Abstract
The prophylactic action of cyclandelate was investigated in a multicentre, randomized, placebo-controlled, parallel group study. A 4-week baseline period was followed by a 4-week placebo phase and a 16-week treatment period with either 1600 mg cyclandelate or placebo. Patients (n = 251) with two to six migraine attacks/month were randomized. Neither the primary study endpoint (reduction of migraine days from baseline to the last 28 days) nor most of the secondary endpoints (reduction in the number of migraine attacks, severity or duration of attacks, frequency of autonomic disturbances, medication for treatment of attacks) showed a difference between cyclandelate and placebo. Cyclandelate, however, was superior to placebo in a global impression of efficacy rated by the patients and the treating physicians. Both treatments were well tolerated. In conclusion, cyclandelate was not superior to placebo in the prophylaxis of migraine with regard to parameters usually used in migraine prophylaxis trials.
Introduction
Patients with frequent and severe migraine attacks require migraine prophylaxis. The aim is to reduce the number of attacks and to reduce pain intensity and duration. The mode of action for most drugs used in migraine prophylaxis is not known. Animal models to test the action of drugs used for migraine prophylaxis are not available. The prophylactic action of β-blockers, the most widely used drugs for migraine prophylaxis, was detected incidentally in patients who were treated for hypertension and suffered from migraine. Several β-blockers, e.g. propranolol and metoprolol, have convincingly been shown to have migraine prophylactic activity (1, 2). Cyclandelate inhibits provoked calcium overload in neurones, calcium-induced contraction of vascular smooth muscle cells and platelet aggregation induced by thrombin, PAF and adenosine (3–7). This pharmacological pattern made prophylactic action in migraine possible.
Earlier pilot studies indicated that cyclandelate indeed might have a prophylactic effect. In a small pilot trial with 40 patients, Nappi et al. (8) showed that 800 mg bid were as effective as flunarizine 5 mg for migraine prophylaxis. Cyclandelate (800 mg bid) was superior to pizotifen (0.5 mg tid) in one trial (84 patients) (9) and equivalent to propranolol in another trial (84 patients) (10). To verify this, a randomized, parallel, double-blind, multicentre study was performed to investigate efficacy and tolerability of cyclandelate compared with placebo and propranolol for the prophylaxis of migraine in 214 patients (11). Patients were treated for 12 weeks with 1200 mg cyclandelate (n = 81), placebo (n = 55) or 120 mg propranolol (n = 78). The number of migraine attacks and duration of headache were compared between the baseline and the last 4 weeks of treatment. Migraine attacks were reduced by > 50% in 37% of the patients with cyclandelate, 30.9% with placebo and 42.3% with propranolol (P = 0.58 cyclandelate vs. placebo). Duration of migraine attacks per month was reduced with cyclandelate (36.8 h) compared with placebo (13.7 h; P = 0.046). Cyclandelate and propranolol were not different in terms of efficacy (P > 0.05). This study was inconclusive in answering the question whether cyclandelate was superior to placebo. The trial was additionally flawed by the fact that during the study sumatriptan became available which had an influence on the duration of attacks. We therefore set out to do a second large, multicentre, double-blind and placebo-controlled trial to show or disprove the prophylactic action of cyclandelate in migraine.
Patients and methods
The trial was a multicentre, randomized, double-blind, placebo-controlled, parallel group study in out-patients suffering from migraine. The trial was performed by 43 neurologists, internists and family doctors in Germany. The study lasted 24 weeks for every patient. A 4-week observation period without medication was followed by a 4-week placebo run-in phase which served as the baseline period. Patients were then randomized and treated for 16 weeks with 1600 mg cyclandelate/day or placebo. Inclusion criteria were: age between 18 and 65, migraine with or without aura according to International Headache Society (HIS) guidelines (12), two to six migraine attacks/month in the last 3 months based on diaries, two to six attacks during the 28 days of the baseline period, duration of migraine at least 12 months, beginning of migraine at age < 50 years and written consent. Exclusion criteria were: other headache than migraine on > 6 days per month, severe illness, intake of migraine prophylactic drugs (e.g. β-blockers) within the last 3 months, use of medication for the treatment of acute attacks on > 10 days per month, pregnancy and breast feeding. The trial was performed in accordance with the Declaration of Helsinki and its subsequent revisions. Ethics Committee approval was obtained and the patients gave their informed consent to participate in the trial. Randomization was performed by a computer generating a list for each centre. Patients were asked to keep a headache diary and make recordings of the presence of headache, intensity of headache, autonomic symptoms and number and brand of medication taken to treat acute migraine attacks.
The primary endpoint was the number of migraine days in the last 28 days of therapy compared with baseline. Secondary endpoints were: number of migraine attacks per 28 days, mean duration of migraine headache per day, headache intensity, intake of medication to treat acute attacks and a global impression of efficacy by the patient and the treating physician. For this purpose the following definitions were used: a migraine day in the clinical record form (CRF) had to have at least 3 h of headache. Headache interrupted by sleep and reoccurring within 24 h after sleeping or intake of a tripan was counted as one attack. A pain-free interval of > 24 h defined a new migraine attack. The intensity of migraine attacks was measured on a four-point verbal scale: mild (1), moderate (2), severe (3), very severe (4). Medication for the treatment of acute migraine attacks was counted. Doses were standardized according to the efficacy of drugs (e.g. paracetamol was multiplied by a factor of 1, codeine by 2, ergots by 3 and sumatriptan by 4). At the time of the trial only sumatriptan was available from the ‘triptan family’ in Germany. The global impression of efficacy was scaled as very effective, effective, moderately effective, not effective. Vital signs were taken at each 4 weeks follow-up visit. A neurological examination, blood counts, biochemistry and ECG were performed at the beginning of the baseline period and the end of the study. The tolerability of study medication was assessed by patients and physicians on a four-point rating scale (very good, good, moderate, poor).
The power calculation was based on the primary study endpoint. Assuming an α of 0.05 (one-sided) and β of 0.20 a 35% difference could be detected between the two groups with cases numbers of n = 100 in each treatment group and a power of 80%. The primary efficacy analysis—reduction of migraine days/28 days—was performed using an analysis of covariance with the consumption of medication for the treatment of acute attacks as covariate. Secondary efficacy variables were evaluated by t-tests or Wilcoxon tests. The Mantel–Haenszel–χ2 test was used to assess qualitative variables (efficacy and tolerability assessments). Changes in lab parameters were evaluated with the Wilcoxon signed rank test.
Results
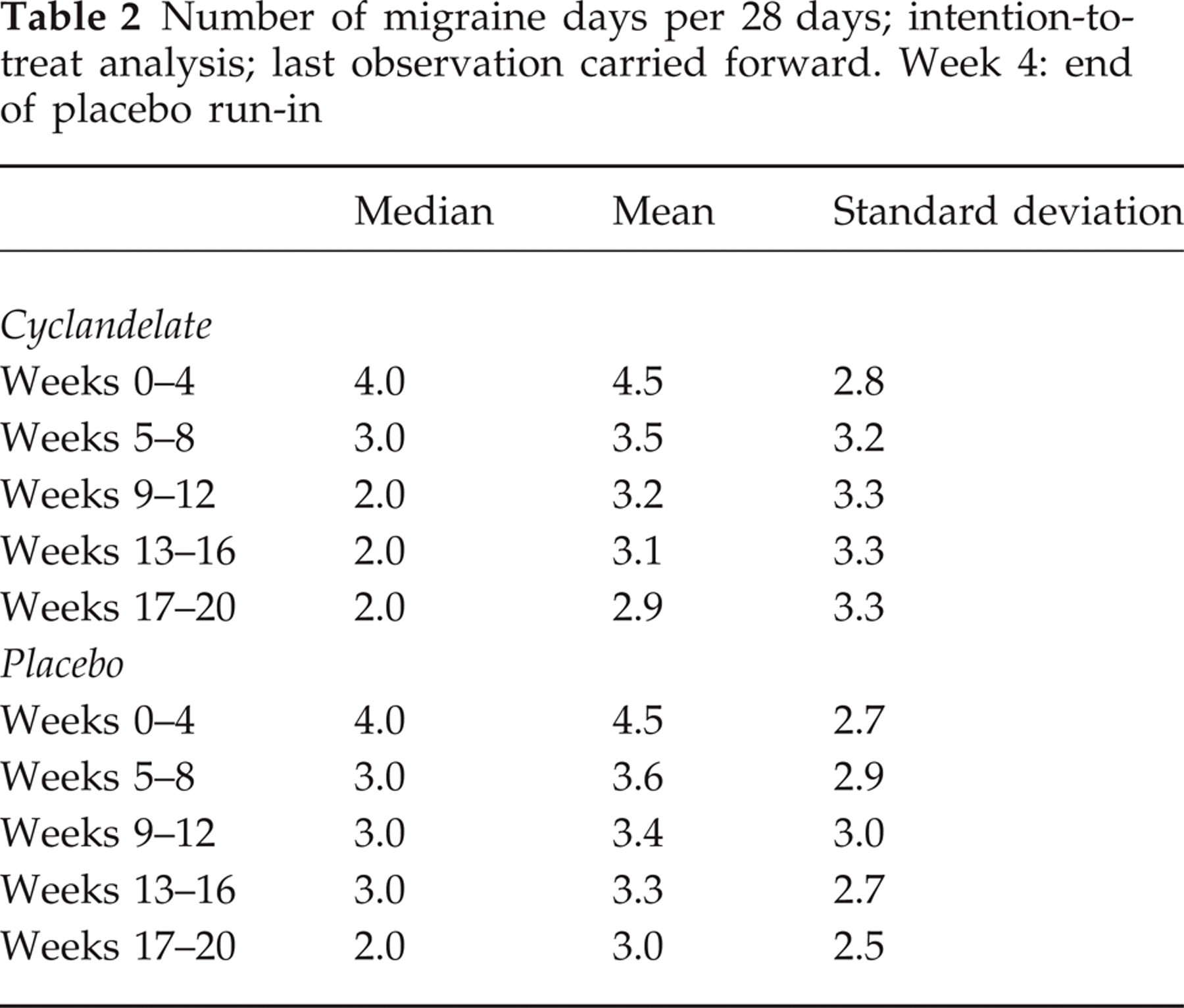
Three hundred and one patients were screened for participation. Twenty-seven patients were excluded before entering the baseline phase, in most cases because the number of attacks was less than two attacks per month. Two hundred and seventy-four patients entered the placebo run-in phase, and 251 patients finally received study medication. For the safety analysis all patients were considered taking a least a single dose of study medication (n = 251). The intention-to-treat population (n = 249) comprised all patients with valid entries into the diary at baseline and having taken at least one dose of study medication (125 cyclandelate, 124 placebo). The per protocol analysis included 185 patients who fulfilled all inclusion and exclusion criteria and participated for at least 8 weeks in the treatment phase (94 cyclandelate, 91 placebo). One hundred and seventy-three patients were treated for 16 weeks. Missing data were handled as last value carried forward. In the following, only data for the intention-to-treat analysis are presented. The two treatment groups were comparable with respect to important biological variables (see Table 1). The number of migraine attacks and days with migraine were identical in the two groups at the end of the observation period and the placebo run-in phase. The primary endpoint, reduction of migraine days between baseline and the last 28 days of treatment, was 1.6 in the cyclandelate group and 1.5 in the placebo group (see Table 2). This difference was not significant (P = 0.90) and independent from the intake of medication for the treatment of acute migraine attacks (analysis of covariance). Cyclandelate and placebo had the same efficacy concerning most secondary endpoints (Table 3). However, a significant difference in favour of cyclandelate was seen in the parameter ‘disability during migraine attacks’ (P = 0.003). This correlates with a significant decrease in the intensity of attacks, from severe in 54.6% of cyclandelate patients at baseline to 43.6% at the end of treatment (P = 0.006). The corresponding numbers were 59.1% and 53.7% in the placebo group. In terms of global impression cyclandelate was rated better than placebo both by the patients and the treating physicians (Table 4).
Global impression of treatment effect by patients (A) and treating physicians (B), numbers (percent)

Difference is significant in favour of cyclandelate (Mantel–Haenszel–χ2 test, P < 0.001).
Secondary endpoints: mean and (
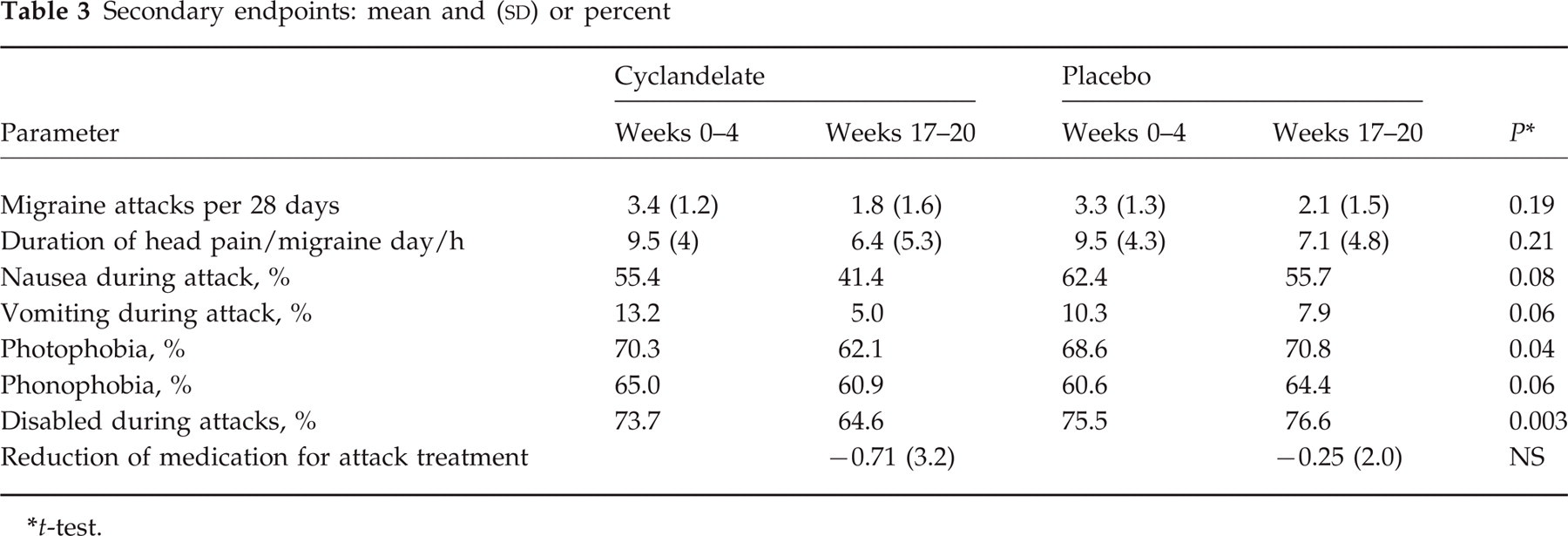
t-test.
Number of migraine days per 28 days; intention-to-treat analysis; last observation carried forward. Week 4: end of placebo run-in
Patient characteristics

All biological parameters (blood pressure, heart rate), biochemistry and ECG remained within the normal range after randomization. The safety analysis included 126 patients in the cyclandelate group and 125 in the placebo group. During the double-blind treatment period 30 patients in the cyclandelate group reported 44 adverse events (AEs) and 32 patients 48 AEs in the placebo group. For the overall number of AEs and AEs related to body systems, no differences were found between verum and placebo. Six serious AEs were reported. In the observation period two patients were hospitalized for hysterectomy and treatment of hypertension. During the placebo run-in one patient suffered a severe traffic accident and another patient was hospitalized because of his headache. In the treatment phase one patient suffered an accident. In another patient a brain tumour was detected leading to death. All together, 83% of patients in the cyclandelate group and 82% in the placebo group rated the tolerability as very good or good.
Discussion
This well designed and conducted trial showed no significant difference between cyclandelate and placebo in the prophylactic treatment of migraine. The trial was performed according to the recommendations of the IHS (13) and powered to find a possible difference. This result is in contrast to earlier studies with cyclandelate (8–10, 14, 15). All these trials had much smaller numbers and were not powered to detect a difference. An earlier study by Diener et al. (11) found no difference between cyclandelate, propranolol and placebo. This trial was difficult to evaluate. During the conduct of the trial sumatriptan was approved. The use of sumatriptan was different between the three treatment arms and might have influenced a secondary endpoint, namely the duration of migraine attacks. In the present trial all important predictors of outcome (migraine frequency, migraine duration and intensity, sex, treatment of acute attacks) were perfectly balanced between the treatment groups.
It is of particular interest, however, that a global impression of efficacy showed a significant effect in favour of cyclandelate, when assessed by both patients and physicians. This indicates that aspects of migraine were positively influenced by cyclandelate which are not picked up by our traditional ways of evaluating the efficacy of migraine prophylactic drugs. The difference in global impression can not be explained by an unblinding to side-effects of cyclandelate. AEs were equally distributed between active treatment and placebo.
In conclusion, this trial failed to show a prophylactic action of cyclandelate in migraine when traditional efficacy parameters were used. The discrepancy of this result with a global outcome measure should trigger further studies to elucidate the possible mechanisms behind this difference.
Footnotes
Acknowledgements
The study was supported by a grant from 3-M Medica, Germany. The protocol was designed by H.C.D.
