Abstract
Sixteen patients, 12 with episodic and four with chronic cluster headache (CH) according to the International Headache Society criteria (1), participated in the study. They were randomly selected to start with one out of two different hyperbaric treatments in a double-blind, placebo-controlled, cross-over study design. Both gases were administered by mask inside a multiplace hyperbaric chamber for 70 min at 250 kPa (2.5 ATA) in two sessions 24 h apart. Active treatment was 100% oxygen (HBO treatment), while placebo treatment was 10% oxygen in nitrogen (hyperbaric normoxic placebo = sham treatment) corresponding to breathing air at sea level. All patients were decompressed on air. The patients documented the number of headache attacks and their degree of severity according to a modified VAS scale (level 0-4, where level 0 = no headache and level 4 = very severe headache). A headache index (HI = sum of (number of attacks times degree of severity)) was calculated for the run-in week prior to and the week after each separate treatment. A treatment was regarded as effective if it reduced the HI by > 50%. Blood samples were taken from the external jugular vein before and during hyperbaric treatment (after 30 and 70 min), 1 day and 1 week after each treatment for analyses of calcitonin gene-related peptide (CGRP), vasoactive intestinal peptide (VIP) and neuropeptide Y (NPY) and in a few patients also endotheline and nitrate. No difference between HBO and sham treatment on the HI or the prophylactic effect was observed in our study. However, 83% of the episodic CH patients and 25% of the chronic ones responded to either of the two treatments with at least 50% reduction of HI or remission for shorter or longer periods. This response rate exceeds an expected high placebo response due to the study procedure. Two episodic CH patients still experienced remission on follow-up 1 year after sham treatment. Five patients reported mild or moderate CH attacks during the sham treatment, and none during the HBO treatment. Changes in neuropeptides, endotheline and nitrate levels did not differ systematically when comparing the two different hyperbaric treatments or with respect to responders and non-responders. We conclude that two HBO sessions were not more effective than two sham treatments in reducing the HI and interrupting the CH period when given in a well-established cluster period or in chronic CH. The hyperbaric condition itself seems effective in reducing the HI, at least in patients with episodic CH, although a powerful placebo response can not be ruled out.
Introduction
Normobaric oxygen given to patients during a cluster headache (CH) attack is known to interrupt mild and moderate pain (2, 3). This is thought to be due to the vasoconstrictor effect of 100% oxygen. Drummond has shown that the pain diminishes and vanishes pari passu with the decrease in cerebral blood flow after inhalation of normobaric 100% oxygen (4, 5). Hyperbaric oxygen (HBO) treatment has been tried in therapy-resistant CH (6, 7) and shown to interrupt the headache attacks. Furthermore, HBO has been reported to interrupt the cluster period for at least 4–6 days (8), when given as an acute treatment. Most of these studies have, however, not been double-blinded. The placebo response reported in studies testing different treatments among CH patients is about 25–35% (9). The pain in CH is suggested to be generated by retroorbital vasodilatation (10, 11) and release of vasoactive peptides (12–14), lowering pain thresholds, the sequence probably being initiated from a hypothalamic pacemaker (15). It is also probable that HBO inhibits the pain due to its vasoconstrictive properties, but the mechanism for this is not known. One hypothesis is that HBO might influence the nitric oxide (NO) regulatory system (cf. (16)). NO is a potent vasodilatator and has been proposed to promote a focal neurogenic (sterile) inflammation (17, 18).
The aim of the present study was to investigate if HBO treatment was more effective in interrupting the period of active CH than hyperbaric normoxic placebo (sham) treatment, and if the HBO treatment would affect the blood levels of some neuropeptides, endotheline and/or the NO system.
Materials and methods
Sixteen patients 20–62 years of age with episodic (three females, nine males) or chronic (one female, three males) CH according to the International Headache Society (IHS) criteria (1) were asked to participate in the study and gave their fully informed consent, which was approved by the local Ethics Committee as well as the Medical Products Agency. They were consecutively selected from the patients attending the Outpatient Department of Neurology at Söder Hospital, Stockholm. For demographic data see Table 1. Inclusion required six headache attacks or more during the week prior to hyperbaric treatment and an expected remaining CH period of>4 weeks. Patients were excluded if they had used any CH prophylactic treatment during the present period, had heart or circulatory disorders, a present/past history of ear disorders or were pregnant or nursing. All patients had a physical examination by an otorhinolaryngologic specialist prior to the hyperbaric treatment.
Demographic data on cluster headache (CH) patients
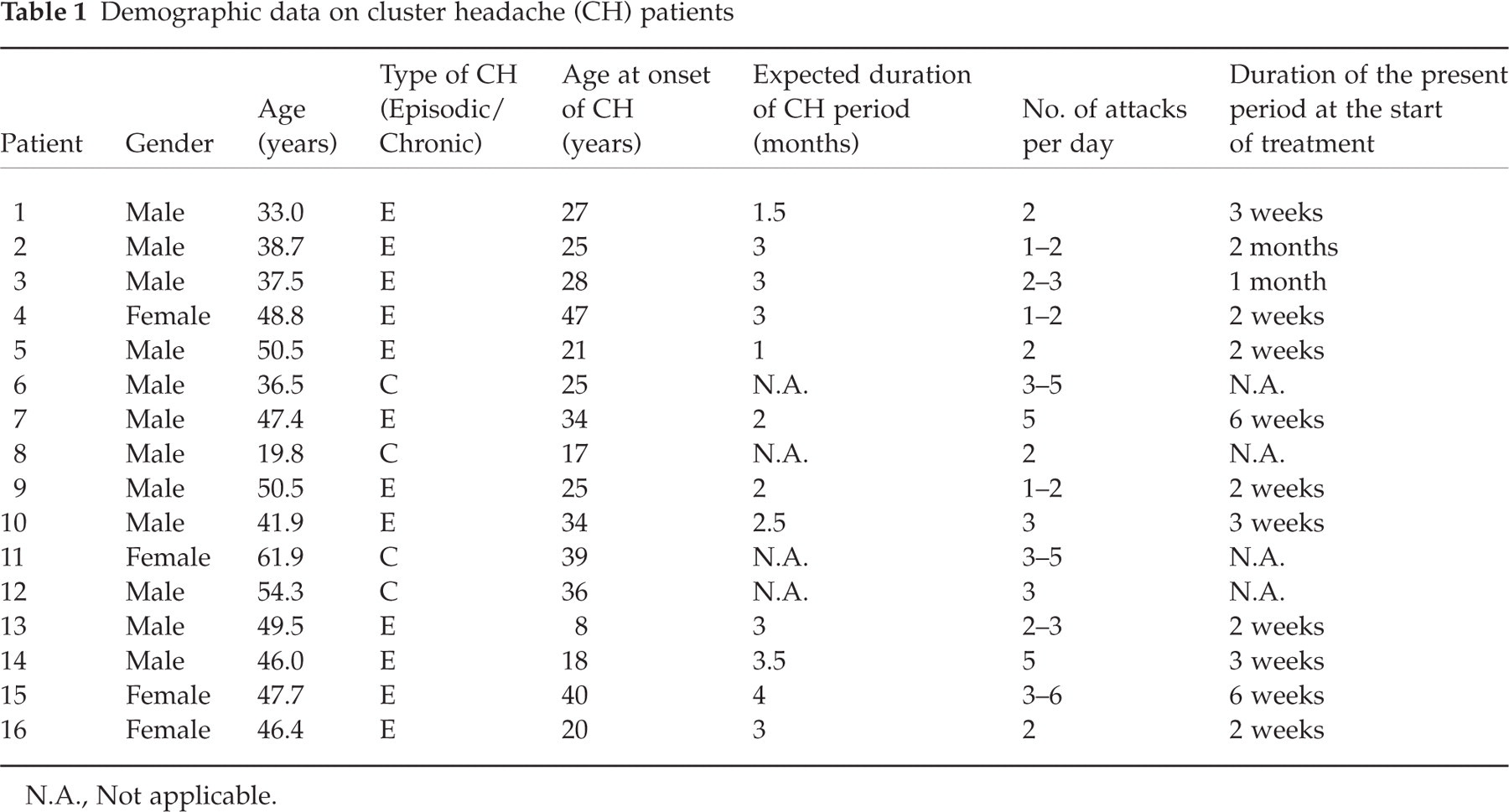
N.A., Not applicable.
The study used a double-blind, placebo-controlled, cross-over protocol (see Fig. 1). Breathing gas 1, active hyperbaric treatment (HBO), was composed of 100% oxygen. Breathing gas 2, hyperbaric normoxic placebo (sham) treatment, was composed of 10% oxygen in nitrogen, which at pressure gives a partial pressure of oxygen corresponding to breathing air at sea level. The gases were given from paired blinded gas cylinders, provided by AGA Gas AB. If the patients fulfilled the inclusion criteria, they were randomly given one of the two breathing gases by mask for 70 min at 250 kPa (2.5 ATA) inside a multiplace hyperbaric chamber in two sessions 24 h apart. The other breathing gas was given in identical sessions 1 week later unless the headache index was reduced by>50%, then the second cross-over treatment was postponed until the patient once again fulfilled the inclusion criteria. All patients were decompressed on air using ‘equivalent N2-pressure’ to calculate safe tables.

The study design. The patients were intended to be treated with both sham and hyperbaric oxygen (HBO) treatment. The treatment procedures were identical with the exception of gas composition. Both treatments included pressure.
All patients kept a headache diary, documenting the number of headache attacks, their duration and degree of severity, the latter according to a modified VAS-scale, where level 0 = no headache, 1 = slight headache, 2 = moderate headache, 3 = severe headache, and 4 = very severe headache. A headache index (HI) (= sum of (number of headache attacks times their degree of severity)) was calculated for 1 week prior to (run-in week) as well as for 1 week following each separate treatment. A treatment was considered effective if the HI decreased by>50%. The differences in HI were tested using the Wilcoxon's sign rank test.
The following escape drugs were allowed: paracetamol and/or codeine.
Patients were asked to have a catheter in their external jugular vein of the painful side during the first session of each treatment. Blood samples were drawn immediately before the first session of each treatment, 30 and 70 min into the hyperbaric treatment, immediately before the second session of each treatment, and 1 week after session 2 of each treatment.
All blood samples, except for the follow-up blood sample 1 week after each treatment, were collected in the hyperbaric chamber, immediately placed in ice, and kept in the hyperbaric chamber during the treatment session. The follow-up blood samples were immediately placed in ice. After the hyperbaric treatment or after the follow-up blood samples were taken, all blood samples were centrifuged at 4°C and 2000
The blood samples were analysed for CGRP, VIP and NPY using radioimmunoassay (RIA) techniques (provided by Dr R. Ekman, Department of Clinical Neuroscience, Section of Psychiatry and Neurochemistry, Göteborg University, Mölndal Hospital, S-431 80 Mölndal, Sweden). Blood samples from episodic CH patients, who were responders to HBO, to sham or were non-responders, were analysed for endotheline using RIA techniques (provided by Professor E. Theodorsson, Department of Clinical Chemistry, University Hospital, S-581 85 Linköping, Sweden), and plasma nitrate using capillary electrophoreses (provided by Dr P. Wiklund, Urology Research Laboratory, Karolinska Hospital, S-17176 Stockholm, Sweden).
Description of RIA techniques used for analyses of CGRP, VIP and NPY
General method
Radioactive ligands were synthesized using a modified chloramin-T method (19), cleaned by means of RP-HPLC using a µ-Bondapak C18 column (3.9 × 300 mm, 125 Å, 10 µm, Waters Code no. 27324), diluted 1:10 in 0.05
Calcitonin gene-related peptide
Samples of 200 µl were incubated with 200 µl CGRP antiserum (K-8429 T10 850213), final dilution 1:28 000 in assay buffer (0.25% bovine serum albumin (BSA), 500 KIE/ml trasylol in 0.05
Vasoactive intestinal peptide
Plasma (1000 µl) was extracted with 2000 µl 96% ethanol, incubated, centrifuged, evaporated and resolved in 2000 µl assay buffer (0.4% human serum albumin, 500 KIE/ml trasylol, 0.1% Triton X-100 in 0.05
Neuropeptide Y
Samples of 100 µl were incubated with 200 µl NPY antiserum (gift from Dr M.R. Brown, UCLA), final dilution 1:250 000 in assay buffer (0.25% BSA in 0.05
Description of RIA techniques used for analyses of endotheline
Sample-handling and extraction techniques were as described by Theodorsson-Norheim et al. (24). Euria-Endothelin-RB-304 RIA kit (Euro-Diagnostica AB, Medeon Malmö, Sweden) was used, which reacts ekvimolar amounts to endotheline 1, 2 and 3. The detection limit was 4 pmol/l (with 10 times concentration 0.4 pmol/l).
Description of the capillary electrophoreses used for NO analysis
All plastics, filters and vials were washed with deionized water and let to dry before use. Plasma samples were diluted in Milli-Q water, centrifuged (Ultrafree-MC filters) at 5000
Results
Effects on CH attacks
The headache indices during the week prior to and after sham treatment and prior to and after HBO for patients with episodic and chronic CH, respectively, are reported in Fig. 2. Sham treatment reduced the HI by ≥50% in six of 12 treated episodic CH patients and in none of four chronic CH patients, while HBO treatment was effective in four of 10 patients with episodic CH and in one of four chronic cases; five patients were non-responders. No patient responded to both treatments with a 50% reduction of the HI. Thus, 83% among episodic and 25% among chronic CH patients responded to either of the treatments with at least a 50% decrease of HI. Two patients received only the first treatment since it gave remission for more than a year. Both of them received the sham treatment. Three patients, two with episodic and one with chronic CH, who responded to the HBO given as the first treatment had also a partial response to sham treatment given as the second treatment. Three other patients with episodic CH did not respond to HBO given as the first treatment, but responded to the sham treatment given as the second treatment. One of them had an expected cluster period duration that should end during the follow-up week after the second treatment.

Clinical effect of hyperbaric treatment. The effect is expressed as headache index; HI = sum of (number of headache attacks times the degree of severity), the latter according to a modified VAS scale (0–4). The two top diagrams show the effect of sham (to the left) and hyperbaric oxygen (HBO) (to the right) treatments among episodic cluster headache patients. The two bottom diagrams show the effect of sham (to the left) and HBO (to the right) treatments among chronic cluster headache patients.
Five patients, four with episodic and one with chronic CH, reported CH attacks during treatment in the hyperbaric chamber, all of them during the sham treatment. All of these episodic CH patients classified the pain as mild (=1 on the modified VAS scale). The chronic CH patient classified the pain as moderate (=2 on the modified VAS scale). During the treatment sessions with HBO none of the patients suffered a CH attack.
The effect on HI was also evaluated with respect to treatment order. Seven of 16 (44%) CH patients responded with a 50% reduction of HI to the first treatment and four of 14 (29%) patients responded to the second treatment.
A statistically significant decrease in HI, using the Wilcoxon's sign rank test, at the 5% level was found after the sham treatment among all episodic CH patients. No statistically significant decrease in HI was observed after HBO treatment among the episodic CH patients. Neither did we find any statistically significant decrease in HI after the first or the second treatment irrespective of gas composition, after sham treatment or after HBO treatment when all patients were pooled.
Treatment was started 2 weeks to 2 months after the onset of the present cluster period. Patients who did not respond to the first treatment were given the second treatment after 1 week, i.e. within the expected remaining duration of the cluster period, with the exception of one patient, whose response to the second treatment may be due to spontaneous remission.
The long-term effect of hyperbaric treatment among the responders lasted from 1 week to>1 year, with a median duration of 1 month (Table 2). Three episodic CH patients who responded to HBO as their first treatment had at least reduced their HI by>50% for 3 weeks, while the patient with the longest remission period was free of CH attacks for 6.5 months. Three other episodic CH patients received sham as their first treatment and two of them became free of CH attacks for>1 year. Four other patients with episodic CH reported a reduction of HI of>50% after the second treatment session. Three of them had received sham treatment and one HBO. The latter had the longest remission period (3.5 months). The only patient with chronic CH that responded to treatment did so when receiving HBO. This treatment reduced the HI by>50% for 2 weeks.
Long-term effect of hyperbaric treatment among patients with episodic or chronic cluster headache

E, Episodic CH; C, chronic CH; HI, headache index (sum of (no. of headache attacks times their degree of severity)); reduced HI, a reduction in HI of <50%; N.A., not applicable.
Plasma levels of neuropeptides
The plasma concentrations of CGRP, VIP and NPY during the different treatments are presented in Table 3. No systematic changes in CGRP, VIP or NPY concentrations were observed during the HBO compared with the sham treatment, or when comparing the responders to the non-responders among patients with episodic CH. The only chronic CH patient who responded to HBO showed higher levels of VIP during HBO treatment than did the non-responding chronic CH patients. The CGRP plasma concentrations in the chronic cases were in the same range as found among the episodic CH patients. NPY levels showed no changes during the separate treatments and did not differ among patients with episodic compared with chronic CH.
Serum concentrations of different neuropeptides in the external jugular vein during normoxic and hyperbaric oxygen (HBO) treatment of patients with episodic or chronic cluster headache
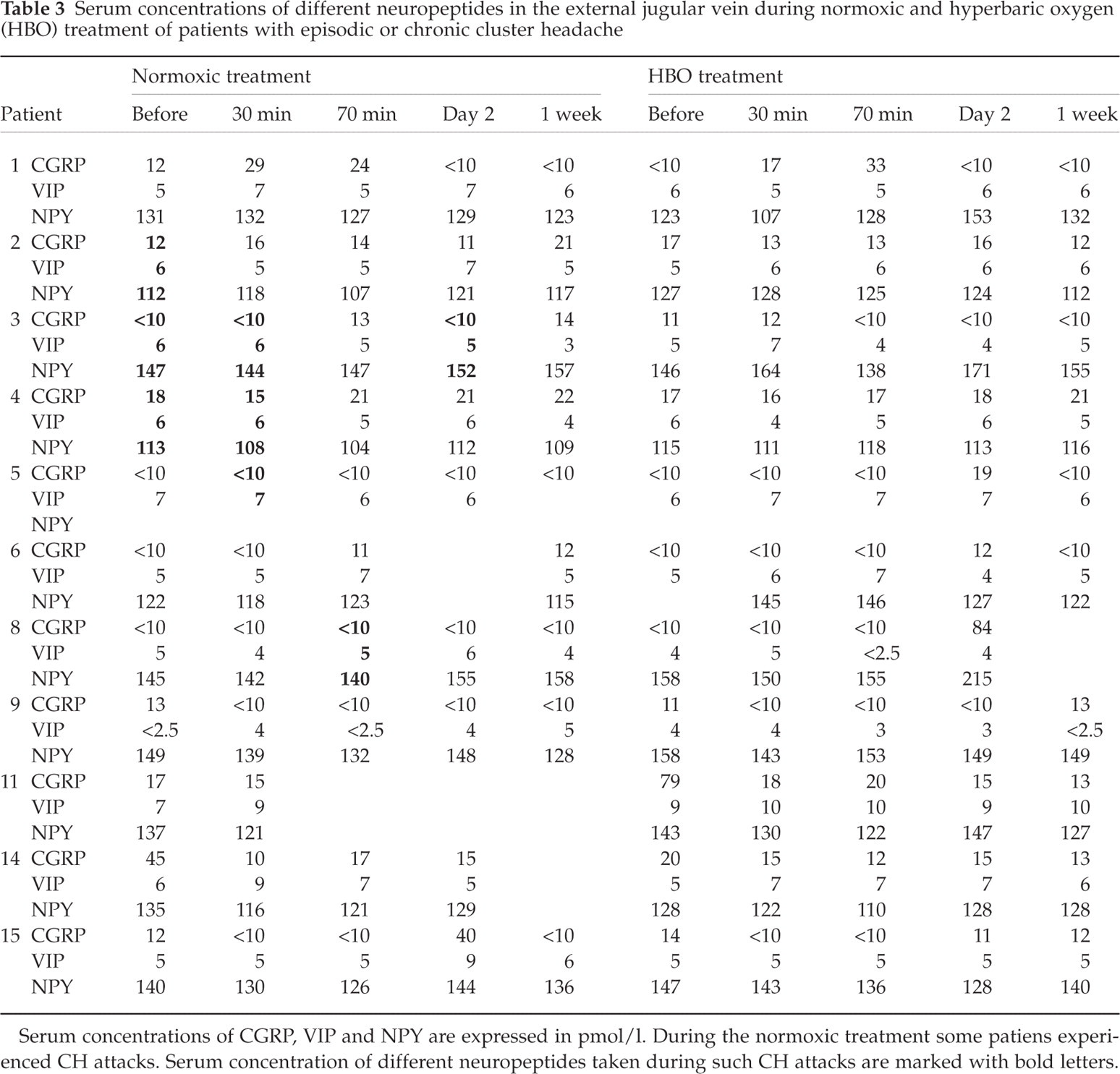
Serum concentrations of CGRP, VIP and NPY are expressed in pmol/l. During the normoxic treatment some patiens experienced CH attacks. Serum concentration of different neuropeptides taken during such CH attacks are marked with bold letters.
Six patients experienced CH attacks during the sham treatment. No consistent trend in the serum concentrations of CGRP, VIP and NPY was observed during these attacks compared with treatment sessions when no pain occurred and compared with basal pretreatment levels.
The changes in serum concentration of endotheline (no. 4) and nitrate (no. 6) for a few selected pilot patients are presented in Fig. 3. The median endotheline level during normobaric conditions was 2.4 pmol/l (range: from below the detection limit to 19) compared with below the detection limit during both sham (0–3) and HBO (all values below the detection limit) treatments. The median nitrate level observed among patients during normobaric conditions was 64.4 µ
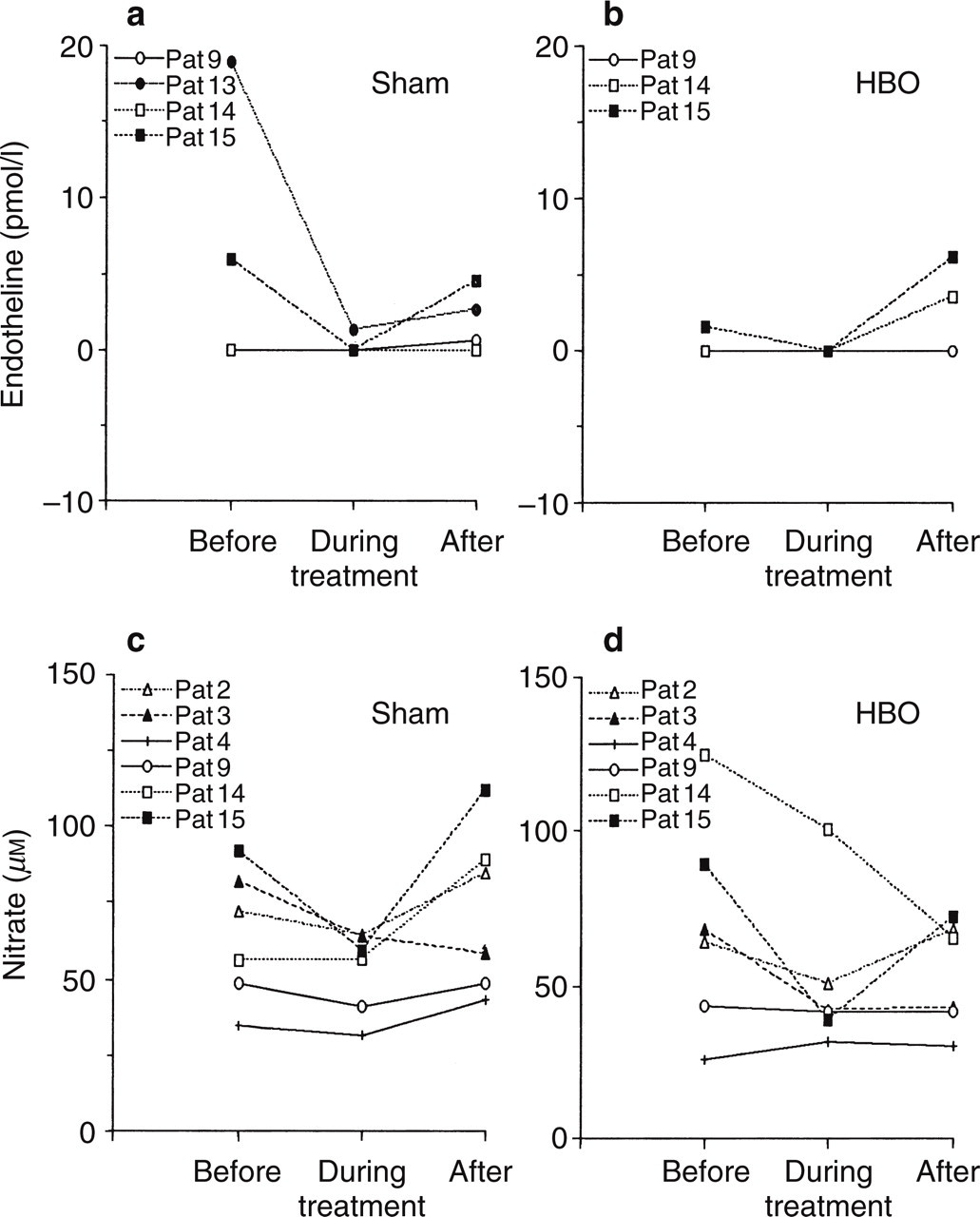
Plasma concentrations of endotheline (a and b) and nitrate (c and d) during hyperbaric treatment found among a few pilot patients with episodic cluster headache (CH), who were responders to hyperbaric oxygen (HBO), to sham treatment or who were non-responders. In (a) and (c) the diagrams show the concentrations immediately before, during (after 30 and 70 min of treatment) and after (after 1 day and 1 week) the sham treatment. In (b) and (d) the diagrams show the concentrations before (immediately before), during (after 30 and 70 min of treatment) and after (after 1 day and 1 week) HBO treatment.
Discussion
Our study is the first study with a double-blind, placebo-controlled and cross-over design investigating the effect of HBO on cluster headache. By using the strict study protocol (Fig. 1) we wanted to avoid both abortive cluster periods and spontaneous remissions, as well as to keep the placebo response under control. On the other hand, the selection of CH patients with long cluster periods may have influenced the outcome of this study. The code for the paired gas cylinders was not opened until all patients had received both treatments except two, who became free of symptoms for more than a year after the first treatment.
The main result of this study is that there was no significant difference between hyperbaric normoxic placebo (sham) and hyperbaric oxygen (HBO) treatment on the HI. Furthermore, there was no difference between the two hyperbaric treatments with respect to the prophylactic effect. Four of 10 episodic CH patients (40%) experienced at least a 50% reduction of HI after HBO treatment, including two who turned into a remission period. This is in line with Di Sabato and co-workers (8), who observed that three out of six patients, i.e. 50%, had an interruption of their cluster period after HBO treatment. They (8) also reported that their patients did not respond to the placebo treatment, while in our study sham treatment significantly reduced HI during the post-treatment week compared with the run-in week, while HBO did not. Besides, altogether 83% of our episodic CH patients experienced some reduction of the HI after hyperbaric treatment. In the description of the placebo Di Sabato and co-workers (8) wrote that ‘the patients … were placed in the same environment without administering HBO. For all patients (either those receiving the effective treatment or the placebo) exposure to the hyperbaric chamber lasted 30 min’. Whether this means that all patients were exposed to hyperbaric conditions or not is, however, obscure. Di Sabato and co-workers treated their episodic CH patients within 10–14 days after the start of the cluster periods. Our patients with episodic CH were treated 2 weeks up to 1.5 months after the start of the cluster period, i.e. our patients had suffered from their cluster period for a longer time before hyperbaric treatment. On the other hand, our patients received an almost five times longer total treatment time than the Italian patients (2 × 70 = 140 min vs. 30 min). Our treatment would then be expected to be more effective than that of di Sabato, but it was not. This may indicate that the time of initiation of the HBO treatment is of importance; the earlier the treatment is initiated the shorter the treatment session can be and/or the more effective it will be. Furthermore, our sham treatment also appeared to reduce the HI. If the placebo treatment in the study by di Sabato and co-workers did not include the exposure to hyperbaric conditions, then this could explain the different outcomes. This would then indicate a preventing effect by the pressure per se (cf. (31)). However, a true preventing effect of oxygen is suggested by the observation that four of our episodic patients experienced cluster attacks during the sham treatment, but none during the HBO one.
Di Sabato and co-workers (32) treated their chronic cluster headache patients in 15 sessions every second day for 4 weeks. All of their patients had a reduction of the number of headache attacks during the treatment period and for at least 2 follow-up weeks. Only one of our four chronic cluster headache patients, i.e. 25%, experienced reduction of the HI by>50% when treated with HBO. However, they received HBO treatment for 70 min in two sessions 24 h apart, which is a much shorter total treatment time. Repeated HBO sessions may be needed to stop the attacks among chronic CH patients (cf. (33)).
Six out of 16 patients responded to sham treatment while five out of 14 patients responded to HBO, which corresponds to 38% and 36%, respectively, for each separate treatment. Possibly a patient who did not experience a positive effect after the first treatment then would expect the second treatment to be effective and vice versa. In studies of sumatriptan treatment of CH patients using a double-blind study design a placebo response of about 25–35% has been reported (9). Ten out of 12 patients (83%) with episodic and one out of four (25%) patient with chronic CH had a>50% reduction of their HI during the week after hyperbaric treatment, irrespective of gas composition. This is well above the expected placebo level among episodic CH patients, which would thus indicate a true effect of the pressure per se or a more potent placebo response. This question cannot, however, be fully answered from the present study.
The neuropeptide levels in CH patients within period but without an ongoing attack were all in the normal range (13, 34). Even when the patients experienced an attack during sham treatment, their neuropeptide levels remained low. This is in contradiction to the results presented by Goadsby and Edvinsson (13), showing increased levels of CGRP and VIP during spontaneous CH attacks. In studies of nitroglycerine provoked CH attacks, the increase in CGRP and VIP was reported to occur during the peak of pain (13). Our patients reporting CH attacks during sham treatment had only mild or moderate pain, perhaps not severe enough to cause detectable increases in peptide levels. Furthermore, hyperbaric treatment might counteract the expected neuropeptide increase during attacks or abort them. The endotheline levels decreased below the detection limit during both sham and HBO treatment. Also the nitrate levels decreased during the sham treatment and even more during the HBO treatment. However, the analyses of neuropeptide concentrations and especially of endotheline and nitrate concentrations must be interpreted carefully because of the small number of samples. Furthermore, the study design may not have been able to catch rapid fluctuations. There is, however, the possibility that the hyperbaric treatments depress the vasoreactivity occurring during CH attacks, thus decreasing the expected outflow of vasoactive/neuroinflammatory substances, the HBO being more effective than the sham treatment (cf. (12)).
In conclusion, two HBO treatment sessions 24 h apart are not better than hyperbaric normoxic placebo treatment when given to CH patients who had a well-established cluster headache period or even a chronic illness. We cannot exclude the possibility that treatment given during the initial phase of the cluster period to episodic patients or a more intense (frequency and duration) HBO protocol would be more effective in blocking the cascade of pain-inducing substances. The present study indicates that the placebo response or the pressure per se may be effective in blocking CH periods or at least reducing the HI. Hyperbaric oxygen seems to have a preventing effect on CH attacks while the patient is under hyperbaric conditions. Future studies must focus on acute hyperbaric treatment among episodic CH patients and prolonged treatment sessions among chronic CH patients.
Footnotes
Acknowledgements
The authors wish to thank the patients who took part in this study and Dr K. Ekbom for initiating this study and for valuable discussions. This study was supported by grants from AGA AB Medical Research Fund and from Glaxo Wellcome. We wish to thank Dr R. Ekman (Department of Clinical Neuroscience, Section of Psychiatry and Neurochemistry, Göteborg University, Mölndal Hospital, S-431 80 Mölndal, Sweden), Professor E. Theodorsson (Department of Clinical Chemistry, University Hospital, S-581 85 Linköping, Sweden) and Dr P. Wiklund (Urology Research Laboratory, Karolinska Hospital, S-17176 Stockholm, Sweden) and their respective teams for analysing the blood samples and for supplying us with description of their techniques. We also wish to thank Dr H. Attorps and colleagues at the Department of Otorhinolaryngology, Söder Hospital, Stockholm, for ear inspection of our patients prior to hyperbaric treatment, nurse Ann-Charlotte Grönqvist for assisting with collecting the blood samples in the hyperbaric chamber and Professor J. Wahren (Department of Surgical Sciences, Section of Clinical Physiology, Karolinska Hospital, S-17176 Stockholm, Sweden) for letting us to use his laboratory facilities.
