Abstract
Taking into account the drawbacks associated with the use of triptans, attempts are being made to explore other avenues for the treatment of migraine. Recently, it has been shown that both α1- and α2-adrenoceptors mediate the constriction of porcine carotid arteriovenous anastomoses, which has effectively served as an experimental model predictive of anti-migraine activity. The present study investigated the effects of a novel α-adrenoceptor agonist S19014 {spiro[(1,3- diazacyclopent-1-ene)-5 : 2′-(4′,5′-dimethylindane)] fumarate} on carotid and systemic haemodynamics in anaesthetized pigs, and on human isolated coronary arteries. Increasing doses of S19014 (1-30 μg/kg, i.v.) produced a dosedependent initial short-lasting vasopressor response and a decrease of total carotid blood flow and conductance. The carotid blood flow and conductance changes were exclusively due to constriction of carotid arteriovenous anastomoses (capillary blood flow increased) and were accompanied by an increase in arterio-jugular venous oxygen saturation difference. Whereas prazosin (100 μg/kg, i.v.) was ineffective, rauwolscine (300 μg/kg, i.v.) attenuated the responses to S19014. The compound did not much affect the distribution of cardiac output to peripheral organs when compared with the vehicle group. Furthermore, S19014 only slightly contracted the human isolated coronary artery and its contractions, contrary to those of sumatriptan, were not increased in blood vessels precontracted with U46619. These results suggest that (i) the systemic and carotid vascular effects of S19014 are mainly mediated by α2-adrenoceptors, and (ii) S19014 could be effective in the treatment of migraine with an improved cardiovascular tolerance.
Keywords
Introduction
Vasodilatation of cranial large arteries and arteriovenous anastomoses has been proposed as playing an important role in the pathophysiology of migraine headache (1, 2). Indeed, to date, all acutely acting anti-migraine agents, i.e. the triptans and ergots, constrict isolated cranial vessels as well as arteriovenous anastomoses within the carotid vasculature (3, 4). While the vasoconstrictor effect of triptans seems to be mediated by the 5-HT1B receptor, that of ergot alkaloids also involves other receptors (5, 6), including the α-adrenoceptors, which mediate the carotid vasoconstriction in anaesthetized dogs (7).
Stimulation of α-adrenoceptors produces contraction of the isolated carotid artery of several species, including the dog (8, 9), rabbit (10, 11) and pig (12). Also, in vivo studies have shown that sympathetic nerve stimulation as well as administration of the α-adrenoceptor agonists, phenylephrine and BHT 933, constrict carotid arteriovenous anastomoses (13), and there is evidence that α-adrenoceptors may regulate vascular tone of carotid arteriovenous anastomoses (14). Thus, it is possible that α-adrenoceptors may provide a target for the development of new anti-migraine drugs (15).
Cordi and his co-workers (16) described a series of compounds with α-adrenoceptor agonist activity and one such compound, S18148 {(5S)-spiro[(1,3-diazacyclopent-1-ene)-5 : 2′-(7′-methyl-1′,2′,3′,4′-tetrahydronaphtalene)] fumarate} decreased carotid and cutaneous blood flows via activation of α2-adrenoceptors (17). The follow-up compound, S19014 {spiro[(1,3-diazacyclopent-1-ene)-5 : 2′-(4′,5′-dimethylindane)] fumarate}, which exhibited a high affinity at both α1- and α2-adrenoceptor subtypes (Table 1), interestingly showed a wide variation in efficacy (maximum effect: Emax) and potency (concentration needed to elicit 50% of Emax: EC50) in contracting rabbit, dog and human saphenous vein (EC50: 18, 79 and 8500 n
Chemical structure of S19014 and its binding affinities (pKi) at human cloned α1- and α2-adrenoceptor subtypes
Over the years, the constriction of porcine carotid arteriovenous anastomoses has served as a predictive model for the anti-migraine efficacy of triptans, which have an agonist action at the 5-HT1B receptor (2, 3, 19). In addition, ergot alkaloids, which act via both 5-HT1B receptor and α2-adrenoceptors (5, 7) and isometheptene, which acts via α-adrenoceptors (20, 21), constrict carotid arteriovenous anastomoses. Therefore, in the present study on anaesthetized pigs we set out to (i) investigate the effects of S19014 on the distribution of carotid blood flow into nutrient (capillary) and non-nutrient (arteriovenous anastomotic) fractions, and (ii) establish the cardiovascular safety of S19014 analysing its effects on cardiac output and its regional distribution to vital organs. The cardiovascular safety was also assessed in the human isolated coronary artery (22, 23).
Materials and methods
Anaesthetized pigs
General
After an overnight fast, 39 domestic pigs (Yorkshire × landrace; 10–14 kg) were sedated with intramuscular injections of azaperone (120 mg) and midazolam hydrochloride (10 mg) and then anaesthetized with sodium pentobarbital (600 mg, i.v.). After tracheal intubation, the animals were connected to a respirator (BEAR 2E, BeMeds AG, Baar, Switzerland) for intermittent positive pressure ventilation with a mixture of room air and oxygen. Respiratory rate, tidal volume and oxygen supply were adjusted to keep arterial blood gas values within physiological limits (pH, 7.35–7.48; pCO2, 35–48 mmHg; pO2, 100–120 mmHg). Anaesthesia was maintained with a continuous i.v. infusion of sodium pentobarbital (12–20 mg/kg/h). This anaesthetic regimen, together with bilateral vagosympathectomy (see below), increases heart rate and dilates carotid arteriovenous anastomoses. Thus, arteriovenous anastomotic blood flow is considerably higher (70–80% of carotid blood flow) in these animals compared with pigs in a conscious state or under fentanyl/thiopental anaesthesia (approximately 19% of carotid blood flow, 14), thereby producing one of the putative features of migraine (1, 2).
Heart rate was measured with a tachograph (CRW, Erasmus University, Rotterdam, the Netherlands) triggered by electrocardiogram signals. A catheter was placed in the inferior vena cava via the right femoral vein for the administration of vehicle and S19014. Another catheter was placed in the aortic arch via the left femoral artery for the measurement of arterial blood pressure (Combitrans Disposable Pressure Transducer; Braun, Melsungen, Germany) and arterial blood withdrawal for the measurement of blood gases (ABL-510; Radiometer, Copenhagen, Denmark). During the experiment, body temperature was kept around 37°C and the animal was continuously infused with physiological saline to compensate for fluid losses.
Carotid blood flow experiments
The common carotid arteries, external jugular veins and vagosympathetic trunks were identified. The vagosympathetic trunks were cut between two ligatures to avoid reflex-mediated cardiovascular changes. Subsequently, the right common carotid artery was dissected free and a hub-less needle, connected to a polyethylene tube, was inserted against the direction of blood flow for the administration and uniform mixing of radioactive microspheres. Another catheter was placed in the right external jugular vein for the withdrawal of venous blood samples to determine blood gases and, subsequently, the difference between arterial and jugular venous oxygen saturations (A-V SO2 difference). Blood flow was measured in the right common carotid artery with a flow probe (internal diameter 2.5 mm) connected to a sine-wave electromagnetic flow meter (Transflow 601-system, Skalar, Delft, the Netherlands) and continuously monitored on a polygraph (CRW, Erasmus University, Rotterdam, the Netherlands).
The distribution of common carotid blood flow was determined with 15.5 ± 0.1 µm (
Cardiac output experiments
A 6F Swan-Ganz thermodilution catheter (Braun Melsungen AG, Melsungen, Germany) was introduced into the pulmonary artery via the right femoral vein to measure cardiac output using a computerized cardiac output monitor (Erasmus MC, Rotterdam, the Netherlands). Another catheter, connected to a pressure transducer (Combitrans Disposable Pressure Transducer), was guided via the left carotid artery into the left ventricle for the injection of radioactive microspheres. Lastly, a catheter was placed into the right femoral artery and connected to a Harvard pump for the withdrawal of reference blood samples during the injection of radioactive microspheres.
The distribution of cardiac output was determined using radioactive microspheres (see above). For each measurement, about 1000 000 microspheres, labelled with one of the isotopes, were injected into the left ventricle against the direction of blood flow over a 10-s period. Starting 10 s before microsphere injection and lasting 70 s, an arterial reference blood sample was withdrawn from the right femoral artery at a constant rate of 6 ml/min. Blood loss during the experiment was compensated by infusing the corresponding volume of haemaccel. At the end of the experiments, the animals were killed as described above and a number of tissues (lungs, kidneys, heart, stomach, small intestine, spleen, liver, adrenals, brain, skin and skeletal muscles) were dissected out, weighed and put into vials for counting radioactivity. As described by Saxena et al. (24), tissue blood flow was calculated by multiplying the ratio of tissue and reference blood sample radioactivity by the blood withdrawal rate (6 ml/min) and normalized to 100 g tissue. Radioactivity in the lungs mainly represents the peripheral arteriovenous anastomotic blood flow (i.e. the non-nutrient part of the cardiac output), although a small amount (1–1.5% of cardiac output) is derived from the bronchial arteries (26). Vascular conductance was calculated by dividing blood flow (ml/min/100 g) or cardiac output (ml/min) by mean arterial blood pressure (mmHg), multiplied by one hundred. Stroke volume (cardiac output divided by heart rate) and nutrient cardiac output (cardiac output minus lung blood flow, i.e. mainly total arteriovenous shunting) were also calculated.
Experimental protocols
After a stabilization period of about 1 h, the animals were divided into two groups, used for either carotid blood flow (n = 27) or cardiac output (n = 12) experiments.
This first group (carotid blood flow experiments) was divided into four subgroups. Whereas the first and second subgroups (n = 6, each) remained untreated, the animals in the third (n = 8) and fourth subgroups (n = 7) received i.v. infusions (rate 0.5 ml/min for 10 min) of prazosin (100 µg/kg) or rauwolscine (300 µg/kg) to block α1- and α2-adrenoceptors, respectively (13). Fifteen minutes later, baseline values of blood pressure, heart rate and total carotid blood flow were collated and the distribution of carotid blood flow into arteriovenous anastomotic and capillary fractions, and A-V SO2 difference, were determined. Subsequently, the animals in the first subgroup received four consecutive i.v. infusions (1 ml/min for 3 min plus 2 min flush) of distilled water (vehicle), whereas those in the second (control), third (prazosin) and fourth (rauwolscine) subgroups received intravenous infusions of S19014 (cumulative doses 1, 3, 10 and 30 µg/kg) over a period of 3 min (1 ml/min) plus 2 min flush. Systemic and carotid haemodynamic variables were reassessed 10 min after each administration of vehicle (first subgroup) or S19014 dose (other three subgroups).
The second group (cardiac output experiments) was divided into two subgroups (n = 6, each). Whereas the first subgroup was treated with four consecutive infusions of 5 ml of distilled water (vehicle), the second subgroup received four cumulative doses of S19014 (1, 3, 10 and 30 µg/kg) at the rate of 1 ml/min for 3 min plus 2 min of flushing. Systemic and regional haemodynamic variables were reassessed 10 min after every administration of vehicle or S19014 dose.
Human isolated coronary artery
Tissue preparation
The right epicardial coronary artery was obtained from eight heart-beating organ donors who died of non-cardiac disorders less than 24 h before the tissue was taken to the laboratory (seven cerebrovascular accident, one hydrocephalus; four male, four female; age 24–57 years). The hearts were provided by the Rotterdam Heart Valve Bank after donor mediation by Bio Implant Services Foundation/Eurotransplant Foundation (Leiden, the Netherlands) after removal of the aortic and pulmonary valves for homograft valve transplantation. The hearts were stored at 0–4°C in a sterile organ protecting solution (UW, EuroCollins, or HTK-Bretschneider) immediately following circulatory arrest. After arrival in the laboratory, the right coronary artery was removed and placed in a cold, oxygenated Krebs buffer solution of the following composition: 118 m
Ring segments of approximately 3-4 mm length were prepared from the blood vessels, excluding macroscopically visible atherosclerotic lesions. The segments were suspended on stainless steel hooks in 15-ml organ baths containing Krebs buffer solution, aerated with 95% O2 and 5% CO2 and maintained at 37°C. After equilibration for at least 30 min and washing every 15 min, isometric tension was measured with a force transducer (Harvard, South Natick, MA, USA) and recorded on a flatbed recorder (Servogor 124, Goerz, Neudorf, Austria). The rings were stretched to a stable pretension of about 15 mN.
Experimental protocol
Segments were exposed to K+ (30 m
Statistical analysis and data presentation
The significance of the difference between the haemodynamic variables within one group was evaluated with Duncan's new multiple range test, once an analysis of variance (randomised block design) had revealed that the samples represented different populations (27). Per cent changes (from baseline values) caused by S19014 (1, 3, 10 and 30 µg/kg) in the animals treated with either prazosin or rauwolscine were compared with the corresponding doses in the control group using Student's unpaired t-test.
The concentration–response curves obtained with S19014 and sumatriptan in the coronary artery rings were analysed using the GraphPad software (GraphPad Software Inc., San Diego, CA, USA) to determine pEC50 values. In the case of a concentration–response curve not reaching a plateau, the contraction in response to the highest concentration was considered as Emax. Emax and pEC50 values of S19014 and sumatriptan were compared by paired t-test. Correlation coefficients were calculated according to Pearson (27). Experiments in the presence of U46619 were compared with paired experiments in the absence of U46619.
All data are presented as mean ±
Compounds
The following compounds were used: azaperone (Stresnil®; Janssen Pharmaceuticals, Beerse, Belgium), haemaccel® (Hoechst Marion Roussel b.v., Hoevelaken, the Netherlands), heparin sodium (to prevent blood clotting in catheters; Leo Pharmaceutical Products, Weesp, the Netherlands), midazolam hydrochloride (Dormicum®; Hoffmann La Roche b.v., Mijdrecht, the Netherlands), phenylephrine hydrochloride (Sigma-Aldrich Chemie b.v., Zwijndrecht, the Netherlands), prazosin hydrochloride (Bufa Chemie b.v., Castricum, the Netherlands), Prostaglandin F 2α tris salt (Sigma-Aldrich Chemie), rauwolscine dihydrochloride (RBI, Natick, MA, USA), S19014 (Institut de Recherches Internationales Servier, Courbevoie Cedex, France), sodium pentobarbital (Sanofi Sante b.v., Maasluis, the Netherlands), substance P acetate (Sigma-Aldrich Chemie), sumatriptan hemisuccinate (Institut de Recherches Internationales Servier), and U46619 (Sigma-Aldrich Chemie).
Except U46619, all drugs were dissolved in distilled water (vehicle); however, a short period of heating was needed to dissolve prazosin. U46619 was dissolved in ethanol and further diluted in distilled water. Solutions of S19014 and sumatriptan were freshly prepared for every experiment. The doses of the drugs refer to their respective salts.
Ethical approval
The local ethics committees dealing with the use of animals and humans in scientific experiments approved the protocol.
Results
Carotid blood flow distribution in anaesthetized pigs
Baseline values
Baseline values in the 27 anaesthetized pigs used for this protocol were: heart rate (104 ± 2 beats/min), mean arterial blood pressure (97 ± 2 mmHg), total carotid blood flow (138 ± 6 ml/min) and total carotid vascular conductance (144 ± 6 10−2 ml/min/mmHg).
Effects of antagonists
Table 2 shows the values of systemic and carotid haemodynamics as well as the A-V SO2 difference before and after pre-treatment with vehicle (control animals), prazosin (100 µg/kg) or rauwolscine (300 µg/kg). Whereas vehicle and prazosin did not produce any changes, rauwolscine elicited only a small, but significant, decrease in mean arterial blood pressure (9 ± 2%).
Absolute values of heart rate, mean arterial blood pressure and total carotid blood flow in anaesthetized pigs, before and after intravenous infusions of vehicle (control), prazosin or rauwolscine
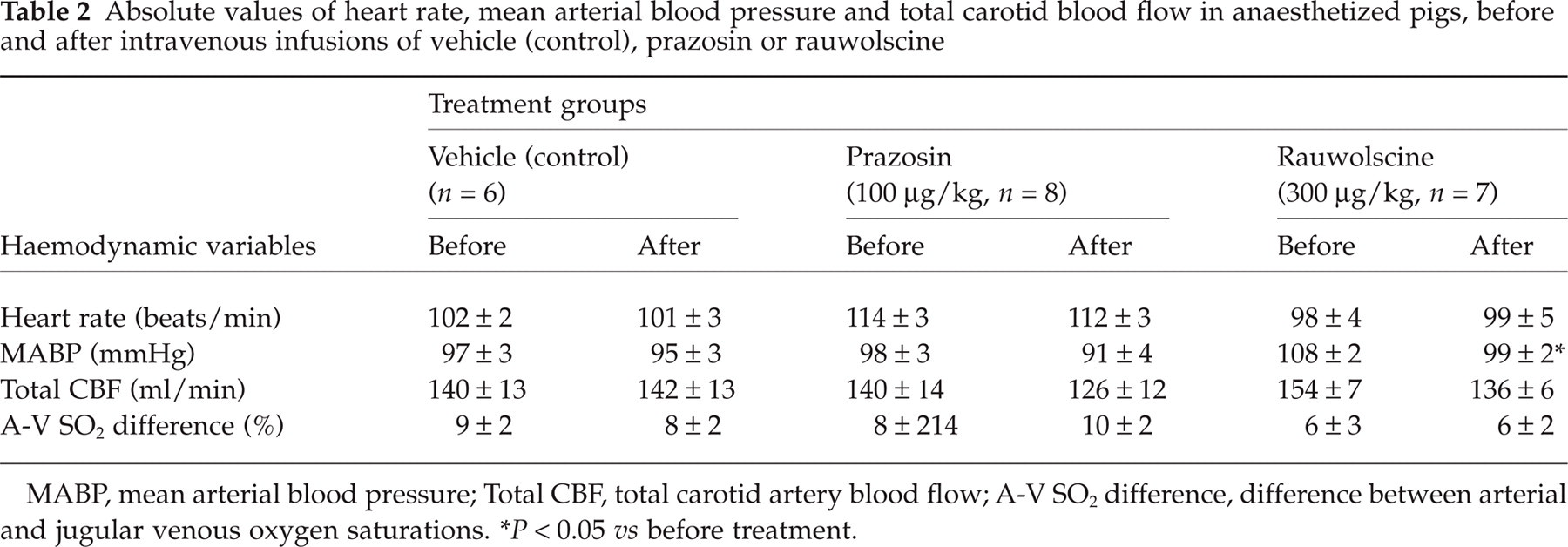
MABP, mean arterial blood pressure; Total CBF, total carotid artery blood flow; A-V SO2 difference, difference between arterial and jugular venous oxygen saturations.
∗P < 0.05 vs before treatment.
Systemic haemodynamics and A-V SO2 difference
In control animals, S19014 (1, 3, 10 and 30 µg/kg, i.v.) produced dose–dependent initial increases in mean arterial blood pressure that lasted for about 3 min (Fig. 1). Prazosin neither affected the magnitude nor the duration of these pressor responses. However, in rauwolscine-treated animals, mean arterial blood pressure increased only with the highest dose of S19014; the lowest two doses of S19014 caused a hypotension that lasted 10-15 min (Fig. 1).

Magnitude (as per cent change from baseline values; left panel) and duration (right panel) of the initial change in mean arterial blood pressure following administration of S19014 (1, 3, 10 and 30 µg/kg, i.v.) in control pigs (n = 6) and in pigs treated with prazosin (n = 8) or rauwolscine (n = 7). All values are expressed as mean ±
Table 3 presents the values of heart rate, mean arterial blood pressure and A-V SO2 differences measured before (baseline) and 15 min after vehicle (4 times 5 ml each) or S19014 (1, 3, 10 and 30 µg/kg). In both vehicle and S19014-treated animals, similar small decreases in heart rate (maximum change 4 ± 1 and 3 ± 1%, respectively) and mean arterial blood pressure (maximum change 7 ± 2 and 5 ± 2%, respectively) were noticed. These small changes were probably time related. However, S19014 caused a significant, but moderate, increase in the A-V SO2 difference after the two highest doses of S19014 and this effect was attenuated by rauwolscine but not prazosin (Table 3).
Systemic haemodynamic effects of vehicle (n = 6) per se and S19014 in the absence (n = 6; control) or presence of either prazosin(100 µg/kg; n = 8) or rauwolscine (300 µg/kg; n = 7)

A-V SO2 difference, difference between arterial and jugular venous oxygen saturations.
P < 0.05 vs baseline.
P < 0.05 vs the corresponding change with S19014 in control animals.
Four infusions of 5 ml distilled water were given after baseline measurements.
Carotid haemodynamic effects of S19014
Absolute values of total carotid, arteriovenous anastomotic and capillary blood flows and conductances in the different groups of animals are shown in Fig. 2. Whereas vehicle was devoid of any carotid haemodynamic effects, S19014 (1, 3, 10 and 30 µg/kg) produced dose–dependent decreases in total carotid and arteriovenous anastomotic blood flows (maximum change 18 ± 2 and 34 ± 5%, respectively) and conductances (maximum change 14 ± 2 and 30 ± 4%, respectively). In contrast, S19014 increased capillary blood flow and conductance (maximum change 40 ± 23 and 49 ± 27%, respectively). These effects of S19014 remained largely unchanged in animals treated with prazosin, but were clearly attenuated by rauwolscine.
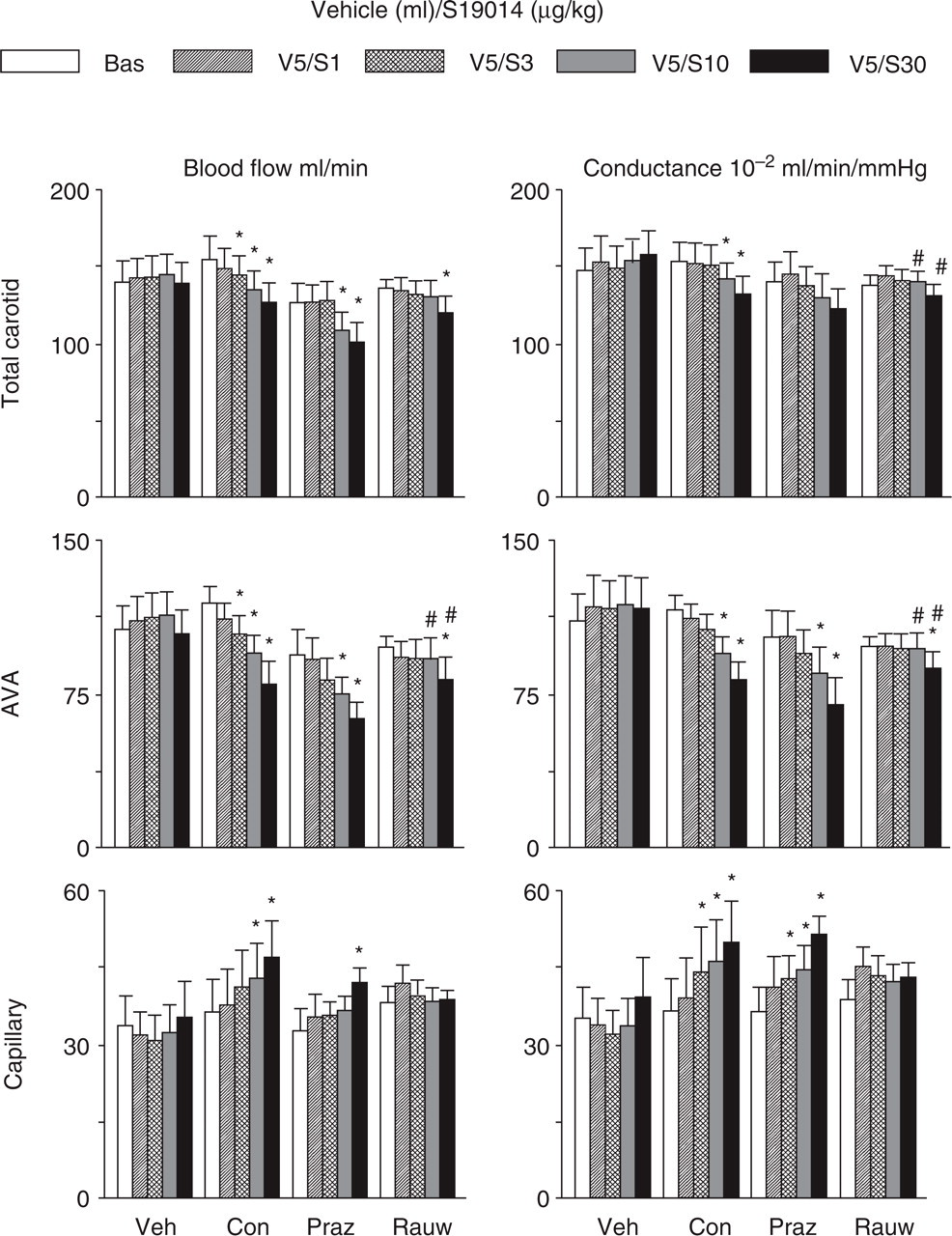
Total carotid, arteriovenous anastomotic (AVA) and capillary blood flows (left panels) and vascular conductances (right panels) measured in groups of pigs receiving either vehicle (Veh; n = 6) or S19014; the latter were either untreated controls (Con; n = 6) or treated with prazosin (Praz; 100 µg/kg; n = 8) or rauwolscine (Rauw; 300 µg/kg; n = 7). The measurements were made sequentially at baseline (Bas) and 15 min following infusions of vehicle (four times each 5 ml, i.v., V5) or S19014 (1, 3, 10 and 30 µg/kg, i.v. as indicated by S1, S3, S10 and S30, respectively). All values are expressed as mean ±
Figure 3 depicts the per cent changes (from baseline values) in carotid arteriovenous anastomotic conductance by S19014 (1, 3, 10 and 30 µg/kg) in control, and prazosin- (100 µg/kg) or rauwolscine- (300 µg/kg) treated animals. While prazosin did not modify the constrictor effect of S19014 on arteriovenous anastomoses, rauwolscine clearly did.

Per cent changes (compared with baseline value) in porcine arteriovenous anastomotic (AVA) conductance 15 min following administration of S19014 (1, 3, 10 and 30 µg/kg, i.v.) in control pigs (n = 6) and in pigs treated with prazosin (n = 8) or rauwolscine (n = 7). All values are expressed as mean ±
The changes caused by vehicle (four doses) and S19014 (1, 3, 10 and 30 µg/kg) in vascular conductance in the different cranial tissues are depicted in Fig. 4. Whereas administration of vehicle did not elicit any changes, S19014 moderately increased the vascular conductance in several tissues, including the skeletal muscle, bone, fat, salivary gland and dura mater, while those in the others (skin, eye, brain, ear or tongue) remained unchanged. These effects of S19014 on tissue vascular conductance were similar in animals treated with prazosin, but relatively less marked in animals treated with rauwolscine, suggesting that the vasodilator effects of S19014 may be partly passive following constriction of arteriovenous anastomoses.

Carotid regional vascular conductances measured in groups of pigs receiving either vehicle (Veh; n = 6) or S19014; the latter were either untreated controls (Con; n = 6) or treated with prazosin (Praz; 100 µg/kg; n = 8) or rauwolscine (Rauw; 300 µg/kg; n = 7). The measurements were made sequentially at baseline (Bas) and 15 min following infusions of vehicle (four times each 5 ml, i.v., V5) or S19014 (1, 3, 10 and 30 µg/kg, i.v. as indicated by S1, S3, S10 and S30, respectively). All values are expressed as mean ±
Distribution of cardiac output
Baseline values
Baseline values in the 12 anaesthetized pigs used for this protocol before any treatment were: heart rate (112 ± 3 beats/min), mean arterial blood pressure (105 ± 1 mmHg), cardiac output (1588 ± 47 ml/min), nutrient cardiac output (1354 ± 49 ml/min), stroke volume (14.3 ± 0.5 ml) and systemic vascular conductance (1511 ± 44 10−2 ml/min/mmHg).
Systemic haemodynamics
The values of systemic haemodynamic variables in anaesthetized pigs collated at baseline and after i.v. treatments with vehicle or S19014 (1, 3, 10 and 30 µg/kg) are presented in Fig. 5. Mean arterial blood pressure did not change, but there were small decreases in other variables in both vehicle- and S19014-treated groups. The maximum changes, which did not significantly differ in the two groups were, respectively, heart rate, 4 ± 1 and 4 ± 2%; cardiac output, 14 ± 3 and 14 ± 2%; stroke volume, 10 ± 3 and 10 ± 3%; and systemic vascular conductance, 10 ± 2 and 11 ± 3%. A small decrease in the nutrient cardiac output (maximal change 14 ± 3%) was also observed in the vehicle subgroup.

Systemic haemodynamic values of heart rate (HR), mean arterial pressure (MAP), cardiac output (CO), nutrient cardiac output (NCO), stroke volume (SV) and systemic vascular conductance (SVC) measured in groups of pigs receiving either vehicle (n = 6) or S19014 (n = 6). The measurements were made sequentially at baseline (Bas) and 15 min following infusions of vehicle (four times each 5 ml, i.v., V5) or S19014 (1, 3, 10 and 30 µg/kg, i.v. as indicated by S1, S3, S10 and S30, respectively). All values are expressed as mean ±
Regional haemodynamics
As depicted in Fig. 6, there were no significant changes in regional vascular conductances in the vehicle group other than a decrease in vascular conductance in the adrenals (maximal change −30 ± 11%), and increases in the brain with the highest dose (25 ± 9%) and skin with the last two doses (maximal change 54 ± 16%); this latter effect was also observed in the animals receiving S19014. In addition, S19014 decreased vascular conductances in lungs (mainly peripheral arteriovenous anastomoses; maximal change 50 ± 5%), skeletal muscles (maximal change − 16 ± 4%) and liver (maximal change 50 ± 5%). Except in the lungs, the changes with S19014 were not significantly different from those in the vehicle group.

Regional vascular conductances measured in groups of pigs receiving either vehicle (n = 6) or S19014 (n = 6). The measurements were made sequentially at baseline (Bas) and 15 min following infusions of vehicle (four times each 5 ml, i.v., V5) or S19014 (1, 3, 10 and 30 µg/kg, i.v. as indicated by S1, S3, S10 and S30, respectively). All values are expressed as mean ±
Human isolated coronary artery
Basic contractile properties
The coronary artery contraction to 100 m
Contractile responses to S19014 and sumatriptan
Both S19014 and sumatriptan induced a concentration-dependent contraction of the human isolated coronary artery (Fig. 7, left panel). Whereas the maximal contraction to S19014 was considerably smaller than that to sumatriptan (3 ± 1 and 19 ± 10% of the contraction to K+, respectively), the difference was not statistically significant (P = 0.13). This lack of significance may be explained by the large variability of contraction to sumatriptan (Emax: 3–77% of contraction to K+), which is in accordance with our previous findings (28). The pEC50 values for S19014 (5.55 ± 0.24) and sumatriptan (5.99 ± 0.14, respectively) were similar. The Emax of S19014 and sumatriptan did not correlate to the endothelial quality of the blood vessel segments as assessed with the relaxation to 1 n

Human isolated coronary artery contractions to sumatriptan (squares) and S19014 (circles), expressed as percentage of the response to 100 m
As depicted in Fig. 7 (right panel), the contractions to S19014 were not different in the presence of U46619 (Emax: 4 ± 1% of the contraction to K+: pEC50 6.55 ± 0.77) compared to that in quiescent blood vessel segments (Emax: 3 ± 1%, pEC50: 5.34 ± 0.24). In contrast, the Emax of sumatriptan was significantly augmented in the presence of U46619 (76 ± 22 vs 33 ± 1% of the contraction to K+, P < 0.05), while the pEC50 remained unaffected (6.77 ± 0.49 vs 6.14 ± 0.23). In the presence of U46619, the maximal contraction to S19014 was significantly lower than that to sumatriptan (P < 0.05).
Discussion
It is generally agreed that both α1- and α2-adrenoceptors play an important role in the regulation of the vascular resistance and blood pressure (29–33). Recently, we showed that both these receptors mediate canine (external) and porcine (arteriovenous anastomotic) carotid vasoconstriction (13, 34, 35). In this context, several lines of evidence demonstrate that vasoconstriction in the carotid vascular bed is predictive for anti-migraine activity (2, 3). Therefore, the present study was designed to assess the anti-migraine potential of the novel α-adrenoceptor agonist S19104 in anaesthetized pigs and human isolated coronary artery, as previously described (22, 36, 37).
Systemic haemodynamics
Other than a moderate initial pressor effect, intravenous administrations of S19014 did not cause significant changes in systemic haemodynamics. This initial pressor effect of S19014 was short-lasting (approximately 3 min) and, being amenable to blockade by rauwolscine but not prazosin, involves α2-adrenoceptors (see Fig. 1). Although pressor responses can be elicited via both α1- and α2-adrenoceptor subtypes (33), the involvement of α2-adrenoceptors in the action of S19014 is in accordance with its higher affinity at α2-adrenoceptor (pKi: 8.33–8.98) than at α1-adrenoceptors (pKi: 7.65–7.80) subtypes (Table 1). Further, it may be pointed out that, in our previous experiments (13), no pressor changes were observed with either phenylephrine (α1-adrenoceptor agonist) or BHT933 (α2-adrenoceptor agonist) in anaesthetized pigs. One of the reasons for this apparent discrepancy may be that, in these experiments, phenylephrine and BHT933 were slowly infused into the carotid artery (13), while we injected S19014 i.v.
Carotid haemodynamics
As reported previously (13), i.v. administration of vehicle, prazosin (100 µg/kg) and rauwolscine (300 µg/kg) did not produce major carotid haemodynamic changes (Table 2). In contrast, S19014 produced a dose-dependent constriction within the porcine carotid vasculature, which was confined to arteriovenous anastomoses; the vascular conductance in the capillary fraction was increased. In accordance with the constriction of carotid arteriovenous anastomoses (1, 2), S19014 increased A-V SO2 difference. Both effects of S19014 were antagonized by rauwolscine and not at all by prazosin, thus establishing the involvement of one or more α2-adrenoceptor subtypes. As mentioned above, the involvement of α2-adrenoceptors in the action of S19014 is in accordance with its higher affinity at α2-adrenoceptor (pKi: 8.33–8.98) than at α1-adrenoceptors (pKi: 7.65–7.80) subtypes (Table 1) and it is possible that S19014 lacks efficacy, being a partial agonist or antagonist action at α-adrenoceptors. Interestingly, it has been reported that the venoconstrictor responses to S19014 are variable in potency and efficacy (18), suggesting that the tissue distribution of α-adrenoceptor (most likely α2-adrenoceptor) subtypes at which S19014 is efficacious may be uneven.
Cardiac output and regional haemodynamics
Acutely acting anti-migraine drugs (triptans and ergot alkaloids) have been shown to decrease cardiac output and systemic vascular conductance in anaesthetized pigs (38). In the present study with S19014, no changes in cardiac output, systemic vascular conductance and vascular conductances in many body organs, including the heart, kidneys, intestines, stomach, adrenals, were observed when compared with the vehicle subgroup. Admittedly, S19014 produced a decrease in the vascular conductance of the liver, but it is difficult to predict whether this decrease in the hepatic blood flow would be important in the clinical setting. Interestingly, a transient decrease in hepatic blood flow has also been observed in humans after i.v. ergotamine, unlike its prolonged constrictor property on large arteries (39).
As found in the carotid artery experiments, S19014 decreased the ‘lung’ blood flow and vascular conductance. This is due to constriction of arteriovenous anastomoses, because the contribution via the bronchial artery to the ‘lung’ blood flow is rather limited (26, 40). Indeed, the nutrient part of cardiac output remained unchanged.
Human coronary artery contraction
To further predict the cardiovascular safety of S19014, we decided to analyse its effects on the human isolated coronary artery as compared with those of sumatriptan. Thus, in quiescent blood vessel segments, i.e. in the absence of U46619, the coronary artery contraction to S19014 and sumatriptan was not significantly different, although contraction to S19014 tended to be less than that to sumatriptan. In the presence of U46619, the contraction to sumatriptan was significantly augmented, while that to S19014 remained unaffected.
The pEC50 value of S19014 in the human coronary artery (5.55) was found to be 100–1000-fold lower than its pKi values at either α1- or α2-adrenoceptor subtypes (Table 1). However, it must be emphasized that agonist pKi values do not necessarily correlate to the potency of a compound in inducing a functional response (41). Secondly, the pEC50 values may have been overestimated, because the contraction observed at the highest concentration of the agonists was considered as Emax when, in some cases, a plateau had not been reached. However, when we calculated the pEC50 values with the curves extrapolated to estimate Emax (sumatriptan, 5.65 ± 0.35; S19014, 5.66 ± 0.44), these did not differ significantly from those reported in the Results section (5.99 ± 0.14 and 5.55 ± 0.24; P = 0.19 and 0.53, respectively, paired t-test). Finally, the fact that the contraction to S19014 was very low in the coronary artery may have possibly affected the accuracy of estimation of the pEC50 values. Although further experiments with subtype selective agonists and antagonists might elucidate which α-adrenoceptor subtype(s) mediates the S19014-induced contraction of the human coronary artery, such experiments would be difficult to interpret because of the small contractions elicited by S19014.
In the present study, the Emax of contraction to S19014 or sumatriptan was not related to the endothelial quality of the blood vessel segments. This suggests that the contraction to S19014 and sumatriptan might not solely depend on blood vessel wall characteristics, such as the presence of atherosclerosis. However, our study sample was fairly small (n = 8) for such an analysis. Moreover, we only included blood vessel segments without macroscopically visible atherosclerotic lesions in our study. Indeed, we have previously demonstrated in a large post-hoc study that the contraction to sumatriptan is larger in human coronary artery segments with intact endothelium than in blood vessel segments with functionally impaired endothelium (42). In contrast, it has been reported that constriction to α-adrenoceptor agonists is augmented in atherosclerotic human coronary arteries (43).
Possible clinical implications
Both in vitro (44, 45) and in vivo (2, 3, 7) experimental models demonstrating cranial vasoconstrictor properties have consistently shown their value in predicting therapeutic potential of drugs in the acute treatment of migraine. Therefore, the results obtained with S19014 in the present experiments suggest that this compound may well have anti-migraine properties. As S19014 had little systemic and regional haemodynamic effects, this compound appears to be well tolerated. However, as is the case with the currently available anti-migraine agents (22, 23, 46), we are aware of the potential liability of α-adrenoceptor agonists in constricting coronary arteries (47, 48). Nevertheless, the distribution of cardiac output to various body organs, including the heart, was not affected by S19014. This is further reinforced in this study, where S19014 was clearly less effective than sumatriptan in contracting the human isolated coronary arteries and, contrary to sumatriptan, its effect was not augmented by the thromboxane A2 analogue U46619. This may be clinically relevant, as the plasma concentration of thromboxane A2 increases in patients with myocardial infarction and unstable angina (49–51). Although the augmentation between 5-HT1B receptor agonists and thromboxane A2 has been well characterized (28), it is not yet clear whether such a mechanism also operates for α-adrenoceptor agonists in the human coronary artery. However, the α2-adrenoceptor-mediated contraction of the porcine isolated ear artery is enhanced by U46619 (52). Thus, the coronary side-effect potential of S19014 may be less than that of sumatriptan. However, considering that S19014 induces constriction of the coronary artery, albeit only to a small degree, the compound might remain contraindicated in patients with coronary artery disease.
Footnotes
Acknowledgements
This study was partly supported from funds obtained from Servier (Courbevoie, France) for a contract research project with Erasmus Pharma B.V.
