Abstract
Introduction
Research in development of new migraine therapeutics is hindered by the lack of suitable, predictive animal models. Cilostazol provokes headache in healthy humans and migraineurs by increasing intracellular cAMP levels. We aimed to investigate whether cilostazol could provoke headache-like behaviours and c-fos expression in rats. In order to evaluate the predictive validity of the model, we examined the response to the migraine specific drug sumatriptan.
Methods
The effect of cilostazol (125 mg/kg p.o.) in female Sprague Dawley rats was evaluated on a range of spontaneous behavioural parameters, light sensitivity and mechanical sensitivity thresholds. We also measured c-fos expression in the trigeminal nucleus caudalis.
Results
Cilostazol increased light sensitivity and grooming behaviour. These manifestations were not inhibited by sumatriptan. Cilostazol also induced c-fos expression in the trigeminal nucleus caudalis. Furthermore, trigeminal – but not hind paw hyperalgesia was observed.
Conclusion
The altered behaviours are suggestive of cilostazol induced headache with migraine-like features, but not specific. The presence of head specific hyperalgesia and the c-fos response in the trigeminal nucleus caudalis imply that the model involves trigeminal nociception. The model will be useful for studying mechanisms related to the cAMP pathway in headache, but its predictive properties appear to be more limited due to the lack of response to sumatriptan.
Introduction
The development of novel anti-migraine drugs is hampered by a lack of suitable animal models. Currently, we are working towards developing a rodent migraine model that is chemically induced, suitable for multiple testing of the same animals, predictive in terms of response to known migraine therapeutics and is established in female subjects in order to reflect the fluctuating hormone levels possibly involved in migraine (1).
Cilostazol is a phosphodiesterase 3 (PDE3) inhibitor. PDE3 is quantitatively one of the most important enzymes responsible for degrading cAMP in endothelial and vascular smooth muscle cells of cerebral arteries (2), and hence cilostazol causes increased intracellular cAMP concentrations (3). PDE3 is also present in the rat trigeminal ganglion, where it is co-localised with calcitonin gene-related peptide (CGRP) in 20–30 % of neurons (4). Moreover, functional inhibition of PDE3 activity by cilostazol in cell lysates of the rat trigeminal ganglion has also been confirmed (4). PDE3 mRNA and/or protein have been shown in the rat middle cerebral artery, basilar artery, dura mater and trigeminal ganglion (5). Cilostazol dilates cerebral arteries, but sensitisation of the trigeminovascular system has also been proposed to play a role in its headache inducing properties (6).
In healthy people, cilostazol has been reported to induce headache in 11 out of 12 and 28 out of 30 persons in two independent studies (6,7). A minority of these headaches fulfilled the International Headache Society Criteria for migraine without aura, but frequently it had migraine-like features such as pulsating pain and aggravation by physical activity (6,7) However, the headache was not reversed by sumatriptan treatment (7). In migraine patients, cilostazol triggered a migraine-like attack in 12 out of 14 patients (8). Importantly, cilostazol induces only head pain and no other pain in humans. This is similar to the NO-donor glyceryl trinitrate (GTN), which is commonly used as a headache trigger in experimental migraine models. However, in contrast to the cAMP activation mediated by cilostazol, GTN acts through the cGMP pathway (9).
Cilostazol is one of the most potent headache inducers in healthy human subjects (6,7) and we hypothesised that oral administration of cilostazol could also induce a headache-like state in the rat, possibly with migraine-like features. To test our hypothesis, we studied the effect of cilostazol on blood pressure and pulse frequency, light aversion, several spontaneous behavioural parameters, mechanical sensitivity and c-fos expression in the trigeminal nucleus caudalis (TNC). Additionally, we investigated whether some of the effects could be reversed by the anti-migraine agent sumatriptan.
Material and methods
Animals
Number and distribution of rats used to complete the study.
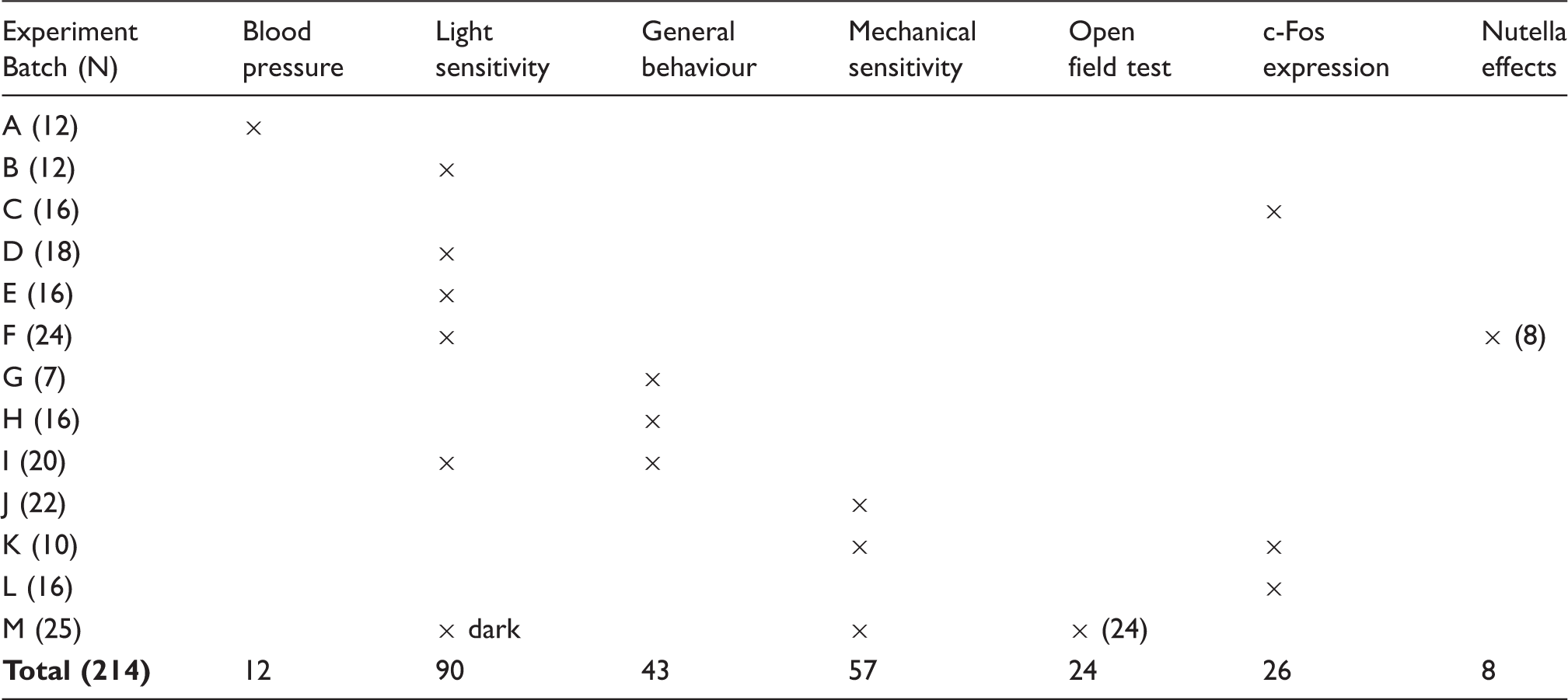
For the light sensitivity and general behavioural tests, randomisation was done with respect to having a balanced cross-over design. For the experiments involving simple comparison between groups, the rats were randomly allocated to treatment or vehicle, but stratified at home cage level so that all cages had an even distribution of treatment and control rats.
Drug administration
Cilostazol was prepared from pletal tablets (100 mg, Otsuka Pharmaceutical Europe, Ltd) by crushing the tablets to a fine powder. The drug was mixed in approximately 0.5 to 1.0 g of Nutella® nut paste for voluntary ingestion by the rat (10). Vehicle was the nut paste alone. The reported pharmacokinetics of cilostazol in female Sprague Dawley rats mimic those of humans, with a tmax of four hours and t½ of 3.7 hours (oral dose of 10 mg/kg) (11). The bioavailabily of 1 mg/kg p.o. was estimated at 37 %, but a dose-dependent increase in bioavailability was also reported (12). Prior to administration, the rats were habituated to eating Nutella. To address whether Nutella itself would change the behaviour of the rats, we measured the effect of the vehicle on general behaviours (see supplementary material). Sumatriptan succinate (Neuraxon, Inc/Sigma Aldrich, USA) was diluted in saline and administered subcutaneously, 2 mg/kg. GTN 5 mg/ml (Hospira, UK) was diluted 1:4 in saline and administered i.p. at 1 mg/kg.
Blood pressure measurements in awake rats
Twelve rats underwent surgery to enable placement of a permanently indwelling catheter, which was inserted inside the femoral artery and advanced to the abdominal aorta (see supplementary material for details). Two rats had to be euthanised due to adverse events. We continuously studied the effects of cilostazol on mean arterial blood pressure (MABP) and pulse frequency (PF) in order to establish a dosing regimen for further studies. The effect of vehicle, 25, 125 and 250 mg/kg cilostazol were studied for 3–7 hours in 10 rats. The same animal received different doses on separate days, adding up to a total of five measurements for each dose.
Light sensitivity
Overview of the five experiments measuring light sensitivity.
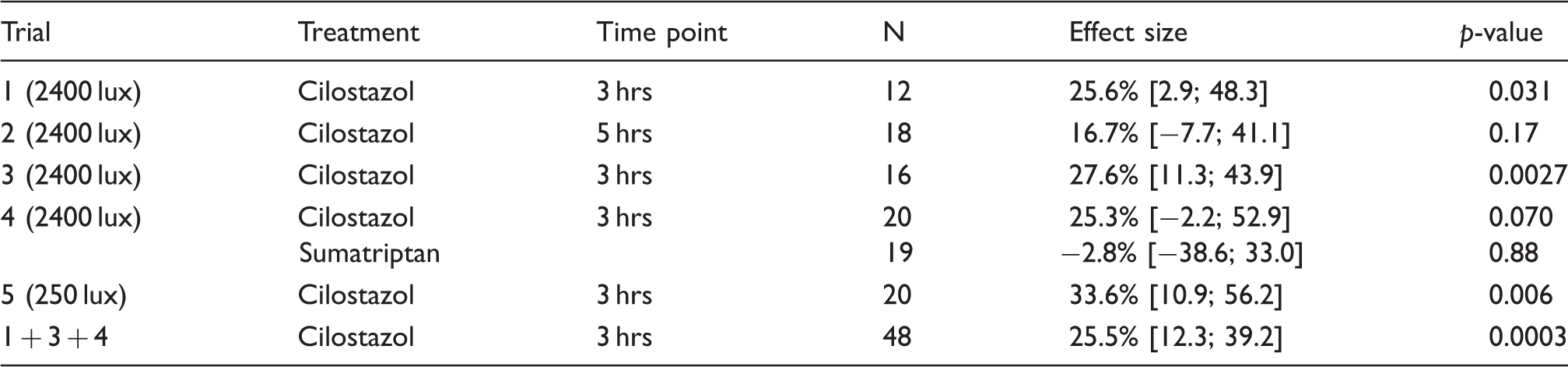
To verify that the light/dark box detected photophobia and not general anxiety per se, we performed the test in the presence of dim red light only, and compared the time spent in the white compartment of the box between a control group and cilostazol treated group, n = 12 and 13, respectively. On another test day, the same rats were also tested in an open field arena, which is a traditional way to measure anxiety (14). The arena consisted of a grey plastic box with the dimensions (L/W/H) 77/57/42 cm, marked with a central zone of 30 × 50 cm. The test was performed in 5 lux illumination. Rats were tested for five minutes and filmed with no observer present in the test room.
General spontaneous behaviours
We used the LABORAS (Metris, Netherlands) setup for automatic registration of rodent behaviour. Simply, the system consists of individual solid floor cages (with the floor covered with bedding material) placed on platforms sensitive to vibrations caused by the animal. Different behaviours (locomotion, immobility, rearing, grooming, eating and drinking) are picked up by their specific vibration patterns (15,16). We hypothesised that locomotion and rearing behaviours could be utilised as markers of “routine physical activities” that are usually decreased during migraine attacks (17). Immobility and grooming were expected to increase after cilostazol treatment. Notably, rats have been shown to increase their grooming behaviour towards sites of trigeminal nociceptive hypersensitivity (18,19). Eating and drinking could be possible markers of nausea also typical for migraine, and these behaviours were hypothesised to decrease (17).
The protocol used was as follows: Rats were habituated to the test cage for six hours on day 1, and then tested on days 3, 5 and 8 in a randomised three-way cross-over design with cilostazol, vehicle and cilostazol + sumatriptan treatment, balanced to have equal distributions in treatments on the different test days. Cilostazol and sumatriptan were given at time 0. The room was left dark, and behaviour analysed in the interval 2–4 hours post dosing. In the initial experiment, 23 rats were studied and all outcome parameters were tested to define what would be relevant to study in the follow up experiment. The follow up experiment included 20 rats.
Mechanical sensitivity
Mechanical sensitivity is a typical outcome parameter used in rodent headache research. Several models show decreased mechanical sensitivity thresholds on the plantar surface of the hind paw (20–22) and/or in the trigeminal dermatomes (23–25). We tested the effect of cilostazol on hind paw withdrawal thresholds using manual von Frey filaments and an automatic Randall Selitto paw pressure device (IITC LifeScience Inc, USA), which has also been used and validated by other laboratories (26–28). For the frontal region (V1 trigeminal dermatome (29)) of the head, mechanical thresholds were measured using an electronic von Frey anesthesiometer fitted with a rigid tip (IITC LifeScience Inc, USA). To our knowledge, this has not previously been applied to the head, but has been validated with respect to the hind paw (28). Rats were tested at baseline, three and six hours after the administration of cilostazol. The three-hour time point is around Tmax and the six-hour time point was chosen to test for delayed onset hypersensitivity. To test the frontal region, the rat was gently restrained in a towel and held in the lap of the experimenter. The tip of the anesthesiometer was applied with increasing force (max 250 g.) to the right and left side above the eye towards the midline until the rat withdrew its head or vocalised. The soft tissue around the eye was carefully avoided. The average of three measurements was considered the threshold. Care was taken not to apply the tip to the exact same location. Five minutes after the facial test, the automatic paw pressure device was applied to the hind paw. Here, the rat was held firmly in the hand and lower arm of the experimenter and the left hind leg extended. The probe was placed on the paw between the foot pads, and increasing pressure was applied (max 450 g) until the rat withdrew the paw or vocalised. Again, an average of three measurements was recorded. We tested the same 25 rats in the frontal region and hind paw using the automatic device. For manual von Frey testing, another set of rats was placed in a clear cage with grid floor (L: 32 cm, W: 17.5 cm, H: 20 cm). The 50 % mechanical sensitivity threshold was assessed by applying von Frey filaments (Aesthesio, Ugo Basile) according to the up-down method (30,31). Rats that did not respond to the 15 g filament were assigned this value. Two groups of 16 rats were tested with this method. As a positive control for all three methods, GTN 1 mg/kg was administered i.p. All testing was performed with the experimenter blinded to treatment.
C-fos expression in the trigeminal nucleus caudalis
In order to confirm the presence of trigeminal activation, we studied c-fos expression in the TNC (32,33). Five hours after cilostazol or vehicle treatment, 26 rats were deeply anaesthetised with pentobarbital (90 mg/kg i.p.) and transcardially perfused with 250 mL PBS, pH 7.4 at room temperature followed by 300 mL cool (approximately 10° C) 4% PFA (AppliChem PanReac, A3697). Brain tissues were collected and post fixed in 4% PFA for 3–14 hours. The rostral border of the brain stem was cut approximately 2 mm rostral to obex and the caudal end defined so that the piece of brain stem was 1 cm long. Upon dehydration in a sucrose gradient, the samples were cut in 40 µm sections on a cryostat (Leica 3050 S) and every seventh section collected to represent one sample. Generally, tissue processing and immunohistochemical staining followed the protocol described in (34), but the primary antibody used was c-Fos-Rb (Merch/Millipore, ABE457) in a concentration of 1:8000. C-fos positive cells were counted in laminae I and II of the trigeminal nucleus caudalis under the 20 × magnification lens (Nikon Eclipse Ni) by an observer blinded to treatment. Sections were anatomically subdivided in three distinct areas: Vi/Vc [0.8 – obex], Vc [obex – spinal cord] and C1–C3 [spinal cord] (35). In each rat, c-fos expression is reported as the average number of positive cells per section within the given area. Cilostazol and vehicle treatment were compared overall, and within each anatomical subdivision.
Oestrous cycle determination
A sample for oestrous cycle determination was collected by vaginal lavage after behavioural testing or just before perfusion for tissue collection. We adapted the method described by Marcondes et al. (36). The samples were evaluated under a light microscope at 10 × magnification (Nikon Eclipse Ni) and categorised into three categories: met/dioestrous, proestrous and oestrous, as previously described (13). Primary readouts were controlled for a possible effect of the oestrous cycle.
Statistics
Light sensitivity and spontaneous behavioural data were analysed using the mixed model procedure in SAS enterprise guide 7.1. A repeated measure model was built with the primary readout as the dependent variable. Independent variables were treatment, but also oestrous cycle, batch of rat and test number (test 1, 2, or 3). The models were checked for possible interaction between treatment and oestrous cycle. Locomotion and drinking data were log10 transformed as the models then had improved Gaussian distribution of the residuals. SAS syntaxes are provided as supplementary material.
A repeated measure two-way ANOVA with Sidak’s correction for multiple testing (GraphPad Prism 6) was used to analyse mechanical sensitivity data. C-fos expression of the vehicle and treatment group was compared using a two-way ANOVA (GraphPad Prism 6) with Fisher’s LSD post hoc test. Data are reported as mean difference between treatments in percent or real values with 95 % confidence intervals, and evaluated at 95 % significance level.
Results
Effect of Nutella on general behavioural parameters
We found no effect of Nutella (vehicle) treatment on locomotion, immobility, rearing, grooming, eating or drinking activity, see supplementary material and supplementary Figure 1(a–f).
Effect of Cilostazol on blood pressure and pulse frequency
At 250 mg/kg, cilostazol caused a clear decrease in blood pressure (up to 17%). The effect on blood pressure was less marked at the lower doses (Figure 1(a)). For all three doses (25 mg/kg, 125 mg/kg and 250 mg/kg), we observed a compensatory increase in pulse frequency (Figure 1(b)). The peak effect on the cardiovascular system appeared around three hours after administration of the drug. This is in agreement with the reported pharmacokinetics (Tmax) in female rats (11,12) Based on these findings, we decided to continue our study with the 125 mg/kg dose. This is a high dose (approximately 45 times the human dose), but cardiovascular responses to this dose are not pronounced in rats.
(a) Mean arterial blood pressure (MABP) visualised as mean change from baseline in percent (±SE) after treatment with cilostazol. Time 0 is baseline calculated as mean pressure throughout the last 30 minutes before administration of cilostazol; (b) pulse frequency (PF) measured simultaneously as MABP. For clarity, error bars are not shown. Ten rats were used to obtain the data. Individual rats received several doses adding to a group size of five non-independent observations.
Light sensitivity
Three hours after cilostazol treatment, the time the rats spent in 2400 lux light decreased by 26 % [12; 39], p < 0.001, N = 48 compared to vehicle treatment. At 250 lux light, the animals decreased their time in light by 34 % [11; 56], p = 0.006, N = 20. Five hours after cilostazol treatment, the effect of cilostazol was less pronounced, 17 % [−8; 41] and not significant, p > 0.1, N = 18. There was no effect of sumatriptan treatment. Figure 2(a–f) illustrates the data obtained in the experiments, and Table 2 summarises the results in terms of effect sizes and p-values.
Data from trial 1-5 in the light sensitivity experiments and the control experiment performed in red light. Time spent in light is shown in seconds (±SE) and the total test duration was 600 seconds. In trials 1, 3, 4 and 5, rats were tested three hours after dosing and in trial 2, rats were tested five hours after dosing. Trial d also shows the effect of sumatriptan after cilostazol treatment. In e, the light intensity was reduced to 250 lux. *: p ≤ 0.05, **: p ≤ 0.01, repeated measure, mixed model including test number and oestrous cycle as covariates. Number of animals is reported in Table 2. (a) Trial 1: at 3 hrs, 2400 lx*,(b) Trial 2: at 5 hrs, 2400 lx, (c) Trial 3: at 3 hrs, 2400 lx**, (d) Trial 4: at 3 hrs, 2400 lx, (e) Trial 5: at 3 hrs, 250 lx**, (f) Control experiment: at 3 hrs, red light.
The mixed model found statistical significance of the test day (a reduction in time spent in light from test 1 to test 2) and batch of rats (differences in baseline light sensitivity) when all 2400 lux data were pooled. It is important to incorporate these variables in the analysis and to balance treatments on the test days to avoid confounding bias. The data for oestrous cycle did not indicate an effect of the cycle nor interaction with treatment, but data were insufficient (too few observations in oestrous and proestrous) to draw definite conclusions. In experiment 4, four out of 24 rats had to be excluded prior to testing, as they failed to enter the light zone of the box in the habituation trial.
When the light/dark assay was run in darkness (red light), we found no difference in the time spent in the white zone of the box between vehicle and cilostazol treated rats. The control group spent 271 s [±13.4] in the white part of the box, and the cilostazol treated group spent 311 s [±17.0], p = 0.08. In the open field test, we used the outcome “time spent in the central zone of the box”. Here, the value for the control rats was 37.1 s [±5.8] and for the cilostazol treated group 36.1 s [±5.5], p = 0.9.
Effect of cilostazol on general behaviour
In the initial study, we found that grooming was increased by 23 %, 365 seconds [98; 633], p = 0.009 and rearing behaviour was reduced by 22 %, 133 seconds [25; 241], p = 0.002 by cilostazol treatment. None of the other behaviours tested were altered. In the follow up study, we confirmed that grooming behaviour was increased after cilostazol treatment by 36 %, 432 seconds [184–681], p = 0.001. Sumatriptan did not inhibit the increased grooming behaviour, p = 0.07 in the initial experiment nor in the follow up study, P > 0.1. In these experiments the test day, stage of oestrous cycle and batch of rats did not significantly influence the model. Data are shown in Figure 3(a–h).
Data on general behaviour measured in LABORAS over three different test days where the rats received vehicle, cilostazol or cilostazol + sumatriptan in a balanced setup. Data are shown in seconds (±SE). The time spent doing each behaviour was analysed in the period 2–4 hours post treatment. In the initial study (a-f) 23 rats received vehicle and cilostazol treatments, but only 15 also got sumatriptan. In the follow up study (g-h), including 20 rats, only grooming and rearing behaviour were analysed. *: p ≤ 0.05, **: p ≤ 0.01, repeated measure, mixed model including test number and oestrous cycle as covariates. (a) Locomotion, (b) Immobility, (c) Grooming**, (d) Rearing**, (e) Eating, (f) Drinking, (g) Grooming**, (h) rearing.
Mechanical sensitivity thresholds
We found that rats displayed increased sensitivity of the frontal region at six, but not three hours after administration of cilostazol. In the cilostazol treatment group, the threshold changed from 175.9 g ± 9.3 at baseline to 148.4 g ± 10.3 at the six hour time point, whereas the vehicle group remained stable from 170.9 g ± 12.0 at baseline to 171.3 g ± 10.3 six hours later, see Figure 4(a). The difference in the cilostazol group was significant with a p-value of 0.004, repeated measure two-way ANOVA, but there was not a significant difference between the cilostazol treatment and vehicle group, p > 0.1. We found no changes in hind paw mechanical sensitivity thresholds either at three or six hours after cilostazol treatment, see Figure 4(b–c).
Data on mechanical sensitivity thresholds measured with an electronic von Frey anesthesiometer in the frontal region of the head (panel a), an automatic Randall Selitto paw pressure device applied to the hind paw (panel b) and manual von Frey filaments applied to the hind paw (panel c). The top row shows the response to cilostazol and the bottom row is the positive control for the matching measurements. Animals were tested at three and six hours after dosing. At six hours, cilostazol induced hyperalgesia is found in the frontal area, but not in the hind paws. *: p ≤ 0.05, **: p ≤ 0.01, two-way repeated measure ANOVA with Sidak’s multiple comparison test.
C-fos expression in the trigeminal nucleus caudalis
Overall, cilostazol significantly increased c-fos expression in the TNC, p = 0.015, see Figures 5 and 6. The difference in average number of c-fos positive cells between the two groups was 7.9 [3.4; 12.5], p < 0.001 in the Vi/Vc transition zone, 4.6 [0.1; 9.1], p = 0.04 in Vc and 1.6 [−2.9; 6.1], p > 0.1 at the level of C1–C3. On average, 31 (range 25–38) sections were counted per rat. Based on the observations from all rats, there was no indication that the oestrous cycle influenced the c-fos response to cilostazol.
Representative photo of the c-fos distribution. Panel 1 is from the interpolaris/caudalis transition zone and shows vehicle (top row) and cilostazol (bottom row) at 4, 20 and 40 × magnification. Panel 2 is from the main body of the trigeminal nucleus caudalis, vehicle (top row) and cilostazol (bottom row) and panel 3 is representing the spinal cord sections C1–C3 again, vehicle (top row) and cilostazol (bottom row). The scale bar is 250 um at 4 × , 100 µm at 20 × and 50 µm at 40 × . Average numbers of c-Fos positive cells per section in laminae I and II of the interpolaris/caudalis transition zone and throughout the length of the trigeminal nucleus caudalis. The animals were sacrificed at five hours after cilostazol treatment. n = 12–13. *: p ≤ 0.05, ***: p ≤ 0.001, two-way ANOVA with Fisher’s LSD post-hoc test.

Discussion
The major results of the present study were that oral cilostazol caused both migraine relevant behavioural changes and c-fos expression in the trigeminal nucleus caudalis. The model will be useful in studies of the cAMP pathway and its role in headache and migraine-related pathophysiology. This is in contrast to the widely used nitroglycerin model (20,33,37), which employs the cGMP pathway. The model that we have presented avoids surgery, injections and oral gavage and, based on the experience from human experimental and clinical use of cilostazol, it most likely does not cause chronic changes to the rats. This allows the experimenter to perform cross-over studies or to do multiple tests with the same rats (40). No animal model perfectly recapitulates all aspects of a complex human disorder. A major limitation of the cilostazol model is the lack of response to sumatriptan, a medication that has been effective for treatment of migraine in humans and also reverses the changes seen in several animal models of migraine (20,22,40). However, sumatriptan also fails to alleviate cilostazol-induced headache in healthy humans (7). Therefore, it may be that cilostazol induces headache via a mechanism insensitive to treatment with sumatriptan. It will be interesting to study the effect of CGRP-antagonists and other classes of anti-migraine drugs in this model.
This is the first study describing a method for voluntary administration of a potential headache inducer served orally mixed in Nutella. We tested the acute effect of the vehicle and found no changes in general behaviour. Because migraine is more prevalent in women, the study was performed in female rats only. Additionally, the pharmacokinetics of cilostazol differ between male and female rats (12), and therefore direct comparison of the two sexes would be possible only after further pharmacokinetic studies.
Dose selections
We performed an initial dose titration study with cilostazol, wherein we assessed the effects on cardiovascular performance. The dose of cilostazol selected, 125 mg/kg, induced a minor decrease in blood pressure of 0–10 %. The larger dose caused a more pronounced decrease and hence was not suitable. Notably, we observed a compensatory increase in pulse frequency at the selected and even the lower dose. The results are in accordance with another study reporting on pharmacokinetics of cilostazol in rats (41). It also compares well with the vascular effects of the dose used in human headache provocation studies (2).
The dose of sumatriptan used here was based on a dose range of 0.6–1 mg/kg i.p. used in other studies (20,22,25). However, we increased the dose because of the short half-life of sumatriptan (42) and long duration required for onset of cilostazol effects on behavioural endpoints. The timing of sumatriptan administration differed between the light sensitivity assay and the general behaviour study. In the former, sumatriptan was given two hours after cilostazol, allowing one hour for the sumatriptan to be effective before testing the rats. In the general behavioural assay cilostazol and sumatriptan were given simultaneously, as it is not possible to disturb the animals once they are in the test cages.
Light sensitivity
Photophobia is a common symptom during migraine attacks (17). As a marker of photophobia, increased light aversive behaviour has previously been described in mice after administration of CGRP (43–46). Photophobic behaviour also increased in the rat after application of inflammatory soup to the dura with prior sensitisation via bisphenol A (47). In healthy human subjects, the headache induced by cilostazol was not accompanied by photophobia (6). In migraineurs, photophobia was present in four out of 14 patients, but was not significantly different from placebo (8). In the present study, cilostazol increased the light sensitivity of the rats. This might be explained by a difference in cilostazol plasma or brain concentration, as we used a dose 45 times higher than in the human studies. We observed a rather large inter-animal and inter-batch variation in the baseline light sensitivity. This stresses the importance of the rats being their own controls. Given that cilostazol induces head pain but no other pain in humans, we suggest that the photophobia seen in the rats is due to the involvement of migraine-relevant pathways. However, it can be argued that light aversion is increased in rats during all cases of unpleasant sensation and therefore our finding is not migraine-specific.
We tested the light sensitivity under both high (2400 lux) and low (250 lux) intensities. The lower intensity did not successfully increase the baseline time spent in the light zone, and both setups were effective in the detection of cilostazol-induced light aversion. Therefore, we recommend the lower light intensity for future studies.
Importantly, control experiments were performed to verify that cilostazol does in fact induce photophobia and not general anxiety per se.
General behaviour
Cilostazol did not have a major impact on the general behaviour of the rats. Their locomotion activity, eating and drinking behaviour were unaltered. Rearing activity decreased in the first experiment, but this could not be repeated in the subsequent experiment. Only grooming behaviour was altered in both the initial and the follow up study. Rats are known to direct their grooming pattern towards a site of pain or irritation (48,49) and several studies report increased face grooming in various models measuring headache-like and facial pain-like behaviours (18,19,50–52). We did not measure face grooming specifically compared to general grooming, as it was not possible in the setup used.
Mechanical sensitivity thresholds
Our data clearly show cilostazol-induced mechanical hyperalgesia in the V1 trigeminal dermatome (frontal region), but not in the hind paw. The hyperalgesia was late and occurred six hours after administration, which is two to three hours later than the peak plasma concentration. We tested the hind paw sensitivity both with manual von Frey filaments and an automatic Randall Sellito paw pressure device. It is suggested that the two methods detect different kinds of mechanical sensitivity, as the von Frey filaments measure cutaneous sensitivity, whereas the Randall Sellito measures deeper tissue sensitivity (27).
Here, we present a model that actually shows head specificity in terms of hyperalgesia in the frontal region, but not in the hind paw. Similar findings have been reported for a spontaneously fluctuating allodynic rat model of migraine-like pain (25). Repeated sumatriptan treatment, used as a model of medication overuse headache, induces both trigeminal and hind paw hyperalgesia (24), as does the application of inflammatory soup to the dura (39). To the best of our knowledge, migraine relevant models using nitroglycerin in “normal” rodents only investigated hind paw or tail hyperalgesia or thermal sensitivity (20–22,37), without paying attention to the trigeminal area. However, in one study performed on spontaneous allodynic rats, trigeminal hyperalgesia was found in response to nitroglycerin (25). In terms of face validity, a rat model displaying trigeminal, but not peripheral hyperalgesia has more validity than the one displaying both or only peripheral hypersensitivity, as this mimics the human model better (1). Though not a part of the diagnostic criteria for migraine (17), patient-reported cephalic allodynia during migraine attacks is a common phenomenon, and to a lesser extent allodynia in more peripheral regions (53,54). The use of an evoked reflex such as the application of von Frey filaments to rodents as a marker of ongoing allodynia during a human migraine attack is somewhat debated, and might not be as valid as spontaneous behaviours in capturing ongoing pain (55,56).
In healthy human volunteers tested with von Frey filaments on the forehead and the arm 90, 150 and 210 minutes after cilostazol administration, no effect was observed on mechanical sensitivity (6). However, a difference might have been detected if a later time point had been implemented. Another explanation is that the dose difference between the rat and human experiment is responsible for the observed cephalic hyperalgesia in rats.
C-fos expression in the trigeminal nucleus caudalis
The c-fos expression study was performed to confirm that the rats did have activation of trigeminal nociceptors after cilostazol treatment. Initially, the experiment was performed with animals housed using a reversed light cycle as in the behavioural experiments. However, the effect of cilostazol was not significant due to a very large variation in both the treatment and vehicle group (see supplementary Figure 2(a–b)). Especially in animals housed in normal light conditions, the cilostazol-induced c-fos expression was robust and significant. It was found in the first 16 rats housed under the reversed light cycle, and subsequently in two separate experiments performed on a total of 25 rats housed under a normal light cycle regime.
C-fos positive neurons were counted in the laminae I and II, as this area is relevant to headache/facial nociception (32,57,58). The deeper laminae were not counted systematically, but superficial observation revealed minimal c-fos expressed in this region. The increase in c-fos positive neurons was localised in the interpolaris/caudalis transition zone just rostral to obex (35) and throughout the TNC to the level of C1. In the C1–C3 spinal cord sections, we did not observe an increase in c-fos expression. The observed pattern of c-fos expression is similar to that reported by Strassmann et al. after stimulation of the superior sagittal sinus or the snout of the rat (35). In another study, they observed that stimulation of the snout and upper lip increased c-fos in the Vi/Vc and Vc (58). Stimulation of the transverse sinus, vibrissal pad and supraorbital vibrissae resulted in c-fos expression at the C1–C2 level (35,58). GTN appears to induce c-fos mainly in the caudal Vc (32). Stimulation of the tongue and lower lip (mandibular branch of the trigeminal nerve) also induced c-fos in the Vi/Vc transition zone, but only strictly dorsal within the zone (35,58). However, in our sections the dorsal/ventral/lateral distribution of c-fos expression was mainly dorso- and ventrolateral. We therefore suggest that our c-fos expression reflects activation of V1 and V2 of the trigeminal nerve through branches innervating relatively rostral parts of the face. Most people report fronto-temporal pain after cilostazol treatment (6,8). The effect of cilostazol on c-fos expression was also evident in the area postrema and nucleus tractus solitarius. This indicates activation of baroreflexes (59) and is due to the effect of cilostazol on blood pressure. We did not investigate the c-fos response to sumatriptan, as we had already shown that sumatriptan did not change the behavioural outcomes. Also, c-fos expression was not studied in other pain relevant brain regions, as our main objective was to establish a behavioural model. However, this could be pursued in follow up studies to further characterise the cilostazol model.
In conclusion, we have developed a novel way of triggering trigeminal nociception in rats by oral administration of cilostazol. Besides cephalic mechanical hypersensitivity and c-fos expression in the TNC, the rats display behavioural changes in terms of increased light sensitivity and increased grooming behaviour.
Article highlights
Cilostazol increases light sensitivity and grooming behaviour. Cilostazol causes hyperalgesia in the trigeminal region, but not in the hind paw. Cilostazol induces c-fos expression in the trigeminal nucleus caudalis. The changes in grooming behaviour and light sensitivity are not reversed by sumatriptan.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research leading to these results has received funding from the European Union’s Seventh Framework programme (EUROHEADPAIN) under grant agreement no. 602633. Also, the work was funded by Candy’s foundation, The Simon Spies Foundation, Kong Christian den Tiendes Foundation, Foundation for Research in Neurology and The A.P. Møller Foundation for the Advancement of Medical Science.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
