster and larger than the P3 component of the first trial in the HSA patient, but not in the healthy control.
Regarding the latency, a loss of habituation (unchanged or decreased P3 latency during the second trial) was seen in 19 (79%) of 24 headache-free HSA patients, in 18 (75%) of 24 migraine patients, but only in four (17%) of their 24 matched controls. This difference was significant (P < 0.001). Regarding the amplitude, a loss of habituation (unchanged or increased P3 amplitude during the second trial) was seen in 17 (71%) of 24 headache-free HSA patients, in 19 (79%) of 24 migraine patients, but only in eight (33%) of 24 matched controls. This difference was also significant (P = 0.002).
A second analysis between HSA patients and their respective healthy controls was performed leaving out HSA patients with a migraine co-morbidity or a family history of migraine (n = 18). With respect to latency, a loss of habituation was seen in 14 (78%) of 18 HSA patients and in three (17%) of 18 controls. Regarding the amplitude, a loss of habituation was seen in 13 (72%) of 18 HSA patients and in five (28%) of 18 controls. The difference was still significant (P = 0.001 for the latency, P = 0.02 for the amplitude, Fisher's exact test). Also in the HSA patients without migraine co-morbidity or family history, a significantly decreased mean P3 latency and a significantly increased mean P3 amplitude during the second part of the measurement could be observed. The mean P3 latency was 441 ± 40 ms in the first trial vs. 430 ± 25 ms in the second trial (P = 0.04). The mean P3 amplitude was 46 ± 20 µV in the first trial vs. 51 ± 24 µV in the second trial (P = 0.04).
The three HSA patients investigated during the headache phase showed an increased P3 latency and a decreased P3 amplitude during the second trial.
Discussion
We present a sample of patients with HSA type 2 which is in concordance with other case series finding a marked male predominance (6, 8, 10, 11, 13) and a mean age of about 40 years (6, 8, 10, 11, 13). Our patients with HSA type 2 had a high co-morbidity with other primary headache disorders. The high co-morbidity with migraine (24%) and benign exertional headache (24%) resembles the results of former studies. Other authors diagnosed migraine in 19–47% (10, 11, 13) and benign exertional headache in 31–40% (10, 11) of patients with HSA type 2. However, our patients had the highest co-morbidity with tension-type headache (44%) which was hitherto thought to be a specific feature of HSA type 1 (6). To date, no systematic experimental studies on the aetiology of HSA type 2 exist. The current hypotheses published in the literature were put forward by authors presenting case reports or case series.
Most authors assumed that HSA type 2 is a vascular headache or a disturbance of arterial blood pressure with dilated intracranial vessels and increased intracranial pressure (5, 6, 11, 14, 19, 20). Other authors impicated an abnormal release of neurotransmitters and vasoactive substances such as neurokinin or serotonin (5,21) or catecholamines (4) in causing this type of headache. Decades ago, patients with HSA were described as ‘anxious personalities’ and ‘under some emotional stress’ (4), but a psychosomatic background of the disease is no longer presumed (14). Finally, some authors emphasized a pathophysiological relationship between HSA type 2 and migraine (10, 22). In addition to the co-morbidity of these two headache disorders, a family history of migraine in 11–30% of patients with HSA type 2 (8, 10) and the familial occurrence of HSA type 2 (8) support such a relationship. Propranolol is effective in the prophylactic treatment of HSA type 2 (8, 11, 14, 23) and is one of the first-line drugs for the prophylaxis of migraine. Recently, a successful preventive treatment with naratriptan, an anti-migraine drug taken immediately before sexual intercourse, has been reported anecdotally (24). Furthermore, patients with HSA type 2 can suffer from vegetative symptoms or unilateral pain otherwise typical for migraine (14).
The most important finding of our study is that patients with HSA type 2 have a loss of cognitive habituation as measured by visual ERP. They share this characteristic feature with migraine patients. Earlier studies had investigated the contingent negative variation in migraine to evaluate cognitive processing. Analysis of the contingent negative variation had revealed evidence for a specific influence of migraine on cognitive processing which was described as a loss of habituation, and, for the time being, had been explained by a cortical hyperexcitability (25, 26). A potentiation of auditory ERP instead of a physiological habituation also pointed to a dysfunction in cortical information processing in migraine (27). According to more recent research, the abnormal information processing in migraine is an interictal cortical dysfunction probably due to inadequate control by the so-called ‘state-setting, chemically addressed pathways’ originating in the brainstem. It is now assumed that a low preactivation level of sensory cortices results, which may play a pivotal role in migraine pathogenesis in conjunction with a decreased brain mitochondrial reserve that favours a rupture of metabolic homeostasis (28, 29). Resulting biochemical shifts are postulated to activate the trigeminovascular system and, thus, to produce a migraine attack (30–32).
Evers et al. investigated visual ERP in 108 patients with migraine (16). A decrease of the P3 latency during the second trial was found in 75% of migraine patients, which is very close to the 79% of HSA type 2 patients and 75% of migraine patients reported here. We have also found this loss of habituation in the P3 amplitude which was increased during the second trial in 71% of the HSA type 2 patients and in 79% of migraine patients. A loss of cognitive habituation is not a unique feature of migraine and HSA, and can also be found in a minority of healthy controls [(16), in this study 17% for the P3 latency and 33% for the P3 amplitude]. Familial influences on cortical evoked potentials have been shown in migraine (33, 34). The existence of first and second relatives with migraine could be an explanation for the dysfunction of cognitive processing in a minority of so far healthy controls and should be controlled for in further studies.
A loss of cognitive habituation seems to be a common feature of migraine and HSA type 2. It was not found in any other primary headache disorder such as tension-type headache, cluster headache, or chronic paroxysmal hemicrania (16, 17). Migraine patients show this loss of habituation only in the interval between headache attacks, but they have a normal habituation up to 24 h before and during the attack (35, 36). This phenomenon was also observed here in the three HSA type 2 patients investigated during an attack.
The mean latencies and amplitudes of ERP components and the mean choice reaction time did not differ significantly between HSA type 2 patients, MO patients, and healthy controls. However, there was a tendency towards an increased P3 amplitude and a decreased mean choice reaction time in MO. Previous studies failed to show significant differences between MO and healthy controls, whereas migraine with aura, tension-type headache, and cluster headache during the cluster period were characterized by an increased cognitive processing time (16, 17).
In conclusion, our data suggest that the pathophysiology of HSA type 2 also involves cognitive processing as measured by ERP. The dysfunction in cortical information processing is very similar to the one observed in MO. It is regarded as a susceptibility marker for either disease and supports a pathophysiological relationship between them.
