Abstract
Background
To differentiate primary headache associated with sexual activity from other devastating secondary causes.
Methods
In this prospective cohort, we recruited consecutive patients with at least 2 attacks of headache associated with sexual activity from the headache clinics or emergency department of a national medical center from 2005 to 2020. Detailed interview, neurological examination, and serial thorough neuroimaging including brain magnetic resonance imaging and magnetic resonance angiography scans were performed on registration and during follow-ups. Patients were categorized into four groups, i.e. primary headache associated with sexual activity, reversible cerebral vasoconstriction syndrome, probable reversible cerebral vasoconstriction syndrome, and other secondary headache associated with sexual activity through a composite clinic-radiological diagnostic algorithm. We compared the clinical profiles among these groups, including sex, age of onset, duration, quality, and clinical course (“chronic” indicates disease course ≥ 1 year). In addition, we also calculated the score of the reversible cerebral vasoconstriction syndrome2, a scale developed to differentiate reversible cerebral vasoconstriction syndrome from other intracranial vascular disorders.
Results
Overall, 245 patients with headache associated with sexual activity were enrolled. Our clinic-radiologic composite algorithm diagnosed and classified all patients into four groups, including 38 (15.5%) with primary headache associated with sexual activity, 174 (71.0%) with reversible cerebral vasoconstriction syndrome, 26 (10.6%) with probable reversible cerebral vasoconstriction syndrome, and 7 (2.9%) with other secondary causes (aneurysmal subarachnoid hemorrhage (n = 4), right internal carotid artery dissection (n = 1), Moyamoya disease (n = 1), and meningioma with hemorrhage (n = 1)). These four groups shared similar clinical profiles, except 26% of the patients with primary headache associated with sexual activity had a 3 times greater chance of running a chronic course (≥ 1 year) than patients with reversible cerebral vasoconstriction syndrome. Of note, the reversible cerebral vasoconstriction syndrome2 score could not differentiate reversible cerebral vasoconstriction syndrome from other groups.
Conclusion
Our composite clinic-radiological diagnostic algorithm successfully classified repeated headaches associated with sexual activity, which were predominantly secondary and related to vascular disorders, and predicted the prognosis. Primary headache associated with sexual activity and reversible cerebral vasoconstriction syndrome presented with repeated attacks of headache associated with sexual activity may be of the same disease spectrum.
Keywords
Background
Headache associated with sexual activity (HSA) was previously named “benign” orgasmic cephalalgia in the 1970s (1,2). However, the clinical features of primary HSA have great resemblance and overlap with other life-threatening secondary causes that may also cause headaches attributed to sexual activity such as subarachnoid hemorrhage (SAH), arterial dissection or venous abnormalities (3–7). Although there have been substantial case reports, large-scale studies that have systematically investigated HSA are lacking, resulting in diagnostic strategies for HSA being based mostly on anecdotal experiences or expert opinions.
The International Classification of Headache Disorders, third edition (ICHD-3) defines
In previous studies that did not exclude patients with a single attack, more than 90 percent of the patients with HSA or RCVS presented with multiple attacks. A case series that included 60 patients with HSA, 55 (92%) suffered repeated attacks (5). As for RCVS, one case series that included 67 patients found that 63 (94%) suffered from repeated attacks (10). Although this could have excluded a minority of patients, in order to decrease the heterogeneity, we recruited a large sample of patients with repeated HSA from 2005 to 2020 in this study. Our aim was to develop a composite diagnostic algorithm for repeated HSA based on exhaustive intracranial neuroimaging work-up and ICHD-3 criteria to avoid misdiagnosis. In addition, we also determined the RCVS2 score (18), which was originally developed to assist in the diagnosis of RCVS.
Methods
Patients and clinical settings
In this prospective cohort study, we recruited consecutive patients presenting with at least 2 attacks of HSA over a 15-year period from May 2005 through January 2020. The study was conducted in the headache clinics or emergency department of Taipei Veterans General Hospital, a national tertiary medical center in the capital of Taiwan. They all received a detailed, questionnaire-based, semi-structured interview and a thorough neurologic examination by board-certificated neurologists when they first visited the clinics or emergency department. The inclusion criteria were (1) patients with at least 2 headache attacks brought on by and occurred during sexual activity, and (2) the head pain increased in intensity with increasing sexual excitement or abrupt explosive intensity just before or with orgasm. All included patients received initial brain magnetic resonance imaging (MRI) and magnetic resonance angiography (MRA) within one month of the first headache attack in order to increase the homogeneity of the recruited patients, and at least one follow-up MRI and MRA 3 months after disease onset. The protocols have been reported in detail previously (15,19). Patients who had primary headache such as migraine occurring after sexual activity were carefully excluded by experienced neurologists after detailed interview and comprehensive diagnostic work-ups (20,21). The cerebrospinal fluid (CSF) analysis was done under discretion of the responsible physician and not systematically. This study protocol was approved by the Institutional Review Board of Taipei-VGH, and all patients provided a signed informed consent to join the study. In this cohort, we followed STROBE guideline (Supplementary 1), and every item was fulfilled except that there existed selection bias because no patient with a single attack of HSA was enrolled and studied.
Data collection at baseline
Demographics; self-reported medical illnesses; headache profiles, including number of attacks, previous attacks, localization, quality (including explosive, throbbing, lancinating, dull, stabbing, or tearing sensation), thunderclap-like pattern, duration of one headache episode and associated symptoms (including nausea, vomiting, photophobia, or phonophobia), and additional headache triggers; comorbidities (e.g., hypertension, other pre-existing headaches); and neuroimaging findings were all recorded. Thunderclap-like headache was defined by a sudden severe headache reaching its maximal intensity in less than one minute.
Neuroimaging studies
Neuroimaging studies including brain MRI and MRA were used to exhaustively identify secondary causes of HSA. The MRI protocol consisted of T1-weighted imaging, T2-weighted imaging, and contrast-enhanced T1-weighted imaging sequences to exclude structural lesions; diffusion weighted imaging (DWI) and apparent diffusion coefficient (ADC) mapping to exclude ischemic stroke or posterior reversible encephalopathy syndrome; gradient echo (GRE) and/or susceptibility weighted imaging (SWI) to exclude subtle intracerebral or subarachnoid hemorrhages; fluid-attenuated inversion recovery (FLAIR) to evaluate white matter hyperintensity lesions or subtle SAH; contrast-enhanced FLAIR imaging to exclude disruption of blood-brain barrier (BBB) (since January 2016); three-dimensional time-of-flight (TOF) MRA to assess segmental cerebral arterial vasoconstriction or other vascular abnormalities such as aneurysm or arterial dissection. Magnetic resonance venography (MRV) was performed in most of our patients to exclude cerebral venous sinus thrombosis or high-grade sinus stenosis (19,22). The procedures of MRI have been detailed elsewhere (15,19,22,23).
Of note, MRI and MRA were performed using 1.5-tesla (prior to 2009) or 3-tesla (since 2009) MRI machines. Sequential MRI and MRA were performed in patients with arterial vasoconstriction on the initial MRA to determine whether the cerebral arterial narrowing was reversible or caused by atherosclerosis or hypoplasia. Follow-up MRAs were conducted until the focal narrowing of cerebral arteries were reversible and back to the normal caliber or 3 months after disease onset (24).
Composite diagnostic algorithm of HSA
Based on the ICHD-3 criteria, we retrospectively confirmed and classified all these patients with HSA into four groups through a composite clinic-radiological diagnostic algorithm (see Figure 1). At first, HSA was categorized into primary and secondary based on the presence of attributable causes on neuroimages. Patients with secondary causes other than RCVS were analyzed as a separate group, i.e. “other secondary HSA”. Patients with imaging-proven reversible vasoconstriction were diagnosed with “definite RCVS”. Patients with negative imaging findings were subcategorized based on the headache trigger; patients with sexual activity as the only trigger were diagnosed with “primary HSA”, and those with multiple triggers, including sexual activity, were diagnosed with “probable RCVS”. However, patients with “probable RCVS”, where the diagnosis was unclear, would probably benefit from a CSF study to rule out secondary causes.

Flowchart of the diagnosis of headache associated with sexual activity (HSA).
Follow-up
All eligible patients were followed up at our headache clinics for at least three months. For those who continued to have headache, we kept on following up them until headache remission. The last follow-up in this study was completed in April 2020. The last 3 patients who were included in January 2020 had headache resolution prior to April 2020. The clinical course of HSA was divided into an episodic course with or without relapsing bouts and a chronic course. In this study, a “bout” was defined as at least two attacks occurring in the acute stage regardless of the frequency and then none for more than 4 weeks despite continuing sexual activity. A “chronic” course was defined as HSA lasting for more than one year with no remission longer than 4 weeks. “Relapse” was defined as two separate bouts occurring with a remission longer than 6 months. Duration in episodic or chronic HSA was defined as the length of a bout, which was differentiated from the duration of one headache episode, which was defined as the duration for each single attack.
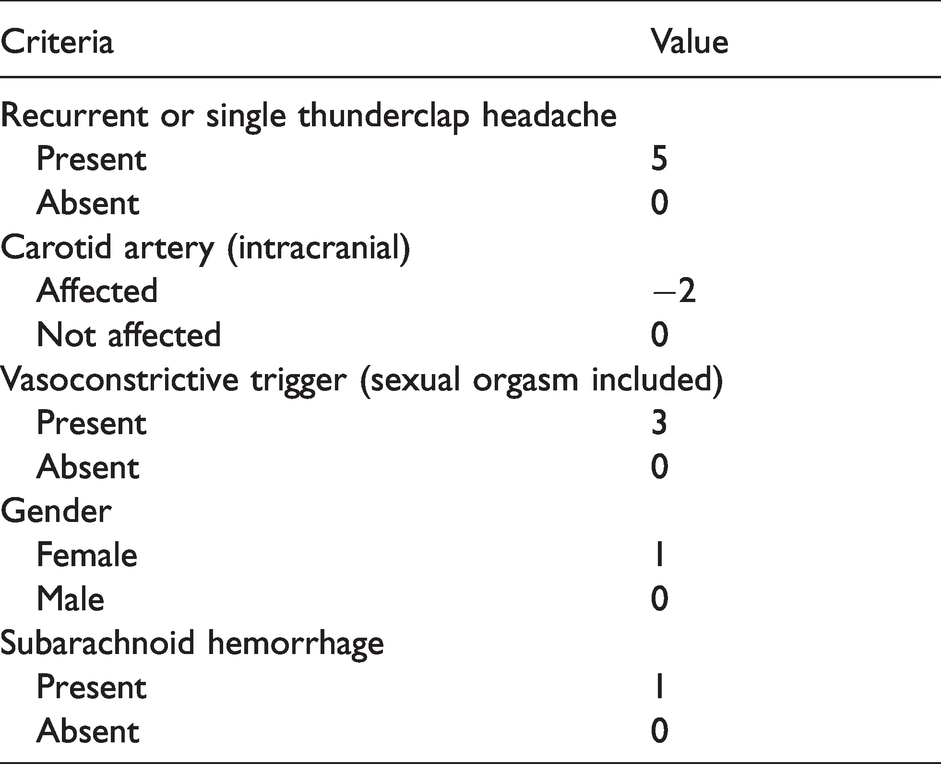
RCVS2 score in patients with HSA
The RCVS2 score was recently developed to assist in the diagnosis of RCVS, especially in distinguishing it from other intracranial arteriopathies, including primary angiitis of the central nervous system, Moyamoya disease, intracranial atherosclerosis, radiation-induced arteriopathy, and secondary vasculitis, with excellent performance (18). The score was composed of 5 items (Table 1), with scores ≥ 5 had 99% specificity and 90% sensitivity for diagnosing RCVS, and scores ≤ 2 had 100% specificity and 85% sensitivity for excluding RCVS. The RCVS2 score was not intended for use in primary headaches. As this score has not been used to differentiate RCVS from other causes of HSA, this study evaluated the distribution of RCVS2 score in our patients.
RCVS2 score criteria.
Statistics
Descriptive statistics are presented as the means ± SD or percentages. Some parameters were not normally distributed, so median values and percentiles are presented. IBM SPSS Version 22.0 for Windows was used for the statistical analysis. For continuous variables, we used the unpaired t test or analysis of variance (ANOVA) test. For categorical or ordinal values, we used chi-square tests or Fisher’s exact tests when appropriate. We also did Kaplan-Meier analysis for the disease course of each group and conducted the log-rank test. All calculated p values were two-tailed, and statistical significance was defined as a p value of less than 0.05. Missing data were handled with pairwise deletion procedure, which eliminates information only when the particular data-point needed to test a particular assumption is missing on an analysis-by-analysis basis. We did not perform a priori sample size estimation but instead include maximally available cases during the study period.
Results
Clinical characteristics of patients classified with composite diagnostic algorithm
During the study period, 251 patients (230 patients from headache clinics; 21 patients from emergency department) were screened. Six patients were excluded due to lacking brain MRI data (n = 5) or loss to follow-up (n = 1). The remaining 245 patients (169 men (69.0%), mean age 41.6 ± 10.2 [range 20–73] years) were enrolled in the analysis and received serial brain MRI and MRA on registration and during follow-ups. The average time from onset to the first brain MRI was 15.3 ± 7.9 days (range 0–30).
Based on our diagnostic algorithm (Figure 1), 7 patients were identified to have imaging-proven “secondary causes” other than RCVS, including aneurysmal subarachnoid hemorrhage (n = 4), right internal carotid artery (ICA) dissection (n = 1), Moyamoya disease (n = 1), and meningioma with hemorrhage (n = 1), which comprised 2.9% of the total group of patients with repeated HSA. The other 238 patients were further categorized into primary HSA (n = 38, 15.5%), RCVS (n = 174, 71.0%) and probable RCVS groups (n = 26, 10.6%). Of note, all 245 patients, even predominantly with secondary causes, had normal neurological examinations at the first visit. Among them, 38 patients (15.5%) had a history of hypertension and 110 patients (44.9%) had other pre-existing headache disorders, the frequencies of which, in order, were migraine (n = 61, 24.9%), tension-type headache (n = 48, 19.6%) and cluster headache (n = 1, 0.4%).
Comparisons between HSA groups
The HSAs were predominantly bilateral (68.6%) and occipital (67.3%), explosive at onset (63.7%), lasting for 4 and 24 hours (36.4%) per attack (Table 2). The mean number of thunderclap-like headache attacks in one bout was 3.8 ± 2.4 (range 2-20). The mean frequency was 3.9 ± 6.3 times a month (range 0.3-20.0). Nausea was the most common associated symptom (n = 74, 30.2%), followed by phonophobia (n = 66, 26.9%), neck pain (n = 54, 22.0%), vomiting (n = 34, 13.9%), and photophobia (n = 32, 13.1%).
Comparisons of patients with HSA.
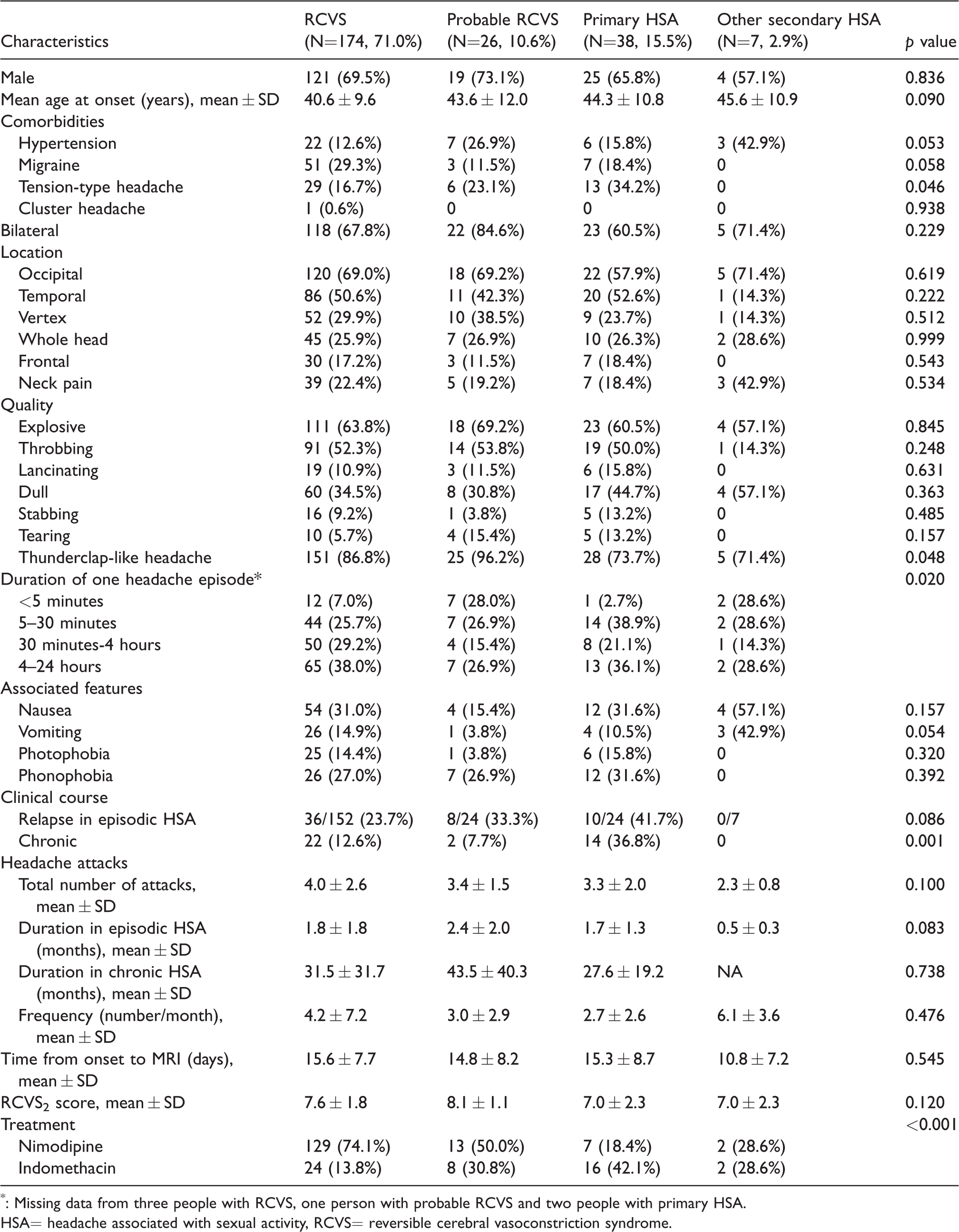
*: Missing data from three people with RCVS, one person with probable RCVS and two people with primary HSA.
HSA= headache associated with sexual activity, RCVS= reversible cerebral vasoconstriction syndrome.
The age, sex ratio, previous headache disorders, comorbidities, headache laterality, headache location, headache quality, frequency of headache attacks, total number of attacks and associated symptoms were not statistically different between the four groups, except that pre-existing tension-type headache was more common in the primary HSA group (34.2%) (p = 0.046). The proportion of HSAs presenting as thunderclap-like headaches in these groups in descending order was 96.2% in the probable RCVS group, 86.8% in the RCVS group, 73.7% in the primary HSA group, and 71.4% in the other secondary HSA group. Remarkably, all patients with other secondary HSA had at least two severe headache attacks without significant focal neurological deficits or meningismus. One patient with aneurysmal SAH even experienced five thunderclap headaches but remained neurologically intact.
RCVS2 score in patients with HSA
The distribution of the RCVS2 scores in the 245 patients with HSA is shown in Figure 2. All patients had scores ranging from +2 to +10. The mean score was similar in patients with RCVS, probable RCVS, primary HSA and other secondary HSA (Table 2). We further evaluated the proposed cut-offs of the RCVS2 score to see if this score could be used to differentiate RCVS from primary HSA or other secondary HSA. With a score of 5 as a cut-off point, the proportion of RCVS2 score ≥5 in these groups in descending order was 96.2% in the probable RCVS group (n = 25), 86.8% in the RCVS group (n = 151), 85.7% in the other secondary HSA (n = 6), 73.7% in the primary HSA group (n = 28), and 57.1% in the other secondary headache group (n = 4) (p = 0.073). Using a cut-off score ≥5, the diagnosis of RCVS had a specificity of 29% (95% confidence interval (CI): 12.9% to 39.5%) and sensitivity of 88% (95% CI: 82.7% to 92.1%); with a cut-off score ≤2, there was 100% (95% CI: 98.1% to 100.0%) specificity but only 2% (95% CI: 0.1% to 9.2%) sensitivity in excluding RCVS.

Distribution of the RCVS2 score in patients with headache associated with sexual activities (HSA).
Clinical course and prognosis
Among the 245 patients with HSA, 207 patients (84.5%) had an episodic course with mean duration of one bout as 2.0 ± 2.0 months (range 0.1–12) and 38 patients (15.5%) ran a chronic course (≥ 1 year) without remission, with a mean duration of 33.5 ± 27.7 months (range 13-120). The proportions of chronic HSA in these groups in descending order were primary HSA (n = 14, 36.8%), RCVS (n = 22, 12.6%), probable RCVS (n = 2, 7.7%), and other secondary HSA (n = 0, 0%). Of patients with episodic HSA, 54 patients (26.1%) experiencing a relapsing bout after a mean disease-free interval of 37.1 ± 42.3 months (median: 24, range 7–204) during follow-up. The relapse rate was similar in patients with primary HSA, probable RCVS and RCVS. The mean duration of HSA of each group was presented as a Kaplan-Meier curve (Figure 3), where primary HSA ran a more prolonged course (log-rank p < 0.001). Two patients (1.1%) with RCVS developed complications; one had convexity SAH, and the other had ischemic stroke. No complications were noted in the probable RCVS or primary HSA group. In our clinic, nimodipine was used as the first-line treatment for 150 patients (61.2%), and one patient used it following indomethacin. Indomethacin was used as a first-line treatment for 50 patients (20.4%). No neurological complications occurred after starting nimodipine or indomethacin. Only one patient discontinued nimodipine due to headache increase (not related to sexual activity), but his HSA resolved in 2 months.

Kaplan-Meier curves displaying the proportion of mean duration of headache associated with sexual activity (HSA) for 4 different groups of patients.
Across the four groups, 151 patients with HSA were treated with nimodipine, in which 144 experienced good response, which was defined as if no further thunderclap headaches occurred in 48 hours after the last dose escalation (25). The proportions of good response in these groups in descending order were other secondary HSA (SAH, n = 2, 100.0%), RCVS (n = 125, 96.9%), primary HSA (n = 6, 85.7%), and probable RCVS (n = 11, 84.6%) (p = 0.130).
Discussion
In this large study of patients with repeated HSA, we demonstrated that HSAs were predominantly secondary and related to vascular disorders (84.5%), with RCVS/probable RCVS (81.6%) being the leading cause and 2.9% having potentially life-threatening secondary causes. Most patients with repeated HSA presented with thunderclap-like onset of the headache (85.3%), even those with primary HSA (73.7%) or other secondary HSA (71.4%). As a result, patients with RCVS, probable RCVS, primary HSA or other secondary HSA were difficult to distinguish by clinical manifestations at disease onset. The RCVS2 score, although excellent in distinguishing RCVS from other intracranial arteriopathies, and response to nimodipine could not differentiate RCVS from primary HSA or other secondary HSA. Considering the risks of missing potentially devastating secondary causes, neuroimaging studies including detailed intracranial vascular imaging are mandatory for diagnostic work-up. Our composite diagnostic algorithm for HSA based on neuroimaging findings and the ICHD-3 criteria performed well in avoiding misdiagnosis.
When we applied the RCVS2 score to our patients, up to approximately 85% of patients with primary HSA and other secondary HSA had cut-off scores above 5, and the specificity differentiating them from RCVS was low, as they all had a high proportion of thunderclap headaches, and all patients had sexual orgasm as a vasoconstrictive trigger. As a result, the RCVS2 score could not be very useful for differentiating RCVS patients presenting as HSA from either primary or other secondary HSA. Instead, detailed intracranial neuroimaging including vascular imaging is mandatory to avoid a devastating situation. However, as we only enrolled patients with “repeated” HSA, this might affect the results of the RCVS2 score.
For patients with primary HSA, although they could completely fulfill the diagnosis of ICHD-3 4.3 primary headache associated with sexual activity, they could actually also fulfill the diagnosis of ICHD-3 6.7.3.2 acute headache probably attributed to RCVS if we only considered headache characteristics other than the duration criterion (8). The distinction between primary HSA and probable RCVS is arbitrary when judged only on the presence of “additional triggers”. In fact, two patients in our cohort could be classified as having primary HSA during the first bout but as having probable RCVS during the relapsing bouts. Our previous study suggested that primary HSA and RCVS may share similar underlying mechanisms and be on the spectrum of the same disorder (9). In this study, we further validated this speculation by demonstrating an indistinguishable clinical picture among primary HSA, RCVS and probable RCVS. This dynamic change or instability of diagnosis poses a great challenge to clinicians.
In concordance with previous reports, HSA presented in two times as many men as women (4,7,9,26,27). In fact, sexual activity has been found to be an important trigger of RCVS, especially in men (11,25,28,29). In a report of 67 French patients with RCVS, sexual intercourse (29%) was the most common trigger (10). In addition, we found an approximately five-fold higher relapse rate in this study cohort (26%) than in other reported RCVS patients (5%) (7,15,25). The high relapse rate in our patients was probably because this study enrolled only patients with sexual activity as a trigger. In our previous study, having sexual activity as a trigger for thunderclap headache was an independent predictor of relapsing RCVS (15). In addition, an intriguing finding was that patients diagnosed with primary HSA had an approximately 3 times higher chance of running a chronic course than patients with RCVS. Hence, correct classification and diagnosis of these patients could help in the prediction of the prognosis. Specific instructions may be needed for these patients to cope with future attacks such as taking pre-emptive indomethacin before sexual activity.
The low complication rate for RCVS in our cohort could be the combination of multiple factors. One possibility is that there is an increased awareness of RCVS in both physicians and general population in our country after education through media and continuing educational programs, and as a consequence, early identification and management. Patients with RCVS overall had decreasing rates of complications year by year. Another possibility may be due to referral bias. As a tertiary referral national medical center, many of our patients with RCVS were referral patients. Patients with complications could have been identified after initial surveys at other hospitals. Moreover, ethnic factors might be another possibility. Large series of RCVS showed relatively lower complication rates in Asians (6.5%) compared to Europeans (37%) and North Americans (81%) (23,30,31).
Our study has some limitations. First, the MR scanner was changed from 1.5-tesla to 3-tesla in 2009 to increase sensitivity. Hence, the resolution of MRA after 2009 may be better. However, the clinical and demographic pictures and the percentage of patients with positive findings in the MRI did not show statistically significant differences except for a higher percentage of male patients after 2009 (Supplementary Table 1). Second, although MRA is the most commonly adopted diagnostic tool for studying RCVS, it has limitations in detecting distal vasoconstrictions (32), and even a follow-up MRA performed at around 2 weeks after disease onset to capture the centripetal propagation of vasoconstrictions cannot completely overcome this limitation (33). As a result, there could be false negative RCVS cases. Nevertheless, the mean interval from disease onset to MRA performance in our study is 15 days, when the vasoconstrictions are usually visible in proximal segments of cerebral arteries (24). Third, the efficacy of the treatment response could not be assessed well because of the possibility of spontaneous resolution of both primary HSA and RCVS. Fourth, we did not correct the p values for multiple comparisons considering the exploratory nature of the study and the small numbers after subgrouping. Fifth, during bouts, patients usually abstain from sexual activity to prevent further attacks. Sixth, the criterion of primary HSA is head pain induced by sexual activity as the only trigger and probable RCVS is headache induced by at least one trigger including sexual activity. As a result, we arbitrarily excluded patients with sexual activity as the only trigger from a diagnosis of probable RCVS and rendered them a diagnosis of primary HSA for the purposes of the study. Seventh, the composite clinic-radiological diagnostic algorithm was a proposal of diagnostic criteria based on preconceived assumptions, it needed further studies to validate. Eighth, neither MRV nor CSF analysis and opening pressure determination were done systematically. There existed the possibility of some missed diagnoses of cerebral venous sinus thrombosis or other secondary causes of headache, and overdiagnosis of the causes that could be detected with an MRI, such as RCVS or other vascular disorders. Nevertheless, in patients with primary HSA, RCVS, or probable RCVS who had received spinal taps (n = 33), we did not notice any abnormalities. In one patient out of 7 with other secondary HSA who had received spinal taps, the CSF analysis revealed xanthochromia and high opening pressure, 320 mmH2O. Finally, as a tertiary referral medical center, many of our patients with HSA visited our hospital after initial surveys at other hospitals and often suffered more than 2 attacks before presentation, so there might be referral bias. In addition, the mean time of the first MRI from disease onset was as late as 15 days, even though we performed MRI as soon as the patients visited us.
Conclusion
Our composite clinic-radiological diagnostic algorithm classified repeated HSAs into four groups, i.e. primary HSA, RCVS, probable RCVS and other secondary HSA. Most cases of repeated HSA are actually RCVS and maybe primary HSA and RCVS are of the same disease spectrum. We could not observe any differences in the initial presentations of these four groups. As a result, exhaustive intracranial neuroimaging is needed. Correct diagnosis of the patients with HSA is important for outcome prediction.
Clinical implications
A comprehensive intracranial neurovascular imaging work-up is needed for distinguishing primary HSA from RCVS. HSAs are predominantly secondary and related to vascular disorders, of which RCVS is the most common. Primary HSA and RCVS presented with repeated attacks of HSA may be of the same disease spectrum. Patients with primary HSA had a 3 times greater chance of running a chronic course (≥ 1 year) than patients with RCVS.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024211028965 - Supplemental material for Diagnosis and classification of headache associated with sexual activity using a composite algorithm: A cohort study
Supplemental material, sj-pdf-1-cep-10.1177_03331024211028965 for Diagnosis and classification of headache associated with sexual activity using a composite algorithm: A cohort study by Po-Tso Lin, Yen-Feng Wang, Jong-Ling Fuh, Jiing-Feng Lirng, Yu-Hsiang Ling, Shih-Pin Chen and Shuu-Jiun Wang in Cephalalgia
Footnotes
Author contributions
SPC and SJW were responsible for conception and design of the study. PTL, YFW, JLF, JFL, SPC and SJW were responsible for acquisition and analysis of data. PTL, SPC and SJW were responsible for drafting the manuscript and figures. All authors edited and approved the final version of the manuscript.
Availability of data and materials
Anonymized data that support the findings of this study are available from the corresponding author upon request from any qualified investigator.
Ethics and approvals
The study protocol was approved by the Institutional Review Board of Taipei Veterans General Hospital, Taiwan.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Brain Research Center, National Yang Ming Chiao Tung University (to SJW & SPC) from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan; the Ministry of Science and Technology, Taiwan [MOST-107-2314-B-010-021 & 108-2314-B-010-022-MY3 (to SPC) and MOST 108-2321-B-010-014-MY2, MOST 108-2321-B-010-001- & MOST 108-2314-B-010-023-MY3 (to SJW)]; [MOST 110-2321-B-010 -005- (to SJW)] in the Ministry of Science and Technology, Taiwan; Ministry of Health and Welfare, Taiwan [MOHW107-TDU-B-211-123001 and MOHW 108-TDU-B-211-133001] (to SJW); and Taipei Veterans General Hospital, Taiwan [VGH-109-C-090 & V109E-005-1 (to SJW) and V109C-139 & V109D52-001-MY3-1 (to SPC)]. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
