Abstract
An adjuvant therapy to prolong the therapeutic window for stroke patients is urgently needed. This randomized, blinded, placebo-controlled study investigated adjuvant intravenous sodium nitrite with recombinant tissue plasminogen activator (rtPA) in middle cerebral artery occlusion (MCAO) with 6 and 2 h of ischemia followed by reperfusion in Sprague—Dawley rats (n = 59). Quantitative diffusion, T1-, T2-weighted, and semiquantitative perfusion imaging were performed before and after reperfusion and at 48 h after ischemia to determine the spatiotemporal evolution of stroke. After 48 h animals were killed and examined to evaluate infarct size and evidence of hemorrhagic transformation. Factor VIII immunostaining was performed to assess vessel morphology. Nitrite treatment (6 h group: 37.5 μmol for more than 90 mins; 2 h group: 26.25 and 1.75 μmol for more than 60 mins) did not reduce infarct volume 48 h after MCAO compared with saline-treated placebo groups after 6 or 2 h of MCAO. Stroke progression from baseline to 48 h, based on the apparent diffusion coefficient and relative cerebral blood flow deficits before and after reperfusion and T2-weighted hyperintensity at 48 h, did not differ between treated and control animals. These results suggest that nitrite is not a protective adjuvant therapy to delayed rtPA administration after ischemic stroke in rats.
Introduction
Thrombolysis using recombinant tissue plasminogen activator (rtPA) is the only treatment for acute ischemic stroke. Because intravenous rtPA increases the risk of hemorrhagic transformation, its use is limited to 3 h after ischemic stroke (NINDS, 1995). If rtPA could be administered safely to patients within 6 h after stroke, it would substantially increase the number of patients eligible for thrombolysis (Kleindorfer et al, 2004). Recent magnetic resonance imaging (MRI) studies indicate that hemorrhagic transformation and poor outcome are associated with blood—brain barrier (BBB) damage after recanalization (Latour et al, 2004). Animal studies suggest that thrombolysis induces ischemia—reperfusion injury through a surge in radical oxygen species and BBB disruption (Szocs, 2004). Therefore, reperfusion injury during thrombolysis is a main target of stroke research.
Sodium nitrite (NaNO2) recently emerged as a promising candidate to reduce ischemia—reperfusion injury because it preferentially releases nitric oxide (NO) at low tissue pH and in the presence of deoxygenated hemoglobin (Cosby et al, 2003) and it crosses the BBB (Pluta et al, 2005). In experimental settings, intravenously administered sodium nitrite has been reported to protect against ischemia—reperfusion injury in the heart and liver (Duranski et al, 2005) but not the kidney (Basireddy et al, 2006), and was recently described as a neuroprotectant (Jung et al, 2006).
On the basis of these encouraging early studies suggesting protection against ischemia—reperfusion injury in other tissues, we examined whether nitrite can reduce reperfusion injury and extend the effective interval of rtPA administration after cerebral ischemia using an established model of reversible middle cerebral artery occlusion (MCAO). Two sets of experiments with delayed reperfusion were used in conjunction with MRI and histology to study possible time-dependent effects of nitrite using several in vivo parameters (i.e., apparent diffusion coefficient (ADC), relative cerebral blood flow (rCBF), T2-weighted MRI, BBB integrity, cerebral edema, and hemorrhagic transformation).
Materials and methods
Study Design
An established model of reversible MCAO in rats (Longa et al, 1989) was used to simulate ischemia—reperfusion injury after rtPA (Pfefferkorn and Rosenberg, 2003). In two experiments, ischemia durations of 6 and 2 h were followed by reperfusion. Magnetic resonance imaging scans were performed before and after therapeutic intervention as well as before killing at 48 h.
To simulate thrombolysis, all animals received intravenous rtPA (10 mg/kg, Alteplase®, Genentech, San Francisco, CA, USA) after MCAO. The infusion of rtPA started 30 mins before reperfusion with a 10% bolus and was maintained for 60 mins (Pfefferkorn and Rosenberg, 2003). This dose is 10 times higher than in humans because the human rtPA has lower activity in rats (Karges et al, 1994).
Experiment 1 (6 h of ischemia). One hour before reperfusion animals received a 10-min bolus of 7.5 μmol (0.075 mol/L) NaNO2 (Sigma-Aldrich, St Louis, MO, USA) diluted in phosphate-buffered saline (PBS; BioWhittaker, Cambrex, MD, USA) followed by an intravenous infusion of nitrite at 0.375 μmol/min for 80 mins (total = 37.5 μmol for more than 90 mins, n = 13). Placebo animals (n = 13) received PBS in lieu of nitrite. Baseline MRI was performed at the fourth hour after MCAO (before any drug infusions) with follow-up scans at 1 h after reperfusion (6 + 1 h) and at 48 h.
Experiment 2 (2 h of ischemia). Two doses of nitrite were investigated: the maximum tolerated dose with minimal effect on blood pressure (n = 7) and a very low dose (n = 6) previously shown to exert maximum protection against ischemia injury in the heart and liver (Duranski et al, 2005). Thirty minutes before reperfusion, treatment animals received a bolus of sodium nitrite diluted in PBS, either 7.5 μmol (0.075 mol/L) or low dose (0.5 μmol, 0.005 mol/L) for more than 10 mins, followed by an intravenous infusion of 0.375 μmol/min (low dose: 0.025 μmol/min) for 50 mins (total 26.25 μmol, low dose = 1.75 μmol). Controls received PBS in lieu of sodium nitrite (n = 10). Baseline MRI was performed at 1 h after MCAO (before drug infusions) with follow-up scans at 1 h after reperfusion (2 + 1 h) and at 48 h. After the 48-h MRI animals were killed. The brains were extracted for histologic analysis.
Animal Preparation
All procedures were performed in accordance with the Animal Care and Use guidelines of the National Institutes of Health. Male Sprague—Dawley rats (n = 59, 300 to 400 g; Taconic Farms, Germantown, NY, USA) were used.
Rat MCAO Model
Anesthesia was induced with 5% isoflurane and maintained at 1.5%. Normothermia (36.5°C to 37.5°C) was maintained using a thermostatically regulated heating pad (American Hamilton Co., OH, USA). The right femoral vein was cannulated for drug infusions and delivery of contrast agent (Gd-DTPA). Transient MCAO and reperfusion were achieved with the intraluminal thread technique (Longa et al, 1989).
Inclusion/Exclusion Criteria
All animals that successfully received treatment or placebo solutions were included in the study. Animals were excluded in case of subarachnoid hemorrhage, defined by hypointensity on T2-weighted imaging at the skull base on baseline MRI. Alternatively, if a region of perivascular enhancement appeared on contrast-enhanced T1-weighted imaging in the area of the right MCA, animals were excluded for suspicion of intracerebral hemorrhage. Rats having a total stroke volume <40 mm3/or regional coverage on less than three imaging slices on diffusion-weighted imaging before reperfusion were excluded as well (‘incomplete occlusion‘).
Assessment of Neurologic Status
Neurologic status was quantified using modification of an existing scale (Garcia et al, 1995) based on four parameters: (a) activity was assessed after 5 mins of observation (0 to 3); (b) symmetry of movement (0 to 3); (c) forelimb outreach when held by tail (0 to 3); and (d) ability to climb/grasp (0 to 3). Scores (a to d) were added resulting in a score between 0 (unresponsive to dead) and 12 (no deficit).
Serial Magnetic Resonance Imaging
Magnetic resonance imaging (Bruker-Biospec, Billerica, MA, USA, 7T/30 cm) was performed using the following sequences: T*2 (for presence/absence of hemorrhage), diffusion (three directions, six b-values = 0 to 1250 mm2/ s), T2 (eight echoes, 15 to 120 ms), T1 (assessment of BBB precontrast), contrast-enhanced perfusion, and T1 (assessment of BBB post-contrast). Multislice image acquisition was performed with a field of view of 2.56 × 2.56 cm, 12 slices (eight for perfusion), a slice thickness of 1.5 mm, and in-plane resolutions of 267 μm for diffusion/perfusion, 200 μm for T2, and 100 μm for T2*/T1.
Image Processing and Analysis
Image analysis and parameter map production (ADC, rCBF, T2) was performed using IDL (Research Systems Inc., Boulder, CO, USA) and MIPAV (BIRSS, NIH, Bethesda, MD, USA). Lesion volumes for acute time points (pre- and post-reperfusion) were calculated based on thresholds of a 23% reduction for ADC and a 57% reduction for rCBF (Meng et al, 2004). Lesion volumes at 48 h were calculated based on a threshold of mean + 2.5 s.d. based on T2 hyperintensity. Edema was assessed using hemispheric volumes from T2-weighted images (edema = ((right—left)/left) × 100).
Histologic Preparation
Whole brains were snap-frozen and cut into 10 μm coronal sections at 1 mm intervals. Cresyl violet staining was performed (Chen et al, 2003) to assess infarct size; hematoxylin and eosin staining was performed to evaluate hemorrhagic transformation.
Vessel Morphology Grading
To quantify the morphologic appearance of microvessels, one 10 μm frozen section was obtained from bregma + 1 mm anteriorly and stained with factor VIII as described elsewhere (Sasaki et al, 1988). Each vessel was graded in the infarct core and at the infarct rim in five field of views at × 20. A masked investigator identified the primary motor cortex (a), caudate putamen (b, c), sensory cortex (d), and nucleus accumbens (e) (Figure 1A) using a stereotactic atlas (Murone et al, 1997). Vessels cut in the transverse plane were not counted. Scores were attributed to each counted vessel in the field of view as follows: 2 = physiological, intact endothelial lining and nuclei, 1 = blurred capillary wall with diffusion of factor VIII to the surrounding tissue, and 0 = disrupted vessel lining. Vessel scores were added for all counted field of views in one hemisphere. The sum of the right-side scores was divided by the sum of scores from the same positions on the left side. Thus, a score close to 0 suggests obliterated vessel morphology, whereas a score of 1 indicates that the stroke hemisphere displays the same morphology as the normal side.
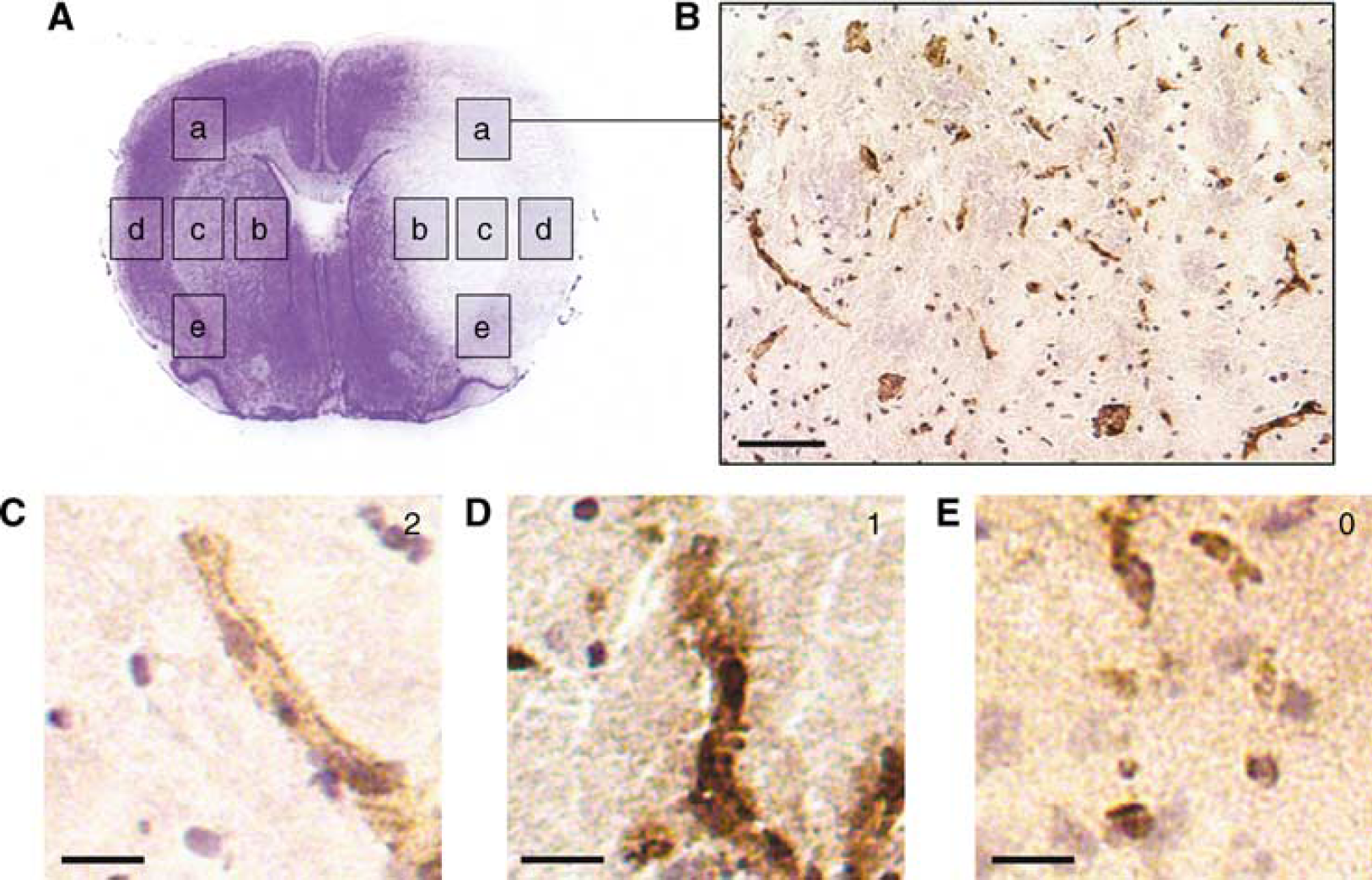
Vessel morphology quantification using factor VIII immunostaining. (
Hemorrhagic Transformation
Hemorrhagic transformation was assessed using a four-point score (Neumann-Haefelin et al, 2002). A score was assigned ranging from 1 (small area of bleeding in a single location identified microscopically but not macroscopically), to 4 (macroscopically detectable space-occupying hemorrhage).
Statistical Analysis
Investigators analyzing data were masked to the distribution of subjects to groups. Data are presented as mean± s.d. All statistical analyses were performed using SPSS (SPSS Inc., Chicago, IL), where P < 0.05 was defined as significant and P < 0.10 was recognized as a trend.
Results
Excluded Animals
Of the 59 experimental animals, two died while under anesthesia before the procedure was completed. Three animals were excluded because of subarachnoid hemorrhage and five animals were excluded because of insufficient baseline stroke volume on MRI.
Outcome
Survival times were not significantly different between nitrite and placebo groups (Kaplan—Meier log-rank test, P > 0.1; survival is given in hours. Six-hour groups: placebo 25.8±18.4, nitrite 37.3±15.0; 2 h groups: placebo 36.8±19.1, nitrite 48.0±0, low-dose nitrite 35.0±20.2). When compared with the appropriate placebo groups, nitrite did not affect infarct volume (Figure 2A), neurologic score (Figure 2B), vessel morphology (Figure 2C), or hemorrhage score (Figure 2D).
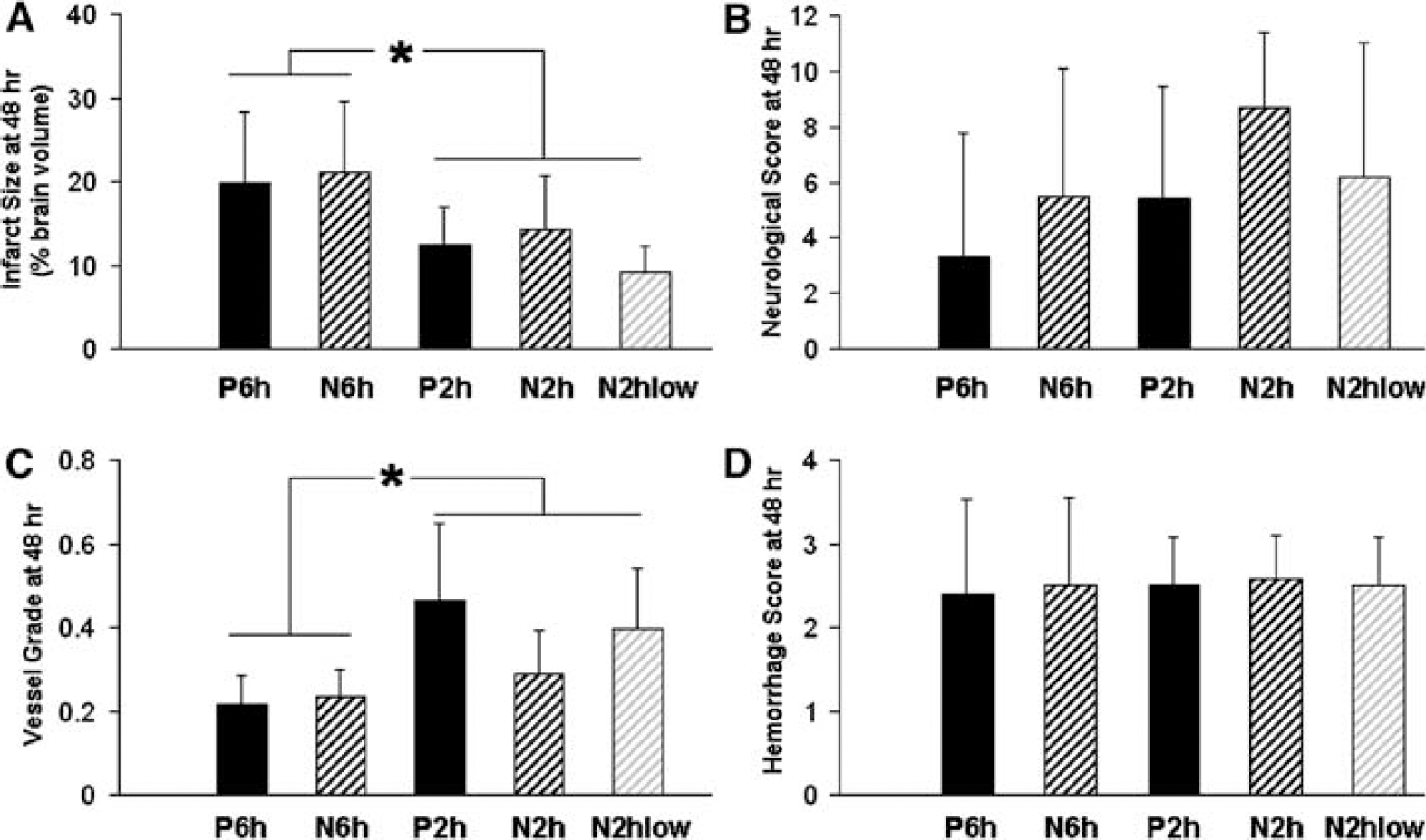
Clinical and histologic outcome at 48 h. (
Because between-group differences were not significant, the pooled data from the 6-h groups (n = 26) were compared with the 2-h ischemia groups (n = 23). As expected, infarct volumes were higher in the 6-h group (Figure 2A, P = 0.04). Further, the 2-h group showed a trend toward better survival (P = 0.07) and neurologic outcome (P = 0.09). Vessel morphology scores in the 6-h group were lower than in the 2-h groups (Figure 2C, P = 0.001).
Magnetic Resonance Imaging
When compared with the appropriate control groups, nitrite did not affect infarct volume. There were no significant differences in the volume of T2 signal increase or cerebral edema. Relative cerebral blood flow levels did not differ among groups. There was a higher incidence of MRI-detectable hemorrhagic transformation in the 6-h groups than in the 2-h groups (P < 0.003).
The evolution of ADC, rCBF, and T2 volumes are depicted in Figures 3 and 4. There were no differences in baseline ADC or rCBF volumes across groups. Final T2 volumes were similar to baseline rCBF volumes. These results indicate similar levels of lesion progression acutely to 48 h, providing additional evidence of no differences between the nitrite and placebo groups.
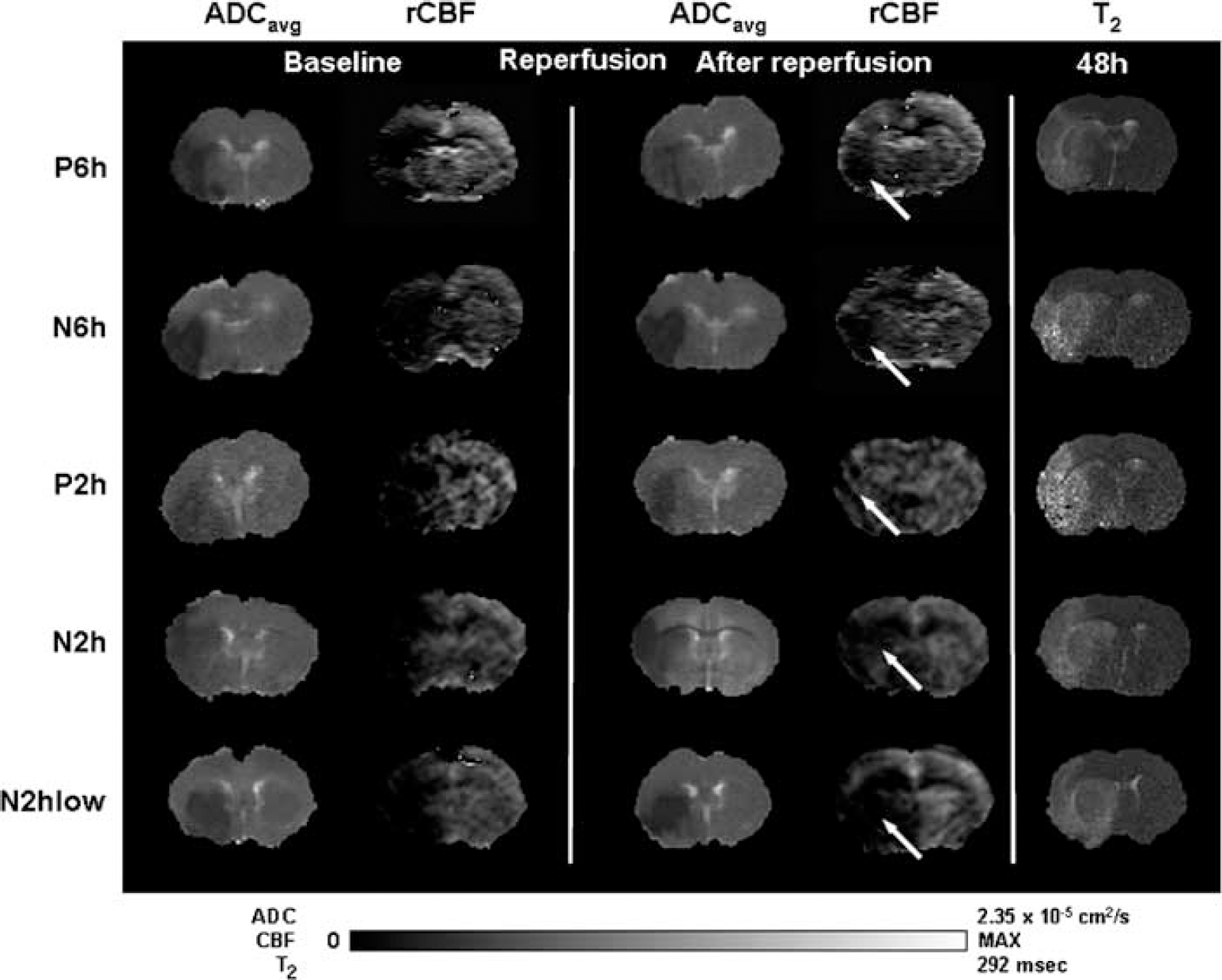
Progression of lesion on MRI in representative animals in the 6- and 2-h ischemia—reperfusion groups. ADC and rCBF parameter maps are shown for baseline and after reperfusion. T2 parameter maps at 48 h indicate final infarct volume. Group assignments (P, placebo; N, nitrite).
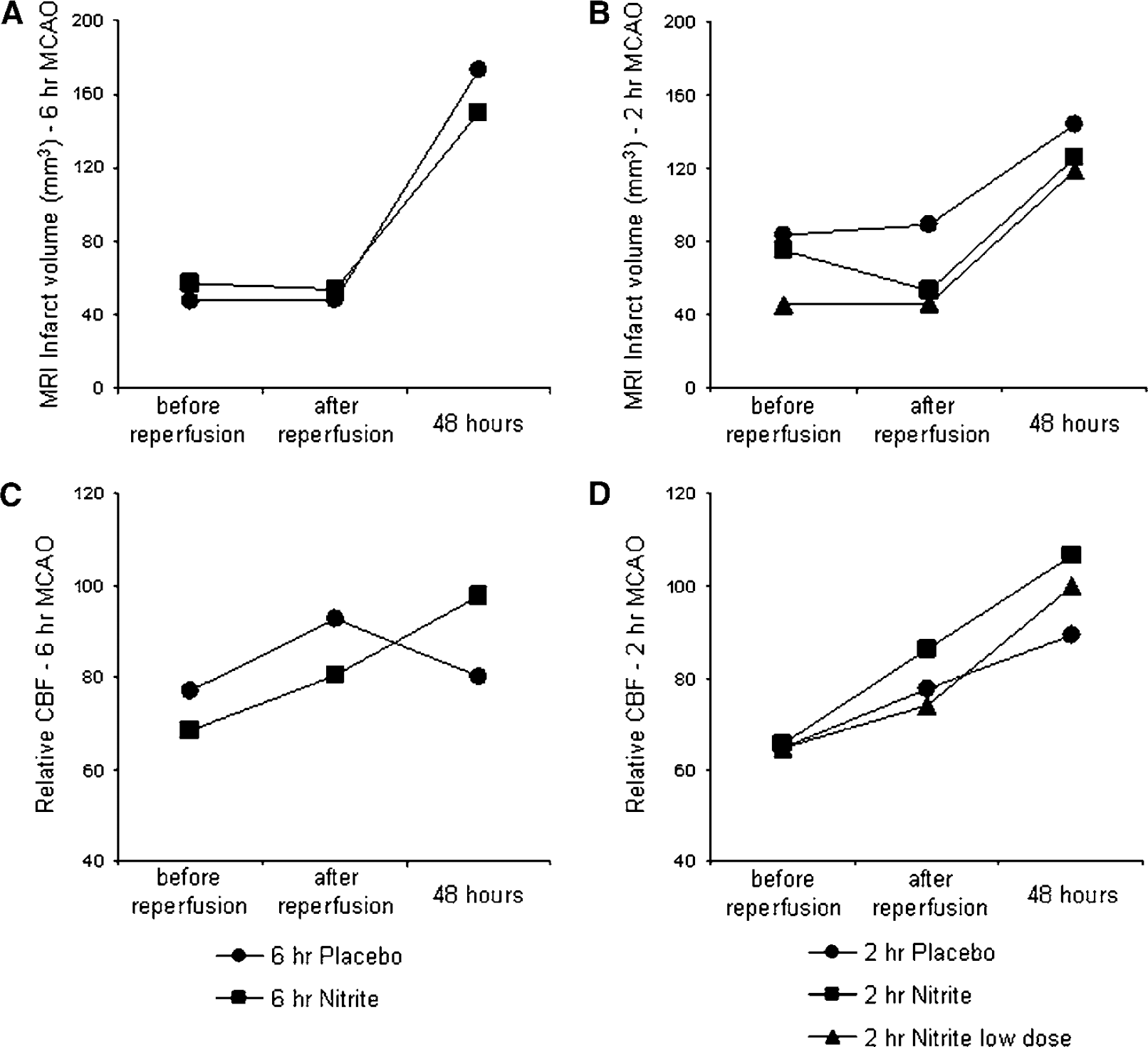
Infarct progression on MRI for (
Discussion
The hypothesis of this study was that nitrite would reduce ischemia—reperfusion injury. However, neither of the treatment regimens, which included two doses and two different durations of ischemia, affected outcome. A potential protective dose should have at least produced a trend toward better outcome in one of the groups. Moreover, because nitrite at the chosen doses did not affect CBF, infarct volume evolution, or vessel health after stroke, it may be concluded that nitrite is not a protective adjuvant therapy when combined with delayed administration of rtPA after ischemic stroke in rats.
The Sources of Nitrite Failure
These results contrast with a recent study reporting a protective effect of nitrite in brain ischemia. In that study, an intravenous bolus of 480 nmol of sodium nitrite delivered for more than 1 min reduced infarct volume by 77% at 24 h (Jung et al, 2006).
Despite certain similarities between Jung et al (2006), important differences may explain this discrepancy. (1) Administration of nitrite. The low dose in the 2-h group was carefully chosen after pharmacokinetic modeling to achieve blood levels identical to the most effective dose in studies of cardiac and renal ischemia—reperfusion as did Jung et al (2006), in their study (480 nmol delivered within 1 min). We did not find this dose-protective. Sodium nitrite in our study was administered as a 50-min infusion, not as a 1-min bolus, which is the dosing used by Jung et al (2006). There was a significant difference in the duration of MCAO (90 mins by Jung et al (2006) versus 120 and 360 mins in our study). (2) Administration of rtPA. Another difference was that we used rtPA to mimic the clinical situation of a contemporary stroke patient, whereas Jung et al (2006) only used MCAO, without rtPA. (3) Study end point. The end point of our study was 48 h, not 24 h, as in the study of Jung et al (2006). (4) Animals used. We used older rats, because younger animals are more resistant to cerebral ischemia (Zhang et al, 2005). The possible role of these differences in the model, duration of ischemia, co-administration of rtPA and nitrite, and study end point, is addressed in following sections.
Duration of Ischemia
The durations of ischemia in this study, 6 and 2 h, were based on recommendations for animal stroke studies (DeGraba and Pettigrew, 2000). Less than 22% of acute stroke patients present within 3 h of stroke onset (Kleindorfer et al, 2004). Thus, positive results in animal models using shorter ischemia times may not translate to the clinic (Dirnagl, 2006). It is often criticized that in brain ischemia research, studies in which neuroprotectants decrease infarct volume (including the study by Jung et al (2006) tend to use short ischemia times. Because time constraints of rtPA treatment limit further improvements in patient care, animal studies increasingly, but not consistently, use longer durations of ischemia. Despite prolonged ischemia, the brain has the potential to be responsive to therapy in experimental models. Studies using permanent ischemia (Bardutzky et al, 2005) and reperfusion injury (Neumann-Haefelin et al, 2002) show that the penumbra, defined by perfusion—diffusion mismatch on MRI, decreases to a minimum 2.5 to 4 h after MCAO. Although this implies that therapy would be minimally effective after that duration, several agents show beneficial effects with reperfusion 4 to 6 h after stroke onset (Pfefferkorn and Rosenberg, 2003; Zhang et al, 2002). In our study, infarct volume in the 6-h ischemia groups was higher than in the 2-h groups. By testing nitrite therapy at different levels of stroke severity, this study showed that nitrite has no beneficial effect in either MCAO model (6 or 2 h).
Co-Administration of Recombinant Tissue Plasminogen Activator and Sodium Nitrite
To address patients with delayed admission (≥ 3 h after stroke symptoms), our study examined delayed nitrite therapy combined with rtPA, because a therapy that works after 6 h, or at least after 2 h of ischemia while co-administered with rtPA could be considered promising for clinical trials (DeGraba and Pettigrew, 2000). Adding nitrite to thrombolysis should reduce reperfusion injury by increasing blood flow and quenching free oxygen radicals, protecting endothelial cells and preventing disruption of the BBB (Pluta et al, 2001). However, we did not detect a beneficial effect. At issue is whether this is due to interactions between these two drugs and if a beneficial effect may be derived by adjusting the timing of nitrite administration. Although nitrite alone has been reported to be neuroprotective up to 90 mins after ischemia (Jung et al, 2006), the absence of an effect in the 6- and 2-h groups suggests that there is a time limit of this potentially beneficial effect. Moreover, BBB integrity deteriorates with ischemia duration. Because both rtPA (Yepes et al, 2003) and NO affect endothelial integrity (Weyerbrock et al, 2003), a detrimental effect on the BBB of rtPA combined with NO released from nitrite cannot be excluded.
Thus, nitrite may have a beneficial effect on brain ischemia and reperfusion when administered early. Further research is warranted to examine possible interactions of nitrite with rtPA in this setting.
Preclinical Stroke Trials and The Problem of Choosing an Arbitrary End point
Some studies exclude animals that do not survive to a specific end point, possibly masking effects of an agent on mortality (Dirnagl, 2006). In a similar model, Pfefferkorn and Rosenberg (2003) have shown a drug-dependent effect on survival beyond 24 h after 6 h of ischemia and reperfusion. The difference of 83% in mortality in their study compared with the present finding of 61% might be attributable to interstrain differences between Sprague—Dawley and Wistar rats. Further, and in contrast to their study, this study assessed mortality for more than 48 h, which showed that animals that perform poorly but survive 24 h, might still die after 24 h. In a retrospective analysis, the 6-h nitrite group would have had significantly better survival than the placebo group (Kaplan—Meier log-rank comparison, P < 0.04) if the experiments had concluded at 24 h, showing that an arbitrarily chosen end point can be decisive in defining whether an agent is considered as neuroprotective or rejected in this experimental model, and perhaps in others.
Experimental Model
The endovascular thromboembolic models have a higher variability in infarct size and outcome than mechanical occlusion models (Beech et al, 2001). Also, a previous comparison of the effects of rtPA between suture and clot models after reperfusion injury showed no difference in neurotoxicity (Meng et al, 1999). Thus, to achieve the goal of this study, we chose the most widely used and recognized standard for assessing novel stroke therapy regimens—the suture MCAO model (Carmichael, 2005; Longa et al, 1989).
Microvessels in Cerebral Ischemia
The density of intact vessels in the ischemic hemisphere was greatly decreased as early as 48 h after stroke; vessels in the ischemic area had more intense staining, possibly due to endothelial membrane damage and better presentation of factor VIII to the primary antibody (Sasaki et al, 1988). Vessel density in our study was decreased compared with the healthy hemisphere 2 days after MCAO. Although vessel morphology scores did not differ between treatment and placebo groups, they were lower in the 6-h ischemia group compared with 2 h of MCAO, suggesting progression of endothelial cell death beyond 2 h. Thus, even 2 h into ischemia, a part of the cerebral microvasculature appears to be salvageable. Moreover, rats with low vessel scores did not necessarily have BBB disruption evident on MRI. Thus, unlike histologic correlates for the BBB, such as endothelial barrier antigen (Fagan et al, 2003), integrin (Tagaya et al, 2001), laminin (Hamann et al, 1996) and collagen IV (Grobholz et al, 2005), morphologic quantification of microvessels using factor VIII might reflect endothelial cell viability rather than BBB integrity. Further evaluation of the significance of factor VIII staining in studies of cerebral ischemia is advocated.
Footnotes
Acknowledgements
The authors acknowledge the help of Drs Christine Hon, Mike Espey, Ms Patricia Zerfas, Mr Daryl Despres, Ms Christl Ruetzler, Mr Stuart Walbridge and the staff of the NIH Mouse Imaging Facility.
Disclosure/Conflict of Interest
The authors state no conflicts of interest of any manner related to the content of this report.
