Abstract
Neurovascular coupling, or the tight coupling between neuronal activity and regional cerebral blood flow (CBF), seems largely driven by the local processing of incoming afferent signals within the activated area. To test if cortical γ-aminobutyric acid (GABA) interneurons—the local integrators of cortical activity—are involved in this coupling, we stimulated the basalocortical pathway in vivo, monitored cortical CBF, and identified the activated interneurons (c-Fos-immunopositive) and the neuromediators involved in this response. Basal forebrain (BF) stimulation induced ipsilateral increases in CBF and selective activation of layers II to VI somatostatin- and/or neuropeptide Y-containing, as well as layer I GABA interneurons. Nitric oxide synthase interneurons displayed weak bilateral activation, whereas vasoactive intestinal polypeptide- or acetylcholine (ACh)-containing GABA interneurons were not activated. Selective cholinergic deafferentation indicated that ACh released from stimulated BF afferents triggered the CBF response, but the latter was mediated, in part, by the local release of GABA from cholinoceptive cortical interneurons, and through GABA-A receptor-mediated transmission. These data show that activation of specific subsets of GABA interneurons and their GABA-A-mediated effects on neuronal, vascular, and/or astroglial targets are necessary for the full expression of the hemodynamic response to BF stimulation. Further, these findings highlight the importance of understanding the cellular networks and circuitry that underlie hemodynamic signals, as only specific subsets of neurons may be activated by a given stimulus, depending on the afferent inputs they receive and integrate.
Keywords
Introduction
Normal brain function is ensured by a constant adjustment of blood supply to neurons displaying increased activity, a process known as functional hyperemia or neurovascular coupling. Recent evidence indicates that this tight coupling, which is at the basis of the signals used in several functional neuroimaging techniques, is driven by the incoming afferents and, essentially, their local processing within the activated area (Logothetis et al, 2001; Lauritzen and Gold, 2003). Although this process requires a thorough interplay between activated neurons, microvessels, and perivascular astrocytes (Hamel, 2006), the identity of the neurons that instigate the hemodynamic response to specific incoming afferent pathways is largely unknown. This is particularly true in the cerebral cortex where only the contribution of glutamate-releasing cells has been investigated in vivo under conditions of sensory (Zonta et al, 2003) or transcallosal (Hoffmeyer et al, 2007) pathway stimulation.
The basalocortical pathway that involves stimulation of basal forebrain (BF) neurons located in the nucleus basalis and adjoining substantia innominata (SI) is a very well-characterized neurovascular coupling pathway. These neurons contain primarily acetylcholine (ACh) and their stimulation induces cortical release of ACh together with increases in cortical activity and cerebral blood flow (CBF; for review and references, see Hamel, 2004). Yet, the activated cortical neurons, likely those that mediate the local hyperemic response, have never been identified. Basalocortical afferents target both pyramidal cells (Houser et al, 1985) and γ-aminobutyric acid (GABA) interneurons (Freund and Gulyas, 1991), the latter being the local integrators of cortical activity (Markram et al, 2004). Moreover, cortical GABA interneurons project avidly to local microvessels and perivascular astrocytes (Vaucher et al, 2000), and are thus strategically positioned to readily adapt CBF to local changes in neuronal activity, as recently suggested from their stimulation in isolated cortical slices, which resulted in vascular responses (Cauli et al, 2004).
In this study, we sought to investigate, in vivo, if cortical GABA interneurons participate in the neurovascular coupling response to BF stimulation, and to identify which interneuron subtypes and neuromediators are involved. Cortical CBF was monitored online during BF stimulation, combined or not with in vivo pharmacologic manipulations or selective ACh deafferentation, followed by identification of the activated interneurons by double immunostaining for c-Fos and markers of different GABA interneuron subtypes. The findings show that cholinoceptive layer I GABA interneurons and those in layers II to VI that express somatostatin (SOM) and/or neuropeptide Y (NPY) are selectively activated by BF stimulation and that the resulting perfusion response requires activation of GABA-A receptors on neuronal, vascular, or astroglial targets for its full expression.
Materials and methods
Animals, Surgery, and Basal Forebrain Stimulation
Sprague—Dawley rats (280 to 320 g, male, Charles River, Montréal, QC, Canada) were used in all experiments that were approved by the animal ethics committee of McGill University, in line with the Canadian Council on Animal Care. Chronic implantation of stimulating monopolar tungsten electrodes (0.35 mm outer diameter; FHC, Bowdoinham, ME, USA) or guide cannulas (Plastics One, Roanoke, VA, USA) was performed stereotaxically in anesthetized rats (ketamine/xylazine, 100/5 mg/kg, intraperitoneally) at Bregma coordinates AP: −0.9 mm, L: 2.7 mm, and V: −7.6 mm (electrodes) or −6 mm (cannulas) below the brain surface. Electrodes were positioned under CBF monitoring to target the site within the SI in the left BF that best increased cortical CBF (Vaucher et al, 1997). On the day of implantation (∼6 days before the experimental day to avoid nonspecific c-Fos activation), the skull bone was bilaterally thinned to translucency in corresponding areas of the frontal cortex (Bregma coordinates AP: +2 mm and L: +/–2 mm) for future positioning of the laser-Doppler flowmeter probes.
On the experimental day, urethane-anesthetized rats (1.1 g/kg intraperitoneal) fixed in a stereotaxic frame were submitted to chemical (glutamate and N-methyl-
Cerebral Blood Flow Measurements
To control for specificity of the BF-evoked perfusion response, CBF was measured in the frontal cortex from both hemispheres with laser-Doppler flowmeter needle-shaped probes (Moor Instruments Ltd, Millwey, UK) connected to a laser-Doppler flowmeter perfusion monitor and positioned in areas free of large vessels. After stabilization (30 to 40 mins) and CBF baseline readings, the left BF was stimulated and bilateral CBF measures acquired. Signals were smoothed to a time constant of 3 secs and analyzed with the DRT4/Win v1.2 software (Moor Instruments Ltd). Rats were removed from the stereotaxic frame and kept in their cage for 1 h while still anesthetized before fixation for c-Fos protein immunodetection.
Immunocytochemical Staining of Activated Interneurons
Rat brains were perfusion-fixed through the ascending aorta (500 mL of 4.0% paraformaldehyde in 0.1 mol/L phosphate buffer) under deep anesthesia (pentobarbital, 100 mg/kg, intraperitoneal), post-fixed by immersion (2 h, 4°C), cryoprotected, frozen, and stored (–80°C) until sectioning as free-floating coronal sections (25-μm thick) on a freezing microtome (SM 2000R, Leica, Montréal, QC, Canada) (Vaucher et al, 2000). Activation was first detected with c-Fos immunostaining alone, followed by identification of activated cells by double immunocytochemistry for c-Fos and markers of astrocytes, pyramidal cells, or different subtypes of GABA interneurons as defined by their co-localized peptides or enzymes (Kubota et al, 1994; Cauli et al, 2004). For this purpose, adjacent sections were first incubated (overnight, room temperature) with one of the following primary antibodies: rabbit anti-glial fibrillary acidic protein (GFAP; 1:300; DAKO, Mississauga, ON, Canada), mouse anti-rat brain pyramidal cells (1:2,000; SWANT, Bellinzona, Switzerland), mouse anti-glutamic acid decarboxylase 67 (GAD67, GABA synthesizing enzyme, 1:20,000; Chemicon, Temecula, CA, USA), guinea-pig anti-vasoactive intestinal polypeptide (VIP; 1:5,000; Peninsula Labs Inc., Belmont, CA, USA), rabbit anti-NPY (1:10,000; Chemicon), rat anti-SOM (1:150; Chemicon), sheep anti-neuronal nitric oxide synthase (1:20,000; Chemicon) or goat anti-choline acetyltransferase (ChAT, ACh synthesizing enzyme, 1:250; Chemicon). After incubation (40 mins) with species-specific secondary antibody (1:200, Vector Labs, Burlingame, CA, USA), the reaction was revealed (ABC complex; Vectastain ABC kit; Vector Labs) and detected with 3,3′-diaminobenzidine (DAB, Vector Labs; brown precipitate). Immunoreactivity to c-Fos was then detected by overnight incubation with a rabbit anti-c-Fos antibody (1:15,000; Oncogene, San Diego, CA) using a biotinylated secondary antibody and the SG reagent (Vector Labs; blue-gray precipitate) or DAB-nickel (dark blue precipitate). Sections were observed under light microscopy and digital pictures taken, calibrated, and edited with MetaMorph 6.1r3 (Universal Imaging, Downington, PA, USA) and Adobe Photoshop 7 (Adobe Systems, San Jose, CA, USA).
Double-Immunocytochemistry of Somatostatin and Microvessels
Sections (n = 4 control rats) were incubated overnight with a rabbit anti-SOM antibody (1:1,000; Peninsula) detected with DAB, and then brain vessels were immunostained with a goat anti-collagen type IV antibody (1:400; Chemicon, overnight) detected with the SG reagent. Some sections were first incubated with the two primary antibodies and then sequentially with species-specific Cy2- (SOM) and Cy3- (collagen IV) conjugated affiniPure IgG antibodies (1:200, 40 mins; Jackson Immunoresearch, West Grove, PA, USA), and observed under confocal microscopy for simultaneous detection (emission intensity at 488 and 543; Zeiss LSM 510; Zeiss, Jena, Germany). Digital pictures were taken (Zeiss LSM Image Browser), calibrated, and edited as above.
Cholinergic Deafferentation
Cholinergic deafferentation of the neocortex was achieved with the selective cholinergic immunotoxin 192 IgG-saporin (saporin, 4.1 μg/2 μL saline (Chemicon), control rats received 2 μL saline, n = 3/group) administered in the left lateral ventricle (Bregma coordinates AP: 0.8 mm, L: 1.4 mm, and 4.3 mm below the skull surface). After 14 days, cortical CBF responses to BF electrical stimulation were measured, and then the animals perfused as above for ChAT, GAD67, and NOS immunostaining.
Pharmacology
Pharmacological manipulations were performed in electrically stimulated rats using parameters (100 Hz, 80 μA, 1 sec on/1 sec off, 20 secs) that yielded a comparable CBF response to that obtained with the longer stimulation used in the c-Fos studies while allowing reproducible CBF increases for at least six consecutive stimulations when performed at 20 min intervals. The role of GABA or co-localized peptides in the induced CBF response was tested in distinct groups of rats through intracisternal injection (3 μL) of drug vehicles or receptor antagonists (10−4mol/L solution in 0.05 mol/L phosphate-buffered saline, pH 7.4, unless otherwise specified) at muscarinic (mAChR, scopolamine, n = 4), GABA-A (picrotoxin, n = 6), GABA-B (CGP 35348, n =4), SOM (cyclo-somatostatin, C-SOM, n = 8), or NPY-Y1 (BIBP 3226, vehicle: 0.05 mol/L phosphate-buffered saline containing 0.04% dimethylsulfoxide, n = 7) receptors. A single drug and its vehicle were tested in each rat. All intracisternal injections were performed under microscopic monitoring using a Hamilton microsyringe connected to a 30G needle inserted through the exposed atlantooccipital membrane, and their location was confirmed by injecting scopolamine at the end of the experiment, a known inhibitor of the BF-mediated CBF increase (Dauphin et al, 1991). The contribution of GABA was also tested with the GAD inhibitor isoniazid (350 mg/kg intraperitoneally in saline, n = 4) (Barbaccia et al, 1996) administered either alone or in combination with scopolamine (intraperitoneally 10 mg/kg in saline n = 5). On the basis of previous studies with picrotoxin (Arancibia et al, 1995), CGP 35348 (Hicks et al, 2004), c-SOM (Bartsch et al, 2005) and BIBP 3236 (O'Shea et al, 1997), and our own studies in which drug effects were measured up to 60 mins after injection, optimal intervals between drug injection and BF stimulation were 10 to 20 mins or, in the case of isoniazid alone or in combination with scopolamine, 45 mins (Barbaccia et al, 1996). Cerebral blood flow responses to BF stimulation were measured first under control conditions, then after vehicle injection and, finally, after drug injection. In a different series of experiments, the effect of picrotoxin on the increase in cortical CBF induced by adenosine (1.5 μL of a 2 mmol/L solution injected intracisternally) was compared with that of vehicle (n = 4/group). Blood gases, pH, and arterial blood pressure were concurrently monitored at each of these steps in representative groups of rats (∼3 rats/compound). All experiments were completed within less than 2 h of the initiation of urethane anesthesia.
The effects of GABA-related compounds and scopolamine (n = 3 to 4 rats/compound) on cortical electroencephalogram (EEG) were recorded before and after drug injection (up to the end of the experiment) in urethane-anesthetized rats fixed in a stereotaxic frame and implanted in the skull with two stainless-steel screws (Bregma coordinates AP: +0.5 cm and AP: −0.3, L: +0.3 cm, for reference and recording electrodes, respectively). Electroencephalogram signals were amplified, digitized, and recorded (AxoScope 10.0 software, Molecular Devices, Sunnyvale, CA, USA). They were visually inspected for the entire recording period and no epileptic-like activity occurred at the doses used. Electroencephalogram recordings were further analyzed at times corresponding to the CBF measurements after vehicle and drug injections. Drugs were from Sigma (St Louis, MO), except CGP 35348 (Tocris Bioscience, Ellisville, MO, USA) and BIBP 3226 (kindly provided by Dr H Doods, Boehringer Ingelheim, Biberach, Germany).
Data Analysis
CBF values were measured in arbitrary units. Changes induced by BF stimulation were taken at the peak response, expressed as percentage changes from baseline, and compared between ipsi- and contralateral sides by paired Student's t-tests. BF-induced ipsilateral CBF responses between control and SAP-lesioned rats were compared by Student's t-tests. In pharmacologic experiments, repeated measures analysis of variance (ANOVA) followed by post hoc Newman—Keuls comparison tests were used to compare physiological parameters as well as the BF-induced CBF responses after control, vehicle, and drug conditions. For all isoniazid-treated rats, paired Student's t-tests were used in comparing values before and after drug injection. In adenosine experiments, Student's t-tests were used to compare CBF responses between vehicle- and picrotoxin-treated rats. Electroencephalogram recordings under vehicle and drug were compared by power spectrum analysis at times corresponding to CBF measurements (Clampfit 9.2, Axon Instruments Inc., Union City, CA, USA) using paired Student's t-tests. In immunocytochemical studies, c-Fos-positive nuclei were quantified on digital pictures from the ipsilateral cortex using MetaMorph 6.1r3, and expressed as the number of stained nuclei per 0.1 mm2 of cortical area. For each subtype of GABA interneurons, c-Fos-positive cells were counted (two to three sections/level/rat) in double-immunostained sections directly under the microscope in layers I, II to IV, and V to VI of the ipsi- and contralateral frontoparietal cortex and, to identify the interneurons activated throughout the cerebral cortex that displayed increased CBF (Vaucher et al, 1997), at three different rostro-caudal Bregma levels (in mm: 1.6 to 1.2, −1.0 to −1.3, and −3.0 to −3.3). Data were expressed as percentage of the total population of labeled interneurons for each marker and compared between the different subtypes of double-labeled cells by one-way ANOVA followed by post hoc Newman—Keuls test, or by Student's t-test when comparing ipsi- and contralateral sides. For each rat, 2860 ± 205 (GAD67), 980 ± 211 (NPY), 2296 ± 212 (SOM), 385 ± 22 (NOS), 2590 ± 218 (VIP), or 1194 ± 109 (ChAT) immunostained cells on average were counted. Data were expressed as means ± s.e.m. All statistical analyses were performed with GraphPad Prism4 (San Diego, CA, USA) and a P < 0.05 was considered significant.
Results
Basal Forebrain Stimulation Selectively Increases Ipsilateral Cerebral Blood Flow
Baseline CBF was very stable and rapidly increased in the ipsilateral cortex on glutamate, NMDA, or electrical stimulation of the BF (Figures 1A and 1B). The stimulus-evoked increases in CBF were comparable between the various stimulation paradigms. They were significantly larger in the ipsilateral cortex as compared with the small vascular responses observed on the contralateral cortex (paired Student's t-test, P < 0.01, Figure 1) and were not accompanied by significant changes in arterial blood pressure, blood gases, or pH (see Materials and methods). Saline microinjections in the BF had virtually no effect on cortical perfusion (Figure 1B). These stimulus-evoked hemodynamic responses fully agree with several previous reports that showed a selective effect of BF stimulation on ipsilateral cortical CBF without alterations in arterial blood pressure and blood gases in both anesthetized and unanesthetized rats (Dauphin et al, 1991; Vaucher et al, 1997).
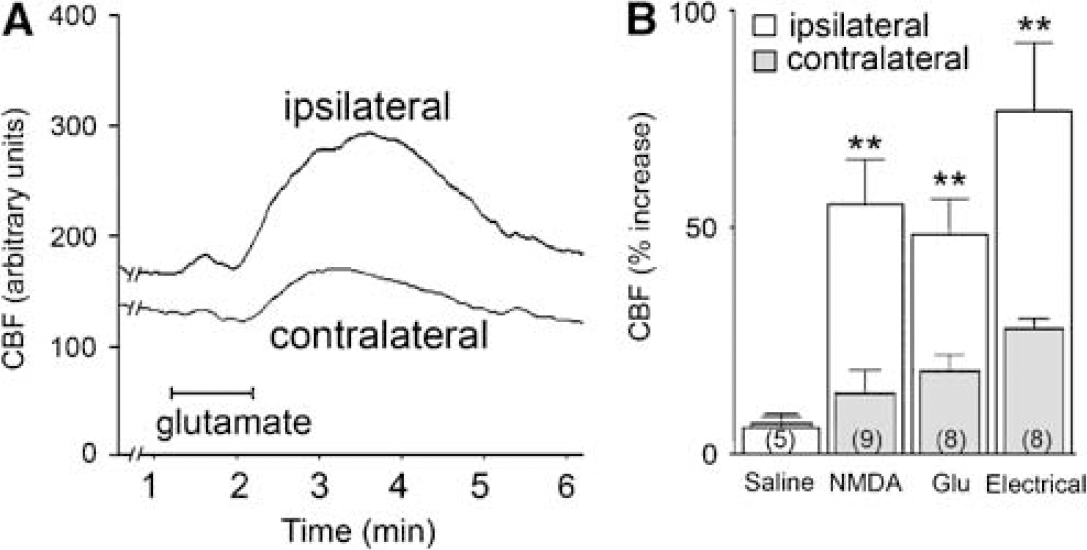
(
Basal Forebrain Stimulation Activates Layer I and Somatostatin- and Neuropeptide Y-Containing γ-Aminobutyric Acid Interneurons
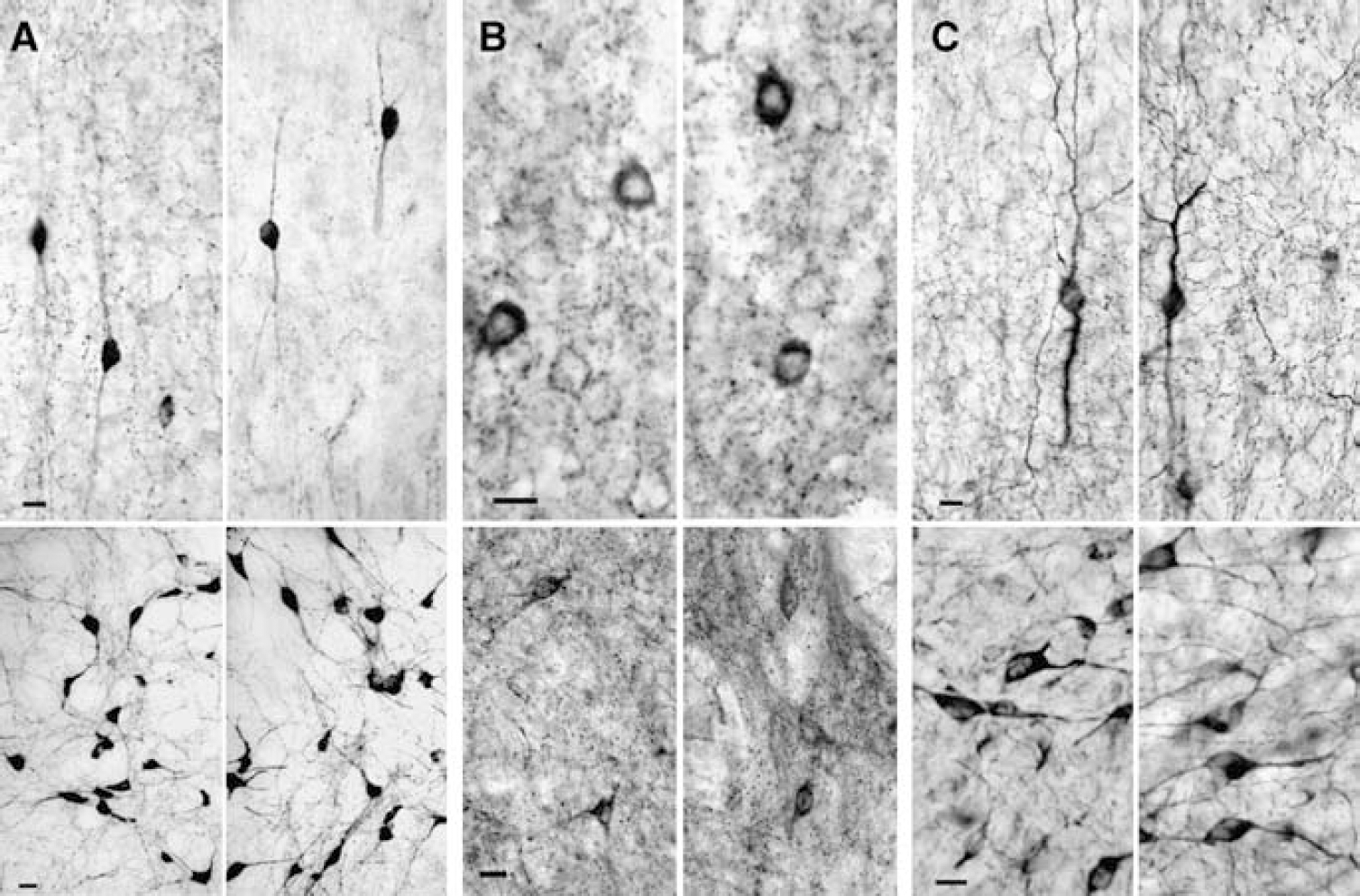
Microscopic observation of c-Fos-immunostained sections from a subset of responsive animals revealed that BF stimulation selectively increased c-Fos immunoreactivity in the ipsilateral cortex (Figures 2A to 2D). The weak proportion of immunostained nuclei in the contralateral as compared with the ipsilateral cortex, together with the absence of cortical c-Fos immunostaining in control rats with saline microinjection or with no current injection (data not shown), indicated that the neuronal activation was stimulus-related. Quantification of c-Fos positive nuclei showed that NMDA stimulation activated on average 73.2 ± 3.5 cells/0.1 mm2 of ipsilateral cortex, with slightly but not significantly less activated cells after glutamate and electrical stimulations (54.2 ± 7.5 and 43.5 ± 8.6 cells/0.1 mm2, respectively, P = 0.079, ANOVA).
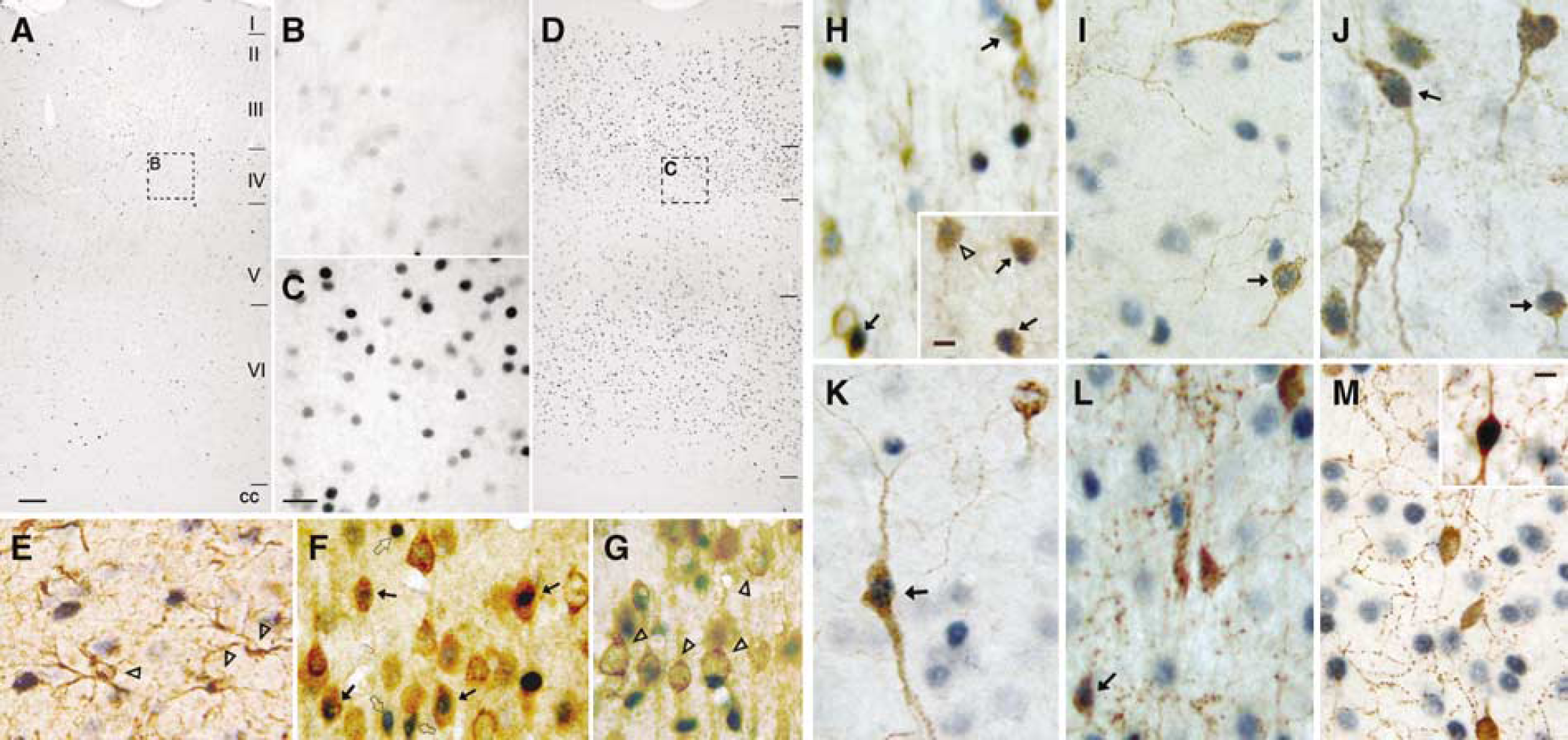
C-Fos immunostaining was minimal in (
Identification of c-Fos-positive cells by double immunostaining showed no co-localization with GFAP (Figure 2E), whereas c-Fos staining was evident in many neurons labeled with the pyramidal cell marker. These immunostained pyramidal cells distributed in layers II to IV and VI of the different cortical levels examined (Figure 2F, not quantified), but not or only rarely in layer V (Figure 2G). A large proportion of c-Fos-positive nuclei were distinct from, thought intermingled with, immunostained pyramidal cells, hence demonstrating activation of cortical interneurons (Figures 2F and 2G). Quantitative analysis of the different subtypes of activated GABA interneurons revealed a significant increase in the number of c-Fos-positive GAD67, SOM, and NPY interneurons throughout layers II to VI of the ipsilateral frontoparietal cortex after chemical or electrical BF stimulation (Figures 2H to 2J and 3A). Specifically, after NMDA microinjection, 45.7 ± 11.2, 32.4 ± 4.2, and 37.3 ± 11.3% of GAD67-, SOM- and NPY-positive cells, respectively, were activated in the ipsilateral parietal cortex, compared with ∼5% in the contralateral cortex. For the small population of cortical NOS interneurons, a subset of SOM/NPY neurons (Kubota et al, 1994; Cauli et al, 2004), there was no side-to-side difference, with a relatively high proportion of activated cells on the contralateral side, and this particularly after electrical BF stimulation (Figures 2K, 3A). Such bilateral activation may point to their stimulation by fibers en passage and alternate pathways that are predominantly recruited by electrical stimulation of the BF (Adachi et al, 1990). In contrast, a minority (>10%) of VIP- and ChAT-containing interneurons was activated by BF stimulation, a proportion that did not significantly differ from that on the contralateral, non-activated side (Figures 2L, 2M and 3A). In cortical layer I, BF stimulation activated a subset of interneurons that co-stained only for GAD67 among all markers investigated, and which corresponded to 42.1 ± 11.8, 29.5 ± 19.8, or 11.3 ± 3.4%, respectively, of GAD cells in this layer after NMDA, glutamate, or electrical stimulation (Figure 2H inset, 3C). This is likely explained by the fact that several layer I cortical GABA interneurons contain calretinin (Kubota et al, 1994), a marker not examined in this study. The same groups of interneurons were activated across the three rostrocaudal levels and cortical layers of the frontoparietal cortex, as shown for GAD67 and SOM neurons (Figures 3B and 3C), indicating that activation of the same interneuron subtypes occurred within the full extent of the cortex displaying increased CBF after BF stimulation (Vaucher et al, 1997).
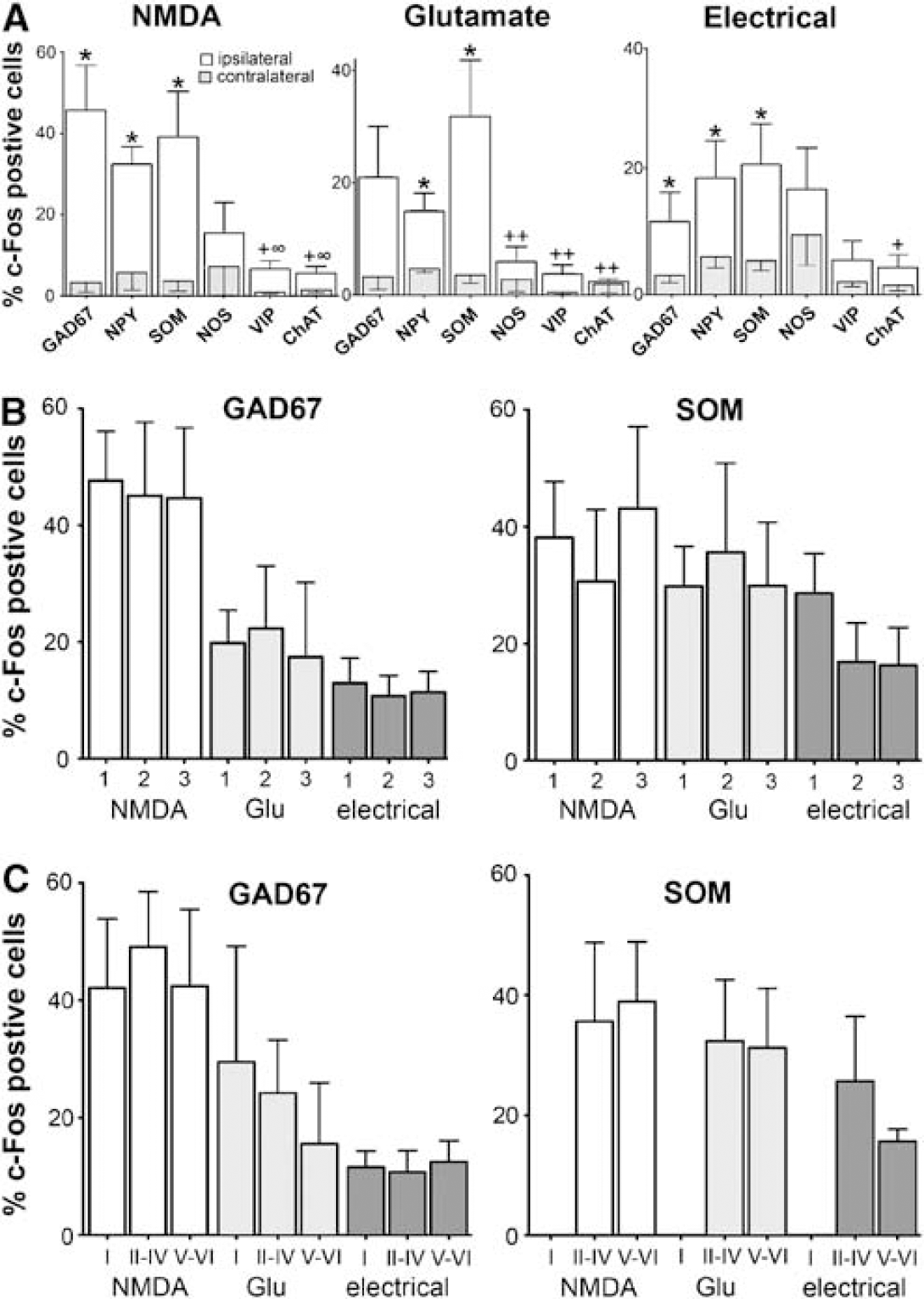
(
Mediators of the Hemodynamic Response to Basal Forebrain Stimulation
Reducing brain levels of GABA by GAD inhibition with isoniazid or blocking GABA-A receptors with Picrotoxin significantly decreased the BF-inducedhemodynamic response in comparison to no drug (–46%, P < 0.05) or vehicle (–28%, P < 0.01) (Figure 4A), with no effect on cortical EEG or baseline CBF, as shown for picrotoxin (Figures 4C and 4D). Yet, Picrotoxin failed to alter the increased CBF response evoked by intracisternal injection of adenosine (+ 27.2 ± 2.3 versus +26.6 ± 5.0% from baseline for vehicle and picrotoxin, respectively, −2.2%, P = 0.92) indicating that its effect on the BF-induced CBF response was specific and not secondary to loss of reactivity of cortical microvessels. Antagonism at mAChRs with scopolamine reduced the CBF response to BF stimulation (–40%, P < 0.05, Figure 4A). In rats that received a combined injection of scopolamine and isoniazid, a 53% inhibition in the BF-evoked CBF response was observed (Figure 4A). In contrast, antagonism at GABA-B, NPY-Y1, or SOM receptors with CGP 35348, BIBP 3226, or cyclo-SOM did not affect the BF-induced CBF response (Figure 4A) or cortical EEG (CGP 35348, data not shown), even when tested up to 60 mins and at doses well within the range where they showed efficacy in other central nervous system-related effects after intracerebroventricular injections (Hicks et al, 2004; O'Shea et al, 1997; Bartsch et al, 2005). Physiologic parameters (blood gases, pH, or arterial blood pressure) were not altered by vehicles and changes were < 5% between vehicles and drugs (scopolamine, picrotoxin, C-SOM, or scopolamine and isoniazid), and at most 15.8 ± 5.7% for isoniazid alone (on pCO2, not significantly different from pre-drug levels, P = 0.10, n = 4).
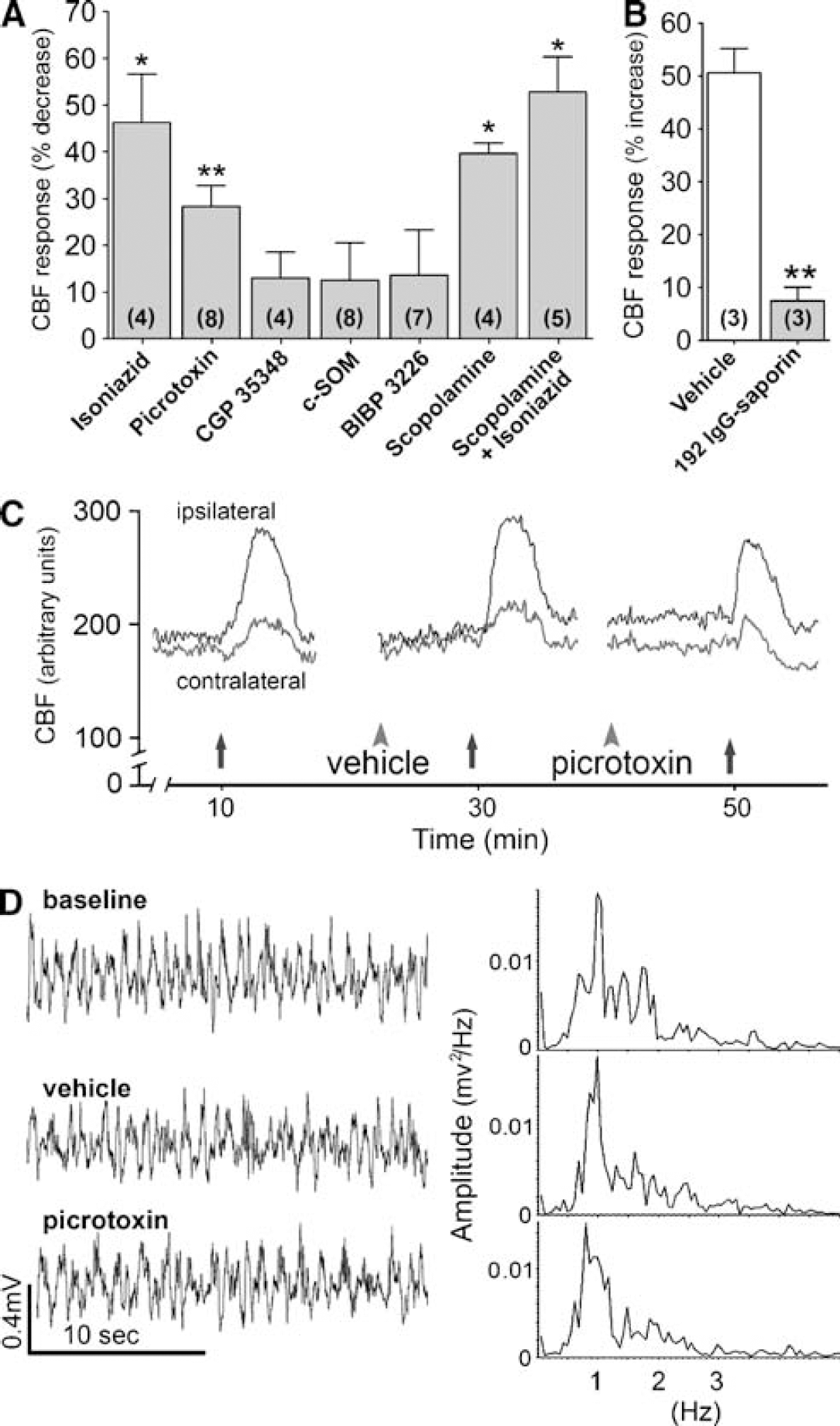
(
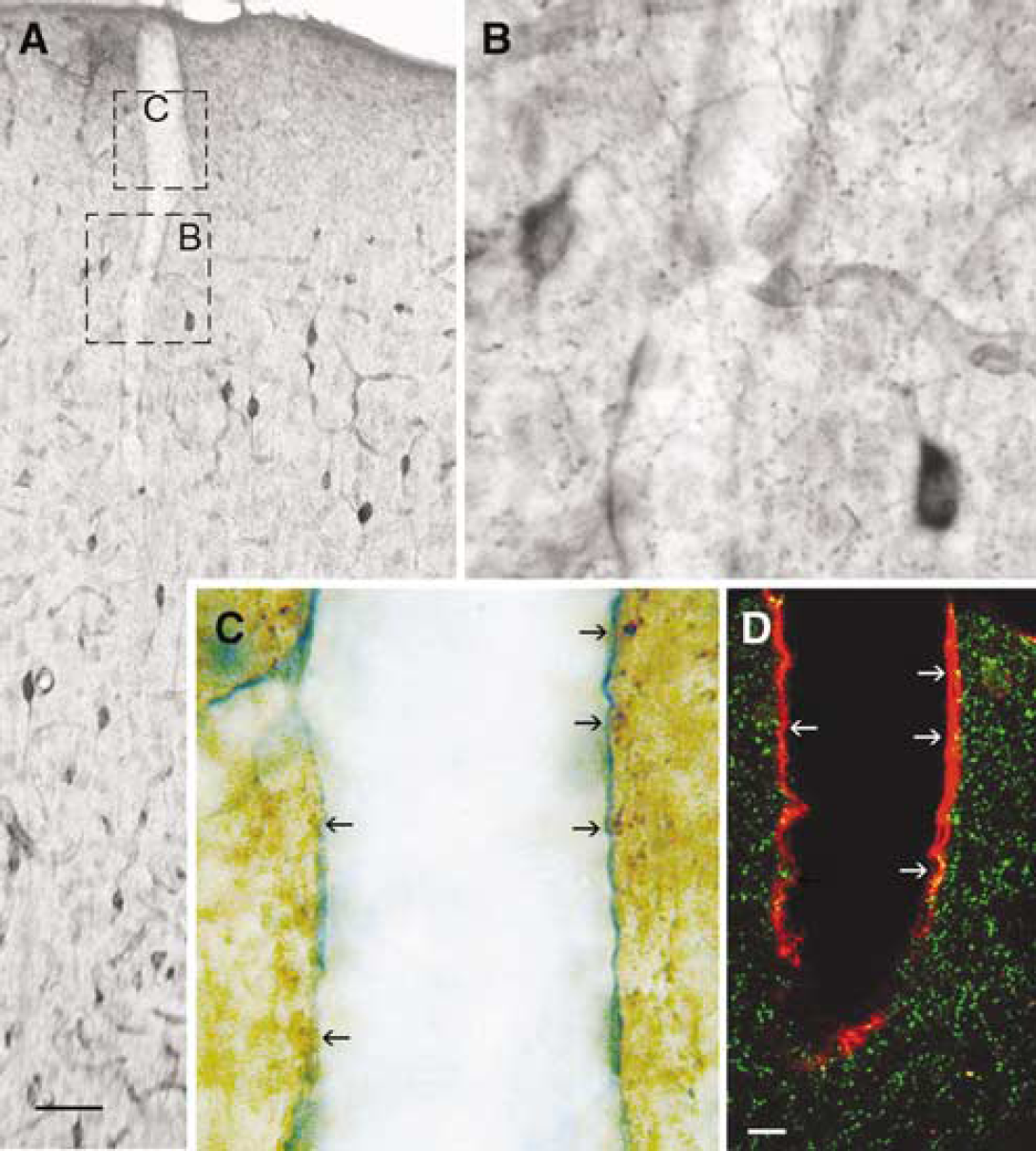
Although these findings showed a role for GABA and ACh in the neurovascular coupling response induced by BF stimulation, the cortical or BF origin of the cells releasing these mediators was not identified. To address this issue, we used rats treated with the selective cholinotoxin saporin. These rats exhibited intact GAD67- and NOS-containing BF neurons and cortical projections, but had marked cortical ACh deafferentation with only minimal loss of ACh neuronal cell bodies in the SI, in agreement with previous reports (Heckers et al, 1994) (Figure 5). In such rats, the CBF response evoked by electrical stimulation of the BF was virtually abolished (–85%, Figure 4B), demonstrating that stimulation of ACh neurons within the BF triggered the CBF response through their basalocortical projections, and that GABA released from activated cholinoceptive cortical interneurons mediated part of this response.
Effects of saline (left panels) or saporin (right panels) on (
Somatostatin-Containing γ-Aminobutyric Acid Interneurons Target Penetrating Vessels in Superficial Layers
Previous studies have shown associations between cortical microvessels and GABA interneurons (Vaucher et al, 2000), and particularly those that contain NPY, NOS, VIP, or ACh (for a review, see Hamel, 2006). Because there was no information available for SOM-containing GABA interneurons that we found to be predominantly activated during BF-evoked CBF increase, we also studied their interactions with cortical microvessels. Double-immunostained sections for SOM and microvessels revealed that SOM interneurons extended numerous varicose nerve fibers across all layers of the neocortex, and primarily in superficial layers where they contacted the walls of penetrating arteries and large microvessels. Fibers were more sparsely distributed in deep cortical layers but perivascular associations were still present (Figure 6), indicating that SOM-containing GABA interneurons preferentially target brain vessels in superficial cortical layers.
Double immunostaining for SOM and cortical microvessels (collagen IV-immunodetected). (
Discussion
Our results show that the cortical neurovascular coupling response to BF stimulation occurred concurrently with activation of layers II to VI SOM- and/or NPY-containing interneurons, as well as layer I GABA interneurons. These anatomic findings together with the pharmacologic data and the virtual loss of the hemodynamic response after cholinergicdeafferentation of the cerebral cortex indicated that the evoked CBF response was driven by basalocortical ACh afferents and mediated, in part, through local activation of specific subsets of cholinoceptive cortical GABA interneurons. Moreover, the results demonstrated that the full expression of the CBF response was dependent on GABA-A receptor mediated transmission.
Selective Activation of Layer I, and Somatostatin- and/or Neuropeptide Y-Containing γ-Aminobutyric Acid Interneurons
Two remarkable findings from the c-Fos experiments were the selective activation of layer I, and broadly distributed SOM- and/or NPY-containing GABA interneurons, as well as the virtual absence of activated pyramidal cells in layer V. The latter was consistent with the recently shown robust m1AChR-mediated inhibition of layer V pyramidal cells (Gulledge et al, 2007). Likewise, activation of layer I GABA interneurons almost certainly reflected the ubiquitous fast nicotinic receptor-mediated depolarization of these electrically coupled cells that, in slices, predominantly inhibited layer II/III interneurons (Christophe et al, 2002, Gulledge et al, 2007), regardless of a similar depolarization in a small proportion of layer II to IV VIP- and/or SOM-containing interneurons (Porter et al, 1999; Gulledge et al, 2007). The prevalent activation of SOM- and/or NPY-containing interneurons across cortical layers also agreed with these cells being the main targets for basalocortical ACh afferents (Cauli et al, 2004), and SOM interneurons being primarily depolarized by mAChR activation (Kawaguchi, 1997). Interestingly, mAChR activation of electrically coupled SOM-containing Martinotti type (Beierlein et al, 2000) or multipolar bursting GABA interneurons (Blatow et al, 2003) elicited synchronized cortical activity that, for the former, correlated with inhibition of the fast-spiking, mainly parvalbumin-containing, interneuron network. Activated NPY interneurons and, particularly, NPY neurogliaform cells (Cauli et al, 2004) could similarly control the activity of other interneuron networks through their widespread electrical connections (Simon et al, 2005).
Altogether, our in vivo findings with c-Fos immunodetection suggest that activation of specific networks of interneurons contributes to the increase in local cortical CBF that occurs, partly, through inhibition of their postsynaptic target interneurons. This conclusion is compatible with the specificity of electrical and chemical connections of different networks of interneurons allowing them to function independently once activated by their respective afferent input (Hestrin and Galarreta, 2005). As such, the weak proportion of BF-activated VIP and ChAT interneurons, which partially correspond to the same interneuron subtype (Chédotal et al, 1994), may result from both their modest innervation by basalocortical ACh afferents as compared with SOM and NPY interneurons (∼30 versus ∼65%, Cauli et al, 2004) and their silencing by ACh-activated interneurons. Moreover, our results agree with and extend, at the cellular level, a recent brain imaging study that suggested that increased activity in inhibitory interneurons initiated the hemodynamic response associated with synchronized cortical activity, as mediated through the BF (Niessing et al, 2005).
Acetylcholine and γ-Aminobutyric Acid as Mediators of the Neurovascular Coupling Response to Basal Forebrain Stimulation
The exquisite efficacy of ACh deafferentation in virtually abolishing the BF-evoked hemodynamic response demonstrated that ACh released from basalocortical afferents initiated this response. The more severe decrease produced by cholinergic deafferentation in comparison to mAChR antagonism (–85 versus ∼40%) further confirmed the involvement of both nicotinic and muscarinic receptors in the BF-induced perfusion response (Biesold et al, 1989; Dauphin et al, 1991). Together with the c-Fos studies, these pharmacologic findings pointed to blockade of ACh receptor-mediated activation of cholinoceptive GABA interneurons contributing to the decreased neurovascular coupling response. This is notwithstanding additional blocking effects of scopolamine on mAChR-activated pyramidal cells (McCormick and Prince, 1986), m5AChR-mediated dilatation of cortical microvessels (Elhusseiny and Hamel, 2000), or astroglial cell responses to cholinergic activation (Seigneur et al, 2006).
The deafferentation data combined with the ability of both isoniazid and the GABA-A receptor antagonist picrotoxin to decrease the BF-induced CBF response, albeit to different extents (46 and 28%, respectively), further indicated that GABA released from cholinoceptive GABA interneurons, and not from BF neurons, mediated part of the neurovascular coupling response. Although it could be argued that our pharmacologic manipulations resulted in incomplete blockade of GABA receptors because we used low doses of antagonists that did not alter baseline CBF or cortical activity (EEG), GABA-A receptors were clearly involved in the CBF response to BF stimulation. Additionally, though intracisternal injection of drugs would favor their rapid access to the cerebral cortex, the final effective dose and volume of diffusion for each drug are not known and, as such, the contribution of other receptors or subcortical sites of action cannot be completely ruled out.
Blockade of GABA-A receptors on pyramidal cells would be expected to disinhibit these cells and increase cortical activity and perfusion. Consequently, the decreasing effect of picrotoxin on the CBF response to BF stimulation was better explained by blockade of GABA-A receptors on and disinhibition of second-order GABA interneurons, the targets of BF-activated cholinoceptive interneurons (Figure 7), leading to decreased pyramidal cell output (Beierlein et al, 2000, Hestrin and Galarreta, 2005). Blockade of the GABA-A receptor-mediated vasodilatation of microvessels (Fergus and Lee, 1997), through their activation on either vascular or astroglial (Bureau et al, 1995) cells, may also account for the decreased CBF response after picrotoxin. In this respect, GABA released from activated layer I interneurons through their superficial arborizations (Kubota et al, 1994, Markram et al, 2004) could act on penetrating arteries that are known to be of prime importance in feeding deeply located microvessels and neurons (Nishimura et al, 2007). What is more, our findings of SOM-containing perivascular fibers across the cerebral cortex and particularly in superficial layers where they contacted penetrating vessels, typical of the axonal arborization of SOM-containing Martinotti interneurons (Markram et al, 2004), suggest that these widely distributed interneurons may be strategically positioned to release GABA or SOM on penetrating arterioles. Further, these findings raise the possibility that the impaired cortical perfusion seen in Alzheimer's disease may be imputed not only to ACh denervation of cortical microvessels (for refs, Hamel, 2004), but also to the selective loss of cortical SOM interneurons (Gaspar et al, 1989).
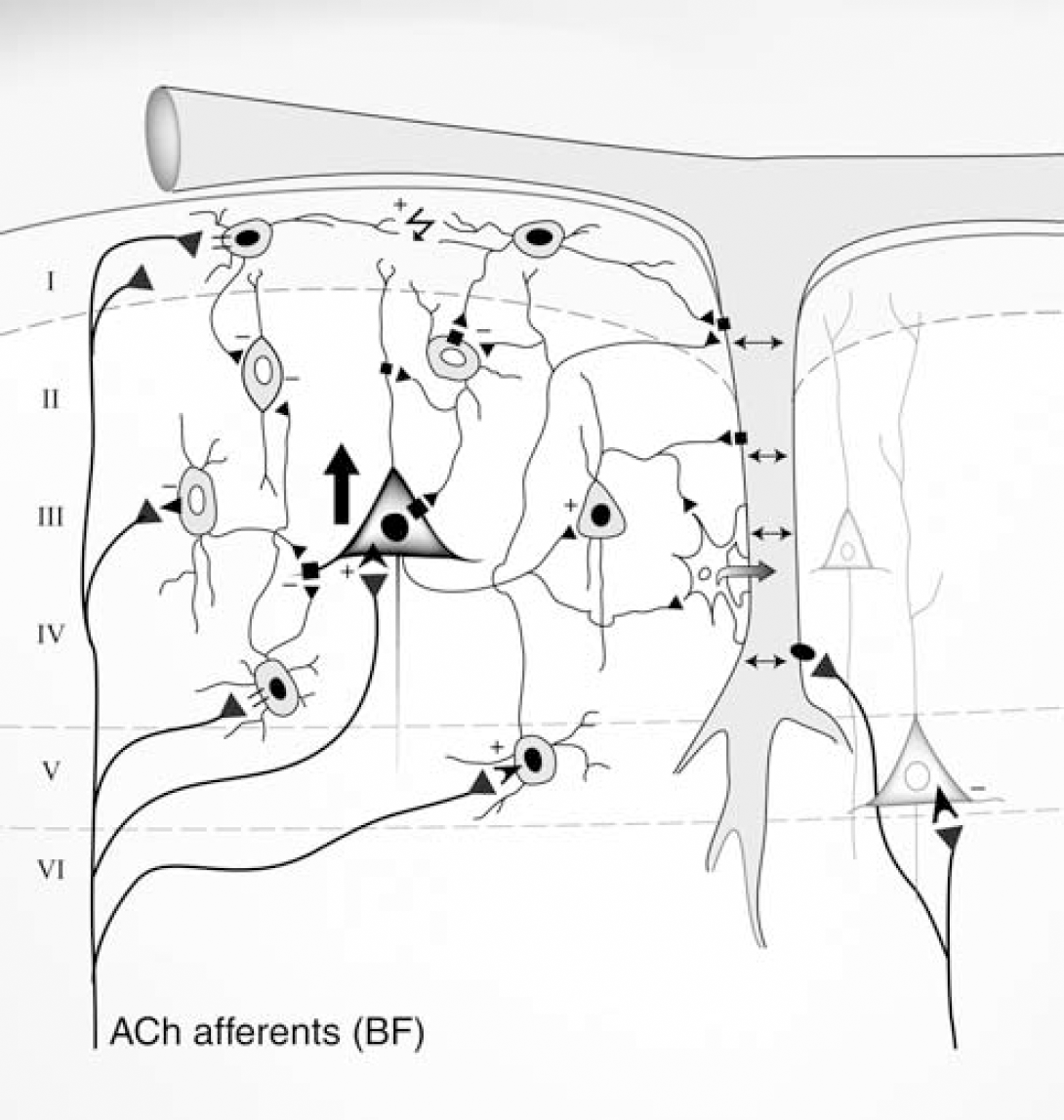
Schematic representation of the role of GABA interneurons in the neurovascular coupling response to BF stimulation. Stimulation of BF ACh afferents activates layer II to VI SOM- and NPY-containing, and layer I, GABA interneurons (black nuclei, c-Fos-positive cells) through nicotinic (=) or muscarinic (◀) receptors, some of interneurons being electrically coupled (↯). In contrast, layer V pyramidal cells would be hyperpolarized (white nuclei, c-Fos-negative cells). GABA released from activated interneurons could act on GABA-A receptors (■) on (i) microvessels and/or perivascular astrocytes (star-like cell attached to the vessel wall) to induce dilatation (↔), and (ii) target interneurons, leading to their inhibition (white nuclei, c-Fos-negative) and, hence, disinhibition of pyramidal cells and increased cortical output (large black arrow, c-Fos-positive cell). Pyramidal cells would release glutamate that induces calcium signaling in astrocytes, leading to vasodilatation (→). Additionally, mAChR-mediated depolarization of pyramidal cells (➤) and m5AChR-mediated dilatation of cortical microvessels (black oval on vessel wall) by ACh released from BF afferents may contribute to the perfusion response. See text for more details.
Contractile receptors for SOM or NPY have been identified in a small proportion of cortical microvessels (Cauli et al, 2004) and it was recently reported that SOM could increase Ca2+ signaling in perivascular astrocytes (Straub et al, 2006), a response normally associated with vascular dilatation. However, SOM and NPY-Y1 receptor antagonism failed to alter the BF-evoked perfusion response. Although their vascular effects may be masked by the dominating dilatory response induced by increased activity in GABA interneurons and pyramidal cells, and the direct vasodilator effect of ACh or GABA on cortical microvessels, it is possible that SOM or NPY release would occur at firing rate higher than that needed to release GABA, and which may depend on the levels of endogeneously released ACh. Yet, the possibility that fine, local vasomotor responses mediated by these peptides occurred that could not be detected when measuring global changes in cortical CBF with laser-Doppler flowmetry cannot be discarded.
Conclusion and Functional Significance
We conclude that the cellular basis of the BF-induced cortical hemodynamic response primarily involves (i) increased activity in layer I, and more widely distributed SOM- and/or NPY-containing GABA interneurons, and release of GABA acting on GABA-A receptors on cortical microvessels and/or astrocytes, (ii) activated pyramidal cells, either directly from BF ACh afferents or after their disinhibition through GABA-A receptor-mediated inhibition of their pre-synaptic GABA interneurons; the contribution of pyramidal cells likely involving glutamate receptor-mediated release of vasoactive astrocytic messengers (Zonta et al, 2003), and (iii) a direct vasodilator effect mediated by ACh released from perivascular BF afferents (Figure 7). Our findings highlight the importance of the cellular basis of the activated neuronal circuitry when interpreting neurovascular signals, and they do not discard the contribution of additional vasoactive molecules derived from neuronal activity, astroglial or vascular processes. Further, they suggest that GABA released from interneurons rather than colocalized peptides is the main signaling molecule, in line with its primary role as a modulator of cortical activity and, hence, of the accompanying changes in local perfusion.
Footnotes
Acknowledgements
We thank Drs M Avoli for expert EEG inspection and analysis evaluation, and BE Jones for helpful discussion.
