Abstract
Clinical and preclinical studies suggest that 5-HT and nitric oxide (NO) mobilization within the trigeminovascular system is fundamental to the initiation of migraine attacks., e.g. m-chlorophenylpiperazine (m-CPP) and glyceryl trinitrate (GTN) induce headache in humans. 5-HT2B receptors are known to mediate NO-dependent vasorelaxation in peripheral blood vessels, raising the possibility that this receptor is implicated in the pathogenesis of the disease. Therefore, we measured the effects of 5-HT2B agonists (m-CPP or BW723C86) or GTN on trigeminal nerves by quantifying Fos expression in the rat TNC. m-CPP (0.1 mg/kg, i.v.) induced time-dependent elevations in Fos-LI in the rat TNC 2 h and 8 h after injection. In contrast, neither intravenous GTN (0.5 μg/kg per min, infused 20 min) nor BW723C86 (0.1 mg/kg, i.v.) increased Fos-LI at 2 h or 8 h after administration. These data are not consistent with the involvement of the 5-HT2B/2C receptors or NO in trigeminovascular activation, and by inference migraine, and suggest the contribution of some other unidentified pathway.
Introduction
The affinities of several migraine prophylactic agents for the 5-HT2C receptor subtype were shown to correlate significantly with the clinically effective daily dose for prophylaxis. Similar correlation was also observed for the pharmacologically similar 5-HT2B receptor (correlation coefficients 0.86 and 0.93 for 5-HT2B and 5-HT2C, respectively (1)). The observations led to the hypothesis that 5-HT2B/2C receptors might be involved in the onset of migraine headache.
Other key observations implicate these two receptor subtypes in the pathogenesis of migraine. First, in a study conducted by Brewerton and colleagues (2), the primary metabolite of trazodone, 1-(m-chlorophenyl)-piperazine (m-CPP) induced head pain in 28/52 subjects, with symptoms associated with common migraine including nausea, photophobia, unilateral pain and throbbing occurring 8–10 h after drug administration. Amongst these, 20 patients had a personal or family history of migraine, 18 of whom developed severe symptoms. Except for the absence of the prodrome phase, the patients described the m-CPP headache as being identical to previous episodes of migraine. At the peak plasma concentration observed in this study (1.8 × 10−7
Activity of m-chlorophenylpiperazine (m-CPP) at 5-HT receptor subtypes

Table adapted from Fozard (33). Data in Table derived from radioligand binding assays (5-HT1, 5-HT2A/2C, 5-HT3 (5, 34–36)), or functional activity assays (5-HT1A, 5-HT1B inhibition of adenylate cyclase, 5-HT1D blockade of 5-HT-induced inhibition of adenylate cyclase (37), 5-HT2C activation of phosphoinositide hydrolysis (37); 5-HT2A/2C blockade of 5-HT-induced phosphoinositide hydrolysis (38); 5-HT2B rat stomach fundus (39). 5-HT3 blockade of 5-HT-induced depolarization of rat vagus nerve (40).
In addition, a systemic fall in 5-HT concentration in platelets and plasma is observed at the beginning of a migraine episode (6, 7), with a concomitant elevation in urinary concentration of 5-HIAA (6, 8). Of all the known 5-HT receptor subtypes, 5-HT binds to the 5-HT2B and 5-HT2C receptors with the highest affinity (9, 10). These receptors are therefore likely to be stimulated in any pathological situation in which the concentration of 5-HT is elevated.
Receptors pharmacologically similar to 5-HT2B/2C mediate endothelium-dependent, L-NAME-sensitive vasorelaxation in isolated sections of pig coronary artery or rabbit jugular vein (11–13). However, based on reverse transcriptase-polymerase chain reaction (RT-PCR) investigations, the receptor through which the m-CPP effects and 5-HT-mediated decreases in blood pressure, is more likely to be the 5-HT2B receptor as mRNA for 5-HT2B but not 5-HT2C receptors in cerebral vasculature and cultured endothelial cells has been detected (14, 15).
Based on these observations, it is postulated that a human analogue of the porcine endothelial 5-HT2B receptor, which similarly mediates nitric oxide (NO)-dependent vasorelaxation, resides in the cerebral vasculature and is important to migraine pathogenesis and consequently the prophylactic actions of 5-HT2B receptor antagonists in migraine (16, 17). Indeed, the non-selective 5-HT2A/2B/2C agonist, DOI (1-(2,5-dimethoxy-4-iodophenyl)-2-aminopropane), causes modest,
In separate studies, intravenous infusion of glyceryl trinitrate (GTN), a NO donor, induced a more profound head pain in migraineurs than in control patients (18, 19). In both groups of patients, GTN rapidly induces head pain, possibly as a result of vasodilatation of major conduit arteries, that abates on cessation of GTN infusion (20–22). The observed effect of GTN to induce migraine-like head pain further corroborates the hypothesis that 5-HT2B receptors, localized in the cerebral arterioles, that mediate NO-dependent vascular relaxation might play an important role in the onset of migraine.
The present study attempts to determine whether 5-HT2B receptor activation or NO modulates trigeminovascular activity in rats. To this end, changes in Fos expression, determined by immunohistochemistry, in laminae I and II of the rat TNC were quantified as an index of activation of the nociceptive fibres implicated in headache. The time course for Fos expression in the rat TNC 2–8 h after i.v. administration of the non-selective agent, m-CPP (0.1 mg/kg), or the selective and efficacious (as defined in isolated rabbit jugular vein (13)) 5-HT2B/2C receptor agonist, BW723C86 (0.1 mg/kg), or GTN (0.5 μg/kg per min for 20 min) were investigated. As a positive control of the immunohistochemistry, Fos-like immunoreactivity (Fos-LI) was confirmed in laminae I and II of rat TNC following direct (electrical) stimulation of the trigeminal ganglia.
Materials and methods
Methods
Male Sprague-Dawley rats (300–350 g) were anaesthetized with pentobarbital sodium (60 mg/kg, i.p.). Cannulae (made from polyethylene tubing, inner diameter 0.58 mm, outer diameter 0.965 mm) were implanted in the right femoral vein for drug delivery. Blood pressure was monitored through an indwelling cannula in the ipsilateral femoral artery. The animals were laid supine on a themostatically controlled heating mat at 37°C. Post-operative anaesthesia was maintained by i.p. infusion of pentobarbital sodium (50 mg/ml, 0.18–0.20 ml/h). In experiments where animals were subjected to lengthy anaesthesia (> 4 h) the trachea was also cannulated (polyethylene tubing inner diameter 1.67 mm, outer diameter 2.42 mm). All animals respired freely.
For experiments requiring electrode placement for unilateral electrical stimulation of the trigeminal ganglia the anaesthetized rat was immobilized in a stereotaxic frame and ventilated with air at 60 strokes per minute, 12 ml/kg following placement of the venous, arterial and tracheal cannulae. Bipolar stimulating electrodes (Rhodes NE-200) were lowered through holes drilled in the skull into each of the trigeminal ganglia using a micromanipulator (displacement coordinates in mm from bregma=3.3, 3.2 and 9.3 along anterior-posterior, lateral and vertical axes, for a 250-g rat). Following electrode placement, animals were maintained under anaesthesia for 3 h before unilateral stimulation of the (left) trigeminal ganglion (0.6 mA, 5 ms square wave pulses at 5 Hz for 5 min). Correct electrode placement was confirmed by jaw and ear movements of the anaesthetized rat during electrical stimulation, and damage to the trigeminal ganglia visible on dissection. The sham (right) electrode was implanted in order to provide a within-animal control for electrical stimulation of the contralateral side.
In other studies, m-CPP, BW723C86, GTN or appropriate vehicle was administered intravenously 10 min following the completion of surgery at a final delivery volume of 1 ml/kg. m-CPP (0.1 mg/kg), BW723C86 (0.1 mg/kg) or saline was administered as a single bolus injection. GTN or PEG vehicle (0.5% v/v polyethethylene glycol) was infused at 0.5 μg/kg per min for 20 min.
Two, 4, 6 or 8 h after injection of m-CPP, BW723C86, GTN or vehicle, or 2 h after electrical stimulation of the trigeminal ganglion, the descending aorta was clamped and the rat head was perfused via the left ventricle with PBS (50 ml/min, 2 min) followed by neutral buffered formalin (10 w/v%, pH 7.4, 50 ml/min, 3 min). Following perfusion fixation, the brain was dissected and immersed in neutral buffered formalin (10%) for immersion fixation overnight. Fixed brains were immersed in a solution of 30% (w/v) sucrose in PBS containing 0.02% (w/v) sodium azide, and stored at 4°C for 2 days. Sections of brainstem (40 μm) were cut using a Jung 1800 cryostat. Brainstem sections were immersed in hydrogen peroxide (0.3% v/v, 30 min) prior to an incubation in primary antibody (polyclonal rabbit anti-Fos antibody, diluted 1:15 000 PBS enriched with normal 3% v/v goat serum, 0.25% v/v Triton X-100, 0.02% w/v sodium azide, for 24 h). Binding of primary antibody was visualized using the Vectastain Elite biotinylated secondary antibody kit in conjunction with diaminobenzidine (DAB) peroxidase staining reagents. Fos-LI in the rat brainstem was determined by counting the number of stained nuclei visible at ×10 magnification in 16–24 sections (40 μm) of the brainstem (0.9–0.6 mm below obex). Results are presented as the mean number for Fos-stained nuclei in 16–24 sections from each animal, averaged over five or more animals per drug treatment. Statistical significance was determined using an unpaired Student's t-test. Negative controls for the staining methodology, generated in the absence of primary antibody, or in the presence of an excess of exogenous Fos peptide, showed no specific staining of brainstem nuclei.
Materials
The following were purchased: sodium azide, m-CPP, 4% neutral buffered formalin, Triton X-100, hydrogen peroxide and normal goat serum from Sigma Chemical Co. (St Louis, MO); pentobarbitone sodium (50 mg/ml solution) from Abbott Laboratories (Abbott Park, IL, USA); polyclonal rabbit anti-Fos antibody and purified Fos peptide from Oncogene (Cambridge, MA, USA); Vectastain ABC elite kit and DAB peroxidase staining reagents from Vector Labs (Burlingame, CA, USA); PBS from Gibco Life Technologies (Rockville, MD, USA); rats from Charles River (Wilmington, MA, USA). GTN (10% w/v solution in PEG) and BW723C86 ((±)1-[5-(2-thenyloxy)-1H-indol-3-yl] propan-2-amine hydrochloride) were generous gifts from Zeneca Inc. (Wilmington, DE, USA) and GlaxoWellcome Medicines Research (Stevenage, UK), respectively.
Results
Effects of electrical stimulation of trigeminal ganglia on Fos-LI
Unilateral electrical stimulation of the trigeminal ganglia produced profound ipsilateral increases in Fos-LI in rat TNC 0.6–0.9 mm from obex (caudal), relative to the unstimulated side into which a sham electrode had been implanted (mean Fos-stained nuclei=381 ± 135 and 51 ± 9 on ipsilateral and contralateral sides, respectively, n = 3). Data from all subsequent studies using chemical stimuli represent numbers of stained nuclei counted from this level of the rat brainstem.
Effects of m-CPP infusion
The mean numbers of Fos-stained nuclei in TNC laminae I and II of m-CPP-treated animals were elevated relative to time-matched saline controls 2 h and 8 h after i.v. injection of m-CPP (0.1 mg/kg, 75% and 207% increase over time-matched controls at 2 h and 8 h, respectively). At the intervening time points (4 h and 6 h after injection of m-CPP or saline) no significant increases in Fos-LI were observed (see Table 2, Fig. 1b).
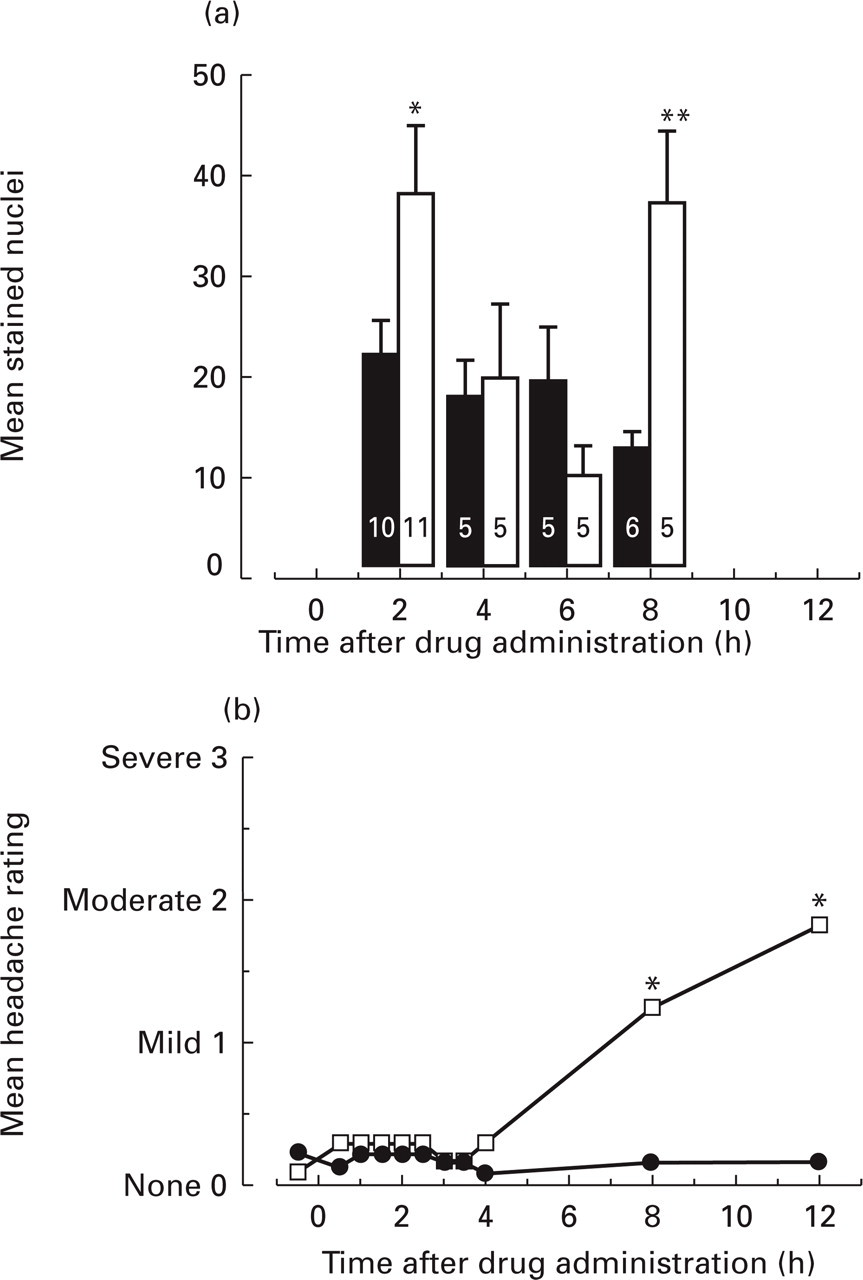
Comparison of the time course for effects of m-chlorophenylpiperazine (m-CPP) on Fos-LI in rat TNC (a) with that for onset of m-CPP-induced head pain in migraineurs and control patients (b). Data presented are mean Fos-LI from five to 10 animals treated with m-CPP (0.1 mg/kg i.v., number of replicates for each group enumerated on columns) and mean headache rating from 52 patients treated with m-CPP (0.5 mg/kg p.o.) or placebo. Differences between m-CPP and time-matched controls were determined using Student's t-test (∗P ≤ 0.05; ∗∗P < 0.005). (a) □, m-CPP (100 μg/kg, i.v.); ▪, saline (1 ml/kg, i.v.). (b) □, m-CPP; •, placebo. Data due to Brewerton et al. (2). (Reprinted with permission from MOSBY inc.
Time course for changes in numbers of Fos-stained nuclei in rat TNC following i.v. injection of m-chlorophenylpiperazine (m-CPP; 0.1 mg/kg), BW723C86, glyceryl trinitrate (GTN) or vehicle

Differences between test and control animals determined by Student's t-test
P ≤ 0.05;
P < 0.005.
Effects of BW723C86
No significant differences were observed between test animals and time-matched saline controls 2 h or 8 h after an i.v. bolus injection of BW723C86 (0.1 mg/kg, delivered in saline solution as 1 ml/kg, n = 5 animals, Table 2).
Effects of GTN infusion
No significant difference was observed between GTN (0.5 μg/kg per min) and vehicle (0.5% PEG)-treated animals at 2, 4, 6 or 8 h following i.v. infusion for 20 min (0.05 ml/kg per min, n = 4–6 animals, Fig. 2a, Table 2).

Comparison of the time course for effects of glyceryl trinitrate (GTN) on Fos-LI in rat TNC (a) with that for onset of GTN-induced head pain in migraineurs and control patients (b). Data presented are mean effects of GTN infusion (0.5 μg/kg per min i.v., for 20 min) on Fos-LI from five to six rats, or head pain in 17 migraineurs or 17 control patients. (a) □, 0.5 μg/kg per min GTN; ▪, 0.5% PEG vehicle. (b) ▵, Migraineurs; •, non-migraineurs. Data due to Olesen et al. (19).
In separate animals, blood pressure was monitored via an in-dwelling cannula in the femoral artery. Infusion of the dose of GTN as utilized in Fos studies (0.5 μg/kg per min for 20 min) via the femoral vein had no significant effect on mean femoral arterial blood pressure. However, at a faster rate of infusion (1 μg/kg per min for 10 min) the same concentration of GTN profoundly depressed mean blood pressure for the duration of the infusion relative to vehicle controls (data not shown). These data confirm the vasoactivity of the GTN used in this study.
Discussion
BW723C86 is a selective, highly efficacious agonist at the endothelial 5-HT2B receptor in the rabbit jugular vein that mediates NO-dependent vasorelaxation (12). Similar agonist behaviour has been demonstrated in the trigeminovascular system where a vasodilatory effect of BW723C86 (62–500 μg) was identified in the pial arterial beds of anaesthetized rats (20–120 μm diameter pial arterioles (23)). In these blood vessels, topically administered 5-HT, α-Me-5-HT, 5-CT, m-CPP or BW723C86 caused significant dose-dependent vasorelaxation relative to artificial controls, while the 5-HT1B/1D receptor agonist, sumatriptan, induced vasoconstriction. These data are consistent with 5-HT2B and 5-HT7 receptor-mediated vasodilatation, and confirm a vasoactive role of BW723C86 in cerebral blood vessels. Furthermore, in the same study the effect of 5-HT was demonstrated to be NO-dependent as it was inhibited by pretreatment with the NO synthase inhibitor, L-NG-nitro-arginine (100 μ
While GTN, infused into the rat femoral vein at 0.5 μg/kg per min, failed to activate the rat TNC, an equivalent dosing regimen in humans is known to significantly increase heart rate and systolic blood pressure, and induce both a transient headache during infusion and a profound migraine-like headache 4–5 h later (24). The same dosing regimen of GTN significantly increased cerebral blood volume and decreased mean cerebral blood velocity with no effect on blood flow through the perfusion territory of the middle cerebral artery, consistent with vasodilatation of large cerebral blood vessels (25). Furthermore, an even lower infusion rate of GTN into anaesthetized cats (0.25 μg/kg per min) increased pial artery diameter (109% and 90.2% of baseline for GTN and saline, respectively) and increased sustained NO release (128% and 75.3% of baseline for GTN and saline, respectively), with no concomitant change in mean arterial blood pressure (26). These reports demonstrate a clear NO-mediated effect in the cerebral vasculature of humans and cats following equivalent or slower rates of GTN infusion. The failure of GTN to elevate systemic blood pressure or Fos-LI in rat TNC in this study may result from a species-specific insensitivity to NO. The reasons for the lack of sensitivity to 5-HT2B/2C receptor agonists or GTN in the trigeminovascular system are not clear, as RT-PCR and immunohistochemical studies have demonstrated that both vascular 5-HT2B receptors and NO synthase enzymes (and, by implication, sensitivity to NO) are present in the rat trigeminovascular system (14, 15, 27, 28). It remains possible that infusion of higher concentrations of GTN or BW723C86 would have caused activation of rat trigeminal neurones in a manner analogous to that seen for the 0.5 μg/kg per min dose in humans or cats. Indeed, in another study elevations in Fos-LI in other regions of rat brain are reported following subcutaneous administration of a higher concentration of GTN (10 mg/kg s.c. (29)).
It is known that reliable induction of Fos requires a strong, prolonged stimulus (30). It is possible therefore that GTN infusion in rats as performed in this study may activate the cell bodies in the TNC, but in a manner that is not sufficient to activate reliably detectable levels of Fos that parallels the transient or later (migraine-like) headaches experienced in humans.
m-CPP is a non-selective 5-HT2B/2C agonist, and induces 5-HT release from the hypothalamus (31). In anaesthetized rats, m-CPP (0.1 mg/kg i.v.) activates the trigeminovascular system, as evidenced by increases in plasma protein extravasation from blood vessels of the dura mater (32). In humans, orally administered m-CPP induces migraine-like headache in patients with a personal or family history of migraine (2). These patients reported mild head pain initially, followed by severe, unilateral, pulsatile headache 3–4 h after drug administration. The failure of the endothelial 5-HT2B/2C receptor agonist, BW723C86 (0.1 mg/kg), but not the lower efficacy agonist, m-CPP, to increase Fos-LI in the rat TNC after administration suggests that the stimulation of endothelial NO synthase enzymes by 5-HT2B receptor activation, if relevant to migraine pathogenesis, may not be sufficient to activate the trigeminal sensory afferent nerves in rats. While it may be argued that the role of 5-HT2 receptors or NO in the trigeminovascular system of rats may differ from that of humans, the similarities between the time course of Fos expression in rats and headache in humans over the 8 h following administration of m-CPP would tend to suggest some commonality between the species. However, the failure to detect similar elevations in Fos-LI following treatment with BW723C86 or the NO donor, GTN, suggests that m-CPP must activate the trigeminal nerve via an as yet unknown mechanism other than the proposed stimulation of the endothelial 5-HT2B receptor and NO synthase.
Clinical observations in migraine patients and controls treated with m-CPP or GTN report a higher incidence of severe or migraine-like head pain in migraineurs than in controls. In one such study, both migraineurs and control reported a mild head pain during infusion of GTN, but a second headache occurring in migraineurs, but not control patients, was observed approximately 3 h after the cessation of GTN infusion (19). It has been suggested this late-onset headache results from a unique hypersensitivity of migraineurs to NO. This idea is further supported by the hypothesis, based on blood flow studies, that migraineurs have a unique cerebroarterial hypersensitivity to NO relative to control and episodic tension headache patients (20). If head pain, and therefore trigeminal nerve activation, is a product of NO hypersensitivity, chemical stimulation of the 5-HT2B and NO pathway in healthy rats devoid of any such predisposition may not be expected to result in trigeminal nerve activation. In support of this, decreases in blood pressure, observed in humans (migraineurs and controls) following GTN infusion at the rate of 0.5 μg/kg per min were not evident in anaesthetized rats in this study. These data are consistent with a lower sensitivity of rats to exogenous NO from GTN. If these findings reflect a generally lower sensitivity of rats to NO, they might explain the failure of BW723C86 or GTN to evoke NO-mediated elevations in Fos-LI.
