Abstract
Postmortem data indicate loss of serotoninergic neurons in Parkinson's disease (PD). We used the serotonin transporter (SERT) radioligand 3-amino-4-(2-dimethylaminomethyl-phenylsulfaryl)-benzonitril (DASB) and positron emission tomography to examine SERT distribution and changes in early PD subjects. We studied five PD subjects (H&Y 1 to 2.5) and eight normal controls. There is reduced SERT binding in PD. The magnitude of DASB binding reductions was greater in the forebrain than in the brainstem regions. There was no asymmetry of diminished SERT binding. DASB binding in the medulla was relatively spared, inconsistent with the description of early prominent pathologic study in these caudal brainstem nuclei.
Introduction
Pathologic, biochemical, and imaging studies indicate loss of serotoninergic neurons in Parkinson's disease (PD; reviewed by Kish, 2003). Changes in non-dopaminergic systems may contribute to non-motor and/or non-dopaminergic features of PD (Chaudhuri et al, 2006; Langston 2006). Braak et al (2003) put forward an intriguing hypothesis in which initial pathologic changes occur in anterior olfactory nucleus neurons and the medulla. Subsequent pathologic changes follow an ‘ascending’ progression, with midbrain and forebrain structures affected later. In ‘Braak’ stage 2, Lewy body (LB) and Lewy neurite (LN) deposition occurs within the raphe nucleus magnus, raphe nucleus obscurus, raphe nucleus pallidus, and the gigantocellular nucleus of the medullary lateral reticular formation; nuclei containing the serotoninergic neurons of the caudal brainstem (Hornung, 2003). The Braak
Earlier efforts to study serotoninergic neurons
Subjects and methods
Subjects
We studied five PD subjects and eight controls. PD subjects met UKPDS brain bank criteria for PD with asymmetric onset and clear response to dopaminergic treatment. No subject was taking medications that would interfere with SERT ligand binding. PD subjects (four men, one woman; mean age 64 years; s.d. 7; range 54 to 71 years; 2 H&Y1, 2 H&Y2, 1 H&Y2.5) were treated with carbidopa/levodopa or a dopamine agonist. None were depressed. Controls (four men, four women; mean age 53 years; s.d. 17; range 35 to 70 years) had no neurologic or psychiatric disease.
PET Imaging
The [11C]DASB-PET studies were acquired as 17 scan frames over a total of 80 mins as follows: 4 × 30 secs; 3 × 1 min; 2 × 2.5 mins; 2 × 5 mins; and 6 × 10 mins. Radiotracer was administered as a bolus plus constant infusion using 70% as a slow bolus over 30 secs, followed by constant infusion of the remaining 30% over the 80 mins study duration. All PET studies were acquired in 3-D mode with interplane septa retracted on a Siemens ECAT HR+ scanner. 2-D transmission scans were acquired for measured attenuation correction followed by segmentation and reprojection. Standard corrections were made for dead-time, randoms, radioactive decay, scatter, and attenuation. Image reconstruction consisted of FORE rebinning of 3-D data into 2-D projections, followed by 2D-OSEM: 4 iterations, 16 subsets. No smoothing filters were used. Resultant images had in-plane and axial resolution of approximately 5.0 to 5.5 mm FWHM. Standard parametric image calculations were performed using an equilibrium analysis (Koeppe et al, 1997). The lateral cerebellar cortex, with very low SERT expression, was used as a reference region assumed to be devoid of specific binding to normalize radiotracer binding measures, yielding estimates of the ratio of volumes of distribution between target regions and the reference region (Ginovart et al, 2001; Kish et al, 2005). The binding potential,
Estimates of the equilibrium ratio of tracer tissue concentrations in a given voxel and the reference region are used to estimate BPND. For [11C]DASB, data taken 60 to 80 mins after injection were used to estimate
The average of the first 4 mins of DASB uptake was used as an index of ligand transport (K1R) across the blood-brain barrier. Parametric K1R images for each subject were reoriented to a common coordinate system based on the stereotactic atlas of Talairach and Tournoux (1998). After reorientation, all images underwent linear scaling and nonlinear warping (Minoshima et al, 1994). The transformation matrix from each individual's K1R images was applied to that subject's ratio of volumes of distribution image set.
A set of 55 standardized volumes-of-interest were defined on the atlas images, including Brodmann areas and subcortical regions. Volumes-of-interest were applied automatically to each subject's DASB image set. Data were averaged for the corresponding areas of each hemisphere. No partial volume correction was applied.
Standard two-sample
Results
DASB-PET imaging revealed reductions in DASB binding throughout the forebrain and brainstem of PD subjects (Table 1, Figure 1). The magnitude of diminished DASB binding was greatest in the forebrain; cingulate cortices and insula were approximately 40% to 50% of normal. Caudal brainstem structures had the smallest reductions: approximately 20% of normal. The amygdala, hippocampus, basal ganglia, thalami, and rostral brainstem nuclei had intermediate reductions: approximately 30% to 35% of normal. In paired forebrain structures, DASB binding was reduced symmetrically. The interhemispheric difference in striatal DASB binding was approximately 3%. Two PD subjects were previously studied with the VMAT2 ligand [11C]DTBZ to quantify nigrostriatal terminal changes (Bohnen et al, 2006) in an asymmetric pattern. In these two subjects, striatal DTBZ binding was reduced markedly and the interhemispheric difference was approximately 20%. In controls, there was no evidence of an age-related change in regional DASB binding, similar to the results of Guttman et al (2007).
Regional DASB binding in PD and control brain
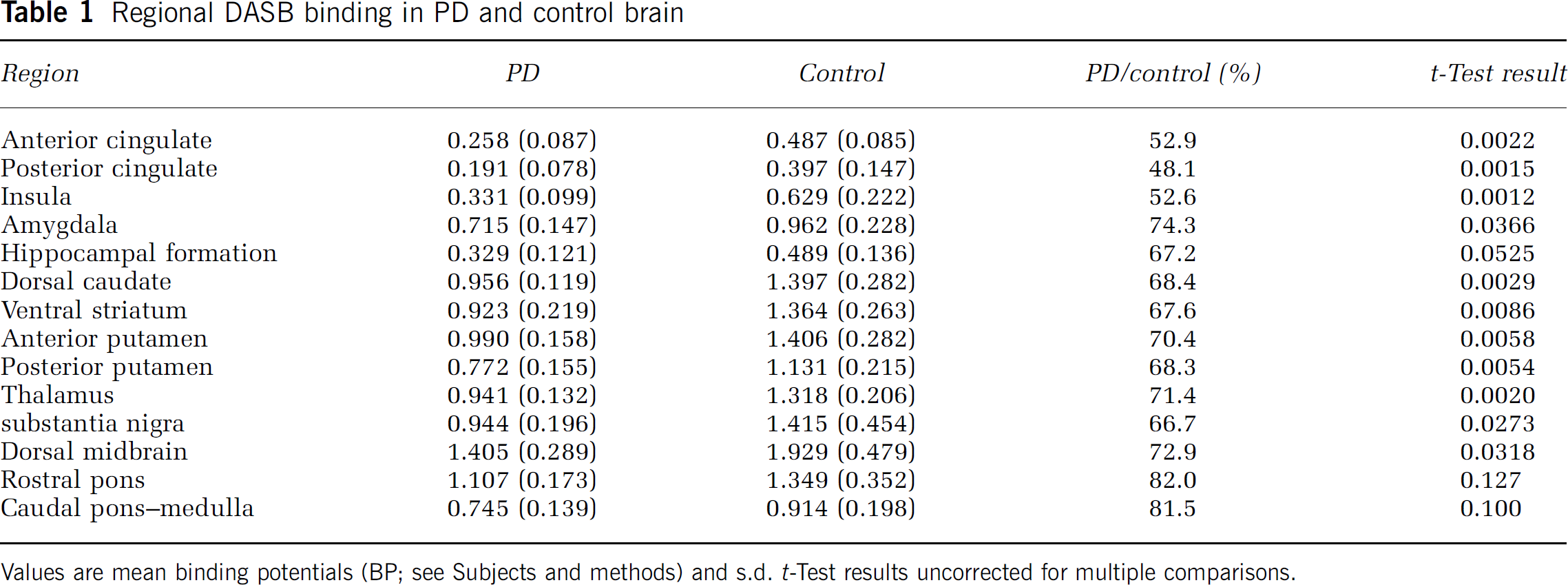
Values are mean binding potentials (BP; see Subjects and methods) and s.d.
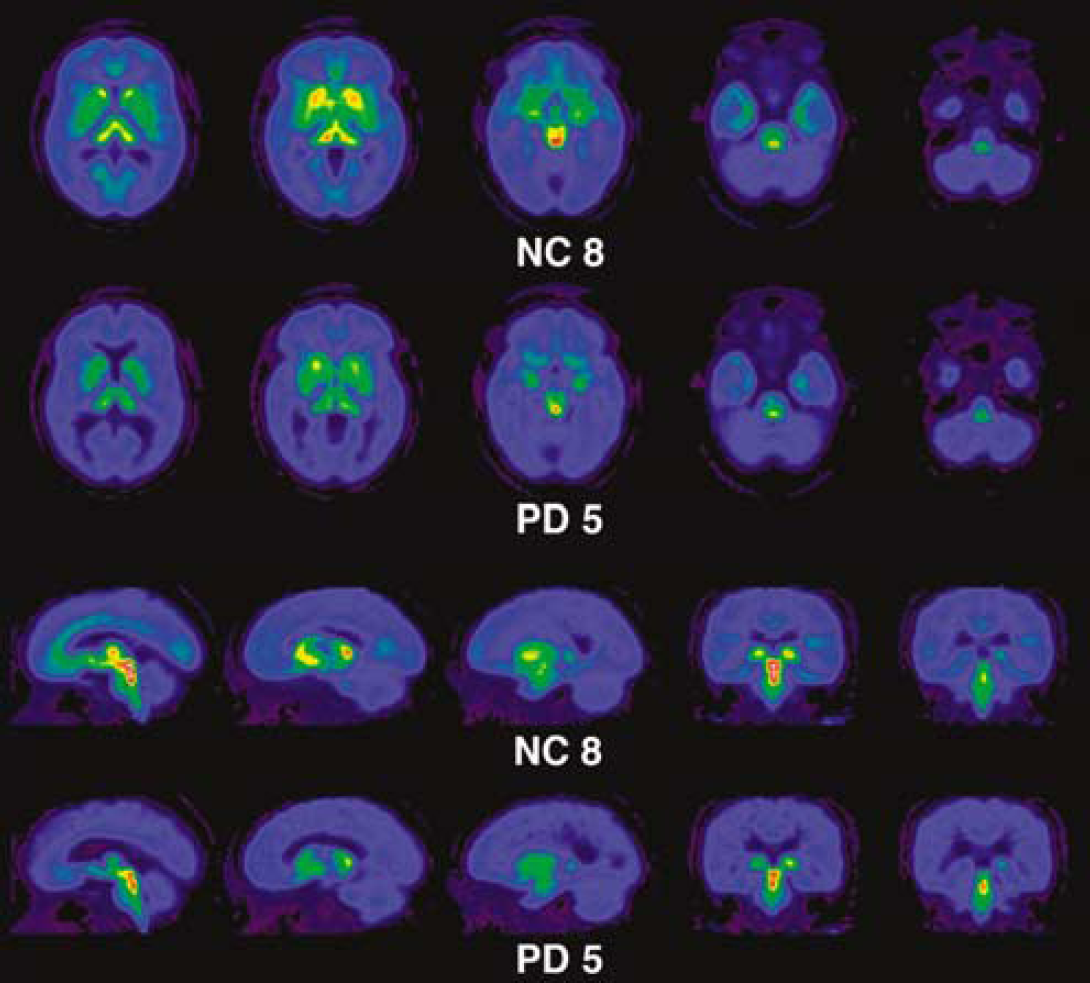
Pseudocolor images comparing DASB binding in representative control (NC8) and Parkinson's disease (PD5) subjects. The control images display the expected distribution of SERT binding. In the PD subject, there is a diffuse reduction with a rostral to caudal gradient.
Discussion
We report diffuse reductions in [11C]DASB binding in PD brain. There was a rostral to caudal gradient in the magnitude of DASB binding reductions, with more rostral regions affected most and caudal brainstem regions affected least. Our results are consistent with loss of SERT-expressing terminals and dysfunction/degeneration of serotoninergic neurons, but may also reflect downregulation of SERT expression. Our results are consistent with prior [123I]β-CIT and [11C]McN5652 studies and broadly consistent with the results of Guttman et al (2007), who describe DASB-PET in PD subjects with more advanced disease. Guttman
The serotoninergic innervation of the CNS arises from the brainstem raphe complex (Hornung, 2003). The rostral group, from the mid-pons to the rostral midbrain, sends ascending projections to the forebrain. The caudal group, from the caudal pons to caudal medulla, innervates the visceral and somatic nuclei of the brainstem and the spinal cord. Braak et al (2003) report early LB and LB deposition in the raphe nucleus magnus, raphe nucleus obscurus, raphe nucleu pallidus, and the gigantocellular nucleus of the medullary lateral reticular formation nuclei—the serotoninergic caudal brainstem complex. Our results suggest relative preservation of caudal brainstem serotoninergic neurons. This result may contradict Braak
Our results show symmetrical reductions of DASB binding in paired structures, unlike the asymmetrical reductions of nigrostriatal terminal markers. The magnitude of inferred serotoninergic terminal loss is less than that of striatal dopaminergic terminal loss, even in early PD (Bohnen et al, 2006). Although SERTs may be upregulated in surviving serotoninergic terminals, our results suggest relative sparing of caudal brainstem serotoninergic neurons in early PD.
Our results support the concept of widespread changes in multiple brain systems in early PD. Degeneration of the nigrostriatal projection is the primary driver of motor dysfunction in PD but non-motor features of PD are major contributors to disability. Changes in serotoninergic systems may contribute to aspects of non-motor dysfunction in PD. Careful correlation of clinical features and serotoninergic system changes may be useful in elucidating the bases for some non-motor dysfunctions and point toward novel useful interventions in PD.
Conflict of interest
The authors state no conflict of interest.
