Abstract
Nitric oxide (NO) may participate in the mechanisms underlying vascular headaches, such as migraine and cluster headache (CH), by triggering neurogenic inflammation and activation of fibres conveying nociceptive inputs to the trigeminal ganglion. Similarly to migraine, the administration of the NO donor glyceryltrinitrate (GTN) to CH patients is a known model of inducing spontaneous-like attacks. We carried out a GTN test (0.9 mg, sublingually) in 18 patients with episodic CH in active phase and 12 controls. The plasma levels of NO metabolite nitrites (NO2 -), after conversion of nitrates to NO2 -, were measured spectrophotometrically at baseline, at the maximum intensity of the induced response (or 45 min after GTN in controls), and 120 min after GTN administration. The basal plasma levels of L-citrulline were also assayed in patients and controls using highperformance liquid chromatography. Basal NO2 - levels, similar in GTN-responsive patients and controls (48.3 ± 10.6 and 44.6 ± 9.5 μmol/l, respectively) were found to be increased significantly at pain peak in patients (76.1 ± 10.2 μmol/l) and after 45 min in controls (78.2 ± 9.6 μmol/l) (P < 0.01 vs. respective baseline values), but not after 120 min, without differences between groups. L-citrulline levels in basal conditions showed no differences between groups (patients 64.8 ± 11.7, controls 67.3 ± 10.8 μmol/l). These data do not support the presence of a basal hyperactivity of the L-arginine-NO pathway in CH patients. Increased NO production may be of importance in the mechanisms leading to CH attacks, but other factors are likely to render CH patients hyperresponsive to NO, and ultimately to cause the occurrence of pain and associated features.
Introduction
While the clinical features of cluster headache (CH) are well defined, uncertainty still exists as to the precise pathophysiological mechanisms underlying the disorder. Autonomic dysfunction and cluster pain have been the aspects receiving most study, with considerable evidence suggesting that both phenomena may originate in the central nervous system (CNS). According to such view, CH patients may be characterized by derangement of the hypothalamic–limbic pathways subserving the autonomic, neuroendocrine and behavioural functions (1–3).
Various theories have been proposed to explain the generation of pain during CH attacks. CH is considered as a primary neurovascular headache, since it most probably involves activation of trigeminovascular pain structures projecting to the trigemino-cervical complex of neurones in the caudal brain stem and upper cervical spinal cord (4, 5). The sensory innervation of intracranial vessels, originating in the trigeminal ganglion, includes several signalling neuropeptides, such as calcitonin gene-related peptide (CGRP), substance P, neurokinin A, pituitary adenylate cyclase activating peptide (PACAP) and vasoactive intestinal peptide (VIP); in particular, a clear association exists between head pain and the release of CGRP (6). However, in addition to classical neuropeptides, other factors appear to be involved in CH pain; among these, the unconventional gaseous transmitter nitric oxide (NO) appears to play an important role. Studies of headache induced by the administration of glyceryltrinitrate (GTN), an exogenous NO donor, and histamine, which induces NO release from vascular endothelium, have suggested that NO may be a key mediator in both migraine and CH (7). There is considerable evidence to indicate that NO release from blood vessels, perivascular nerve endings and brain tissue is an important molecular trigger mechanism in spontaneous headache pain (8). It has also been proposed that patients with vascular headaches may display excess NO production and/or increased response to the activation of NO-ergic pathways (9). The intimate nature of these processes remains unclear, but it is also known that the administration of GTN modulates neuronal activity in several brain areas, particularly the brain stem and hypothalamic nuclei, which are involved in vascular headaches (10). Thus, in addition to its effect at the endothelial level, NO may act centrally, thereby participating in the processes underlying the onset of pain as well as neurovegetative signs and symptoms in migraine and CH (11).
In migraine sufferers, the sublingual (12) or systemic (13) administration of GTN is able to precipitate headache attacks, whose features resemble those of the spontaneous episodes and fulfil the diagnostic criteria for migraine. This method still represents the most reliable and reproducible paradigm of induced headache of the vascular type in humans (14). Even though with different temporal modalities, spontaneous-like CH attacks can also be triggered in predisposed individuals by GTN administration, only in the active phase (15–17).
Nitrites (NO2 –) represent stable inorganic end-products of NO metabolism (18). In this study, we have measured plasma NO2 – levels in a group of CH patients, in basal conditions as well as during a spontaneous-like attack induced using the GTN model, with the aim of further elucidating the involvement of NO pathways in CH pathophysiology. Moreover, since L-citrulline, the stoichiometric metabolite resulting from the conversion of L-arginine to NO, is considered as a specific and reliable index of nitric oxide synthase (NOS) activity in vivo (18, 19), basal plasma L-citrulline levels have also been measured in patients and controls.
Methods
The selected study group consisted of 18 patients, 16 males and two females, presenting with headache at the Headache Centre of the Neurological Institute of the University of Pavia, and enrolled consecutively. All patients were suffering from CH in active phase, according to the International Headache Society criteria (20). All patients had appearances absolutely typical of CH, with a constant presence of autonomic accompanying signs and symptoms during their pain attacks (frequency of two to four attacks/day). Patients were aged between 35 and 63 years (mean ± SD 46.3 ± 8.6). The mean ± SD of symptom duration was of 8.5 ± 3.4 years (range 3–18 years), and that of the pain attacks was of 47.0 ± 11.5 min (range 25–70 min). The latter was calculated on the basis of at least five untreated attacks recorded by patients with a dedicated diary chart. The individual clinical features of the patients studied are reported in detail in Table 1.
Clinical features of patients
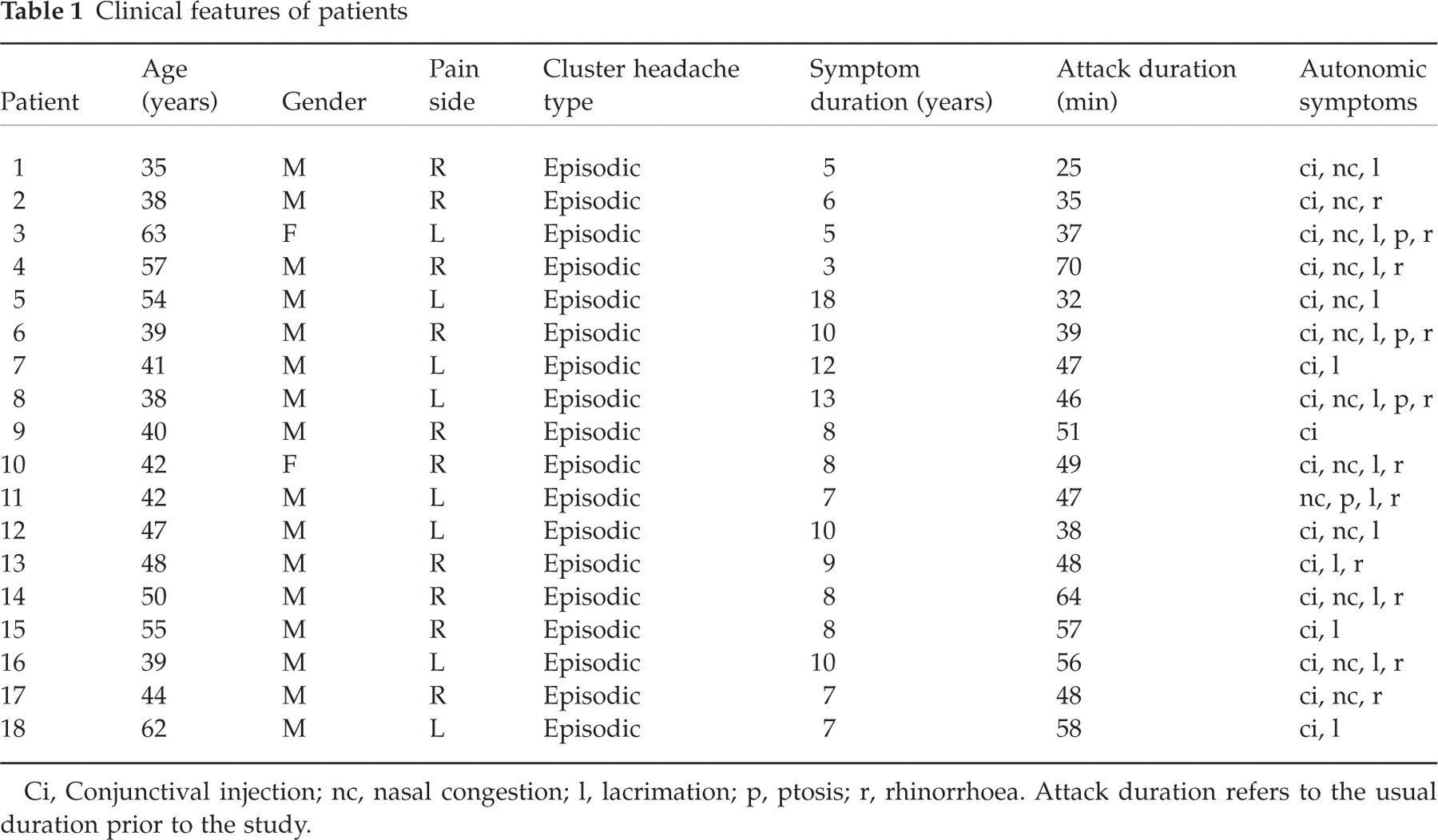
Ci, Conjunctival injection; nc, nasal congestion; l, lacrimation; p, ptosis; r, rhinorrhoea. Attack duration refers to the usual duration prior to the study.
Patients and controls had received a list of potential dietary sources of nitrates/nitrites, and had been instructed to avoid, for at least 48 h before blood sampling, the ingestion of sausages and other canned foods known to contain nitrites as preservatives. At the time of testing, patients were having regular headaches, and none of them had taken any prophylactic medication for at least 1 week prior to the study. After obtaining formal approval by the local Ethical Committee and informed consent by all subjects, a complete clinical history was collected.
As controls, we studied 12 healthy sex- and age-matched subjects (10 males, two females, mean ± SD age of 43.5 ± 8.4 years (range 34–61 years). In particular, none of them had ever suffered from headache or showed any family history of headache. Nine of the 18 patients and six of the control subjects were smokers.
After overnight fast, patients and controls underwent a standard headache-induction test, between 08.30 h and 09.30 h, carried out by administering GTN (trinitrine) 0.9 mg sublingually in headache-free conditions. This dose, currently used at our Department, has been found to induce typical pain attacks in 60–70% of CH patients in active phase (21), a percentage similar to that reported by Ekbom with 1 mg GTN (15). During each test, patients were resting in bed in a supine position, and their cardiopressor parameters (heart rate, arterial blood pressure) were continuously recorded using a vital sign monitor (Dynamap; Kriticon, Orlando, FL, USA). The time of onset of pain, the possible occurrence of general autonomic symptoms, and any changes in the degree of conjunctival injection, width of the palpebral fissure, pupillar diameter and nasal congestion (if present) were also recorded using a dedicated chart. Patients were asked to score the intensity of any provoked headache by means of a visuo-analogue scale (VAS) ranging from 0 to 10 (0 = no pain, 10 = unbearable pain). The duration of the attack, and the severity of headache and autonomic symptoms were assessed and reported in the chart. For ethical reasons, patients were free to ask for a rescue treatment (sumatriptan 6 mg s.c.) whenever required. At the end of each test, patients were also instructed to contact the Headache Centre the following day, to report on the status of their headache. During the test, blood samples (10 ml, in tubes containing sodium EDTA for nitrite and heparin for citrulline) were obtained from the cubital vein in basal conditions, at the peak of the pain response (in the case of patients) or after 45 min (in the case of control subjects), and 120 min after GTN administration.
For NO2 – assay, the other end-products of NO breakdown, nitrates, were converted into NO2 – by enzymatic reduction using nitrate reductase (Sigma, Milan, Italy) in the presence of nicotinamide adenine dinucleotide phosphate (NADPH). The Griess method was then used (22), without deproteinization, by addition to samples of equal amounts of sulphanilamide 1% and naphtylethylenamide 0.1% in phosphoric acid 0.25%, and incubation at room temperature for 10 min. Samples were then centrifuged and stored at −80°C, until final measurement of absorbance by spectrophotometric reading at 546 nm. The intra- and interassay coefficients of variation (CV) were 5% and 10%, respectively.
For L-citrulline measurement, samples were centrifuged at 4°C for 15 min at 2200 g and plasma stored at −80°C until assay. High-performance liquid chromatography (HPLC) equipment (Jasco Inc., Easton, MD, USA series LC-900), coupled with a fluorimetric detector, was used. Amino acids were separated using an inverse phase Waters Nova pak C18 column (4 µm particle size, 3.9 × 150 mm) at 25°C, at a 0.8-ml/min flow (linear gradient). After adding perchloric acid 0.4 N (1 : 1 v/v), samples were centrifuged for 15 min at 4°C. The supernatant was properly diluted with methanol (MeOH), and after addition of ophtaldialdehyde (OPA) the fluorescent derivative was analysed.
Statistical analysis of data was performed using the analysis of variance (ANOVA), where appropriate. Dunnett's correction for multiple comparisons was used to compare plasma NO2 – levels during GTN test with those obtained prior to GTN administration. Differences were considered significant if P < 0.05. Data are expressed as mean ± SD.
Results
Twelve of the 18 patients, all males, experienced a typical, spontaneous-like attack on the usual side, occurring in all cases within 45 min (mean ± SD of latency 32.5 ± 6.3 min, range 19–41 min). Pain peak was reached (and blood samples obtained) after 12.6 ± 2.9 min (range 8–18 min). Only four patients asked for the rescue treatment (sumatriptan 6 mg s.c.), which was administered after the second blood sampling with prompt relief. In the other patients, pain duration (range 30–60 min) and severity were reported as being similar to those of the usual attacks, as were type and intensity of autonomic signs and symptoms. The clinical features of the 12 GTN-responsive patients are reported in Table 2. As expected, none of the control subjects experienced pain or any autonomic signs or symptoms.
Clinical features of the 12 glyceryltrinitrate-responsive patients during induction test
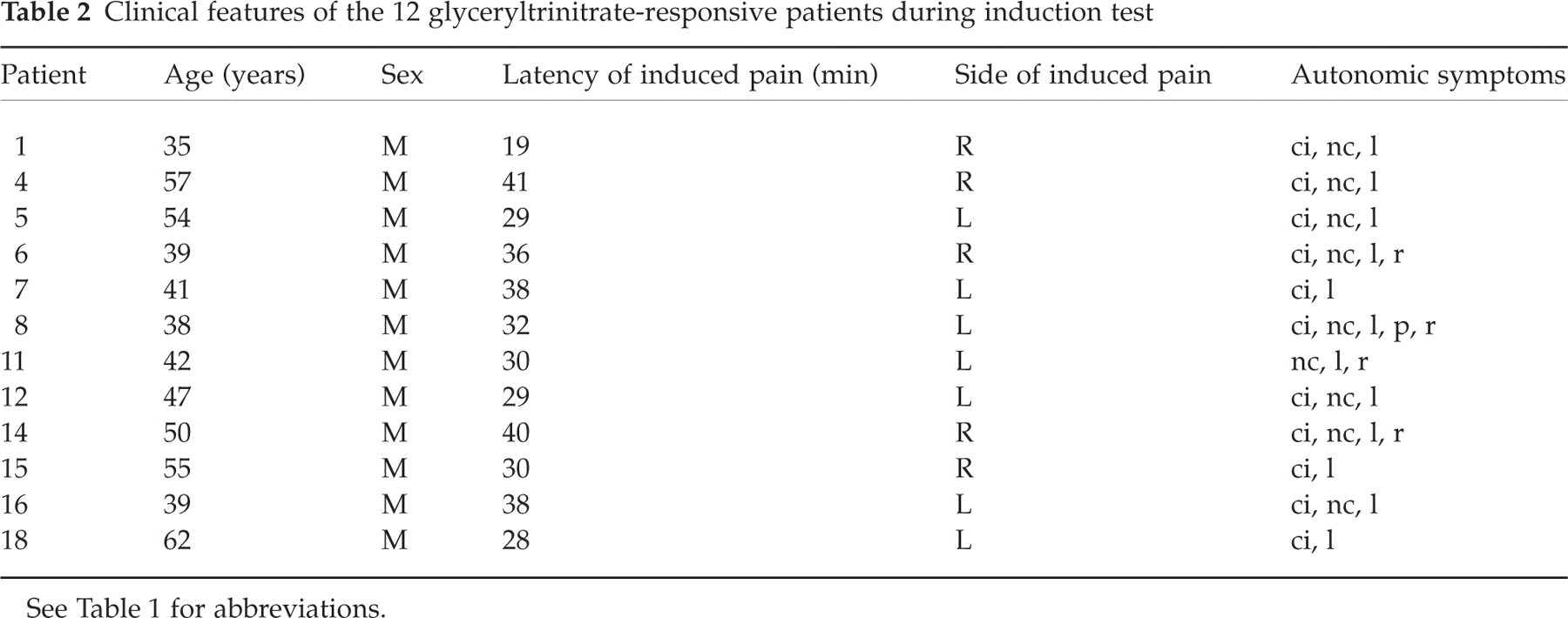
See Table 1 for abbreviations.
Baseline NO2 – levels (Fig. 1) were similar in patients and controls, and were found to be significantly increased at pain peak in the patient group, and 45 min after GTN administration in the control group (P < 0.01 vs. respective basal levels). In both groups, 120 min after GTN administration, NO2 – concentrations were no longer different from their baseline values.

Mean ± SD of plasma levels of NO2 – (µmol/l) in basal conditions and during glyceryltrinitrate test in 12 cluster headache patients (▪) and 12 control subjects (□). ∗P < 0.01 vs. respective baseline values. Patients vs. control subjects: NS.
Similar to NO2 –, plasma L-citrulline levels in basal conditions were not different between groups (Fig. 2). In the six GTN-unresponsive CH patients, basal NO2 – and L-citrulline levels, as well as NO2 – levels during GTN test, were not statistically different from those of GTN-responsive ones (data not shown).
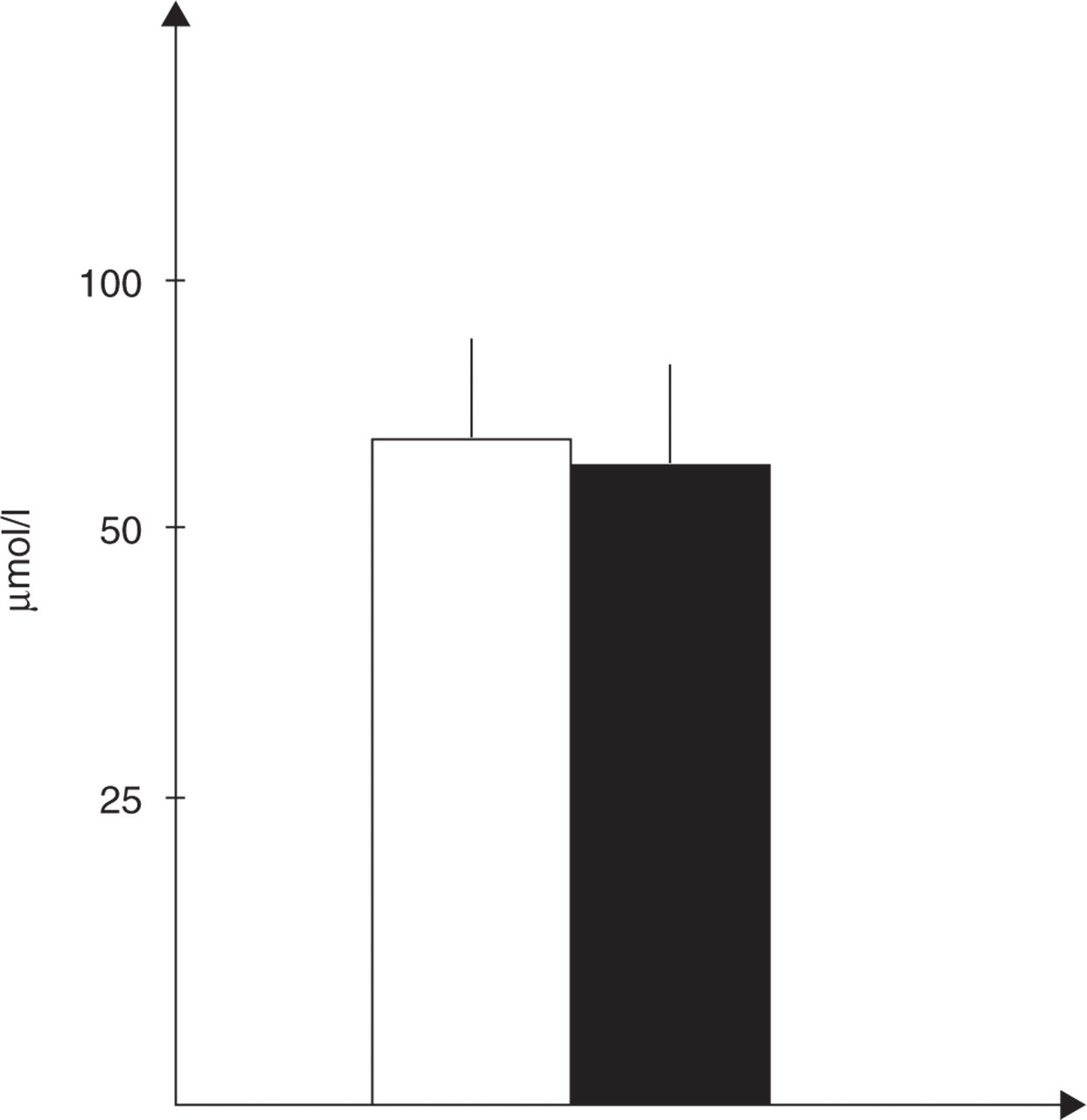
Mean ± SD of basal (interictal) plasma levels of L-citrulline (µmol/l) in 12 cluster headache patients (▪) and 12 control subjects (□). Patients vs. control subjects: NS.
No significant correlations were found between NO2 – or L-citrulline levels and the clinical features of patients, such as age and disease duration.
Discussion
Our findings further confirm the previously reported (15–17, 21) ability of GTN to trigger a spontaneous-like attack in a high percentage of CH patients in active period of disease. The reasons for the observed differences in the clinical response to GTN (which can be variably delayed or even absent) are still unclear, and it has been supposed that the type of response may be related to the individual characteristics of the patients investigated (15, 17). However, in both GTN-responsive and unresponsive CH patients, we found basal NO2 – concentrations similar to those of control subjects. Robust evidence obtained from different experimental models has suggested that L-arginine–NO pathways may be hyperactive in vascular headaches, both during and outside of attacks (23–27), although there are recent inconsistent observations (28). D’Amico et al. found significantly higher NO2 – concentrations in a wide population of CH patients (in either remission or cluster period) compared with controls (27). However, there was no measurement of NO2 – concentrations during spontaneous or induced attacks, the authors suggesting that a basal dysfunction in the L-arginine–NO pathway may be involved in the peripheral mechanisms predisposing subjects with neurovascular headaches to individual attacks. By contrast, in another study Martelletti et al. were unable to find increased NO2 – levels in serum and peripheral blood mononuclear cell supernatants from CH patients in the interictal period (26), an observation apparently consistent with our findings. It is difficult to account for these discrepancies, but it is known that even in spite of dietary restrictions or other precautions, NO2 – concentrations can be significantly affected by several variables, such as alcohol, cigarette smoking, atmospheric pollution and exercise (29). L-citrulline is a metabolite which represents a further specific index of activation of the biological pathway leading to NO production (18, 19). In any experimental paradigm, changes in L-citrulline values, when consistent with those of NO2 –, would strengthen the reliability of the findings with respect to NO metabolism. However, to our knowledge there is no previous report in literature on the levels of circulating L-citrulline in patients with neurovascular headaches. In the present study, as already seen for NO2 –, we were unable to find differences in plasma L-citrulline concentrations between controls and CH patients in the interictal period. Therefore, the concomitant observation of ‘normal’ basal levels of the two principal end-products of NO generation, NO2 – and L-citrulline, would suggest that at least in basal, headache-free conditions there is no increased activation of NO pathways in CH patients.
With particular regard to NO involvement in the attack, NO2 – concentrations in our patients were significantly increased at the time of maximum severity of pain following GTN administration, whereas they were no longer different from their basal values after 2 h. This observation is in line with a previous report in CH patients during spontaneous and NO donor-induced pain attacks (26). In our study, however, a similar trend was also shown by the control group, in which NO2 – concentrations were increased (in the absence of any pain or other symptoms) 45 min after GTN administration, i.e. when the totality of responsive CH patients were already experiencing their GTN-induced attack. It would therefore appear that NO2 – concentrations, which reflect the release and breakdown of native NO plus the amount of NO directly liberated by GTN, increase in a similar manner in normal subjects and CH patients, but are associated with genuine pain attacks only in the majority of the latter group. Indeed, a similar profile of NO2 – concentrations was shown by the six CH patients who did not experience a typical attack after GTN.
There are some limitations to be considered in this study. From a single sample obtained at pain peak it is impossible to infer whether NO2 – peak precedes or follows the onset of headache. In this respect, more frequent sampling would be helpful in further studies. In addition, peripheral blood may not be the best specimen in which to measure changes in NO production. The systemic compartment may be too large and small local changes may not be detectable, unless the investigation is focused, for example, on jugular blood. Furthermore, within a setting of GTN-induced attacks it is difficult to draw reliable conclusions on NO involvement in pain generation, as the provoking agent could mask spontaneous changes. To this purpose, NO changes should be studied also during genuine CH attacks. However, even with these limitations, our data suggest that other factors may sensitize predisposed individuals to the effects of NO (either exogenously loaded using donors, or generated during a spontaneous attack), ultimately causing pain. As in migraine, neurogenic inflammation and activation of the trigeminovascular system may be critical in CH pathophysiology (5, 6, 16), despite some conflicting evidence (30). Neurogenic inflammation is characterized by the release of several vasoactive agents from trigeminal perivascular nerve endings, leading to vasodilation, plasma protein extravasation, and sensitization of sensory afferents conveying nociceptive inputs to the brain stem (31). Increasing evidence indicates that NO may play a considerable role in these events, as it stimulates the release of the potent vasodilating agent CGRP from perivascular trigeminal fibres in cats (32, 33). Consistently, in humans CGRP levels are increased in the external jugular vein during NO donor-induced CH attacks (5, 16), as well as during genuine migraine attacks (34). NO is also present in nerve fibres surrounding cerebral blood vessels (35), induces protein extravasation from the vessels of the dura mater (36), and facilitates nociceptive transmission from the periphery to the CNS (37). Since the simple dilation of cerebral and extracerebral blood vessels does not appear to be a fundamental mechanism in GTN-induced pain in CH (38), GTN may trigger CH pain by stimulating trigeminal nociceptive fibres to release CGRP (16). Dural meningeal vessel dilation may later occur (32), according to a process probably requiring considerable time, if one looks at the usual latency of GTN effect (19–41 min in this study). In line with this view, inhibition of NOS significantly attenuates the activation of the trigemino-cervical complex of the cat (8) and antagonizes neurogenic and CGRP-induced dural vessel dilation (39). NO may also act within the brain stem nuclei of CNS, thereby contributing to the process of central sensitization and the development of pain and autonomic signs and symptoms. GTN-released NO can indeed activate several relevant areas in the CNS, such as the trigeminal nucleus caudalis (10, 40), and modulate the function of neurotransmitter pathways (41, 42). Interestingly, it has been suggested that an up-regulation of the expression of soluble guanylyl cyclase (the NO target enzyme) as well as cytokines in the dura mater may account for the subacute or delayed onset of GTN-induced symptoms (43, 44). Such phenomena may occur in CH, and may be one possible explanation for our finding that CH patients experienced spontaneous-like attacks in spite of a NO availability apparently similar to that of control subjects.
In conclusion, we have found in CH patients in the interictal phase that the plasma concentrations of the two main NO metabolites, NO2 – and L-citrulline, do not differ from those of headache-free subjects. This does not support the presence of a basal hyperactivity of the L-arginine–NO pathway in CH patients. NO2 – values are significantly elevated during the GTN-induced attacks in the patients, but in a fashion similar to those of control subjects at the corresponding time. Therefore, other factors, presently unclear, may render CH patients hyperresponsive to NO, and ultimately cause the occurrence of pain and associated features. Further studies are awaited to better understand NO involvement in CH and other vascular headaches.
Footnotes
Acknowledgements
The authors wish to thank Professor G. Bagetta (Rome) for his assistance with regard to the L-citrulline assay. This work was supported by a grant of the Italian Ministry of Health (Ricerca Corrente, IRCCS C. Mondino).
