Abstract
The pineal hormone, melatonin, has been implicated in the pathophysiology of migraine and several studies have demonstrated its vasoconstrictor properties. In the present study, systemic and carotid haemodynamic effects of melatonin, administered directly into the carotid artery, were investigated in anaesthetized pigs. Ten-minute intracarotid infusions of melatonin (1, 10 and 100 μg kg−1 min−1) produced slight decreases in blood pressure and total carotid and arteriovenous anastomotic blood flows, but nutrient blood flow was not affected. The decrease in carotid blood flow was entirely caused by the hypotension, since no changes in vascular conductance values were observed. It is concluded that melatonin itself is not capable of producing vasoconstriction in the cranial circulation of anaesthetized pigs. Thus, it appears that melatonin has no anti-migraine potential via a vasoconstrictor mechanism.
Introduction
Progress in the field of migraine has led to the introduction of sumatriptan (1–3), which is the first of a completely new class of compounds, now designated as 5-HT1B/1D/1F receptor agonists (4). Sumatriptan effectively aborts migraine headaches (5–7) and constricts, via vascular smooth muscle 5-HT1B receptors, large cranial and extracranial blood vessels (8, 9), including porcine carotid arteriovenous anastomoses (10). The success of sumatriptan in migraine therapy has prompted several companies to synthesize and evaluate new compounds, leading to the development of drugs with similar pharmacological properties to sumatriptan, but with improved pharmacokinetics (for reviews, see 11, 12). Unfortunately, one of the major side-effects of sumatriptan, namely the coronary constriction, has turned out to be a class effect of 5-HT1B receptor agonists (13). Thus, attempts are being directed towards finding other avenues for new drug development in migraine therapy. Selective agonists at different melatonin receptors (14–17) may provide one such avenue.
Melatonin has been implicated in the pathophysiology of migraine (18–20) and abnormality in its plasma levels and urinary excretion has been shown in migraine patients (21–24) and in one uncontrolled study some migraine patients reported headache relief after melatonin infusions (22). Moreover, several investigations have shown that melatonin can contract isolated blood vessels (25–30), but the exact receptor mechanisms remain to be fully understood. Similarly, limited information is available on the ability of melatonin to produce vasoconstriction in the cranial vasculature of anaesthetized animals. Thus, in this study, we investigated the effects of melatonin on carotid and regional (including cerebral) haemodynamics in anaesthetized pigs, with particular emphasis on the carotid arteriovenous anastomoses that have been implicated in the pathophysiology of migraine (31, 32).
Materials and methods
General
After an overnight fast, 11 pigs (Yorkshire × Landrace; female; 10–15 kg) were anaesthetized with azaperone (140 mg, i.m.), midazolan hydrochloride (7.5 mg, i.m.) and pentobarbitone sodium (600 mg, i.v.), intubated and connected to a respirator (BEAR 2E, BeMeds AG, Baar, Switzerland) for intermittent positive pressure ventilation with a mixture of room air and oxygen. Respiratory rate, tidal volume and oxygen supply were adjusted to keep arterial blood gas values within physiological limits (pH: 7.35–7.48; pCO2: 35–48 mmHg; pO2: 100–120 mmHg). Anaesthesia was maintained with a continuous i.v. infusion of pentobarbitone sodium at 20 mg kg−1 min−1. With this anaesthetic regimen, arteriovenous anastomotic blood flow is considerably higher than that in pigs in a conscious state or under thiopentone anaesthesia (33), thereby producing one of the main putative features of migraine, i.e. vasodilatation of carotid arteriovenous anastomoses (see 31, 32).
Catheters were placed in the inferior vena cava via the left femoral vein for the administration of drugs and in the aortic arch via the left femoral artery for the measurement of arterial blood pressure (Combitrans disposable pressure transducer; Braun, Melsungen, Germany) and the withdrawal of arterial blood for determining blood gases (ABL-510, Radiometer, Copenhagen, Denmark). The common carotid arteries, external jugular veins and vagus nerves were identified and both vagi and the accompanying cervical sympathetic nerves were cut between two ligatures, in order to avoid reflex-mediated changes in the carotid vasculature. Another catheter was placed in the right external jugular vein for the withdrawal of venous blood samples. The right common carotid artery was dissected free and a needle was inserted against the direction of blood flow for the administration and uniform mixing of radioactive microspheres. Blood flow was measured in the right common carotid artery with a flow probe (internal diameter: 2.5 mm) connected to a sine-wave electromagnetic flow meter (Transflow 601-system, Skalar, Delft, The Netherlands). Heart rate was measured with a tachograph (CRW, Erasmus University, Rotterdam, The Netherlands) triggered by electrocardiographic signals.
Arterial blood pressure, heart rate and carotid blood flow were continuously monitored on a polygraph (CRW, Erasmus University, Rotterdam, The Netherlands). Body temperature was kept at c. 37°C and the animals were continuously infused with saline to compensate for fluid losses during the experiment. The Ethics Committee of the Erasmus University Rotterdam dealing with the use of animals in scientific experiments approved the protocol for this investigation.
Distribution of common carotid blood flow
As described in detail elsewhere (34, 35), the distribution of common carotid blood flow was determined with 15.5 ± 0.1 (SD) μm diameter microspheres labelled with either 141Ce, 113Sn, 103Ru, 95Nb or 46Sc (NEN Dupont, Boston, USA). For each measurement a suspension of about 200 000 microspheres, labelled with one of the isotopes, was mixed and injected into the carotid artery. At the end of the experiment, the animal was killed, using an overdose of pentobarbital, and the heart, kidneys, lungs and the different cranial tissues were dissected out, weighed and put in vials. The radioactivity in these vials was counted for 5 min in a γ-scintillation counter (Packard, Minaxi autogamma 5000), using suitable windows for discriminating the different isotopes (141Ce: 120–167 KeV, 113Sn: 355–435 KeV, 103Ru: 450–548 KeV, 95Nb: 706–829 KeV and 46Sc: 830–965 KeV). All data were processed by a set of specially designed software (34) using a personal computer.
The fraction of carotid blood flow distributed to the capillary bed of different tissues (nutrient fraction of carotid blood flow) was calculated by multiplying the ratio of tissue and total radioactivities by the total common carotid blood flow at the time of the injection of microspheres. Since little or no radioactivity was detected in the heart and kidneys, all microspheres trapped in the lungs reached this tissue from the venous side after escaping via carotid arteriovenous anastomoses. Therefore, the amount of radioactivity in the lungs was used as an index of the arteriovenous anastomotic (non-nutrient) fraction of carotid blood flow (35). Vascular conductance was calculated by dividing blood flow (ml min−1) by blood pressure (mmHg), multiplied by one hundred and expressed as 10−2 ml min−1 mmHg−1.
Experimental protocol
After a stabilization period of about 1 h, baseline values of heart rate, mean arterial blood pressure, carotid blood flow and its distribution, as well as arterial and jugular venous blood gases were determined. At this point the animals were divided into two groups, which received three intracarotid infusions (0.1 ml min−1 for 10 min) every 20 min of either melatonin (1, 10 and 100 μg kg−1 min−1; n = 7) or its vehicle (distilled water, 50% ethanol, v v−1; n = 4). Ten min after each infusion as well as 30 min after stoppage of the last infusion (recovery), all parameters were reassessed.
Data presentation and statistical analysis
All data have been expressed as the mean ± SE mean. The significance of the changes (from baseline values) induced by the different doses of melatonin or its vehicle was evaluated with Duncan's new multiple range test, once an analysis of variance (randomized block design) had revealed that the samples represented different populations (36). Statistical significance was accepted at P < 0.05 (two-tailed).
Chemical compounds
Apart from the anaesthetics, azaperone (Janssen Pharmaceutica, Beerse, Belgium), midazolam hydrochloride (Hoffmann La Roche b.v., Mijdrecht, The Netherlands) and pentobarbitone sodium (Apharmo, Arnhem, The Netherlands), the compounds used in this study were: melatonin (Sigma-Aldrich Chemie, b.v., Zwijndrecht, The Netherlands) and heparin sodium (Leo Pharmaceutical Products, Weesp, The Netherlands) for preventing clotting of the catheters. Melatonin was dissolved in distilled water, containing 50% ethanol (v v−1).
Results
Systemic haemodynamic changes
As shown in Table 1, 10-min intracarotid infusions of melatonin produced a slight bradycardia, which was also observed after administration of the corresponding volumes of vehicle. Even after a 30-min recovery period, heart rate remained lower in the melatonin as well as the vehicle group. A slight hypotension was observed after the two highest doses of melatonin, whereas the corresponding volumes of vehicle were devoid of this effect. In both groups, mean arterial blood pressure was significantly decreased after a recovery period of 30 min. No changes in the difference in oxygen saturation between arterial and jugular venous blood (A-V SO2) were produced by either melatonin or its vehicle, although after a 30-min recovery period, A-V SO2 was slightly higher when compared with the initial baseline values in the melatonin group.
Systemic haemodynamic values at baseline, after 10-min intracarotid infusions of either melatonin or the corresponding volumes of vehicle and after a 30-min recovery period. Percentage changes from baseline are given in brackets

All values have been presented as the mean ± SE mean.
P < 0.05 vs. baseline.
Carotid haemodynamics
As shown in Fig. 1 (absolute values) and Fig. 2 (percentage changes from baseline), the highest dose of melatonin decreased total carotid and arteriovenous anastomotic blood flow, an effect that persisted during a 30-min recovery period. These decreases in blood flow were not accompanied by changes in total carotid and arteriovenous anastomotic vascular conductance. Melatonin did not change nutrient blood flow, but the highest dose produced a slight but significant increase in nutrient vascular conductance, which did not persist after 30 min. Except for an increase in total carotid vascular conductance after a 30-min recovery period, no carotid haemodynamic changes were observed in the vehicle group.

Values of total carotid arteriovenous anastomotic (AVA) and capillary (nutrient) blood flows and vascular conductances at’baseline (□), after three consecutive 10-min intracarotid infusions of either melatonin ((diagonal lined box) 1, (hashed box) 10 and (dotted square) 100 μg kg−1 min−1; n=7) or the corresponding volumes of vehicle (0.1 ml min−1; n=4) and after a 30-min recovery period (▪) in anaesthetized pigs. All values are presented as the mean ± SE mean. ∗P<0.05 vs. baseline.
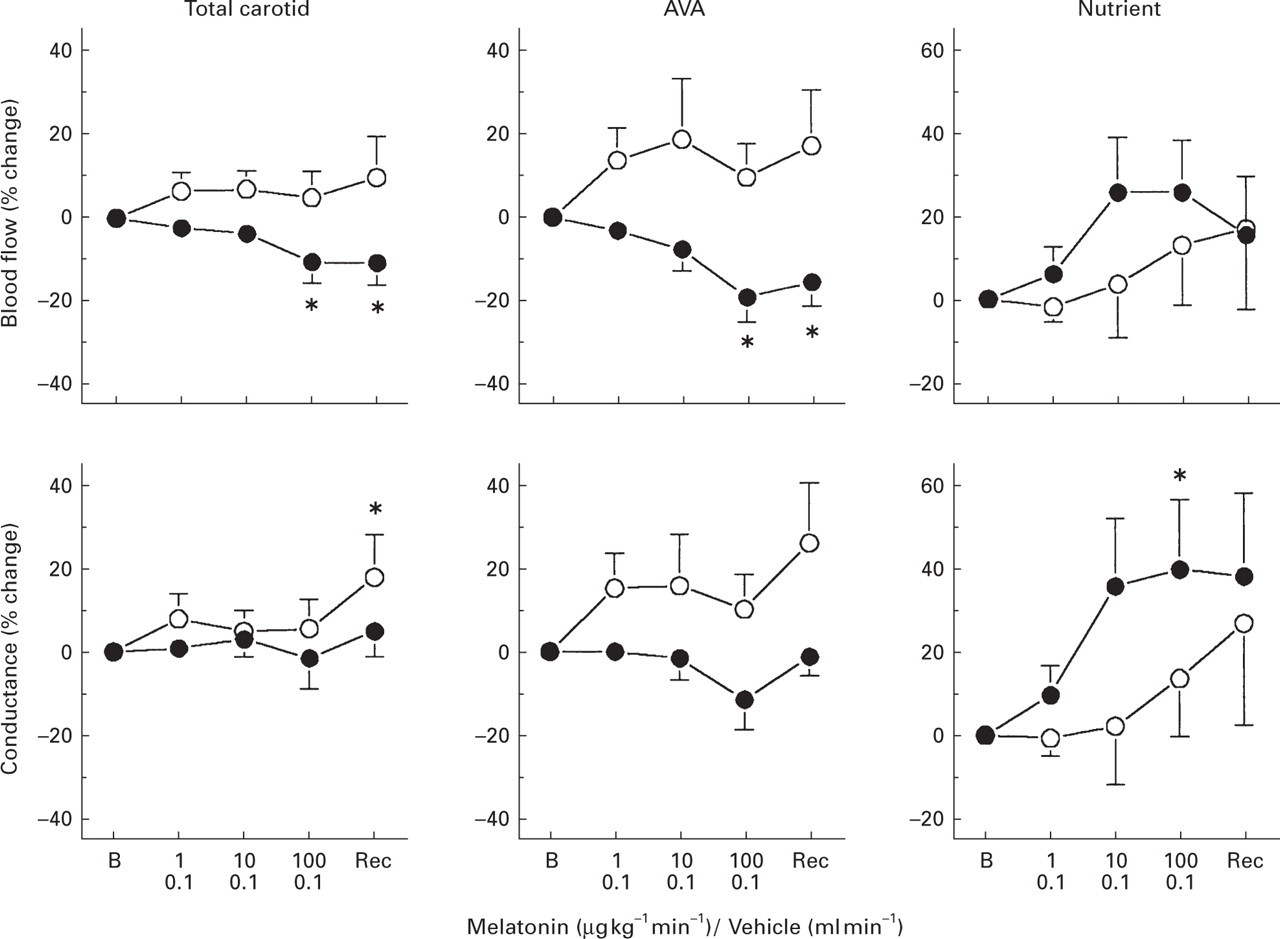
Percentage changes from baseline values caused by 10-min intracarotid infusions of either melatonin (1, 10 and 100 μg kg−1 min−1; n = 7, •) or corresponding volumes of vehicle (0.1 ml min−1; n = 4, ○) in total carotid blood flow and its fractionation into arteriovenous anastomotic (AVA) and capillary (nutrient) fractions in anaesthetized pigs. All values are presented as the mean± SE mean. ∗P < 0.05 vs. baseline. B, Baseline; Rec, Recovery.
The higher doses of melatonin significantly increased ear, fat and salivary gland blood flow (data not shown) and vascular conductances (Fig. 3). No changes were observed in skin, bone, eye, brain, tongue and dural blood flows or vascular conductances. Only an increase in vascular conductance, but not in blood flow, was observed in skeletal muscles.

Values of vascular conductance in ipsilateral cranial tissues at baseline (□), after three consecutive 10-min intracarotid infusions of either melatonin ( 1,
1,  10 and
10 and  100 μg kg−1 min−1; n = 7) or the corresponding volumes of vehicle (0.1 ml min−1; n = 4) and after a 30-min recovery period (▪) in anaesthetized pigs. All values are presented as the mean ± SE mean. ∗P < 0.05 vs. baseline.
100 μg kg−1 min−1; n = 7) or the corresponding volumes of vehicle (0.1 ml min−1; n = 4) and after a 30-min recovery period (▪) in anaesthetized pigs. All values are presented as the mean ± SE mean. ∗P < 0.05 vs. baseline.
Discussion
General
It has been shown that several acutely acting anti-migraine drugs, including the ergot alkaloids and triptans, constrict porcine carotid arteriovenous anastomoses and that a dilatation of these ‘shunt’ vessels may be involved in the pathophysiology of migraine (4, 11, 31, 32). Since the vasoconstriction of cranial and coronary arteries by current acutely acting anti-migraine drugs seems to be mediated by a similar receptor (5-HT1B), attempts are now being directed towards finding other avenues for new drug development in migraine therapy. One such possibility may be via melatonin receptors. Indeed, some investigators have implied a role for this pineal hormone in the pathophysiology of migraine, mainly because of alterations in its urinary excretion and plasma levels in migraine patients (21–24). In keeping with this, an impaired pineal function has been suggested in migraine and melatonin infusion may result in some headache relief (22). Admittedly, information is rather scarce and no placebo-controlled data are available. Nevertheless, melatonin has also been shown to constrict isolated cranial blood vessels (28) and to reduce regional cerebral blood flow in rats (37). However, the cranial circulatory effects of this endogenous ligand have not been studied much in vivo. As discussed below, the results of the present study show that melatonin is devoid of cranial vasoconstrictor effects in anaesthetized pigs.
Systemic haemodynamics
Although bradycardia was observed after higher doses of melatonin, it is not likely to be an effect of the drug itself, since the vehicle (distilled water containing 50% ethanol) produced a similar effect. Additionally, lower doses of melatonin (1–100 ng kg−1 min−1), dissolved in distilled water containing only 1% ethanol, were devoid of heart rate changes (data not shown). On the other hand, low (data not shown) as well as high doses of melatonin (10 and 100 μg kg−1 min−1) produced a decrease in blood pressure, which was not observed after administrations of the corresponding vehicle. In keeping with this, melatonin-induced hypotension has also been demonstrated in normal (38), pinealectomized (39) and spontaneously hypertensive (40) rats. As suggested elsewhere, melatonin can reduce sympathetic nerve activity either at a central or a peripheral level (38, 41, 42) and relax isolated blood vessels (43–46).
Carotid haemodynamic changes
The present results show that only the highest dose of melatonin (100 μg kg−1 min−1) produced a moderate reduction of total carotid blood flow, confined to the arteriovenous anastomoses. This decrease in total carotid and arteriovenous anastomotic blood flow was exclusively due to the simultaneous reduction in blood pressure (see above), since vascular conductance values remained unchanged. In keeping with the latter, no significant changes were observed in the A-V SO2. Accordingly, lower doses of melatonin (1–100 ng kg−1 min−1; n = 3) did not significantly change total carotid and arteriovenous anastomotic vascular conductances (data not shown). Thus, using a wide range of dosing, melatonin, administered directly into the carotid artery, does not constrict the porcine carotid vasculature.
In contrast with the present results, melatonin has been reported to decrease cerebral blood flow in rats (37) and to constrict isolated rat cerebral (29, 47) and caudal (30, 48) arteries, as well as the human coronary artery (28). The lack of vasoconstrictor effect of melatonin in the present experiments may be due to species difference, although the hormone has been demonstrated to constrict porcine coronary artery (28). Similarly, we cannot completely rule out the possibility that melatonin produced a cranial vasodilator response, thereby masking a putative vasoconstrictor effect. Melatonin can relax porcine isolated blood vessels pre-contracted with U46619 (27) and seems to dilate skeletal muscle, ear, fat and salivary gland vascular beds (see Fig. 3). The hormone shows similar binding affinities for its vasoconstrictor (MT1) and vasodilator (MT2) receptors (49) and studies using selective MT1 and MT2 receptor agonists and antagonists may resolve this issue. However, in view of the use of melatonin over a very wide dose range (from 1 ng kg−1 min−1 to 100 μg kg−1 min−1), it seems to us unlikely that the vasodilator effect of melatonin negated the constrictor responses in our experiments in both magnitude and duration in the different regions.
Conclusions
The present results imply that melatonin does not constrict carotid arteriovenous anastomoses. Since this property has been shown to be highly predictive for anti-migraine efficacy, melatonin itself seems unlikely to be effective in the treatment of migraine.
Footnotes
Acknowledgements
This study was supported by a grant from Institut de Recherches Internationales Servier I.R.I.S., Courbevoie, France to Erasmus Pharma B.V.
