Abstract
Introduction
Cluster headache (CH) is arguably the most painful primary headache disorder and belongs to the diagnostic group known as the trigeminal autonomic cephalalgias (TAC’s) (1–3).
Diagnostically, CH exists in two forms; an episodic (eCH) and a chronic variant (cCH), distinguished by the presence of a significant attack-free period in the former (1). The majority of patients (80–90%) have an episodic variant with averagely one or two cluster bouts a year separated by attack free remission periods. The remaining 10–20% suffers from a chronic variant of the disease with attack free periods not exceeding three consecutive months per year (1,4). From clinical experience and previous cross-sectional studies, we know that CH patients can change phenotype over the course of disease (i.e., from primary episodic [pECH] to secondary chronic [sCCH] or from primary chronic [pCCH] to secondary episodic [sECH]) (5,6). Rough estimates of transition-rate have been reported as broad as 2–19% for change into sCCH and 22–32% for change into sECH. Therefore, it still remains uncertain in how many patients this transition occurs (7). Additionally, prior studies have solely focused on identifying possible associated factors for transitioning into the sCCH variant (7,8), leaving possible factors for transition into the sECH variant unexplored. Lastly, clinical observations impel us to believe that a difference in disease severity and response to standard of care treatment can be observed between primary and secondary phenotypes. We addressed these uncovered fields of research by conducting an interview-based study, in which we asked CH patients about their course of disease.
Our aim was to describe the incidence of CH patients changing phenotype and elucidate possible factors associated with transition. Furthermore, we aimed to identify possible differences in clinical features between primary and secondary phenotypes. We hypothesized that approximately 20% of CH patients have changed phenotype since disease onset and that it is possible to identify potential associations for transition as well as a distinction in disease severity between the primary and secondary chronic phenotype.
Methods
We performed a cross-sectional cohort study of patients diagnosed with CH. Data derives from a genetics-study (H22010122), performed at the Danish Headache Center (DHC), a specialized tertiary referral center. Data collection was performed between 2017–2021. All patients filled out questionnaires and underwent a semi-structured interview.
Participants
Patients diagnosed with eCH and cCH were recruited from a register of present and former contacts at the DHC over a four-year period. Patients were included if they had a diagnosis of CH according to most recent current ICHD-criteria (ICHD-3-beta or ICDH-3), validated by a headache specialist, and were able to tell CH attacks apart from other types of headache. There were no criteria for exclusion. During the collection of data between 2017–2021 an update of the ICHD-criteria was introduced, so ICHD-3b-criteria (9) were used for inclusion and categorization of patients until 1 September 2018 and thereafter ICHD-3 was used (1). With the introduction of ICHD-3, diagnostic criteria for cCH were changed with regards to duration of remission period from one to three months consecutively. All patients with CH and affiliated to DHC during 2017–2021 were consecutively invited to participate, so no power-calculation for sample size was performed in advance.
Interview
Authors (NL, MB and RHJ) developed a semi-structured interview composed of questions, based on clinical experience and previous epidemiological questionnaires (3). The interview was assessed by an expert panel of five medical doctors specializing in CH and with research experience. They were asked to evaluate the questions: Are the instructions clear, is the interview coherent, are the questions relevant, is the interview logically divided into sections, and are the questions formulated clearly. The interview was validated by a randomly selected population of five CH patients representing different ages, gender and CH phenotype (eCH and cCH), and comments and suggestions were implemented accordingly.
The interview contained 52 questions in eight sections: headache diagnosis, headache burden, CH phenotype, abortive treatment, preventive treatment, transitional treatment, basic demographic information and comorbidities. Irrespective of phenotype, all patients underwent the same interview and estimated time of completion of the interview was 45 minutes. Interviews were performed by trained CH specialists, including physicians, study nurses or a specially trained medical student. Possible discrepancies between self-reported data and personal medical charts were audited by author MLKS and ASP.
Study endpoints
The primary outcome was defined as number of patients who had changed CH phenotype during their course of disease, that being from pECH to sCCH and pCCH to sECH according to present ICHD-3 criteria, respectively.
Being an exploratory study, the cohort was furthermore described by secondary outcomes, including identification of a broad array of possible associations for transition (sex, age at disease-onset, age at transition, time from onset of disease to time of diagnosis, time from onset of disease to time of transition, level of education, familial disposition to CH (parent, grandparent or siblings reported with a history of CH by the patient), shift of side of attacks (any reported side shift at any time: during an attack, between attacks or between bouts), other known primary headaches (tension-type headache, migraine with and without aura and other trigeminal autonomic cephalalgias) and comorbidities (18 predefined medical and surgical conditions). Secondary outcomes additionally included comparison of disease-severity between the primary and secondary groups regarding attack-frequency, attack-duration without treatment, number of yearly bouts, duration of bouts in weeks and best response to first line abortive therapy (oxygen or triptans), first preventive therapy (Verapamil) and transitional treatment (steroids administered as subcutaneous block of the greater occipital nerve (GON) or orally).
Statistical methods
Data were collected retrospectively. All data management and statistical analyses were performed with SAS® Statistical software, version 9.4 (SAS Institute Inc., Cary, NC, USA). Methods of statistical analysis were discussed with a public biostatistical consultant from the University of Copenhagen.
In order to assess potential indicators for transition, patients with pECH were compared with sCCH patients, and patients with pCCH were compared with sECH patients. Comparison of disease severity was performed by comparison of pECH with sECH and pCCH with sCCH respectively. Additionally, the cohort was split by sex and the same group-comparisons carried out as described above.
Quantitative variables in the study were either age- (e.g. mean age at disease onset), time (e.g. duration of attacks) or frequency-based (e.g. attack frequency per month). For continuous variables we used mean values and standard deviations or medians and interquartile range (IQR) as appropriate.
Group comparison of continuous variables were performed using unpaired t-test or Mann Whitney U test depending on the distribution of data. For categorical data, group comparisons were assessed using chi square test, Fishers exact test or Kruskal Wallis exact test. In group comparisons, a 2-sided p-value below 0.05 was considered statistically significant. Patients with missing data on the examined variables were omitted from analysis. No sensitivity analysis was performed.
Educational level was pooled into three major categories according to International Classification of Education (ISCED) (10); Group 1, Low education, correlating to ISCED level 0-2, Group 2, Medium education, correlating to ISCED level 3-4, and Group 3, High education, corresponding to ISCED-level 5+. Comorbidities were predefined as 18 medical and surgical conditions, based on priorly frequently reported comorbidities for CH and suggested predispositions for developing CH (3,4,6). We defined having a comorbidity as either having a medical history of or currently suffering from the condition. Regarding other simultaneous headaches, migraine with and without aura as well as TACs were pooled into one category each.
Reporting of all findings was conducted according to the Strengthening the Reporting of Observational studies in Epidemiology (STROBE) guidelines (11).
Patient consents and registration
All included patients gave their verbal and written informed consent according to the Helsinki Declaration. The study was approved by both the Capital Region of Denmark Ethical Committee (H22010122) as a part of a larger genetics study and by the Danish data protection agency
Data availability statement
The Danish Cluster Genetics Study contains sensitive information and cannot be shared in full form according to Danish data protection law. Deidentified data that underlie the results of this article can be shared on request.
Results
In total, 766 patients were invited to the study, of which 568 participated in the interview. Eight were unable to answer the full interview and 20 patients were not classifiable regarding phenotype, as they were included within one year after debut of their disease and were not in remission. A total of 540 patients were included in the final analysis, of these 358 (66.3%) were episodic and 182 (33.7%) were chronic patients (Figure 1). In total, 23 patients included before 2018 were classified as episodic according to the ICHD-3b-criteria but would have been chronic according to the present ICHD-3 criteria. All 23 patients would have had a formal change of phenotype due to criteria regarding remission-duration, while no patients would have had a formal change of phenotype due to the changes in criteria for autonomic symptoms. None of the 23 patients reported a transition of phenotype and were thus categorized into the pECH subgroup.
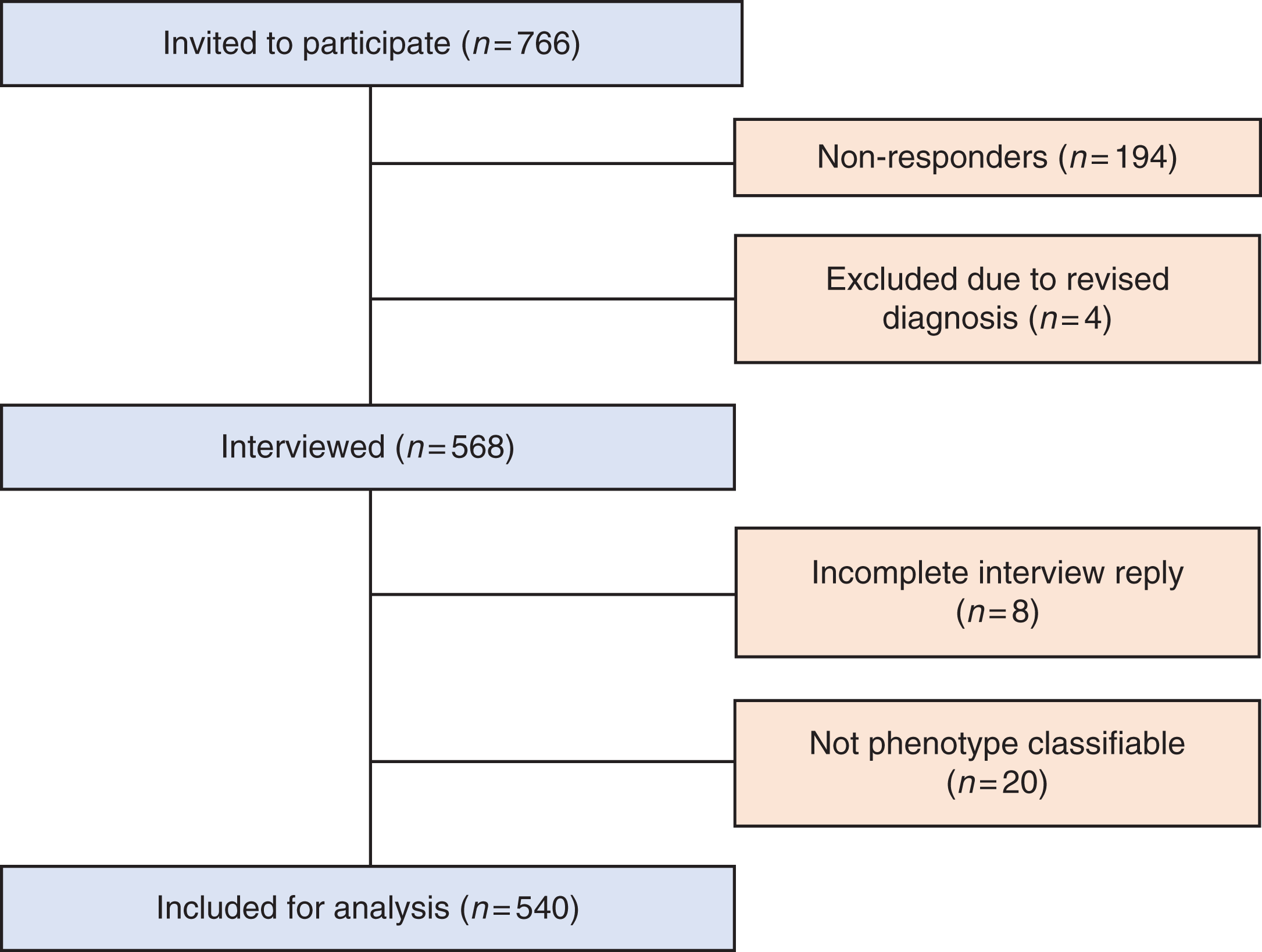
Flowchart of recruitment and inclusion of patients.
According to reported transition during course of disease, patients were categorized in four subgroups: Primary episodic (pECH), secondary chronic (sCCH), primary chronic (pCCH) and secondary episodic (sECH) (Table 1).
Clinical patient characteristics.
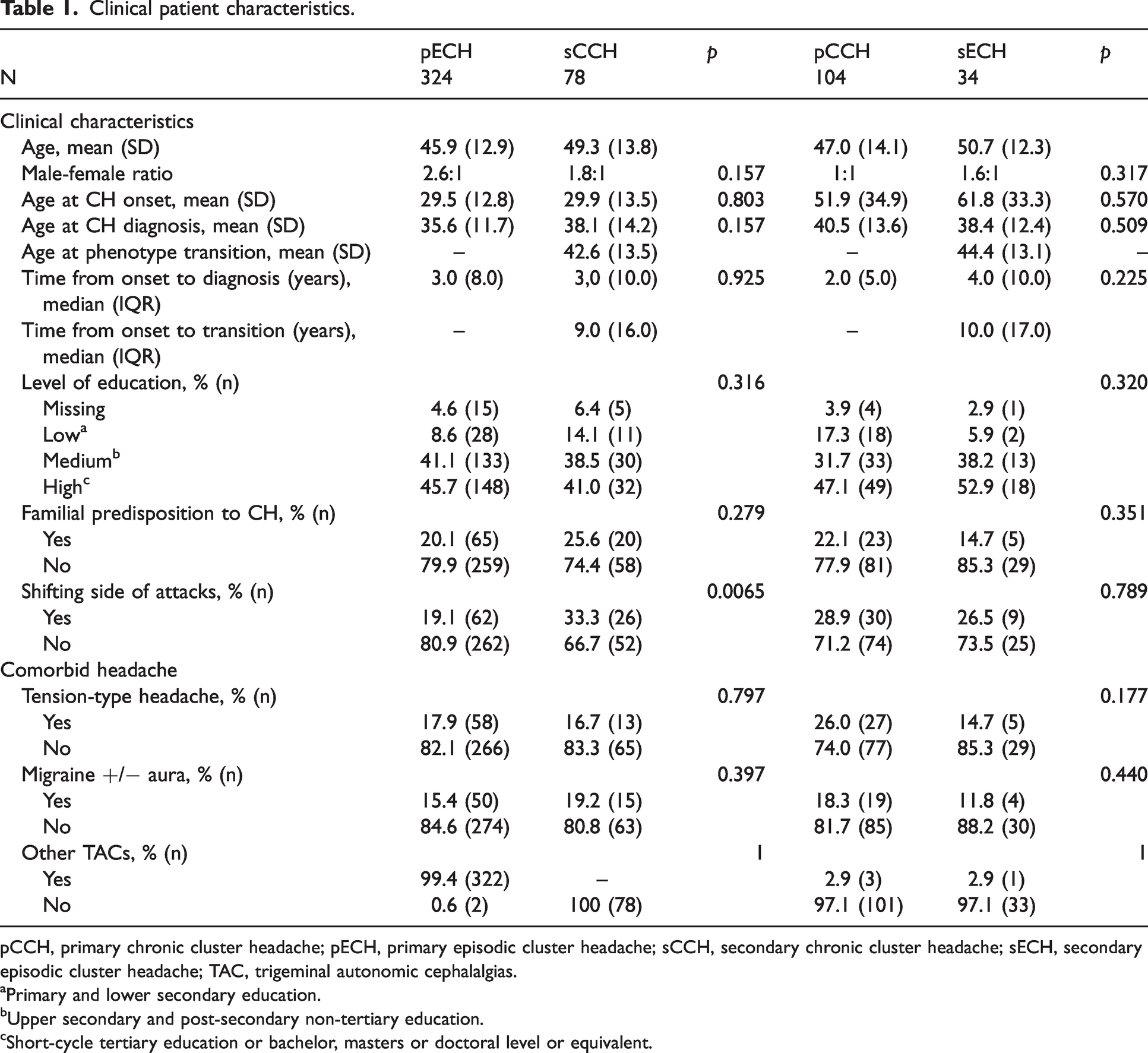
pCCH, primary chronic cluster headache; pECH, primary episodic cluster headache; sCCH, secondary chronic cluster headache; sECH, secondary episodic cluster headache; TAC, trigeminal autonomic cephalalgias.
aPrimary and lower secondary education.
bUpper secondary and post-secondary non-tertiary education.
cShort-cycle tertiary education or bachelor, masters or doctoral level or equivalent.
Change of phenotype
Transition to sECH was reported by 34 (6.3%) patients and to sCCH by 78 (14.4%) patients, constituting a total transition-incidence of 20.7%. The remainder of the population was distributed as 324 (60.0%) pECH and 104 (19.2%) pCCH. In the groups pECH, sCCH and sECH there was a male preponderance with a male-to-female ratio ranging between 1.6–2.6:1, whereas the pCCH group showed an equal distribution of 1:1.
Mean age at onset, mean age at diagnosis and median diagnostic delay were distributed evenly within comparison pairs (Table 1). The mean age at transition of phenotype was 44.3 years (SD: ± 13.1) for the sECH-patients and 42.6 years (SD: ± 13.5) for the sCCH-patients, while the median duration from onset to transition was 10.0 (IQR: 17.0) and 9.0 (IQR: 16.0) years respectively.
A total of 127 patients reported shifting side of attacks, of those 28 (5.2%) during an attack, 44 (8.1%) during a bout if episodic, 47 (8.7%) between bouts if episodic, and 45 (8.3%) between two attacks if chronic (online supplementary data, Table 1). Shifting side of attacks occurred most frequently in the sCCH group (33.3%), while only in 19.1% of the pECH patients, constituting a significant difference between the groups (p = 0.007). We found no differences in the distribution of educational level, comparing pECH with sCCH (p = 0.32) and pCCH with sECH (p = 0.32), as all groups displayed a high level of education (Table 1). Low education ranged between 5.8–17.3% for the four subgroups, most prevalent for the pCCH group. Additional demographic features were distributed equally (Table 1).
Coexisting headaches and comorbidities
For the two most prevalent coexisting headaches, tension-type headache and migraine with and without aura, there were no significant differences between comparison pairs (Table 1). The three most frequently reported comorbidities among the four groups were anxiety (20.0–29.5%), unspecified psychiatric diseases other than depression and anxiety (40.7–52.9%) and prior concussion (41.0–44.7%).
A complete overview of comorbidities is presented in supplementary data, Table 2.
Disease severity in primary versus secondary phenotypes.

pCCH, primary chronic cluster headache; pECH, primary episodic cluster headache; sCCH, secondary chronic cluster headache; sECH, secondary episodic cluster headache.
aMann-Whitney U test.
Disease severity
Looking at attack frequency, sCCH-patients had a significantly increased monthly attack-burden of 60 (IQR: 69.5) compared to 34 (IQR: 80.0) for the pCCH group (p = 0.049) (Table 2). The duration of each untreated attack was shorter for the sCCH group, 60 minutes (IQR: 80.0), compared to 80 minutes (IQR: 90.0) for the pCCH group (p = 0.041).
pECH-patients had a median of 1.0 bouts yearly (IQR: 1.2) lasting 6.0 weeks (IQR: 6.5), while sECH- patients had a median of 1.3 bouts yearly (IQR: 0.95) (p = 0.055) with a duration of 8.0 weeks (IQR: 9.0) (p = 0.10). Both the pECH and sECH group reported a median of two attacks (IQR: 2.0, p = 0.53) daily with an untreated duration of 90 minutes (IQR:16.5) and 75 minutes (IQR: 80.0) respectively (p = 0.73). The remission period in between bouts was significantly shorter, six months (IQR: 7.5), for the sECH group in comparison to 11 months (IQR: 16.5) for the pECH group (p = 0.009).
Treatment response
Regarding response to abortive treatment, we observed no significant differences between the primary and secondary chronic patients; equal for both groups, there were more responders than non-responders to abortive therapy, but only 39.4% pCCH and 37.1% sCCH patients obtained complete freedom of pain (p = 0.73).
Regarding preventive treatment, Verapamil was the most common prior or currently used therapy (pECH 59.6%, pCCH 83.7%, sECH 88.2%, sCCH 83.3%). The second and third most common preventive treatment was Lithium (pECH 3.1%, pCCH 27.9%, sECH 17.7%, sCCH 24.4%) and Gabapentin (pECH 4.0%, pCCH 26.9%, sECH 11.8%, sCCH 16.7%) respectively. For Verapamil, we observed no significant difference in treatment response between the pCCH and sCCH group for both frequency reduction (p = 0.48) and pain intensity reduction (p = 0.91). A substantial share of patients in both groups had no response at all to Verapamil with regards to frequency (pCCH 36.5%, sCCH 43.6%) and pain reduction (pCCH 49.0%, sCCH 47.4%). Of the gathered chronic patient-group 4.3% reported that, despite their chronic phenotype, they had attack-free periods of four weeks or more due to use of preventive medication. Most common response to transitional treatment was freedom of attacks for both chronic groups (pCCH 17.3%, sCCH 20.5%) (Table 3).
Treatment response.

Treatment response to abortive (oxygen, triptans), prophylactic (Verapamil) and transitional (steroids administered as subcutaneous block of the greater occipital nerve (GON) or orally) treatment.
pCCH, primary chronic cluster headache; pECH, primary episodic cluster headache; sCCH, secondary chronic cluster headache; sECH, secondary episodic cluster headache.
aKruskal Wallis Exact test.
In general, episodic patients showed a better treatment response than chronic patients. Episodic patients had an equal response to abortive treatment with the majority (pECH 54.9%, sECH 58.8%) obtaining complete pain-remission, irrespective of being secondary or primary. Treatment response for Verapamil did not differ between the pECH and sECH group, regarding both frequency response (p = 0.22) and pain response (p = 0.53). The most common reported response to Verapamil was no reduction of either frequency or pain for both the pECH and sECH group (Table 3). Transitional treatment most frequently induced freedom of attacks in both the pECH and sECH group, but with a higher prevalence (23.5%) in the sECH patients compared to the pECH patients (12.4%) (p = 0.69).
Sex-specific comparison
Regarding possible associations for transition, we only found significant differences between the subgroups in the male population; A significantly increased mean age at diagnosis in sCCH compared to pECH (sCCH 41.1 years, pECH 35.8 years, p = 0.01) and an increased prevalence of shifting side of attacks in sCCH compared to pECH (sCCH 36.0%, pECH 19.2%, p = 0.01).
Concerning disease severity, males displayed an increased bout duration for sECH compared to pECH (sECH 10 weeks, pECH 6 weeks, p = 0.02). In females, pCCH had a significantly better response to abortive treatment than sCCH (p = 0.01). In addition to this, no differences amongst the subgroups existed within each sex.
Discussion
Transition of phenotype in CH is common and occurred in 20.7% of patients included in this study, on average nine to ten years after onset. Transition-incidence was 6.3% for sECH, while transition to sCCH was more common and occurred in 14.4% of patients. Apart from side shifts no clear predictors for a transition of phenotype could be identified. Comparing disease severity in primary and secondary phenotypes, sCCH patients had significantly more frequent, but shorter-lasting attacks per month than pCCH, and sECH patients experienced shorter remission-periods than patients with pECH. Apart from this, disease-severity did not differ markedly between groups.
The transition-incidence was within the hypothesized range. Our findings hold both a possible overestimation as patients were recruited from a specialized tertiary center, and a possible underestimation due to study-design, where the entire lifetime course of disease for each patient is unknown. Supporting this theory, a previous cohort-study reported a cumulative proportion of patients developing sCCH as 9.3%, 18.1%, 24.5%, and 36% at 5, 10, 20, and 30 years respectively (8). Whether our findings of transition incidence are applicable to the global CH population is difficult to conclude, as reported differences in distribution of CH phenotype amongst different ethnic groups (12) could be due to selection bias and all prior studies of transition have been carried out in Western cohorts.
In our cohort, we found a considerably lower incidence of transition into sECH (6.3%) compared to existing studies, with estimates ranging between 22–32%. However, our findings of transition incidence into sCCH (14.4%) are in line with prior estimates ranging between 2–19% (5,6,8). Interestingly, we found that change of phenotype into sCCH was more common in our population, in contrast to the mentioned studies. A possible explanation for this discrepancy may be the implementation of current diagnostic ICHD-3 criteria, introduced after the other studies were performed. This change formally re-categorizes 6.4% of our cohort’s episodic patients according to ICHD3-b to chronic according to ICHD-3 (6). Additionally, sCCH patients could be thought to maintain their affiliation to DHC while sECH patients resolved to the primary sector, possibly generating a disproportionate distribution of transition.
In general, the four groups (pECH, sCCH, pCCH, sCCH) were very homogenous with few differences in the analyzed demographic parameters. Hence, we did not find any clear predictors for change of phenotype amongst general demographic traits, diagnostic delay, comorbidity with other primary headaches or other comorbidities. Neither could we confirm the previously proposed predictors for secondary chronification, namely high age at disease onset. Additionally suggested predictors have been high frequency of cluster periods, cluster duration exceeding eight weeks and remission periods less than six months (7,8), which we could not explore as our questionnaire was not sensitive to clinical characteristics for any previous phenotype. Shifting side of attacks was the only indicator for transitioning into sCCH. The association was reproduced when looking isolated at the male part of the cohort, but not for the female part, the latter most likely be attributed to the small sample size. No prior studies have investigated the relation between side shift and chronification and it remains uncertain whether side shift is an actual predisposition for transition or simply a frequent trait once the patient has become secondary chronified. To further characterize the distribution of side shifts in CH patients a prospective study is required.
Prevalence of coexisting tension-type headache in the cohort was similar to the background population of 30% (13). The highest prevalence of coexisting tension-type headache was observed in pCCH patients. Migraine with and without aura ranged between 11.7–19.2% in the four groups which slightly exceeds the prevalence of 13% in the background population (14). To further characterize the coexisting headaches as migraine and tension-type headache a prospective study with a diagnostic diary is needed. Our population displayed a much higher prevalence of anxiety (20.6–29.5%) or other unspecified psychiatric diseases (40.7–52.9%) compared to an estimated prevalence in the European background population of 16.6% (15). These findings are in line with prior estimates of comorbid depression in 32% of the CH population in the Danish Cluster Headache Survey (16). No prior studies or clinical experience lead us to believe that there is a causal pathway from psychiatric disease to developing CH, and the finding could rather be interpreted as a confirmation of the significantly increased personal burden in CH-patients (4,17,18).
Regarding education-level, we observed no trends in data for a future change into a secondary phenotype. Concordant with previous studies and supporting the theory of social drifting (19), the pCCH group had the highest prevalence of low education. Interestingly, the majority of patients within all four groups had a high level of education, opposing a prior reported reduced educational level in CH patients (17). A plausible explanation could be selection bias or the cross-sectional nature of the study, where a proportion of the patients have been interviewed before commencement or completion of an education.
Comparing disease severity, an important difference was more frequent, but shorter-lasting attacks for sCCH compared with pCCH, although we found no statistically significant differences in the effect of abortive treatment. We also found shorter periods of remission and a tendency towards longer bout duration for sECH compared to pECH patients, seemingly not explained by differences in the use of preventive medication. This could indicate that even though patients, per definition, change into an episodic phenotype, they remain more burdened than pECH patients. Lastly, an important observation is that fluctuation in disease activity and severity is a common phenomenon for the individual patient, even when remaining the same phenotype (3).
Treatment response was overall relatively poor and underlines the urgent need for more effective abortive and especially preventive treatment. Chronic patients showed reduced alleviation to both abortive treatment and Verapamil treatment compared to the episodic patients in line with prior observations (20–22). These findings confirm clinical experience on insufficient response to treatment among the chronic population and impel us to speculate whether decreasing treatment response could be an association for transitioning into a secondary chronic form.
Strengths and limitations
This study holds several strengths, being the largest cohort examined for transition to date and all patients being recruited from DHC with a validated CH diagnosis by a headache specialist. Additionally, all data were gathered through a semi-structured interview. Still, the study holds some limitations. First, data were collected at a tertiary specialized center, introducing selection-bias due to a possible overrepresentation of severely affected patients. Second, the interview design holds a possibility for recall bias and interviewer bias. However, we minimized this aspect by validation of phenotypes by a headache specialist. Furthermore, possible discrepancies between self-reported data and personal medical charts were audited by author MLKS and ASP. Third, the change of ICHD-criteria during the inclusion period may have affected the categorization of the study population, making data less comparable to previous studies within the field. Fourth, some of the examined subgroups are small in numbers, causing a risk of not exposing significant differences between groups in data-analysis, solely because of small sample-size. Lastly, the questionnaire was not sensitive to multiple changes in phenotype during course of disease, generating a possible underestimation of transition-frequency in individual subjects.
Conclusion
In conclusion, CH is a non-static disorder. In our study, a fifth of the population experienced a change of phenotype. Attack side shift may represent a predictor for transition from pECH to sCCH, but otherwise no indicators of increased transition risk were identified. Further, sCCH have more frequent but shorter attacks compared with pCCH, whereas sECH have shorter periods of remissions compared to pECH. Overall, it was noted that self-reported treatment response was low and possibly related to chronification. As uncertainty about prognosis is of concern to CH patients, disease course remains a desirable topic for future research.
Clinical implications
20% of our cohort of CH patients changed phenotype during course of disease, most often into secondary chronic phenotype. Attack side shift may represent a predictor for transition to chronic phenotype. Secondary chronic patients have more frequent, but shorter-lasting attacks compared with primary patients. Treatment response was poor in all groups and only one third had effective prevention.
Supplemental Material
sj-pdf-1-cep-10.1177_03331024221128287 - Supplemental material for Transition of cluster headache phenotype: An interview-based study
Supplemental material, sj-pdf-1-cep-10.1177_03331024221128287 for Transition of cluster headache phenotype: An interview-based study by Marie-Louise Kulas Søborg, Anja Sofie Petersen, Nunu Lund Malthe Faurschou Wandall-Holm, Rigmor Højland Jensen and Mads Barloese in Cephalalgia
Footnotes
Acknowledgements
The authors would like to thank all study participants and acknowledge the staff at DHC for their contribution to data collection, especially study-nurse Mette Bisgaard and Mette Fisker, medical student Adam Sebastian Pedersen and MD Andreas Hagedorn.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Marie-Louise Kulas Søborg is a current sub-investigator in trial for Lundbeck. Anja Sofie Petersen is a current sub-investigator in trial for Lundbeck and previous sub-investigator in trials for Eli-Lily and CCH pharmaceuticals. Nunu Lund and Mads Christian Johannes Barløse have no conflict of interest to report. Rigmor Højland Jensen has served as principal investigator in studies sponsored by Eli Lily, Novartis, Autonomic Technologies (ATI) and Lundbeck and has given lectures for Eli-Lily, ATI, Lundbeck, Novartis, Allergan, Pfizer, Berlin-Chemie, Norspan, Merck and TEVA. Malthe Faurschou Wandall-Holm has no conflict of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Research Foundation of Rigshospitalet.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
