Abstract
Lipocalin 2 (LCN2) is able to sequester iron-loaded bacterial siderophores and, therefore, is known to participate in the mammalian innate immune response. Of notice, LCN2 was shown to display bacteriostatic effects both in in vitro and in vivo. To reach the brain, bacteria must cross the blood—brain or the choroid plexus (CP)/cerebrospinal fluid (CSF) barriers. Additionally, as the CP is responsible for the production of most of the CSF, responses of the CP mediate signaling into the brain. We show here that in conditions of peripheral inflammation, LCN2 behaves as an acute phase protein in the CP. As early as 1 h after lipopolysaccharide peripheral administration, Lcn2 mRNA levels are upregulated, returning to basal levels after 72 h. Increased LCN2 protein is observed in choroidal epithelia and in endothelial cells of blood vessels in the brain parenchyma. Higher levels of LCN2 are also present in the CSF. These observations suggest that expression of LCN2 at the CP/CSF barrier might be bacteriostatic in the brain, avoiding bacteria dissemination within the CSF into the brain parenchyma. This study shows that the LCN2 is produced by the CP as a component of the innate immune response that protects the central nervous system from infection.
Introduction
The brain is protected from the periphery by the blood—brain and blood—cerebrospinal fluid (CSF) barriers. These barriers restrict the passage of several endogenous and xenobiotic compounds from the periphery to the central nervous system. Although most studies focus on the blood—brain barrier, evidence is increasing on the role of the choroid plexus (CP) in communication between the immune system and the central nervous system. The CP is located within the brain ventricles, and its epithelial cells are responsible for producing most of the CSF. In addition to producing CSF, the CP is also an active site of protein synthesis and possesses receptors for several immune mediators, hormones, and growth factors (Chodobski and Szmydynger-Chodobska, 2001). Therefore, anatomically and physiologically, the CP is well positioned to respond to stimulus induced in the periphery. Unraveling which are the molecular mediators of the CP response to inflammation and infection is, therefore, important to understand how the CP mediates or protects the brain from associated brain damage.
The Gram-negative bacteria lipopolysaccharide (LPS), a major molecular component of the cell wall, has been widely used as a model of inflammation and as a tool to study response to infection. Cells of the immune system are able to recognize LPS through the Toll-like receptors (TLRs), which induce signals responsible for the activation of the innate immune response. One of the proteins shown previously to participate in such a response is lipocalin 2 (LCN2). Lipocalin 2, a member of the lipocalin family of proteins, was initially found in neutrophil granules (Kjeldsen et al, 2000) and later described as an acute-phase protein in the liver (Liu and Nilsen-Hamilton, 1995; Sunil et al, 2007), in blood and peritoneal cells (Flo et al, 2004), and in several epithelial tissues (Cowland et al, 2003). Lipocalin 2 binds to the iron-loaded siderophores, iron chelators secreted by pathogens, and represents an effective defense strategy for the body to control the growth of pathogens (Neilands, 1995). By limiting iron availability, LCN2 exerts a bacteriostatic effect as shown both in in vitro (Goetz et al, 2002) and in vivo (Flo et al, 2004). Thus, limitation of iron availability is a very effective defense strategy for the body to control the growth of pathogens.
In this study, we intended to investigate whether the LCN2 was among the immune modulators induced in the CP in response to peripheral inflammation.
Materials and methods
Animals and Lipopolysaccharide Injection
All experiments were conducted using 8- to 9-week-old C57BL/6 male mice (Charles River, Barcelona, Spain), in accordance with the European Community Council Directive 86/09/EEC guidelines for the care and handling of laboratory animals. Animals were maintained under 12 h light/dark cycles at 22 to 24°C and 55% humidity and fed with regular rodent's chow and tap water ad libitum. To reduce stress-induced changes in hypothalamus—pituitary axis associated with the injection, animals were handled for 1 week before the beginning of the experiment. Animals were administered LPS intraperitoneally at a 5 μg/g body weight dose (Escherichia coli, serotype O26:B6; Sigma, St Louis, USA); a subset of animals was injected with vehicle (0.9% NaCl) alone.
Animals were anesthetized with ketamine hydrochloride (150 mg/kg) plus medetomidine (0.3 mg/kg), transcardially perfused with cold saline, and killed 1, 3, 6, 12, 24, or 72 h after LPS injection. For the mRNA studies, CP isolation was made under conventional light microscopy (SZX7; Olympus, Hamburg, Germany), and tissue was rapidly removed, frozen in dry ice, and stored at −80°C. At least five pools of CP (from three animals each) were prepared for each time point. The experiment was performed twice. Cerebrospinal fluid was collected from the cisterna magna and pooled from several animals. An aliquot of each pool was used to verify the absence of blood contamination and the remainder was immediately frozen.
Quantitative PCR Lipocalin 2 Gene Expression Measurements
Total RNA was isolated from CP using Trizol reagent (Invitrogen, Carlsbad, CA, USA). 500 ng of total RNA were amplified using the Superscript RNA amplification system (Invitrogen) according to the manufacturer's instructions. Choroid plexus RNA was reverse transcribed using random primers of the superscript first-strand synthesis system for reverse transcription PCR (Invitrogen).
Quantitative real-time PCR analysis was used to measure the expression levels of the Lcn2 mRNA transcript. The reference gene, hypoxanthine guanine phosphoribosyl transferase (Hprt), was used as internal standard for normalization (gene abbreviations and gene names are specified in accordance with the HUGO Gene Nomenclature Committee at http://www.gene.ucl.ac.uk/nomenclature/), since we have first confirmed that its expression is not influenced by the experimental conditions.
The oligonucleotide primers for Lcn2 and Hprt were designed using the Primer3 software on the basis of the GenBank sequences NM_008491 and NM_013556, respectively. The real-time PCR reactions, using equal amounts of total RNA from each sample, were performed on a LightCycler instrument (Roche Diagnostics, Basel, Switzerland) using QuantiTect SYBR Green RT-PCR reagent kit (Qiagen, Hamburg, Germany). Product fluorescence was detected at the end of the elongation cycle. All melting curves exhibited a single sharp peak at a temperature characteristic of the primer used.
Immunohistochemistry
Animals were transcardially perfused, under anesthesia, with 4% of paraformaldehyde in phosphate-buffered saline, 12 and 24h after LPS or saline injections. After perfusion, brains were removed from the skull, left 24h in the fixative solution, and then included in paraffin. Immunohistochemistry was performed on coronal sections (10μm) with anti-mouse lipocalin-2/neutrophil gelatinase-associated lipocalin (R&D Systems, Minneapolis, MN, USA) at 1:500 dilution or anti-transthyretin (kindly provided by Dr MJ Saraiva, Institute for Molecular and Cell Biology, Porto, Portugal) at 1:1,000 dilution as primary antibodies. Secondary antibodies, biotinylated (Vector Laboratories Inc., Burlingame, CA, USA) or fluorescent (Molecular Probes, Carlsbad, CA, USA) were used at 1:200 and 1:500, respectively, following standard procedures. For this analysis, we used three animals per group. Samples were analyzed using optical (BX61; Olympus) or confocal (FV1000; Olympus) microscopes.
Protein Measurements
For the detection of LCN2 protein in the CSF, we performed a direct enzyme-linked immunosorbent assay. Two microliters of CSF was used to detect LCN2 levels at the different time points. Lipocalin 2 was detected using the same antibody mentioned above, at 1:300 dilution, followed by a secondary peroxidase-conjugated donkey anti-goat antibody (Santa Cruz Biotechnology, Santa Cruz, CA, USA) at 1:500 dilution, and developed with 2-2′azinobis (3-ethylbenzthiazoline-6-sulphonic acid) diammonium salt (ABTS) (Sigma). The reaction was stopped using 0.1 mol/L citric acid and read at an optical density of 405 nm. The standard curve was made with the recombinant mouse LCN2/neutrophil gelatinase-associated lipocalin (R&D Systems). The detection limit was 50 ng/mL.
Statistical Analysis
Values are reported as mean±s.e. Statistical significance was determined using the non-parametric Mann—Whitney test, with differences considered significant at P < 0.05.
Results
Lipocalin 2 Expression Profile in Choroid Plexus and Cerebrospinal Fluid Levels after Lipopolysaccharide Injection
Choroid plexus Lcn2 expression was very low in basal conditions (and identical at any time point after saline injection), but strongly upregulated from 1 to 72 h after LPS injection, with a peak at 12 h (Figure 1A). In the CSF, LCN2 was below detection level in basal conditions until 3 h after LPS injection (Figure 1B). A robust upregulation was observed at 6 h, remained for at least 24 h and returned to basal levels at 72 h after LPS administration. The same expression profile was observed in the liver (data not shown). Increased expression of Lcn2 in the CP preceded the increase in the CSF protein, suggesting that LCN2 is produced and secreted by epithelial CP cells to the CSF.
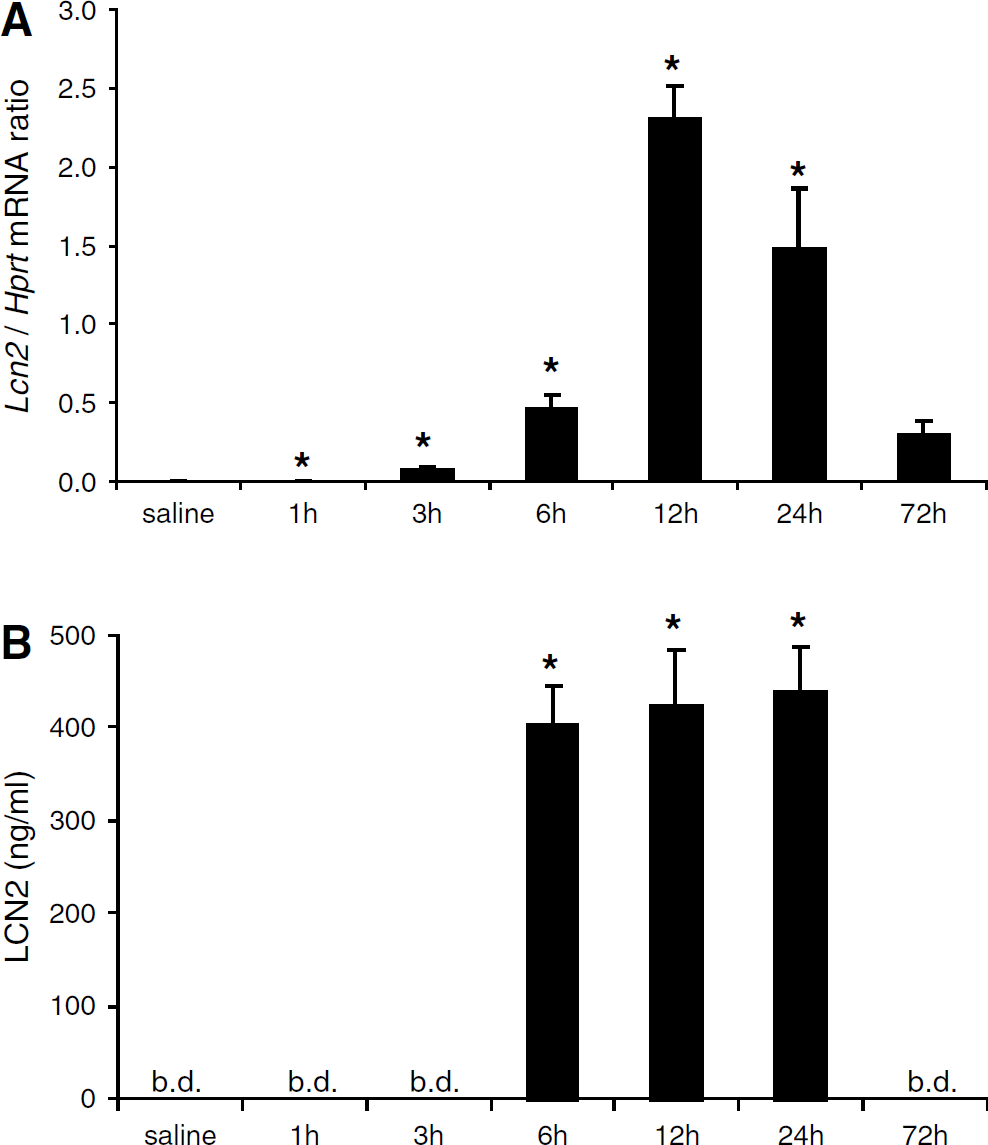
Lipocalin 2 (Lcn2) mRNA expression in the choroid plexus and LCN2 cerebrospinal fluid (CSF) levels are increased in mice injected with lipopolysaccharide (LPS). (
Immunohistochemistry Analysis
We next studied, by immunohistochemistry, the LCN2 protein immunoreactivity in the CP. As shown in Figure 2, although no signal was observed in saline controls (Figures 2A, 2D and 2G) at 12 h (or at any time point after saline injection), at 12 h after LPS administration immuno-positive staining was observed in CP epithelial cells but also in other cells within the CP stroma (Figures 2B and 2E); staining was also observed in endothelial cells throughout the brain parenchyma (Figure 2H). At 24 h, staining was mostly observed in CP epithelial cells (Figures 2C and 2F). In the endothelial cells of the brain parenchyma, a weaker staining was still observed (Figure 2I). Of notice, not all CP epithelial cells showed labeling for LCN2. Double staining for LCN2 and transthyretin, a protein specifically synthesized by CP epithelial cells, confirmed that LCN2 is produced by epithelial cells of the CP (Figure 2J).
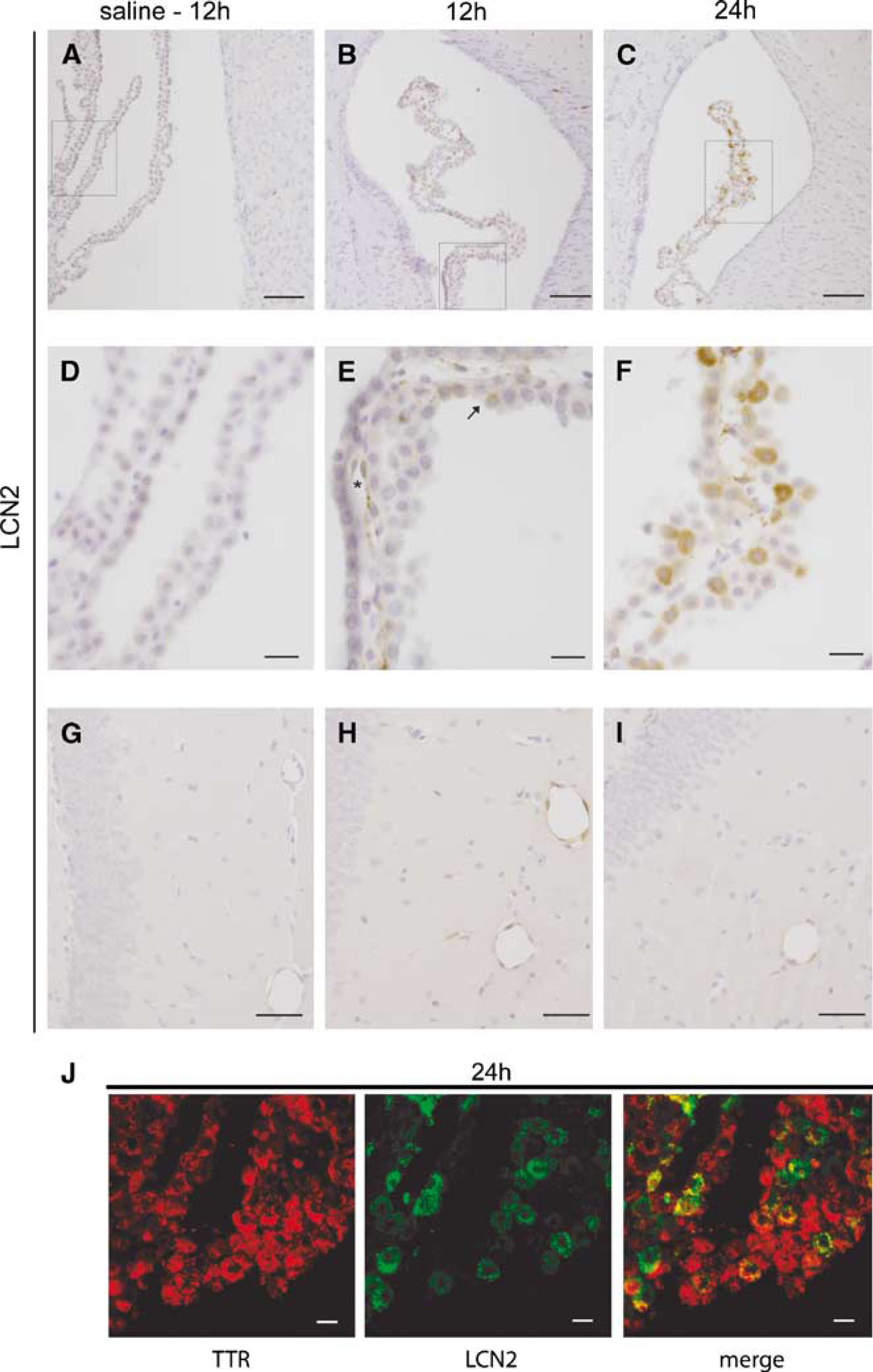
Immunohistochemistry for lipocalin 2 (LCN2) in the choroid plexus (CP) and in blood vessels of the brain parenchyma of mice injected with lipopolysaccharide (LPS). Although no staining is present in (A, D, and G) saline controls, LCN2-positive cells are observed (
Discussion
Although an acute-phase response is well described for the liver, increased evidence is supporting a similar response at the blood—brain barriers (Marques et al, 2007; Quan et al, 1998). This study shows, in vivo, that the gene encoding for LCN2 is quickly upregulated in the CP in response to a peripheral immune challenge. As a result, increased levels of LCN2 are secreted into the CSF. Similarly, increased LCN2 is also observed in blood vessels of the brain parenchyma. Because LCN2 binds to various iron-loaded bacterial siderophores, this increase in LCN2 levels is expected to decrease iron availability for bacterial growth and, therefore, may constitute an important mechanism of neuroprotection against bacteria accessing and disseminating within the brain.
While reporting the blood—brain and blood-CSF barriers as novel sites of LCN2 synthesis, we confirm liver upregulation of Lcn2 mRNA after an LPS inflammatory challenge with a profile similar to that just recently reported (Sunil et al, 2007).
Within the CP, immunohistochemistry reveals positive labeling for LCN2 in epithelial cells only in LPS-treated animals. Interestingly, not all CP epithelial cells are immunoreactive to LCN2 antibodies, suggesting that only certain cells express LCN2 in detectable amounts, at least at a given time. Staining is also observed in blood vessels in the brain parenchyma. It is likely that the other cells of the CP stroma immuno-positive for LCN2 are also endothelial cells of the CP vasculature. These observations together with the fact that endothelial cells of the CP capillaries are fenestrated, and that LCN2 also increases in the blood upon an inflammatory stimulus (Flo et al, 2004) might be particularly relevant in impeding pathogen access into the brain. On one hand, LCN2 might reduce iron access to the bacteria in the blood vessels and, in the case of bacteria entering into the CSF, CSF-borne LCN2 can sequester siderophore-bound iron and prevent bacteria dissemination within the ventricular brain system and into the brain parenchyma. Of notice, it has been recently reported that human neutrophil gelatinase-associated lipocalin, the human homolog of LCN2, is taken up by cells through the receptor megalin (Hvidberg et al, 2005). Since megalin is a scavenger receptor present in the CP and in the endothelial cells of blood vessels, it is possible that neutrophil gelatinase-associated lipocalin bound to iron-loaded siderophore can be taken up by these cells, further preventing iron availability for bacterial growth within the CSF and the brain parenchyma.
This is, to the best of our knowledge, the first demonstration of LCN2 synthesis at the blood—brain barriers in vivo. Previous studies failed to show Lcn2 mRNA in whole brain extracts or several regions of the brain parenchyma (Cowland et al, 2003) under ormal physiological conditions. On the contrary, two other studies have previously reported upregulation of LCN2 expression in whole brain extracts by turpentine (Liu and Nilsen-Hamilton, 1995) and in the basal forebrain of mice intranasally infected with influenza virus (Ding and Toth, 2006). Given this data, it is likely that Lcn2 expression in the CP and in blood vessels is contributing to the overall upregulation of Lcn2 expression observed in whole brain homogenates, particularly since some of these brain regions are often contaminated by CP tissue (Sousa et al, 2007). Of notice, a recent report using LPS stimulation of primary cultures enriched in CP epithelial cells showed induction of LCN2 secretion, which is in agreement with the in vivo data we report here (Thouvenot et al, 2006).
Still to investigate is the molecular pathway that triggers LCN2 expression at these sites. In vitro studies in a lung epithelial cell line stimulated by interleukin-1β (IL-1β) showed upregulation of LCN2, whereas LPS did not (Cowland et al, 2006). It is possible that IL-1β produced in the periphery in response to LPS induces a response in the CP and in blood vessels through IL-1β receptors (Cunningham et al, 1992). This would induce the synthesis of additional IL-1β and other immune mediators, such as LCN2. Alternatively, or in addition, the response might be directly linked to activation of receptors to bacterial endotoxins located at the barriers, as has been shown in vitro for LPS activation of LCN2 expression in macrophages (Meheus et al, 1993) and in lung epithelial cell lines transfected with TLR4 (Cowland et al, 2003). In accordance, several TLRs, including TLR4 and TLR2, are constitutively expressed in the CP and in cells lining the blood vessels (Laflamme et al, 2003).
Irrespective of the molecular pathway that triggers their expression, proteins secreted by the CP toward the CSF may participate in the host's defense against CSF bacterial infections. Interestingly, in the presence of IFN-γ, the supernatant of porcine CP epithelial cells displayed a bacteriostatic effect against Streptococcus suis. This was at least partially attributed to the induction of indoleamine 2,3-dioxygenase activity and the resulting depletion of L-tryptophan (Adam et al, 2004). Given the data we now present, it is also possible that the secretion of LCN2 into the media also contributes to this bacteriostatic effect. Recent studies clearly show that LCN2-null mice exhibit increased susceptibility to infection by bacteria that depend on entherocalin-like siderophore-dependent iron uptake (Berger et al, 2006; Flo et al, 2004). It will be interesting to study the brain response to infection in these mice.
In summary, we describe here a novel acute-phase response protein produced at the barriers between the blood and the brain. By increasing the expression and secretion of LCN2, these barriers contribute to restrict access of bacteria into the brain. This adds CP and endothelial cells of blood vessels as sites in which LCN2 expression is induced by inflammation, and further supports a role for the blood—brain and blood-CSF barriers as part of the innate immune response against central nervous system infection.
Footnotes
Acknowledgements
Marques F and Rodrigues AJ are recipients of PhD, and Sousa JC postdoctoral, fellowships from Fundação para a Ciência e Tecnologia (Portugal).
Disclosure
The authors declare no conflict of interest.
