Abstract
Background
High frequency (HF) stimulation of the sphenopalatine ganglion (SPG) is an emerging abortive treatment for cluster headache (CH) attacks. HF SPG stimulation is thought to exert its effect by physiologically blocking parasympathetic outflow. We hypothesized that low frequency (LF) SPG stimulation may activate the SPG, causing increased parasympathetic outflow and thereby provoking cluster attacks in CH patients.
Methods
In a double-blind randomized cross-over study, seven CH patients implanted with an SPG neurostimulator were randomly allocated to receive HF or LF stimulation for 3 min on 2 separate days. We recorded headache characteristics and autonomic symptoms during and after stimulation.
Results
Six patients completed the study. Three out of six patients (50%) reported ipsilateral cluster-like attacks during or within 30 min of LF SPG stimulation. These cluster-like attacks were all successfully treated with the therapeutic HF SPG stimulation. One out of six reported a cluster-like attack with 3 min HF SPG stimulation, which was also successfully treated with continued HF therapeutic SPG stimulation.
Discussion
LF SPG stimulation may induce cluster-like attacks with autonomic features, which can subsequently be treated by HF SPG stimulation. Efferent parasympathetic outflow from the SPG may initiate autonomic symptoms and activate trigeminovascular sensory afferents, which may initiate the onset of pain associated with CH.
Keywords
Introduction
Cluster headache (CH) is the most frequent and painful of the trigeminal autonomic cephalalgias affecting approximately 0.12% of the general population (1). CH is a devastating disorder, especially in the chronic form where attacks persist for more than 1 year without remission or with remissions lasting less than 1 month (2). The prominent autonomic symptoms (conjunctival injection, lacrimation, nasal congestion or rhinorrhea, eyelid or periorbital edema, forehead and facial sweating, miosis or ptosis) during CH attacks are believed to be caused by increased parasympathetic outflow from the superior salivatory nucleus (SSN) via the sphenopalatine ganglion (SPG) (3,4).
Whether parasympathetic outflow during CH attacks is an epiphenomenon or an integral part of the pain generation is still unknown (5). Blockade of the SPG as a pain treatment has been employed for years (6), and SPG blocks or ablations have been used to alleviate refractory CH (7,8). Emerging studies are targeting the SPG by electrical stimulation as a possible novel therapy strategy in CH. Recently, a multicenter trial was conducted to investigate the therapeutic response in chronic CH patients to high frequency (HF) SPG stimulation using the Autonomic Technologies, Inc. (ATI) Neurostimulation System. An interim analysis undertaken in 20 patients showed that 85% achieved abortive pain relief in ≥50% of attacks treated with electrical SPG stimulation and/or a reduction in average attack frequency to ≤1 attack/week (9). HF SPG stimulation may primarily activate parasympathetic neurons or pre/post ganglionic parasympathetic nerve fibers well above the intrinsic firing rates, which may physiologically block parasympathetic outflow and result in an acute effect on head pain and autonomic symptoms. The HF therapeutic SPG stimulation used in the multicenter study was applied for at least 15 min during an attack. In contrast, low frequency (LF) SPG stimulation may correspond to a more intrinsic pattern, which would possibly lead to increased parasympathetic outflow and subsequent head pain. The rationale for LF stimulation is based on animal studies demonstrating ipsilateral dilatation after SPG activation using electrical stimulation (range 5–60 Hz) of the middle cerebral artery (MCA) (10), small pial arteries (11), internal carotid artery (ICA) (10), anterior carotid artery (ACA) (10) and increased ipsilateral cortical blood flow (12).
In the present study, we hypothesized that activation of the SPG with LF stimulation would induce cluster-like attacks and that induced attacks would be effectively treated with HF therapy stimulation. To test this hypothesis we performed a double-blind randomized cross-over study to investigate headache characteristics following LF (5 Hz) and HF (80–120 Hz) SPG stimulation in seven patients with CH. In addition, we examined the effect of LF and HF SPG stimulation on cranial hemodynamics to assess changes in the cranial vasculature induced by modulating parasympathetic efferent activity.
Methods
Participants
We recruited seven CH patients (2F/5M, mean age 49, range 35–63 years) (Table 1) enrolled in the Pathway CH-1 (CIP-001) study: Sphenopalatine Ganglion Stimulation for the Acute Treatment of Cluster Headache (clinicaltrials.gov: NCT01255813). Briefly, the Pathway CH-1 study is a multicenter, randomized, controlled, interventional, prospective study, which evaluates the safety and performance of the ATI Neurostimulation System when used for SPG stimulation in chronic CH patients who are inadequately treated with available therapies. The rechargeable, multichannel system consists of a small inductively powered SPG neurostimulator (Figure 1), which is powered and controlled by an external remote controller. The current study was initiated in the open label phase of the Pathway CH-1 study in which the patients could treat their CH freely and without limitations. We enrolled all patients irrespective of their headache response to HF therapeutic stimulation in the multicenter study.
The ATI Neurostimulator, a minimally invasive, rechargeable, multichannel neurostimulator, which is implanted transorally and located within the midface. The electrodes are positioned within the pterygopalatine fossa in close proximity to the sphenopalatine ganglion (SPG). Clinical characteristics. Clinical characteristics in cluster headache patients with sphenopalatine ganglion neurostimulator. F: female; M: male; Epi: cluster headache debut episodic; Chr: cluster headache debut chronic.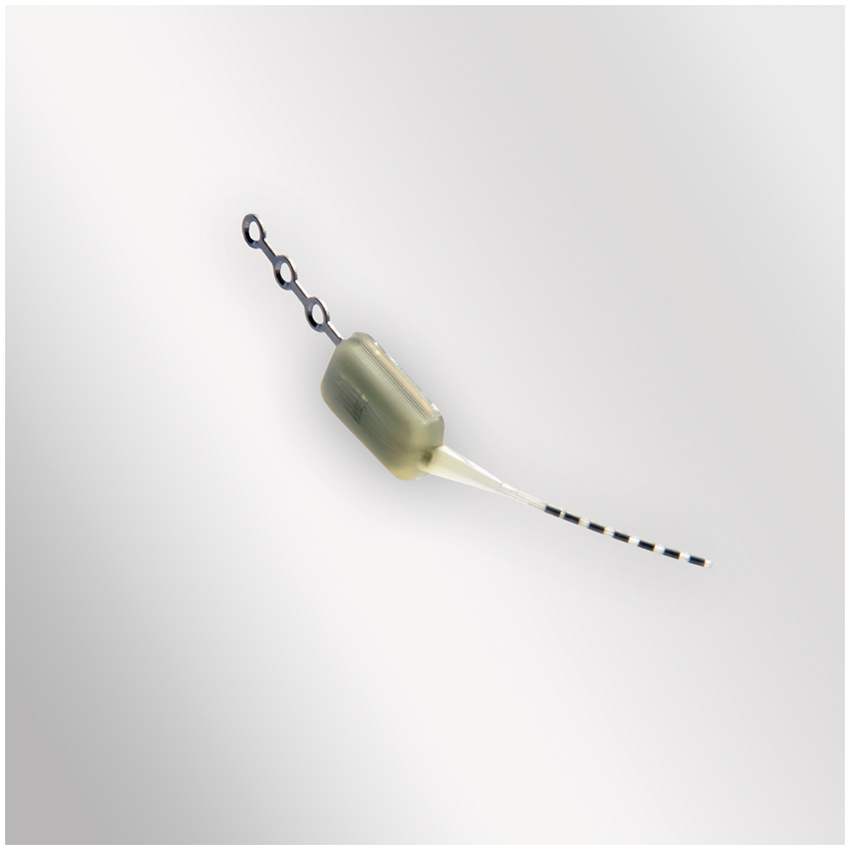

Standard protocol approvals, registrations, and patient consents
The study was approved by the Ethics Committee of the County of Copenhagen (H-2-2010-092), the Danish Medicines Agency (LMST2010123614) and registered at Clinicaltrials.gov (ID: NCT01359631). All patients gave informed consent to participate in the study. The study was followed in accordance with the Helsinki Declaration of 1964, as revised in Edinburgh in 2000.
Experimental procedures
All procedures were performed in-hospital in a quiet room at a temperature of 25℃. The patients were placed in the supine position and allowed to rest for 15 min before baseline recordings were done. Cluster headache occurring less than 3 h prior to initiating the study resulted in postponement of the experimental procedures. Patients were allowed to treat any headaches with the SPG neurostimulator, and/or with their typical acute therapy (oxygen inhalation, sumatriptan and over the counter rescue medication). After each experiment, upon discharge from the hospital, the patients were also asked to complete a headache diary every hour until 12 h after the start of the experimental stimulation procedure.
Each patient was investigated on 2 separate days, where they received either HF or LF stimulation (Figure 2). During each application of SPG stimulation (HF or LF) the stimulation was applied for 3 min and amplitude, pulse width and electrode configurations were held constant, this is referred to as HF or LF experimental stimulation. The applied HF value had already been individually determined during the open-label period in the Pathway CH-1 study as the most suitable frequency for each patient; however, patients may or may not have achieved acute relief with HF therapeutic stimulation. Two investigators (MB or SG) programmed the neurostimulator according to a balanced randomization code, whereas the patients and investigator (HWS) recording headache quality, autonomic symptom responses and vascular changes were blinded to the neurostimulator setting. In-hospital headache characteristics and autonomic symptoms (AS) were recorded at baseline, 3 min, 10 min, 20 min, 30 min and 60 min after stimulation. Velocity of the middle cerebral artery (VMCA), blood pressure and heart rate (HR) were recorded at baseline and at 3 min after start of stimulation. Continuous wave near infrared spectroscopy (NIRS) was recorded at baseline and continuously until 3 min after start of stimulation.
Diagram of the experimental design. LF: low frequency stimulation at 5 Hz; HF: high frequency stimulation ranging from 80 to 120 Hz depending on the individual setting in each patient; VMCA: velocity middle cerebral artery; oxyHb: oxygenated hemoglobin; deoxyHb: deoxygenated hemoglobin; AS: autonomic symptoms.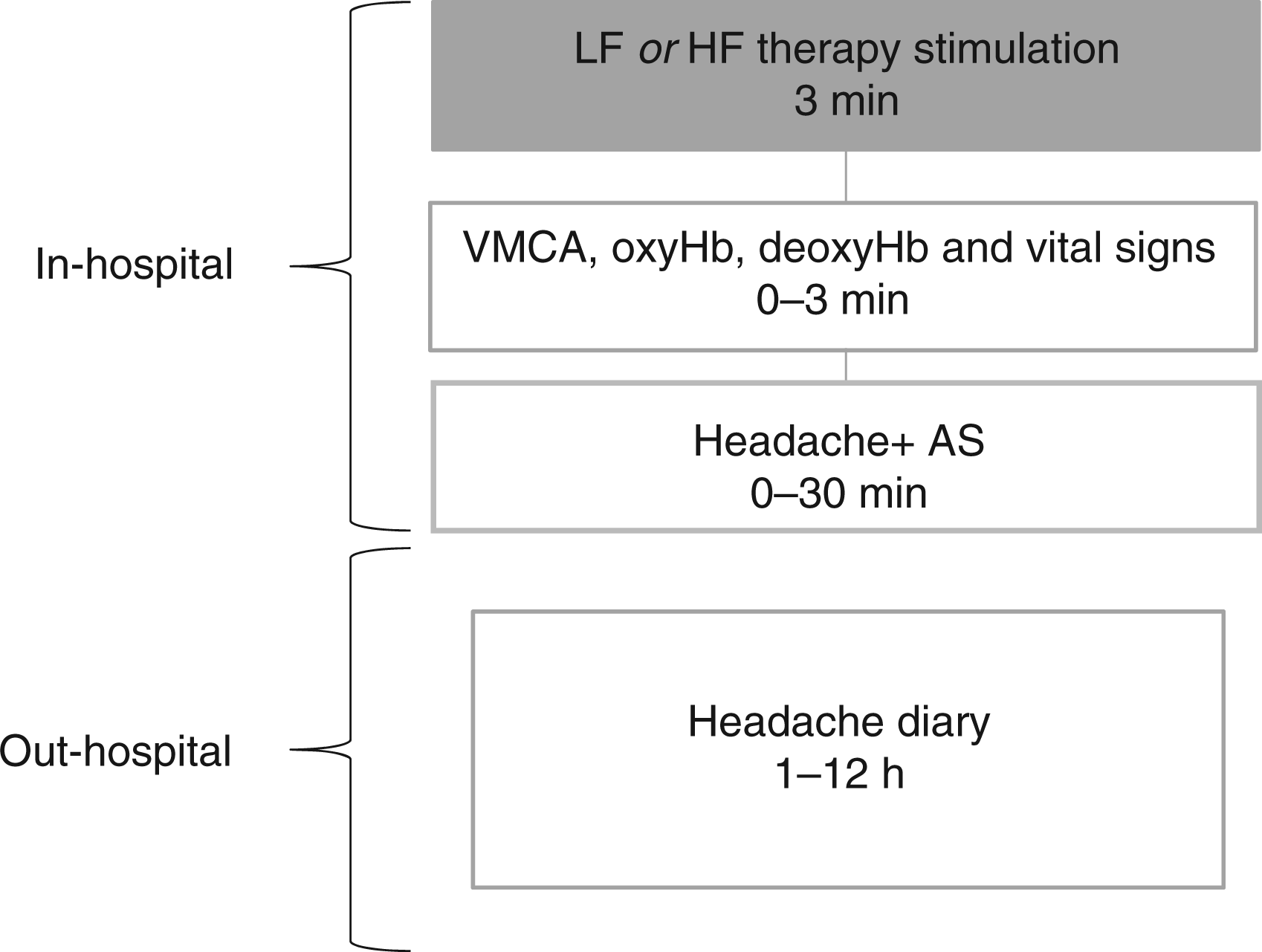
Cluster-like attacks
CH criteria were applied according to the IHS criteria for CH (2). Headache intensity was rated on a categorical pain scale (CPS) from 0 to 4 with; 0-no pain, 1-mild, 2-moderate, 3-severe and 4-very severe. Localization, quality, autonomic features and whether headache mimicked usual CH were also assessed. The out-hospital diary included headache characteristics and autonomic features, whether headache mimicked usual cluster attacks and any rescue medication (including SPG stimulation). The CPS was chosen as the patients were used to rating headache intensity according to this scale from the Pathway CH-1 study.
Vital signs
Blood pressure and heart rate were measured every 10 min using an auto-inflatable cuff and finger pulse oximeter (ProPac Encore®; Welch Allyn Protocol, Beaverton, OR, USA).
Mean middle cerebral artery blood flow velocity (VMCA)
VMCA was recorded bilaterally with transcranial Doppler (TCD) with hand-held 2 MHZ probes (Multidop X; DWL, Sippelingen, Germany) as previously described (13). In brief, four recordings were taken and averaged at each time point. Every TCD recording was performed by the same trained physician (HWS). End tidal CO2 (PetCO2) was recorded simultaneously with TCD recordings using an open mask that caused no respiratory resistance (ProPaq Encore®; Welch Allyn Protocol). All recordings were performed by HWS.
Continuous wave near infrared spectroscopy
Measurement of oxygenated and deoxygenated hemoglobin (oxyHb, deoxyHb) was performed using continuous wave NIRS (NIRS2; TechEn Inc, Milford, MA, USA). The NIRS optodes were placed bilaterally on the forehead with one source (two wavelengths: 690 nm and 830 nm) and two detectors on each side, avoiding the midline sinus. The two detectors were placed at distances of 1 and 3 cm lateral from the source. Thus, the 1 cm detector was measuring frontal scalp territory primarily supplied by the superficial temporal artery, whereas the 3 cm detector was measuring mixed contribution from the scalp territory and the frontal cortex territory supplied by the anterior cerebral artery (ACA) and middle cerebral artery (MCA). All data processing was performed in MATLAB (The MathWorks, Inc., Natick, MA, USA) by JS who was blinded to the stimulation frequencies on the 2 study days. Intensity variations at 690 and 830 nm were converted into relative changes in oxyHb and deoxyHb using the modified Beer-Lambert law (14) with a differential pathlength factor of 6 at both wavelengths. To minimize contribution of the scalp physiology, the data from each 1 cm channel were regressed out from the corresponding measurement at 3 cm (15). After regression, the signal at 3 cm is considered to originate mostly from cortical territory.
Data presentation
All headache data are presented as individual values. Early phase headache is defined as 0–60 min and delayed phase headache as >60 min–12 h after the beginning of stimulation. All vascular values are presented as mean changes from baseline in percent ± SEM. The changes were not tested statistically as the sample number was low. To do so would require inclusion of 12 or more patients in this type of double-blind experimental provocation studies if statistical tests are applied (16).
Results
Six patients completed the study. One patient (patient 4) was excluded from the analysis because of technical issues on the LF day when applying stimulation.
The patients’ individual attack frequency before and after receiving their implantable neurostimulator are shown in Table 1. Two patients had not experienced a spontaneous cluster attack for more than 1 month, and two patients had less than three attacks in the month prior to the present study.
Low frequency 5 Hz stimulation
Headache characteristics after 3 min of sphenopalatine ganglion stimulation.

HF: high frequency stimulation; LF: low frequency stimulation; orb: orbital ipsilateral to neurostimulator; tem: temporal ipsilateral to neurostimulator; thro: throbbing; pre: pressing; uk: unknown; inj: ipsilateral conjuctival injection; rhi: ipsilateral rhinorrhea; oed: ipsilateral eyelid edema; res: a sense of restlessness or agitation; phot: photophobia; lac: ipsilateral lacrimation; swe: ipsilateral forehead and facial sweating; phon: phonophobia; nau: nausea; pto: ipsilateral ptosis; CH: cluster headache attack as patients usual CH attack; SPG: sphenopalatine ganglion stimulation therapy; ox: oxygen inhalation; suma: sumatriptan.
Three out of six patients (patients 1, 2 and 5) reported early phase CH-like attacks with autonomic features after LF experimental stimulation. Two patients (patient 1 and 2) reported attacks starting during the 3 min stimulation and one patient (patient 5) 30 min after the start of stimulation. Each of the three patients that developed cluster-like attacks applied HF therapeutic stimulation (120 Hz). Two patients reported pain freedom and cessation of autonomic symptoms (patients 1 and 2) within 10 min, whereas one patient (patient 5) reported pain relief (from 3 to 1 on the CPS) and cessation of autonomic symptoms within 10 min after HF therapeutic stimulation.
Three patients (patients 2, 5 and 7) reported delayed cluster-like attacks at 2–4 h after LF experimental stimulation start. All patients treated the attacks with HF therapeutic stimulation (range 100–120 Hz) and then reported pain freedom and cessation of autonomic symptoms after a mean of 175 min (range 45–300 min) after HF therapeutic stimulation (Table 2). Patient 7 experienced pain relief 60 min after HF therapeutic stimulation (CPS 4 to 2).
High frequency stimulation
Two out of six patients (33%) reported ipsilateral CH-like attacks after HF experimental stimulation (Table 2).
One patient (patient 3) reported an ipsilateral CH-like attack with autonomic symptoms at 3 min (80 Hz) after the start of stimulation. Seven min after the end of the HF experimental stimulation the patient applied HF therapeutic stimulation (80 Hz). The duration of stimulation was approximately 10 min and thereafter the patient was pain free and without autonomic symptoms.
Patient 3 also experienced a cluster-like attack associated with restlessness but without autonomic features at 4 h after start of stimulation. The attack duration was 3 h and according to the patient it did not mimic a usual cluster attack and therefore was not treated. One patient (patient 2) reported a cluster-like attack with ipsilateral autonomic symptoms in the delayed phase at 120 min after HF experimental stimulation was started. The headache mimicked usual cluster-like attacks and was successfully treated with HF therapeutic stimulation (120 Hz). The patient was pain free and without autonomic symptoms 15 min after pain onset.
Vascular recordings
Because of technical problems, VMCA and vital signs were missing in patient 2 during the HF experimental stimulation day and in patient 6 during the LF experimental stimulation day. VMCA data was not recorded on the non-stimulated side in patient 7 on the LF day because of an insufficient signal. As a result of a technical problem, HR data were missing in subject 1 on the LF day. NIRS data were excluded in patient 1 during the HF day because of motion artifacts.
The effects on VMCA, oxyHb and deoxyHb and vital variables following LF and HF experimental stimulation are shown in Figures 2–4. VMCA decreased following stimulation, which was most pronounced after LF experimental stimulation on the ipsilateral side (−14.0 ± 6.6%). Superficial NIRS recordings showed an increase (3.0 ± 1.1 µM) in oxyHb following stimulation, which was most pronounced on the ipsilateral side following HF experimental stimulation. The NIRS recordings primarily assessing cortical vascular changes showed a moderate increase, which was most pronounced (1.7 ± 0.6 µM) on the ipsilateral side following HF experimental stimulation. Recordings of heart rate revealed a decrease (−6.2 ± 3.1%) with HF experimental stimulation (Figure 5). End tidal CO2 decrease was most pronounced (−2.7 ± 1.3%) with LF experimental stimulation (Figure 5). There was no MAP change from baseline of more than ± 2% (Figure 5).
Changes from baseline (%) for mean middle cerebral artery velocity (VMCA) at 3 min following high frequency (HF) therapy (range 80–120 Hz) and low frequency (LF) stimulation at 5 Hz on the ipsilateral and contralateral side of the SPG neurostimulator. Error bars show SEM. Most pronounced response (−14.0 ± 6.6%) was found following LF stimulation on the ipsilateral side. Continuous wave near infrared spectroscopy changes with normalized relative oxygenated hemoglobin (HbO) and deoxygenated hemoglobin (HbR) values during 3 min high frequency (HF) therapy stimulation (range 80–120 Hz) and low frequency (LF) stimulation at 5 Hz. (a) 1 cm optodes measuring superficial skin changes. (b) 3 cm optodes measuring cortical changes. Y-axis is micromolar (µM) concentration. Error bars show SEM. At 1 cm (a), the most pronounced response 3.0 (±1.1) was found on the ipsilateral side following high frequency stimulation. At 3 cm (b), there was a moderate increase, which was most pronounced (1.7 ± 0.6 µM) on the ipsilateral side following HF stimulation. Changes from baseline (%) for heart rate, end tidal CO2 and mean arterial blood pressure (MAP) at 3 min following high frequency (HF) therapy (range 80–120 Hz) and low frequency (LF) stimulation at 5 Hz on the ipsilateral and contralateral side of the SPG neurostimulator. Error bars show SEM. Most pronounced heart rate change was −6.2 ± 3.1% at HF stimulation. Most pronounced end tidal CO2 −2.7 ± 1.3% at LF stimulation. There was no MAP change from baseline of more than ±2%.


Discussion
The major finding in the present study was that 50% of patients reported ipsilateral CH-like attacks during or within 30 min following LF experimental stimulation of the SPG, and that all of these attacks were successfully treated with HF therapeutic SPG stimulation. Here we shall present and discuss possible mechanisms of generation of CH attack by LF SPG activation, and mode of action of HF therapeutic SPG stimulation.
The pathophysiological mechanisms of CH are complex (4). One of the cardinal features of CH is the prominent autonomic symptoms (2). It is, however, not known whether these autonomic symptoms are caused by trigeminal nociception per se (clinically presented with severe unilateral pain) or if parasympathetic outflow plays a primary role by activating trigeminal afferents and thereby causing or sustaining head pain during cluster attacks. Cluster-like attacks rapidly induced in the current study by LF experimental stimulation may be explained by the release of neurotransmitters from parasympathetic efferents, which in turn directly or indirectly activate or modulate trigeminal nociceptors. Previous clinical evidence has demonstrated that blockade or ablation of the sphenopalatine ganglion reduces attack frequency (17) and provides pain relief (18) in individuals with CH. Previous experimental models have demonstrated: 1. activity in meningeal nociceptors can be modulated by efferent parasympathetic SPG fibers (19); 2. the parasympathetic neurotransmitter acetylcholine can activate unmyelinated C fibers (20); 3. the parasympathetic neurotransmitter nitric oxide (NO) can sensitize peripheral dural nociceptors (21); and 4. activation of SPG can lead to dural plasma protein extravasation (22). Whether LF SPG stimulation could induce cluster attacks in subjects without prior history of CH is unknown. Ongoing studies using SPG stimulation for augmentation of CBF in patients with acute ischemic stroke (23) might elucidate this question, but so far no data are available.
One subject (patient 3) did in fact experience a cluster-like attack induced by 3 min of HF experimental stimulation and subsequently achieved pain cessation after 10 min using HF therapeutic stimulation. It is possible that the therapeutic effect of HF stimulation may be caused via physiological block of parasympathetic activity as a result of depletion of stored neurotransmitters (24). Therefore, HF stimulation may initially cause activation of the SPG before causing a physiological block of the parasympathetic activity in some patients. The duration of the applied HF stimulation to the SPG may be an important factor in determining the effect of the applied stimulus, e.g. short stimulus duration may activate the system, whereas a longer duration stimulus may physiologically block the system. Additionally, the state of the system may also be important in determining the effect of the applied stimulus, e.g. if the stimulus is applied during an attack or in the absence of an attack. In the CH-1 study, HF therapeutic stimulation was applied during the attack, and in this experimental study, LF and HF experimental stimulation was applied in the absence of an attack. LF experimental stimulation was followed by delayed attacks in three out of six patients, whereas two out of six patients reported delayed attacks following HF experimental stimulation. There are no previous experimental data to explain these delayed attacks, which could be caused by a cascade of events following LF or HF stimulation, including spontaneous or recurrent attacks. At present we can only speculate on the nature of these attacks, which should be investigated in future studies.
Recently, Akerman et al. (25) showed that stimulation of the SSN using frequencies between 10 and 50 Hz leads to firing of second order neurons in the trigeminal nucleus caudalis (TNC) in rats, which can be explained by an increased parasympathetic outflow via the SPG. Interestingly, the model included a response to oxygen inhalation, known to abort CH pain (26) and showed that oxygen therapy had no effect on direct activation of trigeminal afferents. However, following SSN stimulation, which leads to parasympathetic outflow via the SPG, oxygen inhibited neuronal firing in the TNC and attenuated corneal/lacrimal sac/duct blood flow. This suggests an antinociceptive mechanism by oxygen leading to a blockade of the effects of parasympathetic outflow, possibly because of faster degradation of nitric oxide (27). Collectively, previous and the present experimental data suggest that cranial parasympathetic outflow may in fact initiate and/or sustain head pain during CH attacks.
It is important to note that sensory nerve fibers from the maxillary division of the trigeminal nerve, which pass through the SPG, theoretically may be activated by both LF and HF. No subjects reported pain from the maxillary distribution during stimulation, so it is unlikely that electrical stimulation of the SPG directly activates pain fibers at the SPG. However, sensory nerve fibers from the maxillary division converge in the TNC on second order neurons with afferents from the first division of the trigeminal nerve, which may hypothetically lead to antinociceptive modulatory changes. Thus, a similar antinociceptive mechanism in CH has been suggested to apply to occipital nerve stimulation (ONS) (28), as occipital nerve fibers also converge on second order neurons with afferents from the first division of the trigeminal nerve in the TNC.
CH shows both circadian and circannual periodicity suggesting a role for the hypothalamus (29), and imaging studies have shown activation in the ipsilateral posterior hypothalamus during CH attacks (30). Nociceptive information from the dura and facial skin are conveyed to the hypothalamus directly via the so-called trigeminohypothalamic tract (THT) (31). Furthermore, hypothalamic deep brain stimulation using 180 Hz in CH patients provokes increased blood flow in both the ipsilateral posterior inferior hypothalamic grey matter, at the site of the neurostimulator tip, and in the ipsilateral trigeminal system (32). However, stimulation of the hypothalamus has never been shown to induce cluster attacks, and LF stimulation at the hypothalamus has, to our knowledge, not been investigated in CH patients. Deep brain stimulation of the posterior hypothalamus has been used in selected patients with chronic CH (33), but there may be a delay of up to 3 months after initiation of stimulation before the beneficial effect emerges (33). Interestingly, most CH patients in our study have used the ATI Neurostimulation System frequently because of a high number of CH attacks. This frequent acute application of SPG neurostimulation therapy also may induce long-term preventive benefits as well. Thus, acute application of SPG stimulation may cause a reduction in the frequency of CH attacks because of slow central neuromodulatory changes, similar to what might occur after occipital nerve stimulation (34). However, the immediate abortive changes following SPG stimulation in the present study are not likely to directly involve central neuromodulatory changes. Furthermore, it seems unlikely that HF stimulation induces a conduction block in the SPG, as this is usually obtained by HF stimulation in the kHz range (35).
Perivascular parasympathetic nerve fibers originating from the SPG are found at rostral pial arteries, the internal carotid artery (ICA) (36), the STA (37) and the middle meningeal artery (MMA) (38). The present data indicate that LF SPG activation using electrical stimulation in humans induces dilatation of the ipsilateral MCA as shown indirectly by a decrease in VMCA. The continuous wave NIRS recording indicated an increase in oxyHb at the superficial and cortical compartment during stimulation on the ipsilateral side at both LF and HF stimulation, which can be seen on cortical CBF increase (39). CBF increase will result in a VMCA increase, which implies that the possible MCA dilatation is larger than the possible CBF increase in this study. Overall, the human vascular data show some interesting findings, which are partly in line with animal models, but need to be reproduced in studies with a larger number of patients. Other imaging modalities such as magnetic resonance imaging may investigate other vascular compartments of interest, such as the middle meningeal artery; however, TCD and NIRS provide faster imaging possibilities which better suit this type of experiment.
The present study has some limitations. First, the number of patients is low and the patients were heterogeneous in response to SPG stimulation therapy and baseline cluster attack frequency, which makes it difficult to arrive at a definite interpretation of the study. In addition, it was necessary to exclude one subject because of technical issues, which made the patients’ lack of headache response impossible to interpret. The present study used a double-blind cross-over controlled design, but blinding may have been hampered by patients who can distinguish their normal HF stimulation from the LF stimulation used for this study. Additionally, the study uses 5 Hz LF stimulation frequency, based on results from previous animal studies (10,12,40,41), but it is possible that 10 Hz or another LF would more effectively induce CH-like attacks, associated autonomic symptoms and vascular effects.
In conclusion, in this first study LF 5 Hz SPG activation using experimental electrical stimulation appeared to induce cluster-like attacks with autonomic features, which were terminated by HF therapeutic electrical stimulation. These findings suggest that efferent parasympathetic outflow from the SPG contributes to a nociceptive mechanism in CH. The present SPG stimulation study design will be developed further for use in future experimental and pharmacological studies of CH.
Clinical implications
Low frequency neurostimulation of the sphenopalatine ganglion may induce immediate cluster-like headache attacks with autonomic symptoms. Efferent parasympathetic outflow from the sphenopalatine ganglion may contribute to a nociceptive mechanism in cluster headache. Neurostimulation of the sphenopalatine ganglion is a new human experimental headache model.
Footnotes
Acknowledgements
The authors wish to thank lab technicians Winnie Grønning Nielsen and Lene Elkjær for their excellent and dedicated assistance. We thank Professor Jes Olesen and David A. Boas for valuable input to the paper.
Funding
The study was supported by the Lundbeck Foundation via the Lundbeck Foundation Center for Neurovascular Signaling (LUCENS), The University of Copenhagen, the Augustinus Foundation, the Toyota Foundation and Autonomic Technologies, Inc.
Conflict of interest
The study was funded by Autonomic Technologies, Inc. Dr. Jensen is an advisory board member for ATI, Allergan, Neurocore and Medotech, and has received honoraria and lectured for MSD, Pfizer, Allergan, Berlin-Chemie, and Norpharma. Dr Ashina is a consultant and/or scientific advisor for Allergan, Amgen and Alder, and has received grant support and honoraria for lecturing from Merck, and honoraria for lecturing from Allergan, Pfizer, GlaxoSmithKline, Norpharma and AstraZeneca. There are no conflicts of interest for the remaining authors.
