Abstract
Several studies suggest that patients with migraine respond physiologically to stress differently from controls, yet experimental data are scarce. In order to evaluate the reactivity to stress in migraine, we recorded the quantitative electroencephalogram (qEEG) during non-noxious and noxious ischaemic arm stress in two groups of healthy controls and compared the results with the effects of non-noxious ischaemic arm stress in a group of patients with migraine. In the controls, non-noxious mild stress did not produce any qEEG change but noxious stress induced a significant decrease of the alpha power. By contrast, in migraine patients the non-noxious mild stress was sufficient to induce a significant decrease of the alpha power in all brain regions. The results of our study show that migraine sufferers display a lower threshold to physical stress and confirm previous studies indicating that migraine is a disease characterized by a state of altered neuronal excitability.
Keywords
Introduction
There are several lines of evidence suggesting that individuals with migraine respond physiologically to stress differently from healthy controls. For example, migraine patients were reported to be more sensitive to mental stressors, as revealed by a larger increase of EEG beta activity compared with controls during arithmetic reasoning (1). According to some authors, patients with migraine display abnormal autonomic nervous system responses to physical stressors, like cold pressor test or Valsava manoeuvre (2, 3). Finally, cardiovascular recovery patterns after cognitive stress are significantly different in patients with migraine compared with healthy controls (4).
However, some contrasting findings still exist. The results of the studies that examined autonomic nervous system responses to stress in migraine are contradictory (5–7) and the pathophysiological mechanisms underlying the abnormal response to stress in migraine are still a matter of debate.
We decided to further investigate the response to stress in migraine using the ischaemic arm test (8, 9), a test that evaluates the sensations induced by a sphigmomanometer cuff inflated around an arm. This test is characterized by a biphasic response: a few minutes after the beginning of the test the subject feels only a light discomfort in the arm (non-noxious ischaemic stress). Prolonging the stress, an acute painful sensation is produced (noxious ischaemic stress). To reduce the variability in the subjective response to stress, we measured the effects induced by the experimental procedure on quantitative electroencephalogram (qEEG). This technique is considered of great research value for the study of experimental pain in humans (10).
Taking into account all these considerations, this study was aimed at comparing the effects of non-noxious and noxious ischaemic arm stress on the qEEGs in healthy controls and in patients with migraine.
Methods
Patients
Nineteen migraine patients (n = 19, 3 males, 16 females, mean age 28.8 years±9.4
Demographic and clinical characteristics of migraine patients and healthy controls

Healthy controls were members of the staff and medical students. Two controls groups were used for the comparison with migraine sufferers. The first control group (n = 16, 5 males, 11 females, mean age 28.4 years ± 4.5
Testing protocol
Ischaemic stress of the left arm was performed according to the tourniquet technique, which we have extensively used in previous studies (12, 13). Briefly, the left arm was extended vertically and venous blood was drained through an Esmarch bandage. A sphygmomanometer cuff was then inflated up to a pressure of 300 mmHg around the left arm for a period of 3 min in order to produce mild discomfort (non-noxious mild stress). To obtain the noxious stress, the sphygmomanometer was maintained inflated up to 10 min. All the subjects gave their informed consent to the study.
EEG recordings
Electroencephalograms were recorded using a 20-channel EEG (Esaote Biomedica, Galileo Star System, Verona, Italy) with the patient in a comfortable supine position on the examination bed, controlled by a technician to prevent drowsiness and limb movements. Nineteen standard scalp electrodes of average reference were applied to the scalp in accordance with the 10–20 international system (Fz, F1, F2, F3, F4, F7, F8, C3, C4, Cz, P3, P4, Pz, T3, T4 T5, T6, O1, O2). Impedance was less than 2 kohm in each active lead. Data were collected for each subject and digitized at a sampling rate of 240 Hz with a low frequency filter of 1.5 Hz, a high frequency filter of 70 Hz and a notch filter in each channel.
EEGs were recorded for two 3-min periods at rest (baseline) and during the ischaemic arm stress. Epochs of 2.0 s, free of artifacts, were selected off-line and processed by a Fast Fourier Transformation (FFT) and the spectra obtained were averaged to obtain a mean power spectrum for each channel. Means of 20 epochs for each band were calculated in each subject. The absolute power of the spectrum in the delta (0.5–3.5 Hz), theta (4.0–7.5 Hz), α-1 (8.0–10.0 Hz), α-2 (0,5–10,10–12), β-1 (12.5–20.0 Hz) and, β-2 (20.5–32.0 Hz) bands was calculated. Artifacts control was based on the frontal epochs: if the power spectrum density was larger than 200 μV, it was rejected for further statistical analysis. In order to compensate for data skewing, a logarithmic transformation of the absolute power in each electrode was calculated. Regional power refers to the frontal region (F3 + F4), to the parietal region (P3 + P4), to the temporal region (T5 + T6) and to the occipital region (O2 + O1).
Statistics
Statistical comparisons were performed using the software package Statistica. Absolute power values were compared using Student's t-test,
Results
At rest, we did not find any difference between controls and migraineurs for all the EEG bands, as assessed by comparisons of both the power spectra and absolute power values among brain loci of the different groups.
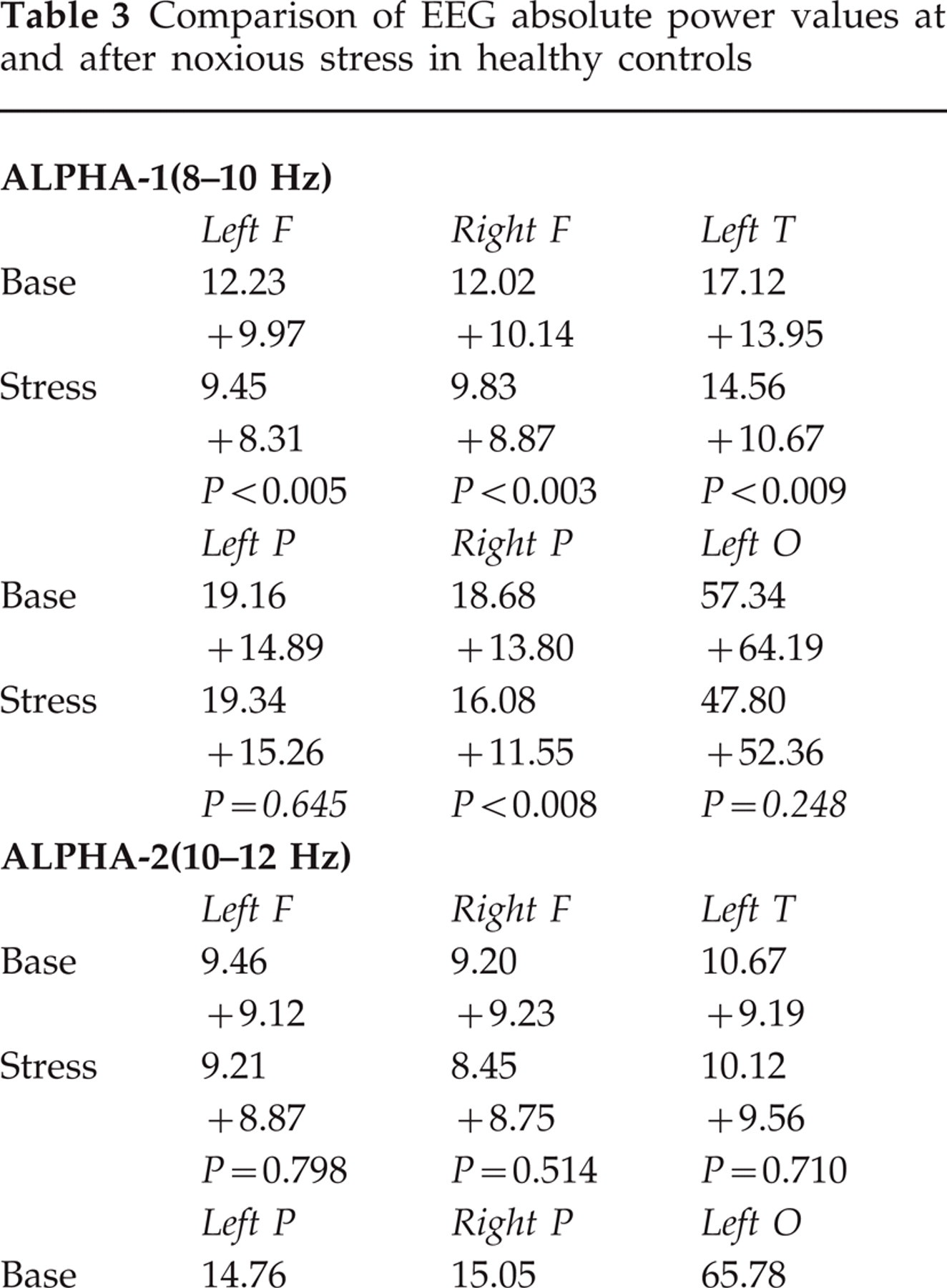
Figure 1 shows the comparison of baseline vs. stress power spectra in the frontal, temporal, parietal and occipital regions after non-noxious and noxious stress in the controls and after non-noxious stress in migraineurs. The controls who underwent non-noxious ischaemic stress did not show any significant modification (P > 0.05 in all comparisons) of the EEG in all the frequency bands (Table 2). By contrast, the controls who underwent noxious stress showed a clear-cut decrease of the alpha-1 power bilaterally in the frontal (left, P < 0.005; right, P < 0.003) and temporal (left, P < 0.009; right, P < 0.03) regions and in the right parietal (P < 0.008) and occipital (P < 0.01) areas (Table 3). The alpha-2 power was significantly decreased in the left (P < 0.007) and right (P < 0.03) parietal regions and in the right temporal area (P < 0.02) (Table 3). No significant change of beta-1, beta-2, theta and delta powers was found.

Frequency spectra in normal subjects who underwent the non-noxious stress and the noxious stress and in migraineurs who underwent the non-noxious stress (F, frontal; P, parietal; T, temporal; O, occipital).
Comparison of EEG absolute power values at rest and after noxious stress in healthy controls
Comparison of EEG absolute power values at rest and after non-noxious stress in healthy controls

Although the migraine patients underwent the same non-noxious stress as the controls, their EEG was similar to the controls who underwent noxious stress (Fig. 1). In fact, the migraineurs showed a significant reduction of the alpha-1 power in all the frontal (left, P < 0.006; right, P < 0.004), parietal (left, P < 0.008; right, P < 0.002), temporal (left, P < 0.05; right, P < 0.001) and occipital (left, P < 0.007; right, P < 0.003) regions, whereas alpha-2 was significantly reduced only in the right parietal locus (P < 0.02) (Table 4). In other words, whereas non-noxious mild stress did not produce any EEG change in normal subjects, it induced a decrease of the alpha power in migraine sufferers.
Comparison of EEG absolute power values at rest and after non-noxious stress in patients with migraine

Discussion
The findings we obtained in normal subjects are in agreement with previous studies showing that experimentally induced pain is associated with quantitative EEG changes. In fact, despite some conflicting results, due in part to the high individual variability of brain electrical activity, a relationship between experimental pain and quantitative EEG changes is now widely accepted (10). After intracutaneous brief noxious stimulation (phasic pain), a significant increase of delta and theta frequencies was observed (14). Under the noxious stress of cold-pressor test (tonic pain), healthy individuals exhibited a significant increase of delta and beta frequencies associated with a reduction of alpha power (15). In our study (tonic pain), we could not observe any change in the delta and beta powers but we confirm the reduction of the alpha power. The reduction of only the alpha power is probably related to our different experimental conditions.
The important new finding of the present study is concerned with the non-noxious mild stress. In fact, whereas normal subjects did not show any quantitative EEG change during non-noxious mild stress, migraine sufferers showed alpha power changes which were similar to those induced by noxious stress in the controls. These results indicate that whereas non-noxious mild stress does not influence the quantitative EEG in normal subjects, it affects the alpha power in migraine patients. Thus the threshold for stress-induced EEG changes is lower in migraine sufferers compared with controls. These results are in accordance with the concept that migraineurs are more sensitive to stress.
A large number of studies have demonstrated the presence of quantitative EEG abnormalities in patients with migraine (16–18). At rest, three major abnormalities are mentioned with some consistency: interhemispheric asymmetry of the alpha rhythm, diffuse or focal slowing and increased fast activity. The migraine attack is associated with a unilateral reduction of the alpha activity accompanied by a less severe reduction of the theta activity. Nyrke et al. demonstrated that abnormalities in alpha rhythm show a clear temporal relationship with the migraine attack: the asymmetries of the alpha rhythm increased significantly before the onset of the prodromal symptoms and clearly outlasted the headache attack (19).
In our study abnormalities in EEG alpha power after mild stress clearly differentiated migraine patients from healthy controls. These data indicate the presence in patients with migraine of a functional disorder of the brain centres that generate the alpha activity. In the past, alpha EEG activity was considered to reflect a passive or ‘idling’ state of the brain. On the contrary, recent findings indicate that EEG alpha activity is functionally related to several brain functions, such as memory and primary sensory processing, and suggest that alpha rhythm is a timing mechanism for cognitive processing (20, 21). These functions may be altered in patients with migraine (22).
Our data suggest that the generators of the alpha EEG activity are involved in the pathophysiological mechanisms of migraine.
Several neurophysiological studies have suggested that migraine is a disease characterized by an altered neuronal excitability (23–25). This finding may represent the basis of susceptibility to migraine attacks. In accordance with previous studies, our study showed that a mild stress produces EEG changes in migraineurs but not in controls and indicated that non-noxious mild stress, which is ineffective in normal subjects, is capable of affecting the excitability of neurones in migraine patients. The altered neuronal excitability in migraine has been related to a loss of inhibitory subcortical processes which normally suppress painful stimuli (26, 27). Further studies are needed to elucidate the precise pathophysiological mechanisms that generate the abnormal neuronal excitability observed in migraine patients.
Footnotes
Acknowledgements
Supported by grants from the Ministry of University and Scientific and Technological Research and the National Research Council ‘Coordinate Project on Trigeminal Pain’.
