Abstract
Background
Migraine research has highlighted the pivotal role of nitric oxide (NO) in migraine pathophysiology. Nitric oxide donors such as glyceryl trinitrate (GTN) induce migraine attacks in humans, whereas spontaneous migraine attacks can be aborted by inhibiting NO production. The present study aimed to investigate how GTN triggers migraine through its three nitric oxide synthase (NOS) isoforms (neuronal NOS (nNOS), endothelial NOS (eNOS) and inducible NOS (iNOS)) via a suspected feed-forward phenomenon.
Methods
Migraine-relevant hypersensitivity was induced by repeated injection of GTN in an
Results
Non-selective NOS inhibition blocked GTN-induced hypersensitivity. This response was partially associated with iNOS, and potentially nNOS and eNOS conjointly. Furthermore, we found that the GTN response was largely dependent on the generation of peroxynitrite and partly soluble guanylate cyclase.
Conclusions
Migraine-relevant hypersensitivity induced by GTN is mediated by a possible feed-forward phenomenon of NO driven mainly by iNOS but with contributions from other isoforms. The involvement of peroxynitrite adds to the notion that oxidative stress reactions are also involved.
This is a visual representation of the abstract.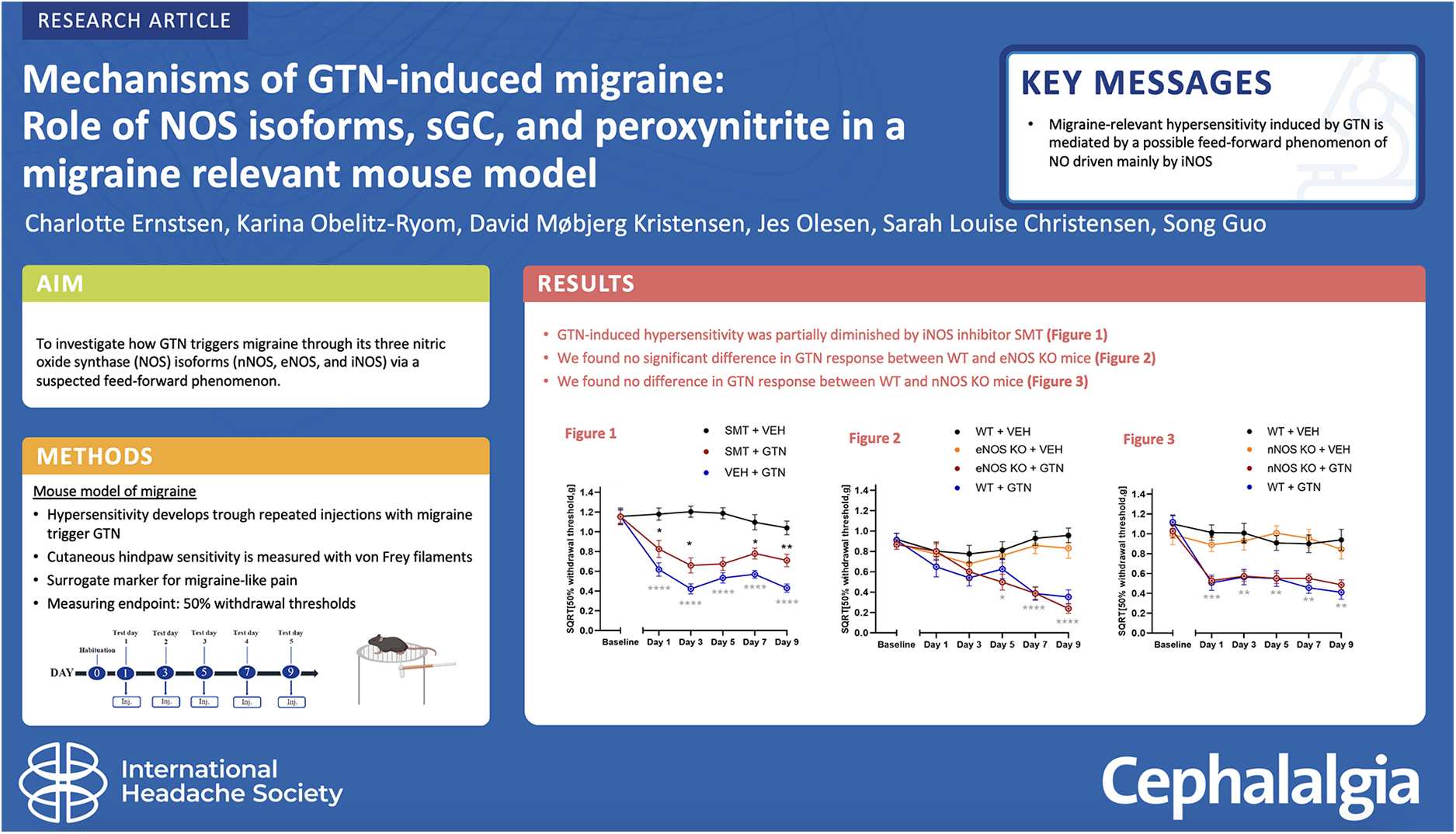
Introduction
In recent decades, there has been significant progress in our understanding of the signaling mechanisms of migraine, a condition that affects one billion people worldwide (1). These advances were achieved using human and animal provocation models (2). It was shown that nitric oxide (NO) is a crucial signaling molecule in migraine, as evidenced by the induction of migraine-like headaches in humans and animals using NO donors such as glyceryl trinitrate (GTN) and the therapeutic efficacy of inhibiting NO production in animals (3,4). Spontaneous migraine attacks in humans can be aborted by
In human provocation models, GTN infusion for 20 minutes caused an immediate headache during and shortly after infusion, as well as a delayed migraine with a peak after 5.5 hours that fulfilled the international diagnostic criteria for a migraine attack (3). This model was translated into a mouse model using either single or repeated injections of GTN. The model has been validated by its response to receptor-specific migraine drugs i.e., sumatriptan (5-HT1B/1D receptor agonists) and olcegepant (calcitonin gene-related peptide (CGRP) receptor antagonist) (4,10,11). These studies posed a crucial question: What happens in the hours long lag phase between immediate headache and delayed migraine? So far, this has not been clarified. NO mechanisms may also be active throughout spontaneous migraine attacks because they respond to treatment with
Methods
Experimental animals
We used a total of 216 wild-type (WT) C57Bl/6J BomTac mice (Taconic, Denmark) along with 96 transgenic knockout (KO) mice aged 7–12 weeks (20–29 g) with an equal distribution of both sexes. Mice not born in-house had a minimum acclimatization period of one week before the initiation of experiments. Age-related variations were not observed. Consistent with previously published protocols (13), mice received uniform conditions of care. They were housed in a room with controlled temperature and lighting (lights on at 7:00 am, 12 h light/dark photocycle) and had
Transgenic mice and genotyping
Adult nNOS KO mice (JAX strain 002986) for experiments were delivered from the Jackson Laboratory (Bar Harbor, ME, USA). eNOS KO and WT controls were bred in-house by mating of homozygous eNOS KO (JAX strain 002684) to WT C57Bl6/J BomTac. Subsequent breeding for homozygous eNOS KO and WT animals was continuously monitored through genotyping. The PCR protocol and primers used was based on a standard protocol from JAX (https://www.jax.org/Protocol?stockNumber = 002684&protocolID = 23416) (14).
Experimental design and protocols
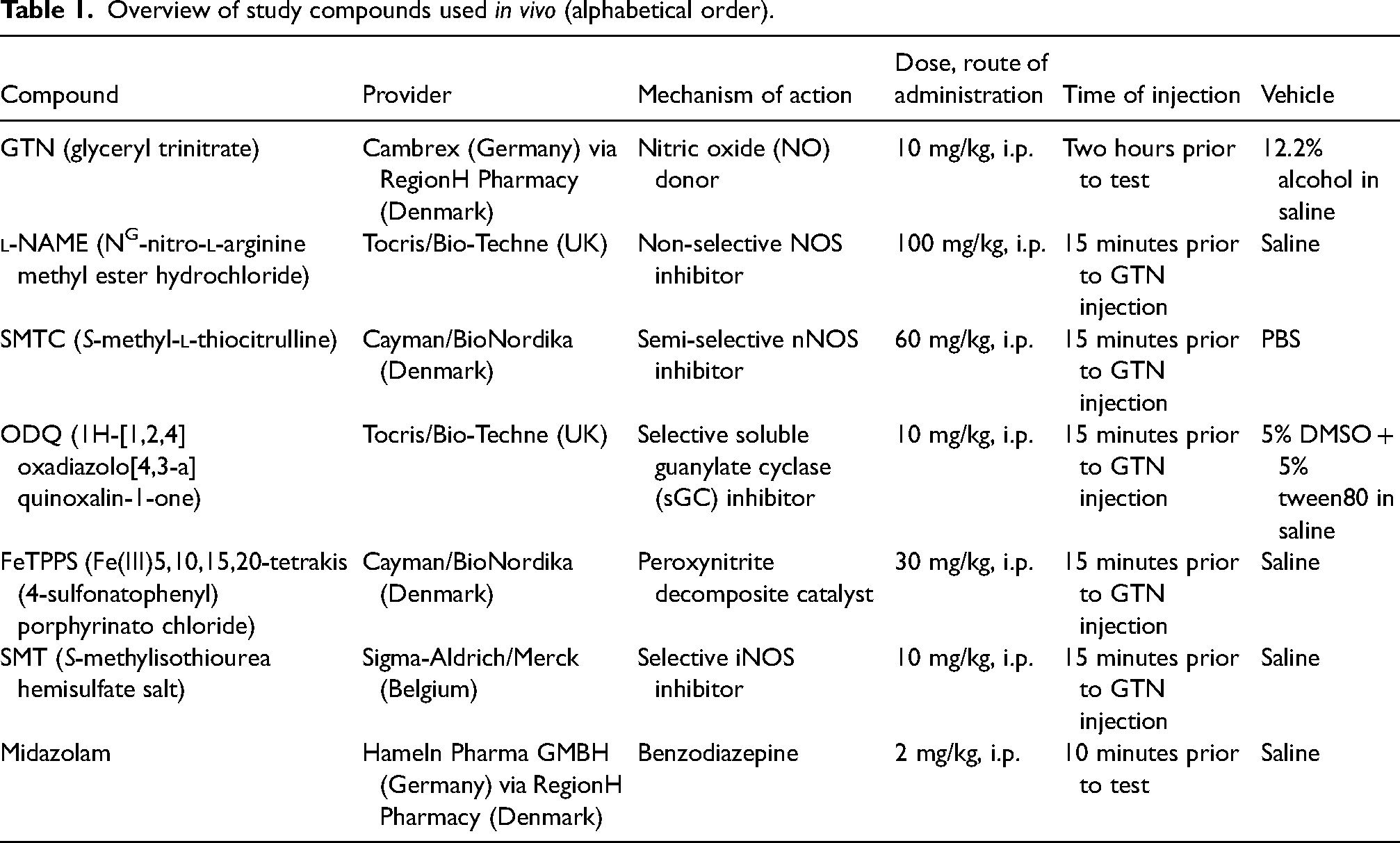
We employed a well-established mechanistic mouse model of migraine using GTN as migraine trigger (4,10) and assessed effects by quantifying cutaneous sensitivity, as described previously (4,15). This model uses GTN injection followed by assessment by von Frey filaments as a proxy for migraine-associated pain (4). Group sizes were twelve mice in all experiments, except for eNOS, where the group size was 14. Each experiment was conducted independently in separate cohorts of mice. The study protocol is outlined in Figure 1, whereas Table 1 summarizes the experiments and compounds administered. Mice of both sexes were tested every other day over a nine-day period, encompassing five test days. Inhibitory compounds were given as pre-treatment 15 minutes before GTN injection on each test day. Baseline cutaneous sensitivity was measured prior to injections on every test day (i.e. 48 hours after the last injection), and acute responses assessed two hours post-GTN injection. All assessments were conducted in low-light conditions (20–30 lux) between 8:00 am and 3:00 pm by an experimenter blinded to the experimental conditions.

Design and experimental timeline of test paradigm for glyceryl trinitrate (GTN) injection. Following 1 day of habituation (day 0), five tests were performed every other day over the course of nine days. All compounds were injected intraperitoneally (i.p.). Pre-treatment of NG
Overview of study compounds used
Behavioral tests
Cutaneous sensitivity to tactile stimulation
Cutaneous sensitivity was evaluated on the left plantar region of the hind paw starting at 0.16 g using von Frey filaments, with a range of 0.008–2.0 g (excluding 1.4 g; sourced from Ugo Basile, Gemonio, Italy). For our assessment, we employed the up-down method (16). The 50% withdrawal threshold in grams was calculated using a freely accessible online tool (https://bioapps.shinyapps.io/von_frey_app) (17) and subsequently square root transformed (SQRT) for improved normal distribution for statistical analyses. For the test, mice were placed in a transparent plexiglas chamber with a mesh net bottom (IITC Life Science, Woodland Hills, CA, USA). To familiarize them with the testing environment, mice were allowed to acclimate in these chambers for 45 minutes before the first test (day 0 and a 30-minute habituation period for subsequent tests (18).
Motor function (Rotarod)
We evaluated general motor function using a rotarod (Rotarods Advanced; IITC Life Science) to ensure that potential side effects of the study drugs, such as impaired motor function or sedation, did not bias the cutaneous sensitivity test with von Frey filaments. The rotarod assessment was conducted before the initial drug injection and immediately following the last von Frey test on test day 9. Each mouse was given a single attempt on the rotarod, starting from 0 rpm and gradually accelerating to 30 rpm within 45 seconds, with a maximum duration of 150 seconds. The duration spent on the rotarod was recorded. Midazolam (2 mg/kg, intraperitoneally (i.p.)) or saline (i.p.) was administered as a positive or negative control, respectively, 10 minutes prior to testing. Mice were randomized into treatment groups when receiving midazolam.
Test compounds
Compounds were administered i.p. in the lower right quadrant of the abdomen. They were diluted in saline (Fresenius Kabi, Germany) and injected at a volume of 10 ml/kg unless otherwise specified. Details regarding compounds, vehicles, concentrations and doses can be found in Table 1.
The NO donor GTN (7.89 mg/ml in 96% ethanol) was administered to mice at a dose of 10 mg/kg after dilution in saline to a concentration of 1 mg/ml (4,15). For vehicle treatment in the GTN model, an equivalent volume of ethanol was dissolved in saline.
Statistical analysis
Mice were randomized balanced according to 50% withdrawal thresholds measured at baseline, sex and home cage. Treatment groups and sex were equally divided throughout the test day (19). Group sizes were based on our previous work with these models (10,13,20) where 12 animals per group provided sufficient power to detect intermediate effects (18,21). Withdrawal thresholds of 50% were SQRT for improved normal distribution and data were analyzed using two-way repeated measures analysis of variance (ANOVA). A subsequent Tukey’s post-hoc test was performed comparing all groups on each of the five test days.
Results
NG- nitro-l -arginine methyl ester hydrochloride (l -NAME) blocks GTN-induced hypersensitivity
We tested whether

NG
nNOS may be involved in GTN-induced hypersensitivity
The effects of semi-selective nNOS inhibitor

Glyceryl trinitrate (GTN)-induced hypersensitivity was partially diminished by neuronal nitric oxide synthase (nNOS) inhibitor
To rule out speculative effects of eNOS inhibition by SMTC, we investigated the role of nNOS on GTN-induced hypersensitivity using genetically modified mice that lack the nNOS enzyme. In contrast to our experiment using the semi-selective chemical nNOS inhibitor (SMTC), nNOS KO mice showed no difference in GTN response compared to WT (
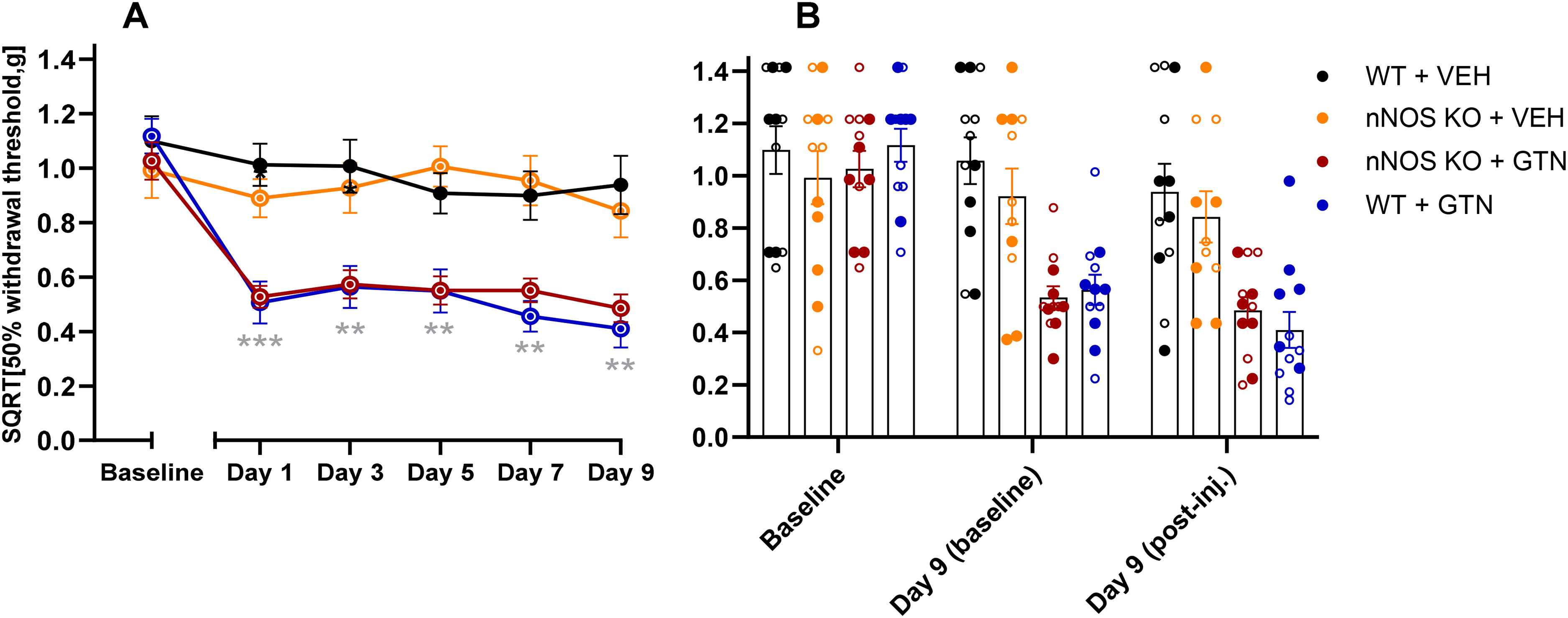
Glyceryl trinitrate (GTN)-induced hypersensitivity in both wild-type (WT) and neuronal nitric oxide synthase (nNOS) knockout (KO) mice, but we found no difference in GTN response between WT and nNOS KO mice. (A) Responses two hours after administration of GTN (10 mg/kg, i.p.) or vehicle (VEH) (i.p.) to nNOS KO mice (n = 12 per group) on five test days. A grey asterisk (*) indicates difference between positive and negative control groups (WT + GTN, blue vs. WT + VEH, black). Repeated measure two-way ANOVA with Tukey's post-hoc comparison between groups. (B) Descriptive representation of individual data points for baseline and day 9 of (A) with the mean ± SEM (○ = males, ● = females).
eNOS is not involved in GTN-induced hypersensitivity
We also investigated the role of eNOS on GTN-induced hypersensitivity using genetically modified mice that do not express the eNOS enzyme. eNOS KO mice showed no difference in GTN response compared to WT on all test days (
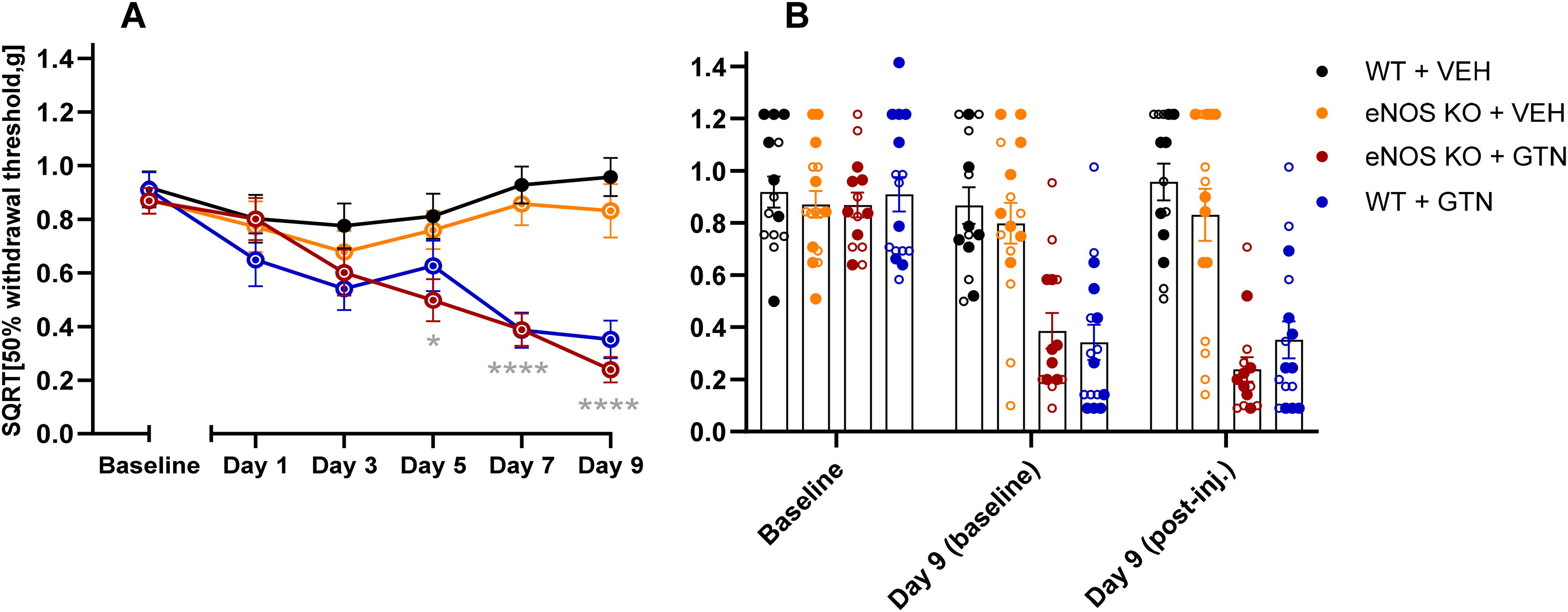
Glyceryl trinitrate (GTN) significantly induced hypersensitivity in both wild-type (WT) and endothelial nitric oxide synthase (eNOS) knockout (KO) mice on day 5, 7 and 9 (
iNOS is partially involved in GTN-induced hypersensitivity
The effects of selective iNOS inhibitor

Glyceryl trinitrate (GTN)-induced hypersensitivity is partially dependent on inducible nitric oxide synthase (iNOS). (A) Responses two hours after GTN (10 mg/kg, i.p.) or vehicle (VEH) injection (i.p.) in wild-type mice (n = 12 per group) on five test days.
GTN-induced hypersensitivity is partially dependent on sgc
The effects of sGC inhibitor 1H-[1,2,4]oxadiazolo[4,3-a]quinoxalin-1-one (ODQ) on GTN response was studied in WT mice. We found a significant difference between the test group (ODQ + GTN) and the positive control group (VEH + GTN) on test days 7 and 9 (Figure 7) (
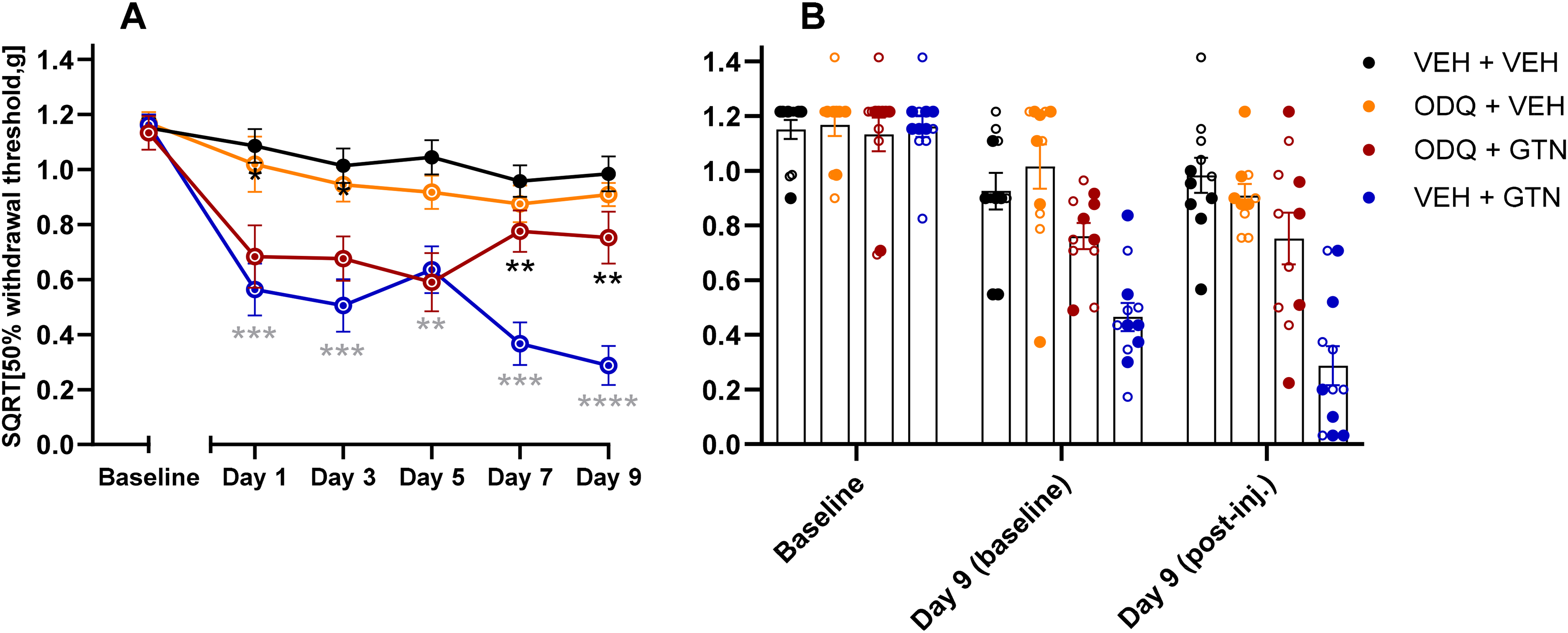
Soluble guanylate cyclase inhibitor 1H-[1,2,4]oxadiazolo[4,3-a]quinoxalin-1-one (ODQ) diminishes glyceryl trinitrate (GTN)-induced hypersensitivity on later test days. (A) Responses two hours after GTN (10 mg/kg, i.p.) or vehicle (VEH) injection (i.p.) in wild-type mice (n = 12 per group) on five test days. ODQ (10 mg/kg, i.p.) or VEH was given on day 0. A black asterisk (*) indicates difference between test group (ODQ + GTN, red) and positive control group (VEH + GTN, blue), whereas a grey asterisk (*) indicates difference between positive and negative control groups (VEH + GTN, blue vs. VEH + VEH, black). Repeated measure two-way ANOVA with Tukey's post-hoc comparison between groups. (B) Descriptive visualization of individual data points for baseline and day 9 of (A) with the mean ± SEM (○ = males, ● = females).
GTN-induced hypersensitivity is partly mediated via generation of peroxynitrite
The effects of Fe(III)5,10,15,20-tetrakis(4-sulfonatophenyl)porphyrinato chloride (FeTPPS), a peroxynitrite decomposite catalyst, on GTN response was studied in WT mice. We found a significant difference between the test group (FeTPPS + GTN) and the positive control group (VEH + GTN) on all test days (

Fe(III)5,10,15,20-tetrakis(4-sulfonatophenyl)porphyrinato chloride (FeTPPS), a peroxynitrite decomposite catalyst, diminishes glyceryl trinitrate (GTN)-induced hypersensitivity. (A) Responses two hours after GTN (10 mg/kg, i.p.) or vehicle (VEH) injection (i.p.) in wild-type mice (n = 12 per group) on five test days. FeTPPS (30 mg/kg, i.p.) or VEH was given on day 0. A black asterisk (*) indicates difference between test group (FeTPPS + GTN, red) and positive control group (VEH + GTN, blue), whereas a grey asterisk (*) indicates difference between positive and negative control groups (VEH + GTN, blue vs. VEH + VEH, black). Repeated measure two-way ANOVA with Tukey's post-hoc comparison between groups. (B) Descriptive visualization of individual data points for baseline and day 9 of (A) with the mean ± SEM (○ = males, ● = females).
Motor function was unaffected by all combinations of test compounds
To assure that the von Frey tests were not biased by impaired motor coordination, this was examined in mice before and after injection of study compounds using the Rotarod test. We found that motor function was unaffected in any of the KO mice or by any of the compounds used in the study. We found no differences between the negative control groups (vehicle) and the test groups (
No adverse effects were observed following treatment with GTN, SMTC, ODQ or FeTPPS in any of the experimental mice. All mice appeared healthy and had normal stools and stable weight throughout the experiment, as well as normal motor function.
Discussion
We investigated the possible feed-forward phenomenon of GTN-induced hypersensitivity in a mouse model of migraine. GTN-induced hypersensitivity was fully dependent on NOS enzymes but neither of the isoforms alone had potent inhibitory effects. Furthermore, both sGC and peroxynitrite generation contributed to the effect. Overall, our results demonstrate the importance of the NO feed-forward phenomenon and suggest that iNOS may be the most important mediator of this response. Downstream pathways involving sGC and peroxynitrite generation both appear to be important, suggesting that GTN-induced hypersensitivity in mice is mediated via multiple pathways rather than one. The GTN induced feed-forward phenomenon and the complex interaction of multiple pathways may explain the gap of several hours between GTN infusion and induction of migraine-like headache in humans.
Feed-forward phenomenon of NO
GTN can liberate NO in tissues with and without active NOS enzymes. Both nNOS and eNOS are constitutively active (22), whereas iNOS is not constitutively active but is induced by bacterial infection and pro-inflammatory cytokines (23). We suggest that an amplifying wave of iNOS and/or nNOS/eNOS induction (possibly uncoupled) may cause the delayed provoked migraine via subsequent high endogenous NO production leading to oxidative stress (Figure 9). In spontaneous migraine, non-selective NOS inhibition effectively aborted attacks (5). It has previously been suggested that GTN administration in rats may cause a delayed upregulation of iNOS mRNA level within dural meningeal macrophages after two hours, followed by a corresponding increase in protein expression after four to 10 hours (24).

Proposed nitric oxide (NO) feed-forward mechanisms based on present study and cited literature. Additionally, TRPA1 is closely linked to these pathways.
NO and NOS
Over the past two decades, inhibiting NO production through NOS inhibition has been investigated as a therapeutic strategy. Two proof of concept clinical trials showed that a non-selective NOS inhibitor was effective in acute migraine attacks and chronic tension-type headache (5,25). Several preclinical studies have also validated the efficacy of non-selective NOS inhibition in animal models of migraine (26). The non-selective NOS inhibitors have poor oral absorption, a short duration of action and cause significant side effects including increased blood pressure (27). Thus, the development of non-vascular NOS inhibitors may be a more promising strategy for migraine.
Based on present and previous preclinical studies, targeting iNOS may appear most effective because of its involvement in dural inflammation that may contribute to trigeminovascular activation (28). However, two phase II trials using a highly potent and selective iNOS inhibitor, GW274150, were negative for acute and preventive migraine treatment (29,30). Perhaps NOS isoform involvement is different in spontaneous vs. GTN induced migraine. On the other hand, iNOS inhibition as a therapeutic target may not need to be abandoned solely because of the lack of effect from a single chemical entity (GW274150). Targeting eNOS for migraine treatment has always been problematic because it seems unsafe as a result of its primary presence in vascular endothelium, essential for healthy cardiovascular function (31). Furthermore, our results do not support eNOS as a viable drug target because eNOS KO did not prevent hypersensitivity by GTN. Targeting nNOS is a promising option as a result of its expression in both the central and peripheral neuronal pain pathways of migraine (22,32,33). Two clinical trials using a combined nNOS inhibitor and 5HT-1B/1D receptor agonist, NXN-188, suggested that this drug may be effective in migraine treatment (34,35). Yet, a follow-up trial showed that NXN-188 did not have a substantial effect on migraine headache in adults with migraine with aura (36). This study was limited by a high drop-out rate in a relatively small sample. Thus, the effectiveness of NXN-188 in migraine remains uncertain. In summary, specific NOS targeting has had limited success, reinforcing our conclusion that NO acts via synergistic effects of the NOS isoforms to induce migraine. This is supported by a prior study investigating the effects of each NOS isoform on neurogenic and CGRP-induced meningeal vasodilation, suggesting a synergistic effect of these isoforms (37).
sGC
NO activates the sGC pathway that catalyzes the conversion of GTP to second messenger cGMP (31). The sGC pathway is a relevant signaling mechanism in migraine pathophysiology because the phosphodiesterase 5 inhibitor, sildenafil, induces migraine attacks in adults with migraine (7). Preclinical experiments using different pharmacological agents have likewise identified the sGC pathway as a key signaling mechanism regulating migraine pathophysiology (26). The phosphodiesterase 5 inhibitor sildenafil induced both acute and chronic hyperalgesia in mice (4) and a selective sGC stimulator, VL-102, induced cephalic and hindpaw hyperalgesia that was sensitive to both acute and preventive migraine treatments (38). Furthermore, the sGC inhibitor ODQ semi-effectively blocked migraine-associated acute and chronic hypersensitivity induced by GTN, even without exogenous NO stimulation. This suggest a potential link between endogenous NOS activity and chronic migraine maintenance (38) or the fact that GTN is acting directly on the sGC-cAMP pathway without NO. Despite the need for a ten-fold higher dose and less robust results, our findings partly reproduce the findings that ODQ inhibits GTN-induced hypersensitivity.
Peroxynitrite
NO can induce oxidative stress through cGMP-independent pathways, potentially contributing to migraine pathophysiology. One such pathway involves the formation of peroxynitrite, a downstream product of NO (39). Clinical studies have revealed disturbances in platelet levels of peroxynitrite-related proteins among migraine patients (40,41). Furthermore, inhibition of peroxynitrite production has been demonstrated to both prevent and terminate nociceptive responses, supporting its significance as a target for pain management (39,42). A recent preclinical study, focusing on peroxynitrite in a migraine model, demonstrated its involvement in modulating ongoing and evoked responses in dural-trigeminocervical neurons, as well as their response to CGRP (43). Another recent study showed that compounds modulating peroxynitrite alleviate the behavioral, neuronal, and molecular alterations induced by repeated stress and nitric oxide donors (44). The induction of oxidative stress pathways by GTN, is in line with previous studies in rats and mice showing that GTN-induced responses are dependent on TRPA1 channel activation (18,45,46). GTN and NO do not activate TRPA1 directly, but likely via the endogenous reactions leading to the formation of electrophilic agents like oxygen species or nitroxyl (47,48). Our findings strongly support that the peroxynitrite pathway is important in migraine. This opens a possible role for oxidative stress and mitochondrial function (44,49,50) in migraine susceptibility. Thus, peroxynitrite and other reactive oxygen species may be targets for migraine and migraine-like headache disorders.
Strengths and limitations
We used a well-validated mouse model to investigate signaling pathways of GTN (4,15,18). The compounds administered in our experiments had no impact on motor coordination, assuring the reliability of von Frey measurements.
We acknowledge some limitations of our study. Although GTN can release NO in tissues (51), it can also directly act on the sGC-cAMP pathway without involving NO (52). Therefore, it is possible that GTN may not be a strong NO liberator, and its effect could be through direct activation of sGC. The modest effect of blocking sGC seen in this study suggests a more important role of NO liberation in this particular model. However, other more genuine NO donors might function differently from GTN and more accurately mimic NO generation through the three NOS isoforms.
Our contrasting results with pharmacological nNOS inhibition and nNOS KO may be a result of compensatory mechanisms in nNOS KO mice. It is known that in some genetically modified mice, backup and compensatory mechanisms can maintain functions (53). Ideally, we should have investigated all three NOS both chemical inhibitors and KO for better comparison. This was not possible because we were not able to acquire a selective chemical inhibitor for eNOS or a KO mouse strain for iNOS. Also, we did not explore the combination of chemical inhibitors and KO mice, which would be interesting as future experiments.
Regarding eNOS, we cannot say with full certainty that it does not have a role in GTN-induced hypersensitivity. The given dose of the semi-selective nNOS inhibitor SMTC was based on literature, where 10–200 mg/kg has been shown to be effective in various mouse models (54,55). SMTC is a potent NOS inhibitor with selectivity toward nNOS compared to eNOS and iNOS with
We used plantar tactile sensitivity as primary outcome, although some suggest periorbital sensitivity is more relevant (18). Both anatomical areas show increased mechanical sensitivity to various migraine-inducing compounds (4,18,38,57). To our knowledge, there have been no studies demonstrating exclusive hypersensitivity in the cephalic region of mice without affecting the plantar area. Migraine-specific drugs without general analgesic effects (58,59). In our experience, periorbital sensitivity is more variable and has a narrower effect window (60,61), necessitating larger experimental groups thus reducing feasibility (18,38). Likewise, other potential endpoints such as light sensitivity or grimacing, also require larger mouse cohorts because of increased variability (62).
Conclusions
The induction of migraine-relevant hypersensitivity by GTN involves a NOS mediated feed-forward phenomenon in which subsequent high NO-levels are maintained and engaged in both cGMP-dependent and cGMP-independent pathways via sGC and peroxynitrite generation. Our results suggest that inhibition of one NOS enzyme is not a likely migraine drug target, whereas antagonizing soluble guanylyl cyclase or peroxynitrite may be more promising.
Migraine-relevant hypersensitivity induced by GTN is mediated by a feed-forward phenomenon of NO driven mainly by iNOS but with contributions from other isoforms. Peroxynitrite and oxidative stress reactions are also involved in migraine-relevant hypersensitivity induced by GTN. Our results suggest that inhibition of one NOS enzyme is not a likely migraine drug target, whereas antagonizing soluble guanylyl cyclase or peroxynitrite may be more promising.
Footnotes
Author contributions
CE, JO, SLC, DMK and SG contributed to the conception, design and planning of the experiments. SG, CE, KOR and SLC contributed to data acquisition by performing the
Data availability
The datasets used or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Song Guo is supported by the BRIDGE – Translational Excellence Programme (bridge.ku.dk) at the Faculty of Health and Medical Sciences, University of Copenhagen, funded by the Novo Nordisk Foundation. Grant agreement no. NNF20SA0064340 (2021 fellows). Also, this work was supported by Candys Foundation, Migrænefonden, Torben og Alice Frimodts Fond and Ragna Rask-Nielsens Grundforskningsfond.
Ethical statement
Ethical approval was obtained from the Danish Animal Experiments Inspectorate.
