Abstract
The administration of nitroderivatives in cluster headache (CH) sufferers is the most reproducible experimental paradigm to induce spontaneous‐like pain attacks. Previous uncontrolled studies have reported that the local use of anaesthetic agents in the area of the sphenopalatine fossa is able to extinguish nitroglycerin (NTG)‐induced pain in CH. The present study, carried out according to a double‐blind placebo‐controlled design, included 15 CH patients, six with episodic CH (mean ±
Introduction
Cluster headache (CH) is a type of primary headache characterized by a peculiar temporal pattern (circadian occurrence of pain attacks, and circannual onset of active phases), an almost constant localization of pain (reported as severe and of non‐throbbing quality), and the presence of ipsilateral autonomic signs and symptoms involving oculo‐cephalic functions (conjunctival hyperemia, lacrimation, ptosis, miosis, nasal stuffiness and/or rhinorrea) (1, 2). The diagnostic criteria of the Inter-national Headache Society (IHS), which are almost exclusively based on the clinical description and periodicity of symptoms, allow for the distinction of two main CH subtypes, namely an episodic form and a chronic form (3).
While the clinical features of CH are usually well‐defined, uncertainty still exists as to the precise pathophysiological mechanisms underlying the disorder. Autonomic dysfunction and cluster pain represent the aspects which have received most study, with considerable evidence suggesting that both phenomena may originate in the central nervous system (CNS). According to such a view, referred to as the ‘central’ hypothesis, CH patients may be characterized by derangement of the hypothalamic‐limbic pathways subserving the autonomic, neuroendocrine and behavioural functions (1, 4, 5).
Various theories have been proposed to explain the generation of pain during CH attacks. An involvement of the sphenopalatine ganglion was originally suggested, following Sluder's first description of a syndrome closely resembling CH and referred to as ‘sphenopalatine ganglion neuralgia’ (6). According to these observations, the sphenoidal sinus has since been regarded as a region of considerable importance in the pathophysiology of CH, and several studies have been carried out on the treatment of this disorder by targeting the spheno-palatine ganglion and its afferent and efferent parasympathetic connections (7–11).
Several agents have been used in CH as a symptomatic approach, and inhaled 100% oxygen, ergotamine tartrate, and the more recently introduced 5‐hydroxytryptamine(5‐HT) receptor agonists, such as sumatriptan, appear to be effective in CH (12, 13). Despite this, in up to 15% of cases CH attacks remain refractory to any medication. The use of substances provided with local anaesthetic properties in CH has so far received little consideration (see 14 for review). Cocaine and lidocaine are two drugs widely used for their anaesthetic effects. Cocaine is provided with sympathomimetic activity via modulation of the uptake of noradrenaline in nerve endings, whereas lidocaine appears to exert its effects via conduction‐blocking properties. Both drugs have been proposed as alternative agents in acute treatment of CH, by local intranasal administration, based on their effects on either spontaneous or provoked attacks in open studies (15–17).
These observations, in addition to their considerable clinical relevance, have further supported the view that the sphenopalatine ganglion may be importantly concerned in CH pathophysiology. However, in none of the studies mentioned above were the effects of both cocaine and lidocaine investigated in the same group of patients. Moreover, all the available data are derived from studies carried out according to uncontrolled experimental designs. We have therefore utilized the commonly adopted model of nitroglycerin (NTG)‐induced headache (18) to test the effects of the intranasal application of lidocaine or cocaine or placebo in a group of CH patients. An additional aim of the present study was to obtain indirect information on the pathogenetic mechanisms of CH, and in particular to elucidate further whether the sphenopalatine region may be actively involved in the process of pain onset during CH attacks.
Methods
The selected study group consisted of 15 patients, 13 males and two females, presenting with headache at the Headache Centre of the Neurological Institute of the University of Pavia, and enrolled consecutively. All patients were suffering from CH in the active phase, according to the IHS criteria (3). Their mean ±
Clinical features of patients; ci = conjunctival injection; nc = nasal congestion; l = lacrimation; p = ptosis; r = rhinorrhea. Attack duration refers to the usual duration prior to the study

At the time of testing, patients were having regular headaches, and none of them had taken any prophylactic medication for at least 1 week prior to the study. After obtaining formal approval from the local ethical committee and informed consent from all patients, a complete clinical history was collected. Then, a thorough othorhinolaryngological examination as well as an anterior rhinoscopy were preliminarly performed by an experienced othorhinolaryngologist, in order to rule out the presence of endonasal diseases or malformations. Otorhinolaryngological supervision was also provided during all test procedures.
Patients then underwent a standard headache‐induction test, carried out by administering NTG (trinitrine) 0.9 mg sublingually in headache‐free conditions. This dose, currently used at our department, has been found to induce typical pain attacks in about 70% of CH patients in active phase, a percentage similar to that reported by Ekbom with 1 mg NTG (18). Those patients experiencing a typical attack following NTG administration underwent the test in two further sessions. An interval of at least 2 days was allowed between the three study sessions. During each test, patients were resting in bed in a supine position, and their cardiopressor parameters (heart rate, arterial blood pressure) were continuously recorded using a vital signs monitor (Dynamap, Kriticon, Florida, USA).
The time of onset of pain, the possible occurrence of general autonomic symptoms, and any changes in the degree of conjunctival injection, width of the palpebral fissure, pupillar diameter and nasal congestion (if present) were also recorded using a dedicated chart. Patients were asked to score the intensity of any provoked headache by means of a visuo‐analogic scale ranging from 0 to 10 (0 = no pain, 10 = unbearable pain).
In all cases, once a spontaneous‐like headache attack became established (pain intensity of at least 5 on the scale, usually 5–10 min after the very onset of pain), patients were asked to extend their head 45 degrees. A cotton swab previously immersed in a 10% solution of cocaine hydrochloride (1 ml, mean amount of application 40–50 mg), or 10% lidocaine (1 ml), or saline was then introduced into the nostrils, placed in the region corresponding to the sphenopalatine fossa of both sides, and left there for at least 5 min. We chose to administer the drugs on both sides since in the previous studies treatments used ipsilaterally to the symptoms had failed in relieving pain in a variable proportion of patients. Procedures were made according to a double‐blind design; all nine patients received randomly the three treatments and completed the study. The time elapsed until obtaining pain relief, and the magnitude of headache and autonomic symptoms were assessed and reported in the chart. The pain extinction time was recorded for all treatments; patients were defined as responders when a decrease of 50% or over in pain intensity was observed after treatments.
The administration of local anaesthetics was never repeated, even in the case of incomplete relief after several minutes. However, for ethical reasons, patients were free to ask for a rescue treatment (sumatriptan 6 mg s.c.) whenever required. At the end of each test, patients were also instructed to contact the Headache Centre the following day, to report on the status of their headache.
Statistical analysis was made using the non‐parametric Wilcoxon ranks test to compare to baseline the VAS values at different times after treatments. The Mann–Whitney U‐test was used to compare pain intensity at different times between treatments, as well as the pain extinction times of different treatments. Differences were considered significant if P < 0.05. Data are expressed as mean ±
Results
During the first session of the study, an overall number of 15 induction tests were performed. As shown in Table 2, nine patients (two with the episodic form, seven with the chronic form) experienced a typical, spontaneous‐like attack on the usual side, occurring in all cases within 45 min (mean ±
Clinical features of the nine NTG‐responsive patients during induction test. See Table 1 for abbreviations

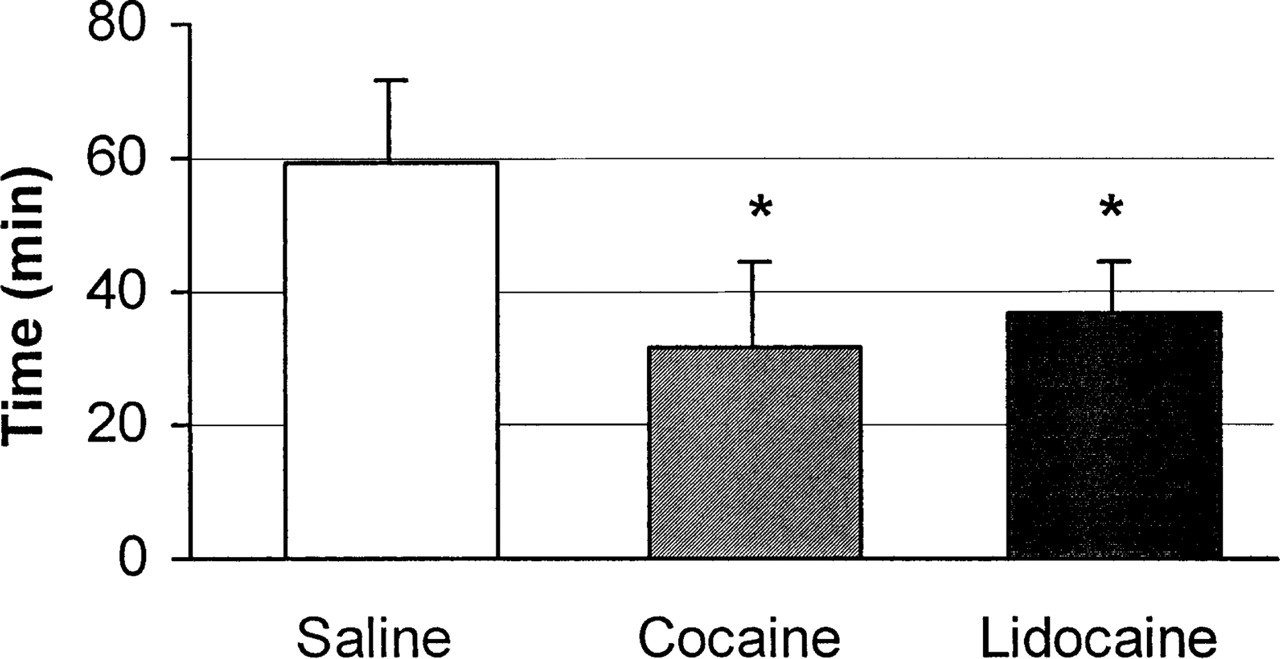
As shown in Fig. 1, all patients responded promptly to both anaesthetic agents, as pain intensity decreased within the first 2 min and reached 3.5 for cocaine and 4 for lidocaine after 5 min (both P < 0.01 vs. respective baseline values). In the case of saline, pain intensity further increased after intranasal application (P < 0.01 vs. baseline, P < 0.001 vs. both drugs). There was no significant difference between cocaine and lidocaine at all times, although a trend towards a better effect of cocaine was observed after 5 min (P = 0.07 vs. lidocaine). Complete cessation of pain (Fig. 2) occurred after 31.3 ± 13.1 min for cocaine, 37.0 ± 7.8 min for lidocaine, and 59.3 ± 12.3 min for saline (P < 0.01 saline vs. both drugs). The duration of pain in the case of saline treatment was not significantly different from that of the usual, spontaneous attacks. Fig. 3 shows the number of pain‐free patients and number of ‘responders’ at different times. After 10 min, there was no pain‐free patient with any treatment, while responders were 3/9 for cocaine, 1/9 for lidocaine and 0/9 for saline; after 35 min all patients were pain‐free with either cocaine or lidocaine, while all were still experiencing pain with saline.

Mean ±
Mean ±

Number of pain‐free patients and responders at different times following treatments. □= pain‐free; b= responders;▪= pain.
None of the patients asked for rescue treatment, even though pain intensity was as high as 9 in one case treated with saline. Following the administration of both anaesthetics, a decrease was also observed in the intensity of autonomic signs and symptoms, which paralleled the reduction in pain severity (data not shown). Nasal congestion was attenuated more slowly, and complete resolution was obtained 5–10 min later than the other complaints. However, congestion of turbinates was mild in all patients, and in no case was the use of intranasal adrenergic decongestionants required to facilitate the access of solutions. Two patients found unpleasant the taste of the substance later identified as lidocaine, but the administration of both lidocaine and cocaine was generally well tolerated, and did not result in any significant side‐effects. None of the patients experienced a spontaneous CH attack during the 5 h following lidocaine and 6 h following cocaine administration.
Discussion
The application of cocaine to the area corresponding to the sphenopalatine fossa has been shown to be effective at extinguishing pain attacks in patients suffering from sphenopalatine neuralgia or other poorly defined related disorders (6, 7), which were likely to be CH forms. Other authors have also reported successful pain treatment in patients suffering from episodic or chronic CH using intranasal lidocaine during spontaneous attacks (16, 17). It is well established that spontaneous‐like CH attacks can be triggered in predisposed individuals by the sublingual administration of NTG (18), which still represents the most reliable and reproducible paradigm of induced headache of the vascular type in humans. Also in migraine sufferers, the sublingual (19) or systemic (20) administration of NTG is indeed able to precipitate headache attacks, whose features resemble those of the spontaneous episodes and fulfil the IHS criteria for migraine. In addition to its effect on spontaneous attacks, intranasal lidocaine has been reported to be useful in relieving NTG‐induced pain in CH patients (16). The effectiveness of locally applied cocaine under the same experimental paradigm has also been observed (15).
It is currently accepted that NTG acts via the production of the potent vasodilating mediator nitric oxide (NO) at the vascular level (21). Over the last few years, it has been proposed that patients with vascular headaches may display excess NO production and/or increased reactivity to the activation of the nitrinergic pathways (22). While the intimate nature of these processes remains unclear, it has been proposed that NO may be involved with a crucial role in the mechanisms leading to pain generation in both CH and migraine; in addition to its effect at the endothelial level, NO may also act centrally (brainstem nuclei), thereby participating in the processes underlying the onset of neurovegetative signs and symptoms in migraine and CH (23). Similar to CH patients, NTG‐induced attacks in migraine sufferers have been recently shown to be significantly relieved by the intranasal application of lidocaine (24), further suggesting that these headache disorders may share common pathogenetic mechanisms.
The present study confirms, in a double‐blind, placebo‐controlled design, the previous observations that cocaine and lidocaine acting in the sphenopalatine region are both effective at reducing pain intensity in CH attacks (15–17). Compared to the previous studies, the effect of anaesthetic drugs was found in 100% of our patients. Whether this depends on the fact that in our study lidocaine and cocaine were administered bilaterally is not known. The issue of CH pain laterality affecting the clinical response to anaesthetic administration has been previously addressed by Kudrow et al. (24). In all of our nine NTG‐responsive patients, appearance of symptoms within or between cluster periods concerned consistently one side. In a previous study, the variable pattern of pain side resulted in the total failure of the bilateral alcohol infiltration of the sphenopalatine ganglion (8). A similar unsuccessful treatment with lidocaine was reported in the case of bilateral migraine (24). On the basis of our findings, it would therefore appear that the bilateral nasal administration of lidocaine and cocaine in CH patients would prove effective regardless of pain localization. Also in cases of strictly consistent pain side within and/or between clusters, anaesthetization of parasympathetic fibers of both ganglia may be beneficial.
Another possible explanation for the rate of pain relief found in our patients may be related to the procedure of drug administration used in our study, which involved anterior rhinoscopy. In the case of the administration of drugs by drop instillation, or using spray preparation, drugs may indeed face restricted access to the most critical region of the nasal mucosa (i.e. that corresponding to the sphenopalatine area), due to nasal obstruction, an accompanying phenomenon which is known to occur frequently in CH (2). At the same time, the different means of drug administration (cotton swab as opposed to nasal dropper or spray) may also account for the fact that while the latencies of cocaine and lidocaine‐induced attenuation of pain in our patients was prompt and similar to that previously reported (15–17), complete pain disappearance occurred considerably later.
The possible mechanisms of action of lidocaine and cocaine in easing CH pain are a matter of current debate. A reduction of afferent nociceptive inputs to the spinal trigeminal nucleus, as well as the block of nerve terminals of the glossopharyngeal nerve have been proposed (17). However, the hypothesis which has received most consideration is that both anaesthetic agents may act at the level of the sphenopalatine ganglion (16), which is thought to be importantly involved in pain generation (7–11). In particular, at variance with lidocaine (which is devoid of sympathomimetic activity), cocaine exerts systemic noradrenergic effects, which may account for the slightly more rapid effect compared to lidocaine observed in this study.
Cocaine has definite vasoconstrictive properties (25), and at least 50% of its peak plasma levels persist for approximately 3 h after intranasal administration (26). This may also account for the prophylactic‐like effect reported by our patients after cocaine application (pain‐free interval of at least 6 h), similar to that previously reported by Ekbom (18). However, both the findings of previous studies (15, 16) and the present observations suggest that its therapeutic effects may be mainly due to its anaesthetic activity. In this respect, it is of interest that the analgesic effect of cocaine has been recently found to involve activation of endogenous opioid peptide activity in the brain, whereas the activation of NO pathways (such as that following NO precursors) results in inhibition of the opioid system (27).
Our findings are also in agreement with the recently reported effectiveness of the sphenopalatine ganglion blockade in CH patients using radio‐frequency lesioning (28). Recent experimental evidence further supports the role of the sphenopalatine ganglion in the pathogenesis of CH. Stimulation of ganglionic parasympathetic fibres has been found to result in vasodilation of pial vessels both in the animal and in the human (29). Moreover, it has been demonstrated that in rats the large cerebral vessels are surrounded by nerve fibres originating in the sphenopalatine ganglion; these fibres contain the NO‐producing enzyme NO‐synthase (NOS), and are known to mediate vasodilatory phenomena (30, 31).
In conclusion, while further suggesting that the sphenopalatine ganglion participates in the mechanisms of pain, our findings indicate that local anaesthetic agents are effective on induced CH attacks. The greater effectiveness of both cocaine and lidocaine seen in the present study compared to previous reports may pertain to the bilateral mode of nasal administration, as well as to increased access to the sphenopalatine area allowed by the use of rhinoscopy. While the present findings need to be confirmed in spontaneous CH attacks, lidocaine and cocaine may be considered as useful alternatives to conventional drugs in the symptomatic treatment of this disorder. In addition, intranasal delivery of headache medications is presently receiving increased attention (14), as it offers considerable advantages, due to the rapid onset of action, comfortable self‐administration, and the possibility for the drug to be absorbed in spite of symptoms (nausea, vomiting) which limit or preclude oral intake.
Bearing in mind the risks of cocaine addiction (which are further increased in a highly disabling disorder like CH) (32), the established relationship between cocaine abuse and the development/worsening of vascular headaches (33, 34), and the relatively poor availability of the drug, the use of lidocaine appears to be by far preferable. However, in view of the absence of major acute side‐effects, the administration of cocaine, if restricted to particular cases (i.e. only patients with episodic forms of CH, and refractory to all of the current symptomatic treatments), may also be useful.
Footnotes
Acknowledgement
We wish to thank Dr E. Marchioni (Pavia) for his helpful advice with regard to statistical analysis of data.
