Abstract
Arteriovenous anastomoses (AVAs) may open up during migraine attacks. In studies with anaesthetized and bilaterally vagosympatectomized pigs, triptans reduce AVA blood flow and increase the arteriovenous O2 difference (AVDO2). To investigate whether subcutaneous sumatriptan 6 mg could induce changes in the AVDO2, we measured the AVDO2 in the external jugular vein in healthy subjects. We also measured the AVDO2 in the internal jugular and cubital veins. There were no changes in AVDO2 after subcutaneous sumatriptan, probably because AVA blood flow is limited in humans with an intact sympathetic nervous system.
Introduction
Increased arterial pulsation in the temporal region during migraine attacks was observed by Heyck in the 1950s. Low blood flow was indicated by pale skin and high blood flow by prominent veins. He then measured the oxygen arteriovenous difference (AVDO2) in the external jugular vein in seven migraine patients during attacks and found lower AVDO2 during attacks than outside attacks and in controls. Heyck concluded that AV shunts open up during a migraine attack (1, 2); Heycks AV shunt theory for migraine (1).
In animal studies, Saxena et al. have reported that the carotid vasoconstriction of ergotamine is confined to the arteriovenous anastomoses (AVAs) (3–5), and ergotamines' mode of action seemingly shed new light on the shunt theory (5). Studies on AVAs have identified an ‘atypical’ receptor for methysergide in the carotid vascular bed (6, 7) and provided incentive for the development of sumatriptan (8). Ergotamine or triptan treatment of migraine attacks may lead to AVA blood flow decreasing; this could be the treatment's mode of antimigraine action (3).
AVAs are large precapillary communications between arteries and veins and are present in most structures, including the cheeks, lips, forehead, nose, ears, nasal mucosa and eyes (9) as well as dura mater (10). Blood flow in the fingers is overwhelmingly arteriovenous anastomotic and the finger blood flow can be as high as 130 ml/min per 100 g (11). Sumatriptan has a selective vasoconstrictor action on AVAs of the human hand and fingers (12). It can be questioned whether there is more than a minimal AVA blood flow in the human head. If a considerable amount of AVA blood flow were present, a drug such as sumatriptan, which closes AVAs (4), would result in an increased AVDO2.
We therefore hypothesized that if subcutaneous sumatriptan 6 mg closed AVAs in the human head, this would lead to more nutrient blood flow through the capillaries and would thereby result in an increase of the AVDO2. This could elucidate the action of sumatriptan.
Materials and methods
We recruited 16 healthy subjects (12 male and four female), mean age 27 years (range 21–35 years). Exclusion criteria were: a history of migraine or any other type of headache (except episodic tension-type headache less than once a week); headache within 24 h before study start; any daily medication apart from oral contraceptives; pregnancy or breastfeeding; and serious somatic or psychiatric diseases. The study was approved by the Ethics Committee of the County of Copenhagen (KA-20060069), Danish Medicines Agency and the Danish Data Protection Agency and was undertaken in accordance with the Helsinki Declaration of 1964, as revised in Edinburgh in 2000. The study was registered on http://www.clinicaltrials.gov and monitored by the Good Clinical Practice unit at Copenhagen University Hospital. All subjects gave written informed consent to participate.
Catheters were inserted in the radial artery, cubital vein and external and internal jugular veins. The catheters in the internal jugular vein were placed with the tip high in the superior bulb of the vein to avoid admixture from the external jugular vein. The subjects rested in the supine position for 30 min before baseline measurements. Blood samples were taken at −10, −5, 15, 30, 60 and 90 min and were immediately analysed on an ABL 5 (Radiometer, Copenhagen, Denmark). All catheters were flushed with isotonic saline 0.9% immediately after sampling and were emptied of saline immediately before sampling. Cutaneous temperature (GM CTD87-M 992172) was measured in the frontal region of the head between the eyes at −10, −5, 15, 30, 60 and 90 min. At 0 min subcutaneous sumatriptan 6 mg was administered by a medical doctor in the right thigh. This study is part of a larger study measuring different neuropeptides.
Statistics
Baseline values for pO2, pCO2 and pH are stated as median ± range. AV differences over time for pO2 (AVDO2), pCO2 (AVDCO2) and pH (AVDPH) were analysed with univariate analysis of variance (ANOVA) with the fixed factors subject and time. If overall differences were found, Dunnett's test was applied to characterize which time points were different from baseline. Baseline was beforehand defined as an average of −10 and −5 min. All analyses were performed with
Results
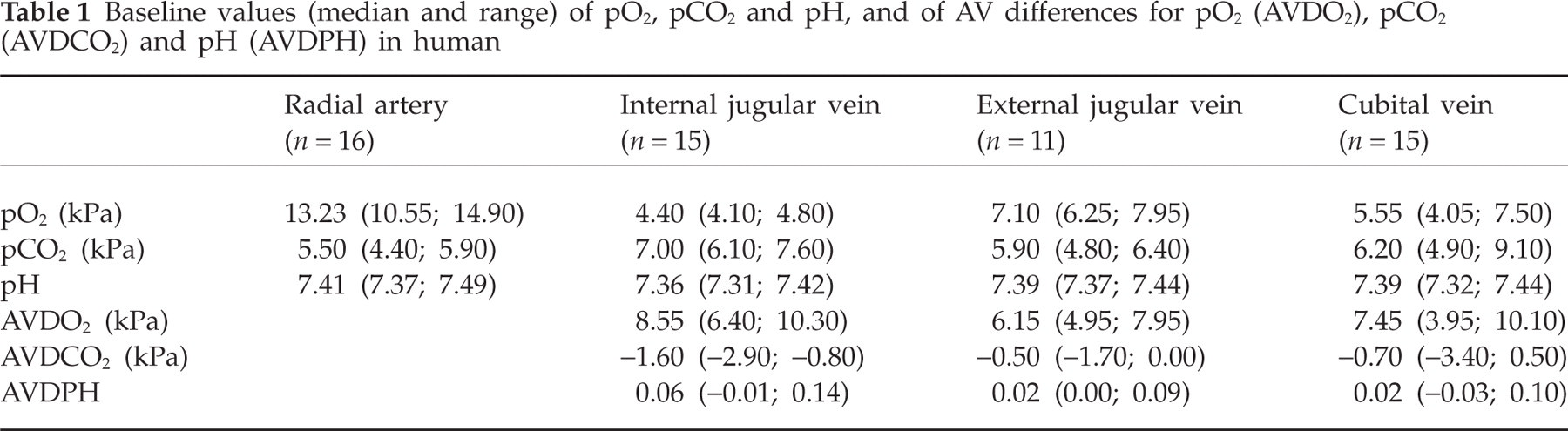
Sixteen subjects completed the study. Due to technical problems, it was not possible to have all catheters in all subjects (see Table 1), and there are a few outliers. Data are therefore presented as median and range. Baseline values for pO2 are shown in Table 1. The median and range AVDO2 in the cubital vein 7.45 kPa (3.95; 10.10), the internal 8.55 kPa (6.40; 10.30) and the external jugular vein 6.15 kPa (4.95; 7.95) (Fig. 1) were unchanged over time (P > 0.05) (ANOVA). Baseline values for pCO2 and pH are shown in Table 1. The AVDCO2 and AVDPH in the cubital vein, the internal and the external jugular veins were unchanged over time (P > 0.05) (ANOVA) (not shown). The mean baseline cutaneous frontal temperature was 34.6°C, and there was a slight increase to 34.8°C at 90 min.

Effect of sumatriptan, 6 mg subcutaneously (s.c.) given at time 0, on pO2 and arteriovenous differences in pO2 (AVDO2) in the radial artery (n = 16), internal jugular vein (n = 15), external jugular vein (n = 11) and cubital vein (n = 15). Baseline is an average of samples taken at time −10 and −5 min. There was no difference over time after sumatriptan (P > 0.05, ANOVA). Data are mean and range (kPa).
Baseline values (median and range) of pO2, pCO2 and pH, and of AV differences for pO2 (AVDO2), pCO2 (AVDCO2) and pH (AVDPH) in human
Discussion
The main result in the present study is the lack of change after subcutaneous sumatriptan in the AVDO2 between the arterial blood and external jugular venous blood. In addition, the AVDO2 was unchanged for internal jugular and cubital veins.
The AVDO2 in the arm was investigated because sumatriptan in a previous study decreased AVA blood flow in hand and fingers (12). Infusion of sumatriptan directly into the brachial artery caused a 50% decrease in AVA blood flow in the underarm and hand (12) during continuous sodium prusside infusion. However, this effect was seen only with the maximum infusion of 1000 ng/kg/min and not with the dose of 100 ng/kg/min (12). The brachial artery had a blood flow of 150 ml/min, and it can be calculated that the resulting plasma concentration of sumatriptan for 100 ng/kg/min would be 95 ng/ml. In the present study a subcutaneous dose of sumatriptan 6 mg could result in a peak concentration of 84 ng/ml (13). Subcutaneous sumatriptan 6 mg, the clinically used dose in migraine, is therefore unlikely to have an effect on AVA in hand and fingers. Similarly, we found no effect on AVDO2 in the cubital vein.
Cerebral blood flow in migraine patients is unchanged after subcutaneous sumatriptan (14), and one would thus not expect a change in AVDO2 in internal jugular vein after sumatriptan. In contrast, if the AVA blood flow is considerable in the extracerebral tissues in the head, one would presume an increase in AVDO2 between artery and external jugular vein after sumatriptan due to closing of AVAs. In anaesthetized and bilaterally vagosympatectomized pigs, sumatriptan caused a 70% decrease of AVA blood flow (4). The arterial-jugular venous AVDO2 saturation increased from 5.1% to 11.4% after sumatriptan 100 µg/kg (4). Thus, with a clinically relevant dose the AVDO2, saturation was doubled. This effect is probably due to the high AVA blood flow in the carotid bed in anaesthetized and bilaterally vagosympatectomized pigs. Thus, at baseline the nutrient flow was 20% and the AVA blood flow was 80% of total carotid blood flow (4). In conscious animals, however, the AVAs are constricted under strong influence of the sympathetic neuronal tone, thereby shunting only a small (< 3%) fraction of the total carotid flow (15).
The present study has shown that sumatriptan, at the clinically maximum dose (16), does not change the AVDO2 in the external jugular vein. This could in theory be due to a dose of sumatriptan without any closing effects on AVAs in the head in humans (see above). However, this is unlikely because sumatriptan is a very potent and selective constrictor of the carotid vascular bed in dogs, with generally no effect on other vascular beds (17). The ED50 for the increase in carotid resistance was 39 µg/kg intravenously (17), which corresponds to a dose of approximately 3 mg in humans, and we used sumatriptan 6 mg subcutaneously. The most likely reason for the lack of effect is that AVA blood flow is only minor in humans, as in conscious pigs (15). The tone in AVAs in pigs is maintained by sympathetic norepinephrine-containing nerves via α1-adrenoceptors (18). Lack of tone can explain the effect of sumatriptan in anaesthetized and bilaterally vagosympatectomized pigs. With a very low proportion of AVA flow, any vasoconstrictory effect of sumatriptan would go undetected when AVDO2 is measured.
In conclusion, no effect of subcutaneous sumatriptan could be detected when measuring AVDO2 in the external jugular blood in healthy volunteers, probably because the AVA blood flow is very sparse. Ideally, a study with administration of sumatriptan should be repeated during acute migraine attacks before firm conclusions can be drawn regarding the involvement of AVA in migraine.
Acknowledgements
The study was supported by grants from The Foundation for Research in Neurology, the University of Copenhagen, the Danish Headache Society and The Lundbeck Foundation for Neurovascular Signalling (LUCENS).
