Abstract
The efficacy of acute therapies for migraine can be measured in many ways. Traditional endpoints (such as reduction in pain from moderate or severe to mild or absent) are used for regulatory purposes, but do not reflect all components of the migraine syndrome, nor, necessarily, what is most valued by patients and clinicians. There is also a pharmacokinetic–pharmacodynamic disconnection for these traditional types of endpoint, suggesting that they teach us little about how these drugs work. More rigorous, but nonetheless pain-score based, endpoints are reviewed. The biases that can attach to measures such as therapeutic gain and number needed to treat, in the context of migraine therapy, and the limitations of these measures for use in meta-analysis, are discussed. The clinical subtleties of these endpoints are numerous: understanding patients' ability to distinguish between multiple headache types, the best timing of treatment relative to the start of an attack, and measuring clinical outcome may be statistically difficult, but yet may also provide more clinical utility than pain-score analyses. The three therapeutic strategies (Step, Stepped-within-attack, and Stratified care) are reviewed and the place of 5HT1B/1D agonists within them, based on the currently best available evidence, is identified. Consideration should be given to more real-life studies, to measuring drug efficacy after early administration during onset of headache, and to greater sophistication in our approach to the necessarily gestalt measures of patient satisfaction and treatment preference.
Few would dispute that the well-controlled clinical trial is a sound source of evidence-based information, although there are other sources to look to (1). The problem is that the pleiomorphic nature of the migraine syndrome and the sheer diversity of patient preferences still require us to introduce nonevidence-based variables into our interpretation of clinical trials data and into management of the individual patient. We are provided in the literature with a wide variety of clinical trial endpoints, which describe different aspects of drug efficacy.
The principal endpoint that has been used for assessing 5HT1B/1D agonist efficacy has been the proportion of patients converted from pain scores of two or three to pain scores of zero or one (‘3/2–0/1 pain endpoint’; Fig. 1). This endpoint is typically reported for single doses of active and placebo therapy at 1, 2 or 4 h postdose, and forms, in most countries, the endpoint that justifies the efficacy requirements for approval of a new drug. While this endpoint is important, it is the intention of this paper to demonstrate that other factors are useful measures in the complicated area of assessing satisfactory acute therapy in migraine. Silberstein (2) has demonstrated the difference between what patients want and artificial efficacy time points used for regulatory purposes (Table 1). Although Shakespeare is unquestionably immortal, the situation is no longer as simple as choosing a drug that fulfils the quotation:
The top 10 features of acute migraine therapies ranked in terms of importance by n = 500 patients, using mean values calculated from a discontinuous scale ranging from zero (not important at all) to 10 (of more importance)
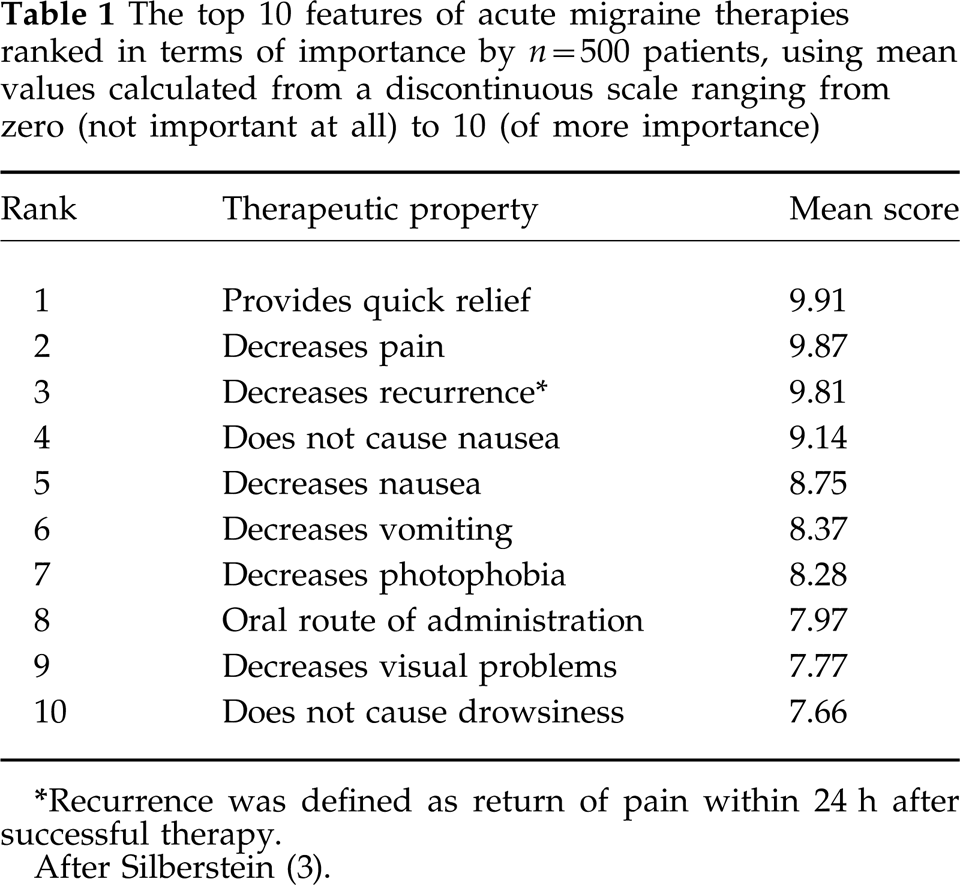
Recurrence was defined as return of pain within 24 h after successful therapy.
After Silberstein (3).

The conventional measures of efficacy for an acute therapy for migraine (3/2–0/1 pain response). Headache response is recorded when a patient enters the clinical trial with moderate or severe pain (pain score 2 or 3), and is converted to no or mild pain (pain score 0 or 1) after treatment with either active drug or placebo. Headache recurrence is recorded when responding patients have an increase in pain back to either score 2 or 3, but not necessarily the same score as when they entered the study, within a time limit (typically 24 or 48 h after previous successful therapy). Rescue medications may be used and are recorded, but do not reduce the response rate if used more than 2 h after the test therapy.
‘O true apothecary! Thy drugs are quick.’ (3)
Furthermore, what do we mean by quick; statistically, numerically, or clinically quick? While previous clinical studies may establish statistical norms for drug efficacy, studies where clinical significance is measured by what the individual patient considers clinically meaningful may give paradoxical results. Should we search for a drug, drug combination, unorthodox route of administration, or unapproved dose-size that the patient perceives as providing satisfactory treatment, according to whatever personal algorithm the individual employs, but that may not be statistically quick for the 3/2–0/1 pain endpoint?
Pharmacokinetic–pharmacodynamic dissociation for 5HT1B/1D agonists
A comparison of the effects of the pharmacokinetics and pharmacodynamics for a series of compounds (such as 5HT1B/1D agonists) requires that a standard set of endpoints be used; only in this way can relative assessments of the clinical relevance of differences in pharmacodynamics be made.
Pharmacokinetic estimates for the triptans (Table 2), such as the time of maximal plasma concentration (Tmax) or half-time of elimination in hours (T1/2), may not predict the time to onset of effect for standard endpoints (3/2–0/1 pain endpoint) at 1, 2 or 4 h. Furthermore, absolute dose (oral bioavailability (%) × administered dose size), while sometimes related to consistency of response for other drugs, again may not be predictive for response to ‘triptans’. The only hint of a relationship between pharmacokinetics and efficacy is that the two drugs with longest half-times of elimination (naratriptan, and the currently investigational drug frovatriptan) seem to have the lowest headache recurrence rates; however, this correlation does not hold for all the other drugs in this class.
Published pharmacokinetic variables for oral 5HT1B/1D agonists. All other drugs penetrate the central nervous system more than sumatriptan
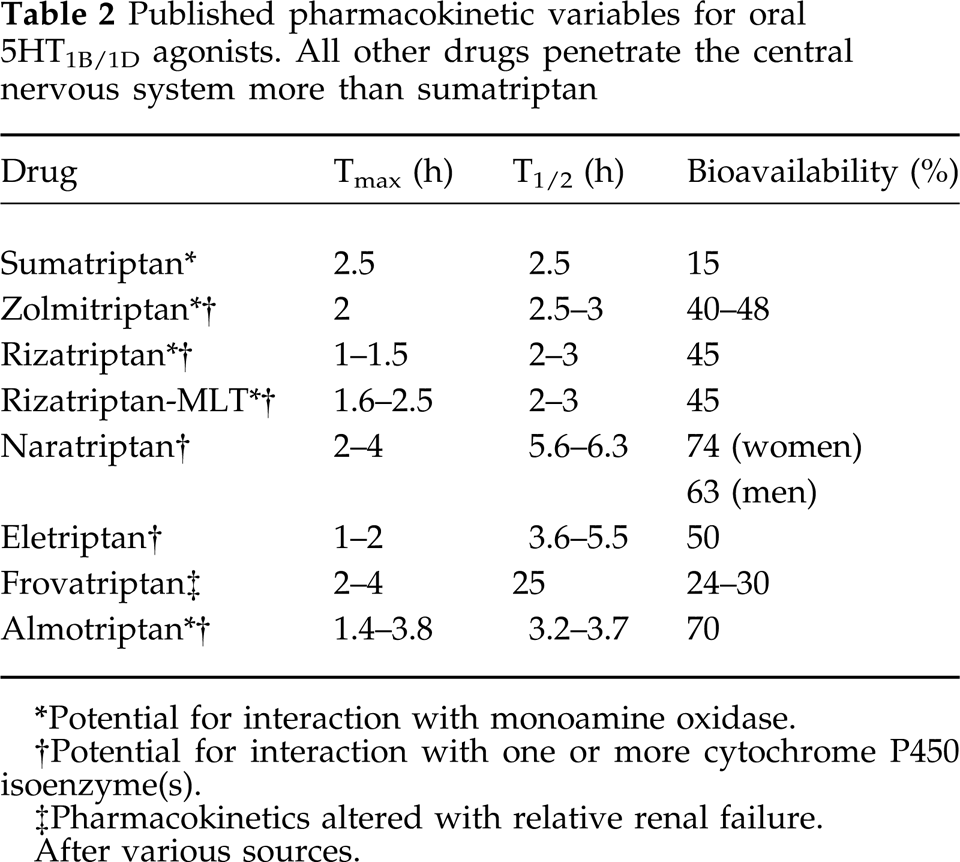
Potential for interaction with monoamine oxidase.
Potential for interaction with one or more cytochrome P450 isoenzyme(s).
Pharmacokinetics altered with relative renal failure.
After various sources.
Evolution of endpoints in the acute treatment of migraine
More rigorous efficacy criteria?
The argument that traditional 3/2–0/1 pain response does not represent an endpoint that is clinically useful has led to suggestions for composite endpoints. This view is that successful therapy is characterized not only by such an endpoint, but also that the same patient should neither experience recurrence (return of headache to grade 2 or 3 within 24 h of dosing; Table 3) nor take any rescue medications (Fig. 2). A further suggestion is that patients should only be counted as treatment successes if they have no pain 2 h after therapy, and have no recurrence, and use no rescue medication (Fig. 3). One difficulty with this composite endpoint is that the set of treatment success criteria do not apply uniformly to all patients; for example, patients with a natural history of migraine attacks lasting 48–72 h might nonetheless appreciate a therapy that treats them within 4 h, and might not object to taking repeated doses of that drug or a rescue medication for the remainder of their attack.
Published pharmacodynamic measures for oral 5HT1B/1D agonists

Not published.
Ahrens et al. (21).
Drug currently under investigation, efficacy rates drawn from small-sample literature reports, and not from standardized approved labelling.
Note: placebo response rates are not shown. After Tepper and Rapoport (4).
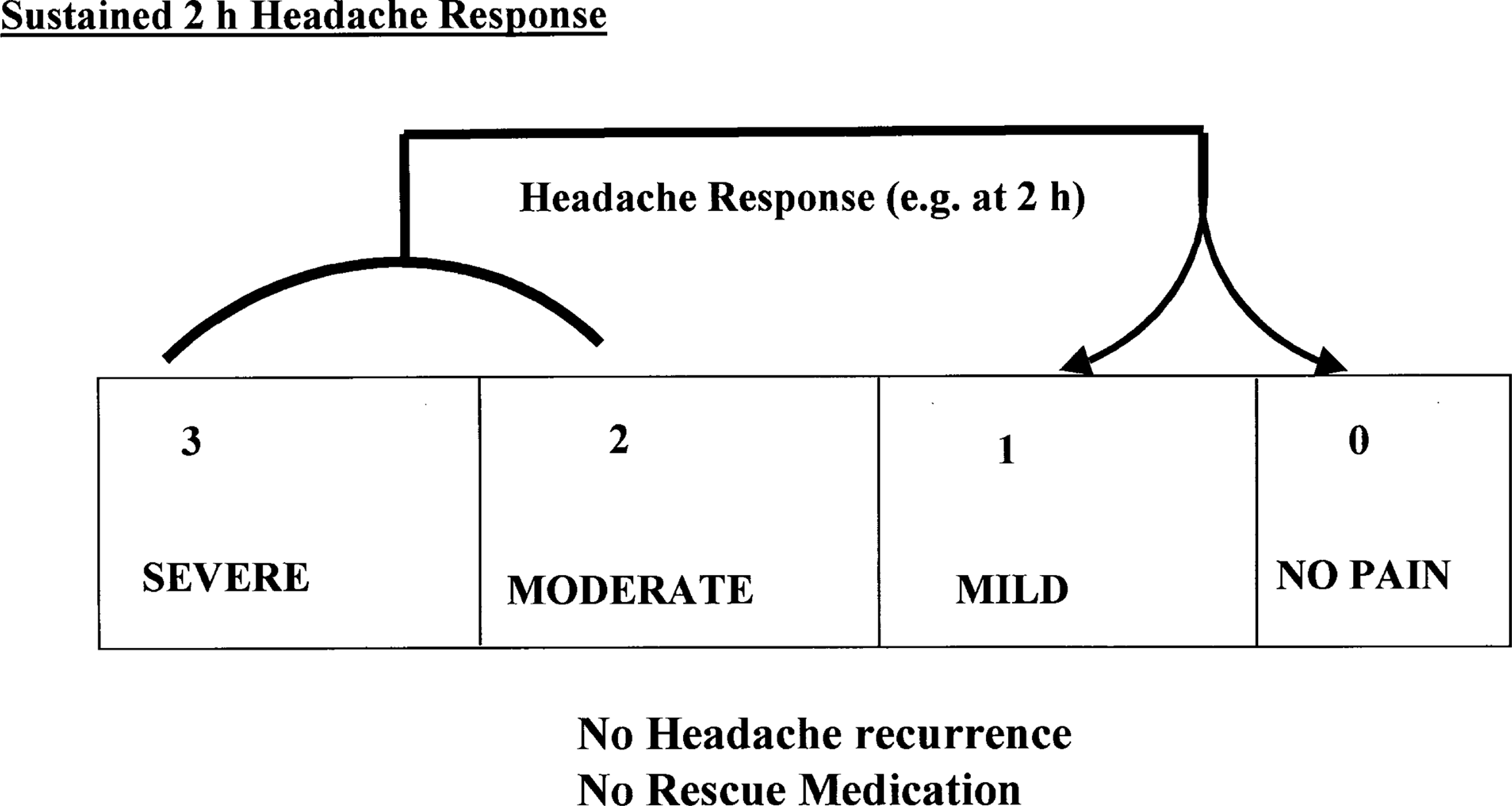
The proposed endpoint Sustained 2 h headache response. Patients are initially converted from score 2 or 3 to score 0 or 1, as in Fig. 1. However, treatment failure is recorded, in spite of the initial response, when either there is any degree of recurrence, or the patient uses rescue medication at any time during the period over which recurrence could be reported.

The proposed endpoint Sustained pain-free response. Unlike Figs 1 and 2, patients must initially respond to the test therapy with conversion from pain score 2 or 3 to pain score 0 (i.e. no pain). Any change from this pain-free status during the prolonged observation period (e.g. 24 h) is recorded as recurrence, and treatment failure. Similarly to Fig. 2, use of rescue medication during this period is also a criterion for treatment failure.
Bias associated with and without prior ‘triptan’ exposure
The traditional efficacy percentages (whether as in Table 3, or the more rigorous, composite endpoints described above) are liable to several factors that could introduce bias. These include placebo-response rates that might vary from study to study, or change with time. Temporal bias might a priori include the fact that triptan-naive patients were treated in early clinical studies, unlike later trials, after the older triptans were already on the market. Similarly, earlier studies could have recruited more severely affected patients than later studies. In both cases, efficacy data of the type shown in Table 3 might apply to a different population for older drugs than for newer ones.
The bias, if any, associated with the recruitment of patients that are not triptan-naive might be related to the fact they have entered a clinical trial of a newer triptan because they were dissatisfied with their older drug. The expectation might therefore be that patients who are unresponsive to one triptan will be less likely to respond to any other. Clinical, anecdotal observations suggest that there are many patients who will respond to one triptan but not another. Studies in patients who are not triptan-naive, by analogy with other pharmacological classes of drug, might be expected to generate dose–response curves shifted to the right, or with depressed maxima, in comparison with unselected patient populations. This type of bias may also sometimes appear in active comparator studies and crossover studies when there are unbalanced rates of dropouts between treatment groups (6–9).
Therapeutic gain (TG) and number needed to treat (NNT)
If it is hypothesized that later studies recruited patients who were less severely affected than those in earlier studies, then it might be expected to be easier for newer drugs than older drugs to achieve the reasonably similar rates of response of the type shown in Table 3.
Traditionally, in fields other than the acute treatment of migraine, it has been assumed that contemporaneous placebo response rates would control for the severity of disease among patients recruited to a study, provided that a genuine randomization took place, and that double-blinding was successful. Factoring active-treatment response rates against contemporaneous placebo response rates may be accomplished in a simple arithmetic manner in the calculation of two variables, the therapeutic gain (TG) and number needed to treat (NNT). These are defined as (10):

where ARR is the active response rate, and PRR is the placebo response rate, i.e. the TG is the incremental efficacy rate for active beyond placebo treatment, and

i.e. the reciprocal of the TG. The NNT is conceived of as the number of patients that must receive the active treatment for at least one of them to truly benefit from that therapy. The lower the NNT, the more effective the therapy. The NNT is used widely when conducting meta-analyses of multiple clinical trials, and when making comparisons between drugs with the same desired effect (11).
Therapeutic gain and NNT have been used by Tfelt-Hansen (12) to compare different formulations of sumatriptan, and by Goadsby (13) for comparisons among oral 5HT1B/1D agonists. Table 4 provides an independently developed, but reasonably similar, set of TG and NNT data. These and other authors have also developed from published data the analogous measures of therapeutic harm (TH), and number needed to harm (NNH). These are the incremental increase in frequency of defined types of adverse events, and its reciprocal, respectively.
Therapeutic gain (TG) and number needed to treat (NNT) for acute treatments of migraine, using published efficacy data from well-controlled clinical trials, based on the 3/2–0/1 pain response at 2 h

ARR, active response rate; PRR, placebo response rate; s.c., subcutaneous; p.o, per oral; i.n., intranasal; p.r. per rectum (suppository).
The appearance of data above does not automatically indicate that the dose or route of administration has regulatory approval in all jurisdictions.
In some cases, efficacy data are drawn from small-scale published trials and not from labelling approved by regulatory authorities.
The use of TG and NNT would initially seem to be well-suited to meta-analyses of migraine studies, which have often been conducted using standard techniques (14). Such comparisons, using US labelled efficacy rates for placebo and active therapies, suggest little difference between oral triptans (except naratriptan), and superiority of the subcutaneous formulation of sumatriptan over all other approved drug/formulation combinations in this pharmacological class (Fig. 4), and yet, these seemingly innocuous arithmetic procedures have nonetheless touched off a good deal of controversy when applied to acute therapies for migraine.

Therapeutic gain (TG) for seven US approved 5HT1B/1D formulations for the acute treatment of migraine. TG has been calculated from labelled efficacy rates for active- and placebo-treated (A and P, respectively) patients, whose sample sizes are shown.
Problems with analyses using TG and NNT
First, if the underlying measurement of efficacy (e.g. a 3/2–0/1 pain response) may be criticized for being an abstraction of clinical reality, then so can any TG or NNT that is secondarily calculated from it. Thus, like the underlying endpoints themselves, TG and NNT do not bring us any closer to measuring therapeutic properties that patients value most.
Secondly, TG and NNT as measures of efficacy are very sensitive to placebo response rate (PRR), and PRR is notoriously variable in migraine clinical trials. To illustrate this point, Klein (15) has drawn attention to ‘megatriptan’, an imaginary drug to which almost all migraineurs respond. Suppose that megatriptan is used at two hypothetical clinics where response rates vary due to the many well-defined, nonpharmacological factors that are beyond the scope of this article to discuss. Let us say that at Clinic A, ARR = 95% and PRR = 45%, and at Clinic B, ARR = 95% but PRR = 15% because of a more holistic approach by clinicians in the former. Megatriptan will then be reported to have TG of 50% or 80% in Clinics A and B, respectively. This might suggest that megatriptan works best in Clinic B, and yet that is the place where migraineurs are being least well-served! Overall, the point is that a difference in TG can easily reflect a different PRR, not a different ARR; perhaps stating what the ceiling effect is for the active drug would be more meaningful.
Thirdly, good studies of inter- and intra-study site consistency within multicentre trials of acute migraine therapies have not yet been published. This would require a comparison of efficacy rates (and their variability) for placebo and active treatments between clinics, as well as tracking each clinic chronologically during the performance of several studies. These are further factors confounding the unsceptical acceptance of TG, without analysing PRR and ARR.
Fourthly, TG and NNT have been criticized because prescribing also depends on tolerability and on finding the best compromise between efficacy and tolerability for each patient. The TH and NNH statistics again only relate to precisely defined, underlying frequencies; to calculate TH or NNH from overall adverse event rates is equivalent to ranking all adverse events to be of similar clinical significance, and an arbitrary decision has to be made whether to count unique patients with any adverse event, or the total number of adverse events when these are multiple in a particular patient. As Van der Kuy and Lohman (16) have pointed out, most clinicians would worry less about dry mouth than coronary ischaemia. Table 5 compares NNT and NNH for those drugs for which we have standardized measures of tolerability that are available in the public domain.
Comparison of number needed to treat (NNT) and number needed to harm (NNH) for some selected oral 5HT1B/1D agonists.

Data are averaged for a number of clinical trials.
Lower NNT and higher NNH are favourable treatment characteristics.
After Goadsby (13), but independently estimated from several small-scale studies (overall adverse event rates, in terms of unique patients, are often not tabulated in labelling).
These difficulties in interpreting TG and NNT can be even more complex when comparing active treatments. The chronobiological differences between early and more recent clinical trials (see above) are compounded with arithmetical manipulation. Spierings (17) has raised a further objection that crossover studies are better suited to TG calculations than parallel-group studies because the patient populations will be more similar for comparison between treatments.
Although imperfect, these arithmetical methods could be equally well applied to recurrence rates, or other measures of efficacy. All are agreed however, that well-designed head-to-head studies are superior to meta-analysis for finding differences between active treatments.
Factors in the treatment of migraine that are endogenous to the patient
Ability to distinguish headache types
Most migraine patients also suffer from headaches of other types. Concomitant episodic tension-type headache is probably the most common. Most clinical trials of acute therapies for migraine require that the patient is able to distinguish between these types of headache, and thus will use the test medication only for the treatment of migraine. While this is a laudable goal for the assessment of new therapies, is it realistic in ordinary clinical practice?
The IHS criteria are for the identification of a patient as a migraineur. Indeed, some patients with tension-type headache will apparently fulfil the IHS criteria for migraine without aura (IHS 1.1) when these criteria are strictly applied. For example, the common patient with a moderate bilateral headache of more than 4 h duration, with some pulsating quality, some relief by resting, with either phono- and photo-phobia or both, and a negative neurological examination, may be experiencing an attack of migraine or episodic tension-type headache. The difficulty in differentiating migraine headache from tension-type headache at an early stage of the attack, in the many patients who experience both types of headache, creates further difficulties in using IHS criteria (this is discussed further, below).
Timing of treatment during an attack
The Spectrum Study (18, 19) compared the efficacy of oral (50 mg) sumatriptan in all headache types experienced by diagnosed migraineurs. These headache types included migraine itself (IHS criteria 1.1 and 1.2), migrainous headache (IHS 1.7), and tension-type headache (IHS 2.1). It was found that the response to sumatriptan in migraineurs was as good for their migraine attacks as for their tension-type headache. Furthermore, sumatriptan was also effective in the treatment of mild headache. In clinical trials, patients are usually required not to treat a headache until a headache score of 2 or 3 is reached.
The importance of this finding relates to headache treatment early in a migraine attack. A guiding principle in the treatment of any painful syndrome is that therapy works better if administered at the onset of pain than when the painful state is well-established. If we instruct our patients to treat their migraine attacks at the earliest possible time with our prescribed triptan, then we shall also be asking them to treat their headache precisely at the time when they are going to be least able to distinguish migraine from among their other headache types. Thus, the Spectrum data, as far as it goes, is reassuring because it makes a large step towards the ordinary clinical situation and away from the artificial constraints of clinical trials.
However, as might be expected, there are also unclear aspects to the Spectrum study. First, the study concentrated only on the pain endpoints, and not the other components of the migraine syndrome. Secondly, it would have been very interesting to know whether the nonresponders (both to active and placebo treatments) went on to develop a typical migraine attack or some other type of headache. Thirdly, it was not reported whether there were any patients who were defined as tension-type headache at the time of treatment, but who actually went on to have a migraine attack; such patients would pose a significant problem in the interpretation of these data.
Patients' satisfaction
Göbel et al. (20) have explored those variables that patients rate in their therapies. Consistency of response over multiple attacks, and rapidity of effect with each attack are always highly rated (see above). Thus, these factors do not correspond to those reported by research physicians in the field of headache for the various therapies. Patients may have preferences for routes of administration, too. This can depend on whether they are vomiting, deeper psychological attitudes or habit. The preference for suppositories in France and other countries, but for tablets or injections elsewhere, have been well-described in the past.
Sheftell, Ramadan and Weeks have proposed a novel set of scales for the measurement of patient satisfaction (Fig. 5). This method is an attempt to not only identify the various factors that contribute to patient satisfaction, but also to assess their relative weights. These factors may include not only responsiveness to test therapies, but also rate of relief, adverse events, consistency of response, and route of administration. This method is currently in validation studies, and may prove to be a useful tool for real-life studies, whose need is discussed below.

A novel method for measuring patient satisfaction (after Sheftell, Ramadan and Weeks). This set of scales is intended to facilitate communication between doctor and patient, and to provide standardized measures that may be compared between alternative treatments used in Step, Stepped-within-attack, or Stratified care strategies. Patients complete their responses within 24 h after the migraine attack, and mail them to the study site separately. The format shown is intended for use with at least three consecutive attacks, although Section 7 of the sheet can be adapted for other chronological purposes. The response sheet is accompanied by a second sheet, explaining how the form should be completed and providing some printed example responses. Panel A, Demography and drug identity; panel B, route of administration and initial response; panel C, recurrence; panel D, function, adverse effects of drug and consistency of response (used when three attacks have been treated); panel E, drug assessment and global measure of satisfaction.
Other factors
We know almost nothing about the safety of the new therapies in pregnancy, the breast-fed neonate, and children. Inter-racial differences in migraine presentation and therapeutic effectiveness of these agents has also not been explored.
Choosing a triptan to prescribe
So which is the best triptan? The best answer to this question is that it is the wrong question to ask as a primary care physician! Assuming that you have taken a good headache history from the patient and that secondary headache from other organic disease has been excluded, then the objective should be to create the best patient-therapy combination. There are three principal strategies that one can employ to arrive at this combination: Step-Care, Stepped-within-attack Care, or Stratified Care (Fig. 6).

Typical approaches when using Step, Stepped-within-attack (formerly Staged) or Stratified care strategies in the clinical management of patients with migraine.
Step Care involves a programme of therapies to be used during successive headache attacks. This programme is pursued to its completion regardless of responsiveness that has been found for each attack. Over several attacks, patient responsiveness to the variety of pharmacological classes of agent can be assessed and compared. If more than one effective therapy is found, the selection may also be based on concomitant tolerability findings. In inconclusive situations, the programme can be repeated, perhaps using fewer therapies, to resolve small differences. Consistency of response to a selected agent or agents should then be monitored. Older forms of step-care halted the programme as soon as an effective therapy had been found; this did not permit optimization for patient preference among different effective therapies.
Stepped-within-attack (formerly Staged) Care involves the use of a variety of agents during the same attack. The agents are usually chosen in order of increasing probability of efficacy, but this is typically accompanied by an increasing likelihood of adverse events, whether due to the new drug alone or as an interaction with the earlier therapies. Definitive rescue therapy is prospectively chosen. Escalation from drug to drug is based upon criteria that define the absence of response to the previous drug(s).
Stratified Care involves the judicious choice of therapy with the greatest probability of effectiveness. This choice is based on degree of severity, upper gastrointestinal aspects of the migrainous syndrome, age of the patient, documented drug allergies, etc. If the treatment fails, then the patient moves directly to rescue therapy, which has been defined in the same way as for Stepped-within-attack Care, and the choice of first-line therapy is refined accordingly before the next attack (22).
The dearth of real-life studies
Phase III and phase IV studies are usually designed to be closer to the situation of ordinary clinical care than phase I or II studies, but, even so, the use of informed consent documents, inclusion/exclusion criteria, and rigid monitoring schedules and procedures are unlike the ordinary practice situation. The circumstances surrounding clinical trials inevitably create abstractions of reality.
Two clinical trial techniques have recently been introduced in an attempt to overcome the abstraction problem. These measurements are: (a) testing drug efficacy at early stages of the attack; and (b) asking patients to express preferences among multiple treatments.
Treatment of a migraine attack at an early stage requires, essentially, that a change in pain score from mild pain to no pain (i.e. score 1 to score 0) becomes the measure of efficacy. Clinically and neurologically, this is a worthwhile therapeutic pursuit because, like many pain syndromes, treatment may be easier before kindling effects have occurred. From a statistical perspective, and in comparison to the usual pain score change from 3/2 to 0/1 as a measure of efficacy, such early treatment endpoints contain greater variability, and are likely to exhibit larger placebo effects. From the point of view of the patient, an additional problem is that it may be quite difficult to distinguish a migraine attack from her or his other types of headache at a time when the complete syndrome has not yet developed (see above). Overall, in comparison to traditional endpoints, the clinical trialist must accept higher placebo response rates, and studies with larger patient populations will be needed, because the size of treatment differences between groups will be smaller than usual.
Patient expression of treatment preference is a valuable thing to learn, not only for the individual's clinical management, but also at the population level for epidemiological and economic reasons. Patients' treatment preferences may arise from the integrated, complex pattern of biological phenomena that they experience (this pattern depends on disease-state and its variability); wanted and unwanted effects of the test treatment; and subtle cultural and individual psychological factors. We must accept that it may be impossible to quantify all the component factors that cause patients to prefer one treatment over another, and that, for the patient, the pattern is more than the sum of all its components, i.e. a true gestalt (23). All we can do is measure the patient's expressed treatment preference, and we should do so in a manner that gives the patient the best chance of experiencing alternative therapies in as unbiased a manner as possible. It behoves us to permit the patient to compare all therapies that are acceptable from the standpoint of pharmacology and safety. So far, very few such studies have been reported.
Concluding remarks
Each company presents data that attempt to sway clinicians into believing that one triptan is better than another, while in fact a hard look at the evidence matches clinical experience, which clearly demonstrates that patient responses are highly individual, with no triptan, on balance, demonstrating superiority over the others.
Successful treatment of migraine patients sine qua non requires obtaining a thorough history, excluding organic causes of headache, and making the right diagnosis. Identification and elimination of headache-provoking phenomena comes before selecting a (some) drug(s). Choosing how to treat the patient pharmacologically, whether using a Step, Stepped-within-attack or Stratified care strategy, requires finding a balance between what the patient (not the doctor) views as satisfactory, a reasonable compromise between efficacy and tolerability, and careful follow-up. Lastly, real-life studies of treatment early in migraine attacks and patient treatment preferences are in their infancy.
