Abstract
Proteolytic disruption of the extracellular matrix with opening of the blood—brain barrier (BBB) because of matrix metalloproteinases (MMPs) occurs in reperfusion injury after stroke. Matrix metalloproteinase inhibition blocks the early disruption of the BBB, but the long-term consequences of short-term MMP inhibition are not known. Recently, a method to quantify BBB permeability by graphical methods was described, which provides a way to study both early disruption of the BBB and long-term effects on recovery in the same animal. We used a broad-spectrum MMP inhibitor, BB1101, to determine both the usefulness of the Magnetic resonance imaging (MRI) method for treatment studies and the long-term effects on recovery. Magnetic resonance imaging studies were performed in control (N = 6) and drug-treated (N = 8) groups on a dedicated 4.7-T MRI scanner. Adult Wistar—Kyoto underwent a 2-h middle cerebral artery occlusion followed by an MRI study after 3 h of reperfusion, which consisted of T2- and diffusion-weighted techniques. Additionally, a rapid T1 mapping protocol was also implemented to acquire one pre-gadolinium-diethylenetriaminepentaacetic acid baseline data set followed by postinjection data sets at 3-min intervals for 45 mins. The same animal was imaged again at 48 h for lesion size estimation. Data was postprocessed pixel-wise to generate apparent diffusion coefficient and permeability coefficient maps. Treatment with BB-1101 significantly reduced BBB permeability at 3 h, but failed to reduce lesion size at 48 h. Behavioral studies showed impairment in recovery in treated rats. Magnetic resonance imaging allowed for the monitoring of multiple parameters in the same animal. Our studies showed that BB-1101 was an excellent inhibitor of the BBB damage. However, results show that BB-1101 may be responsible for significant deterioration in neurologic status of treated animals. Although these preliminary results suggest that BB-1101 is useful in reducing early BBB leakage owing to reperfusion injury in stroke, further studies will be needed to determine whether the later detrimental effects can be eliminated by shorter time course of drug delivery.
Introduction
Matrix metalloproteinases (MMPs) are expressed in ischemic brain tissue as part of a molecular cascade that is initiated by the loss of blood supply by thrombus or an embolus (Mun-Bryce and Rosenberg, 1998b). Thrombolysis with recombinant tissue plasminogen activator (rtPA) restores the flow of oxygenated blood to the ischemic region. However, treatment with rtPA needs to be started within 3 h of stroke onset because reperfusion causes a transient opening of the blood—brain barrier (BBB), allowing rtPA and other substances in the blood to enter the brain (Tsirka, 1997; Wang et al, 1998). It has been shown that disruption of the BBB, which occurs early after the onset of reperfusion, results in the extravasation of rtPA into the brain, where it increases the risk of bleeding (Kelly et al, 2006). Blood—brain barrier disruption is mediated by the action of MMP at the neurovascular unit (Rosenberg et al, 1998), initiated in part by intrinsic expression and activation of MMPs by central nervous system astrocytes and microglia in response to hypoxia/ischemia.
Matrix metalloproteinases, a subfamily of metalloproteinases, are traditionally recognized as matrix-degrading enzymes involved in tissue remodeling during development and homeostasis, and have been implicated in normal and pathologic functions within nervous system including stroke (Yong et al, 2001). Matrix metalloproteinase-2 is constituently expressed by astrocytes, MMP-9 is secreted mainly by blood vessels, neurons, and inflammatory cells, and MMP-3 is mainly secreted by neurons, microglia, and pericytes. Numerous studies suggest that opening of the BBB in focal cerebral ischemia results from activation and expression of MMPs. After the early transient BBB opening, there is a second phase of severe BBB injury causing hemorrhagic transformation, which is associated with marked elevation of MMP-9 expression.
Recently, we have shown that a constitutive MMP, gelatinase A (MMP-2), which is activated by membrane-type metalloproteinase early after the onset of reperfusion, disrupts the tight junction proteins (Yang et al, 2007), allowing substances such as rtPA in the circulation into the brain. There is increasing evidence that synthetic inhibitors to MMPs are able to prevent the disruption of the endothelial cells, preserving the integrity of the BBB, preventing the extravasation of rtPA, and extending the therapeutic window for the use of thrombolytics. Matrix metalloproteinases have beneficial actions, in addition to the detrimental ones that are found in the early stages of reperfusion; they are important in the recovery phase where they facilitate angiogenesis (Lee et al, 2006) and neurogenesis (Wang et al, 2006
Magnetic resonance imaging (MRI) offers the means to measure BBB permeability and infarct size without killing the animal so that long-term behavioral studies can be performed. An MRI-based method to estimate the blood-to-brain transfer constant, ki (or barrier permeability coefficient) using standardized techniques has been described (Ewing et al, 2003). The MRI technique for estimating barrier permeability coefficient, ki, is based on a graphical analysis method (Blasberg et al, 1983). This technique involves quantifying distribution of gadolinium-diethylenetriaminepentaacetic acid (Gd-DTPA), an MRI contrast agent, in the brain tissue. Recently, we have used the MRI-based graphical analysis method for testing the BBB-blocking properties of propylene glycol (Sood et al, 2007). Therefore, the noninvasive MRI method for quantifying BBB permeability appears to be an appropriate technique for investigating the effect of BB-1101, a broad-spectrum MMP inhibitor, on BBB disruption.
Several laboratories have showed in different models that broad-spectrum MMP inhibitor, BB-94 (Batimastat), blocked the BBB damage, but they did not study the long-term effects of the drug (Paul et al, 1998; Pfefferkorn and Rosenberg, 2003). It is possible that the therapeutic window for the use of MMP inhibitors could be narrow, and that understanding the long-term consequences of the use of these agents will be critical. We have used the MRI-based multiple time graphical analysis method to study the effect of a synthetic MMP inhibitor, BB-1101, on early BBB permeability. Additionally, MRI has been used to measure stroke infarct size at 48 h and behavior over 4 weeks. In this way, the full impact of the drug in multiple stages of injury and recovery was investigated effectively.
Thus, the aims of this study were (1) to investigate the BBB-blocking property of BB-1101 using MRI at 3 h after middle cerebral artery occlusion (MCAO) in a rat ischemic stroke model; (2) to investigate the effect of BB-1101 on lesion size at 48 h after MCAO using MRI; and (3) to evaluate the effect of BB-1101 on neurologic function by performing behavioral studies in treated and control rats.
Materials and methods
Middle Cerebral Artery Occlusion
The study was approved by the Local Animal Research Committee and conformed to the National Institutes of Health guidelines for use of animals in research. Wistar—Kyoto rats weighing 250 to 300 g underwent 2 h of MCAO followed by reperfusion for 3 h. Rats were anesthetized with 2% isoflurane during the surgery. The MCAO technique has been described previously (Sood et al, 2007). Briefly, neck vessels were exposed through a midline incision. After isolation and ligation of the branches of the right external carotid vessels, a 6–0 silk suture was loosely tied around the external carotid artery stump. A section of 4–0 monofilament nylon suture was introduced into the external carotid artery and advanced to occlude the origin of the middle cerebral artery. The suture around the stump was tied down onto the nylon suture with the end of the suture protruding slightly. The middle cerebral artery was occluded for 2 h. The thread was slowly pulled back to the external carotid stump for 3 h of reperfusion.
Magnetic Resonance Imaging Technique
Magnetic resonance imaging study was performed in a rat brain MCAO model of ischemic stroke. Animals were anesthetized using isoflurane gas (induction dosage 2 to 3%; maintenance dosage 1.5 to 2%), and a mixture of O2/N2O gases (2:1) was delivered during the entire duration of the study. Real-time monitoring of physiologic parameters (heart rate and respiratory rate) was performed during the entire duration of the study. A PE-50 polyethylene catheter was placed into the femoral vein of each animal for injection of Gd-DTPA during the entire duration of the study.
In this study, 23 male Wistar—Kyoto rats weighing 250 to 300 g were divided into two groups: the first group consisted of 12 rats that had MCAO but were not treated with the drug and served as controls, whereas the second group consisted of 11 rats and were treated with BB-1101. The treated animals were injected intraperitoneally (i.p.) with 30 mg/kg of BB-1101 diluted in 1 mL of 0.9% saline. The drug was injected at 0 (before MCAO), and at 24 and 48 h after MCAO. The experimental design is shown in Figure 1. Rats were imaged at 3 h after MCAO for estimation of BBB permeability and then at 48 h after MCAO for estimation of ischemic lesion size. Each rat underwent behavioral study weekly for duration of 4 weeks. Of the 11 rats treated with BB-1101, data from only six rats were used for permeability analysis. This was due to technical reasons related to animals; two rats did not have visible evidence of ischemic lesion (determined on diffusion-weighted MRI) probably because of failed surgery, and three had evidence of intracerebral hemorrhage. Data from four control rats was excluded owing to technical reasons related to MRI problems; evidence of artefacts on MR images owing to magnetic shimming issues or gradient instability resulted in inability to perform analysis. Infarct size estimation was performed in five BB-1101-treated rats, as in the treated group two rats (in addition to those not used for permeability analysis) did not survive till the 48 h time point.
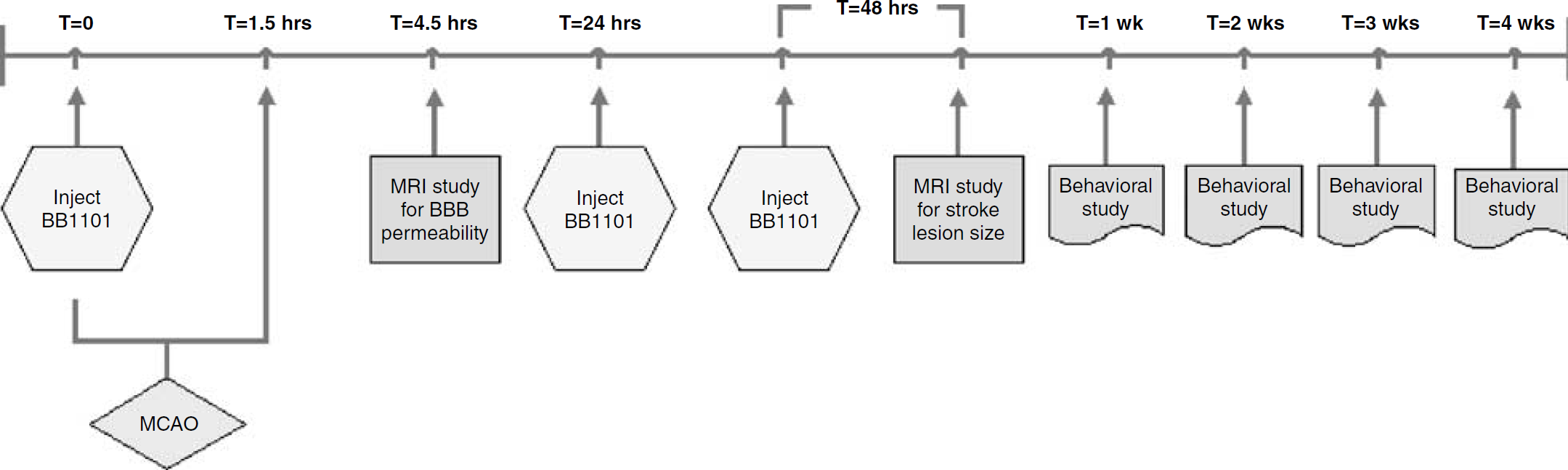
Flow chart providing an overview of the study. See text for more details on MCAO surgery, BB1101 injection dose, and MRI protocol and behavior assessment.
Rats were then transported to the MRI room, placed in a dedicated rat holder, and moved to the isocenter of the magnet before the imaging session. Magnetic resonance imaging was performed on a 4.7 T Biospec® dedicated research MR scanner (Bruker Biospin, Billerica, MA, USA).
The MRI study was implemented in two sessions. In the first session, animals were brought to the MRI suite for imaging at 4.5 h after MCAO. At this stage, data were acquired for quantifying BBB permeability. The details of the scan parameters used in this MRI protocol are described below. Initial localizer images were acquired using the following parameters: 2D FLASH (fast low-angle SHot), repetition time (TR)/echo time (TE) = 10/3 ms, matrix = 256 × 128, field of view (FOV) = 6.4 cm, one slice per orientation. After the localizer images were acquired, T2- and diffusion-weighted imaging was performed using parameters described in Sood et al (2007). The MR protocol for acquiring data for the Patlak plot method was then implemented. In this acquisition, a reference baseline acquisition using the fast T1 mapping protocol was obtained before injecting the contrast agent. A 0.1 mmol/L/kg portion of Gd-DTPA (938 Da) (Berlex Lab, Montville, NJ, USA) was injected as a bolus into the femoral vein via the indwelling catheter, followed by imaging with rapid T1 mapping protocol for a total time of 45 mins, resulting in a total of 14 time points. Gadolinium-diethylenetriaminepentaacetic acid is approximately twice the molecular weight with [14C]sucrose (342 Da), whereas it is small in comparison to [14C]dextran (50 to 90 kDa) (Mun-Bryce and Rosenberg, 1998a).
At 48 h, the rats were brought back for imaging of lesion size. Magnetic resonance imaging was performed using the following parameters: initial localizer images were acquired using parameters as described earlier. After the localizer images were acquired, T2- and diffusion-weighted imaging was performed with the following parameters: T2-weighted-2D turbo RARE (rapid acquisition with relaxation enhancement), TR/TE =4,000/65 ms, matrix = 256 × 128, number of averages = 22, echo train length = 8; diffusion-weighted imaging-2D diffusion weighted RARE, TR/TE = 2,000/31.2 ms, matrix = 64 × 64, number of averages = 15, d = 5 ms, D = 20ms, b = 0 and 927 secs/mm2, diffusion gradient along the slice select direction. Common acquisition parameters were FOV = 3.2 cm × 3.2 cm, slice thickness = 2 mm, slicegap = 0 mm, number of slices = 14, and receiver bandwidth = 100 kHz. The slice prescriptions for T2- and diffusion-weighted acquisitions were matched for all slice locations.
The acquired data from two MRI sessions was transferred to a dedicated computer workstation for postprocessing, image analysis, and archiving. Postprocessing of the raw data involved generating apparent diffusion coefficient (ADC) maps from diffusion-weighted imaging images, T1 maps from the raw data, reconstruction of permeability coefficient maps, construction of the Patlak plots, and lesion volume estimation.
All the data processing was performed using in-house software written in 64-bit MATLAB (Mathworks, Natick, MA, USA) and implemented on a 64-bit processor (AMD64, Advanced Micro Devices, Sunnyvale, CA, USA) workstation running Red Hat Enterprise Linux v3 (64 bit). Image analysis was performed using ImageJ (NIH, Bethesda, MD, USA) and MRVision (Winchester, MA, USA) software.
Image analysis was performed using the methods described previously (Sood et al, 2007). Before analysis of permeability coefficient maps, Kalman filtering was performed on raw and processed data at two stages: first, on the series of inversion recovery echo images (12 frames per time point per slice) and second, on T1 maps over the entire time series, using a technique described previously (Taheri and Sood, 2006). The permeability maps were also used for estimating area of leakage, that is region of BBB disruption. The area of leakage was calculated as a product of number of pixels with high permeability and pixel size.
Lesion size estimation was performed on ADC maps reconstructed from multislice diffusion-weighted images acquired at 48 h using MRVision software 1.67 (MRVision). The multislice diffusion-weighted acquisition was prescribed such that whole rat brain coverage was obtained. All the acquired slices were used for lesion size analysis. Apparent diffusion coefficient maps were reconstructed pixel-wise from diffusion-weighted data such that pixel signal intensity values were proportional to absolute ADC values. Lesion size was measured using various ADC thresholds and was defined as area with ADC values < 80% of the mean contralateral hemisphere values on the ADC map (Hoehn-Berlage et al, 1995; Figure 2). The analysis was performed by a single operator and the procedure was repeated twice. Lesion size, reported by the software, was calculated as number of pixels × pixel size, where the number of pixels were pixels with values less than the ADC threshold value. Lesion size was estimated per slice and reported as sum over the total number of prescribed slices. These estimates were saved as a text file, read into Excel spreadsheet (Microsoft, Redmond, WA, USA) and used for further analysis.
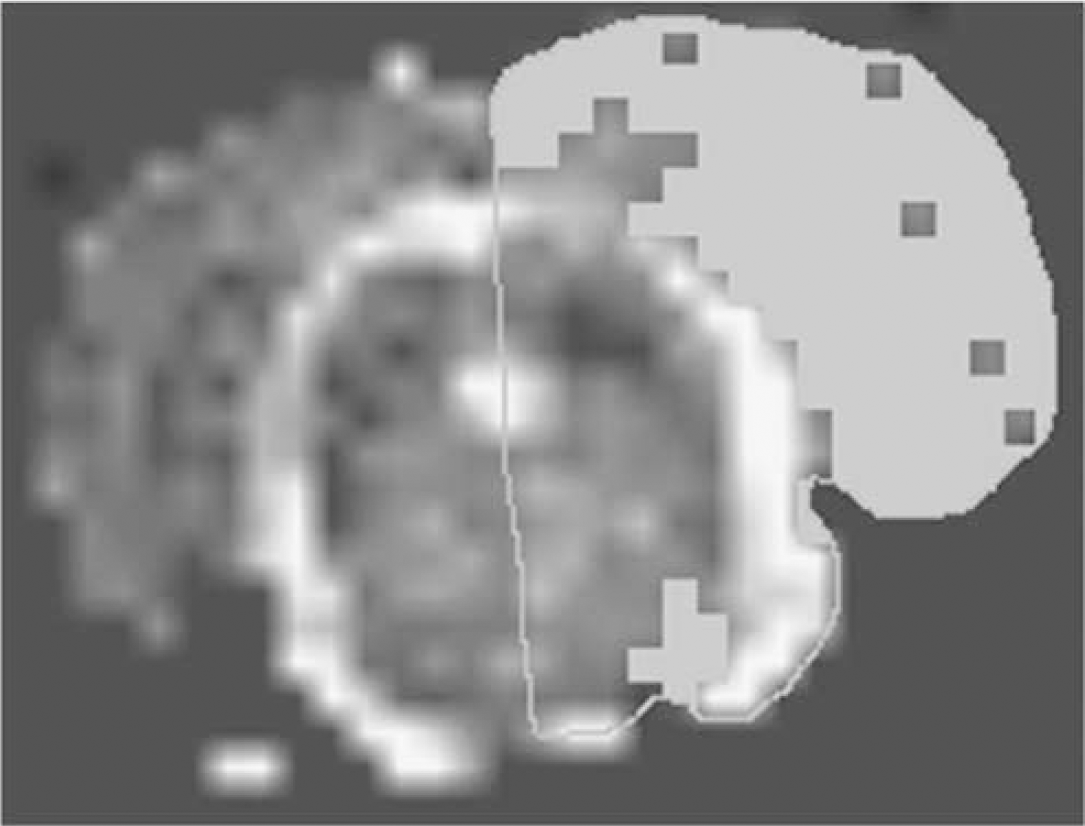
Lesion size estimation from ADC map using MRVision software. Thresholding technique was used to extract lesion pixels on the ipsilateral hemisphere with ADC values < 80% of the contralateral side.
Behavior Studies
The effect of BB-1101 on neurologic function was evaluated using behavioral studies. Behavioral assessments were made every week after surgery for 4 weeks by evaluating the animals using tests that measure neurologic deficit. Briefly, the rats were tested and scored for neurologic deficits as follows: 0, no apparent deficit; 1, failure to extend left forepaw; 2, decreased grip of the left forelimb while tail is pulled or twisting entire body toward contralateral side; 3, spontaneous movement in all directions, circling to left only if pulled by tail, or falling to the contralateral side; 4, spontaneous circling to the left or no spontaneous walking with reduced level of consciousness or inability to walk spontaneously; and 5, death of the animal. Each test was graded as 1 if positive and 0 if negative.
Statistical Analysis
Permeability coefficient data obtained from MRI experiments were analyzed using a one-way analysis of variance statistical test. Permeability coefficient values obtained from region of interest analysis of the ipsilateral and contralateral side and infarct lesion sizes from control and treated groups were read into Microsoft excel software (Microsoft) for statistical data analysis. The results obtained from analysis of variance statistical analysis for the MRI technique was presented as mean ± standard error of mean (s.e.m).
Results
Figure 3 shows a series of T2-weighted images, diffusion-weighted contrast images, and color-coded permeability coefficient maps for two representative rats: treated (top row) and control (bottom row). The T2-weighted images showed the lesion in relation to the anatomic detail in the rat brain.
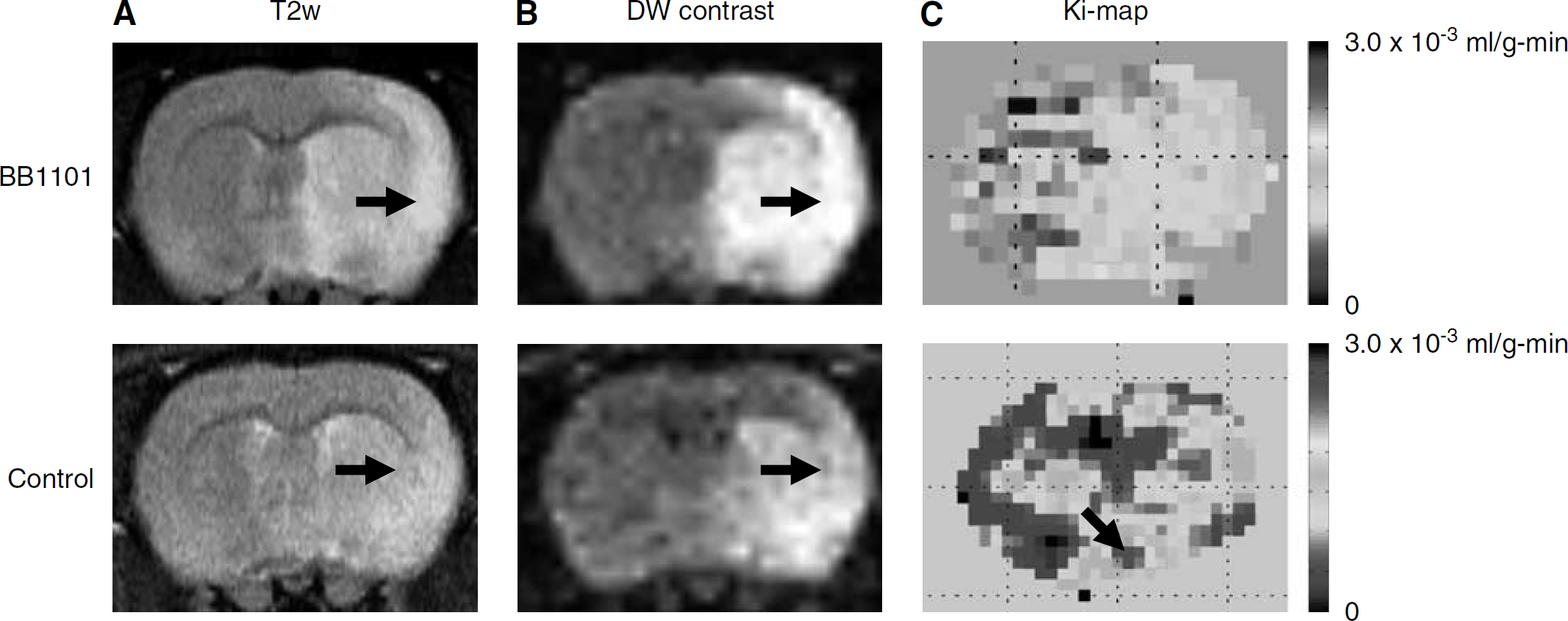
(
In the control group, ADC values in the rat brain were estimated to be 0.46 ± 0.19 × 10−3 (mean ± s.d.) and 0.95 ± 0.21 × 10−3mm2/sec on the ipsilateral and contralateral hemisphere, respectively. Apparent diffusion coefficient values estimated in treated rats on the ipsilateral and contralateral side were 0.42 ± 0.06 × 10−3 and 0.79 ± 0.04 × 10−3mm2/sec, respectively. The ADC value in the ipsilateral region of interest dropped 48.4% of the matching contralateral areas in control group and 53.1% in BB-1101-treated rats, respectively, after 3 h of reperfusion.
Figure 4 shows a graph of mean permeability coefficient values in control group and treated group, estimated using MRI. There was a significant (P < 0.05) reduction observed in mean permeability coefficient values on the ipsilateral side in the drug-treated rats as compared with the untreated rats.
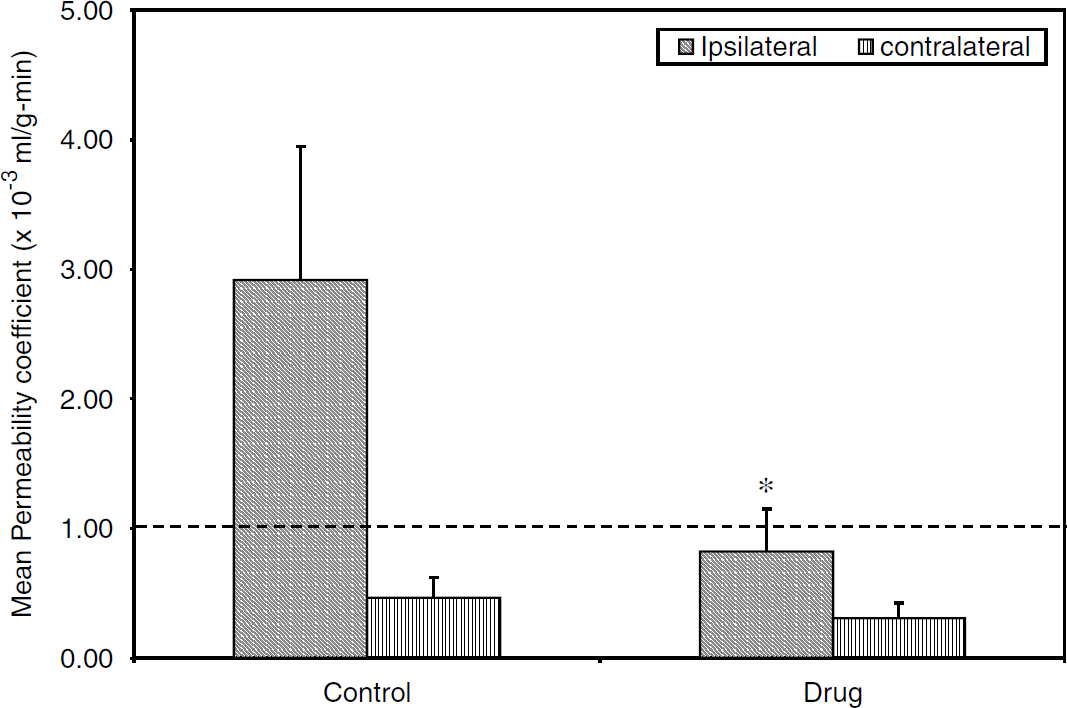
A plot of mean permeability coefficient estimates in treated (N = 6) and control (N = 8) rats obtained using the MRI technique. The dashed line represents the upper limit of the range (0 to 1 × 10 to 3 mL/g per min) of permeability coefficient values in healthy tissue. Rats treated with BB1101 showed a significant reduction in permeability coefficient values on the ischemic side as compared with the untreated rats (analysis of variance, *P < 0.05, *indicates significant reduction). No significant difference was observed on the contralateral side between the control and treated rats. The Y-axis error bars represent s.e.m.
The area of leakage estimated from enhancing pixels on the permeability map in control and drug-treated rats was 7.70 ± 1.70 and 1.24 ± 0.78 mm2, respectively. There was a statistically significant (P < 0.05) difference in the area of leakage between control and treated groups.
Figure 5 shows a plot of individual lesion size measured at 48 h in control and treated rats. The stroke lesion size in the control and treated group was 213.4 ± 49.3 and 223.7 ± 69.6 mm2, respectively. There was no significant difference in lesions size between the control and treated groups.
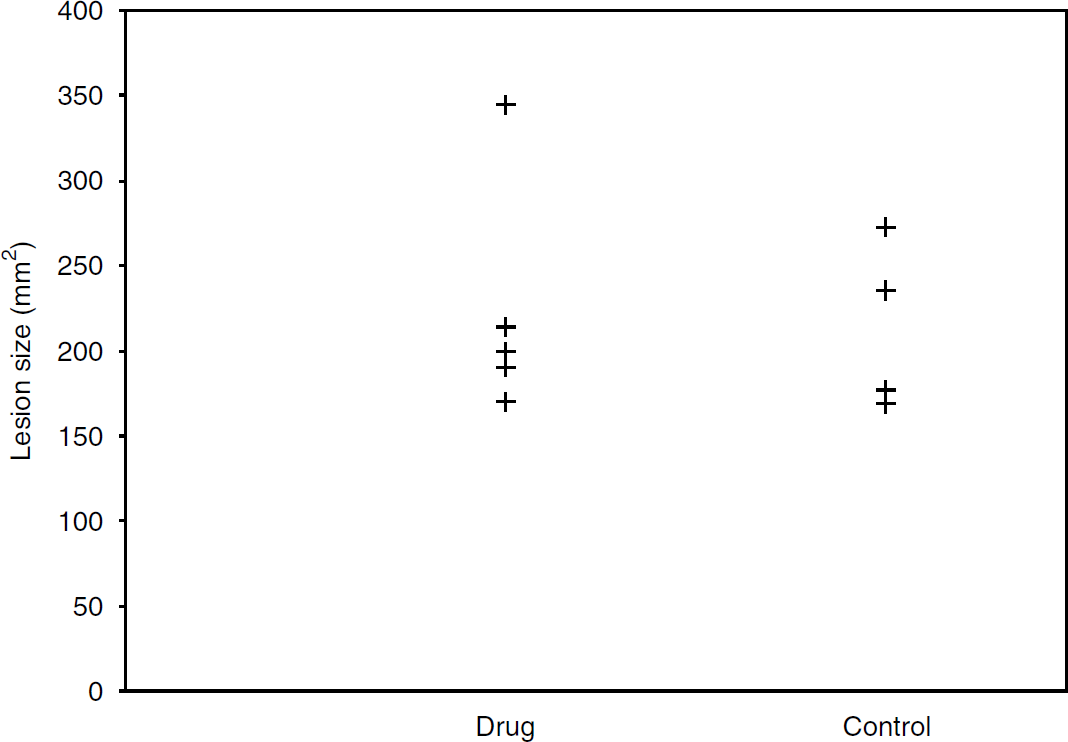
Stoke lesion size in treated and control groups at 48 h after a 90-min MCAO. The lesion size in drug-treated and control group was 223.7 ± 69.6 and 213.4 ± 49.3 mm2, respectively. There was no significant difference in lesion size observed between the two groups.
Behavioral study results, shown in Figure 6, indicate that there was a statistically significant increase in neurologic deficit in the third and fourth week in rats treated with BB-1101 compared with control (P < 0.05).
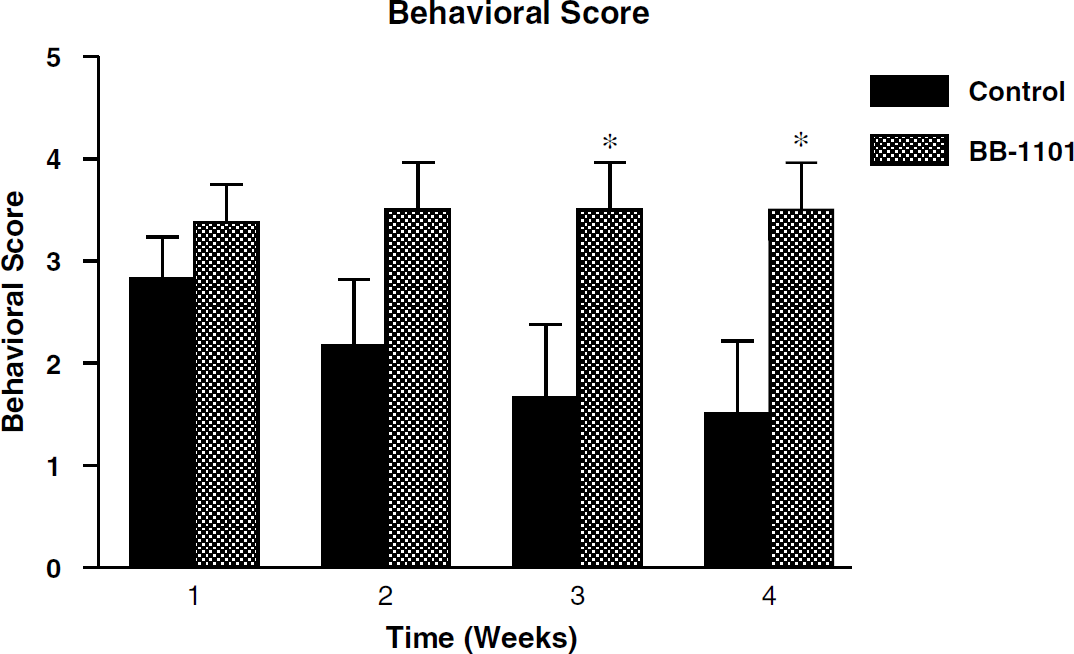
Behavioral scores in rats treated with BB-1101 and control group. Scoring was based on a 5-point scale and was done at weekly intervals for a duration of 4 weeks. There was a statistically significant increase in behavioral scores in the third and fourth weeks. *P < 0.05.
Discussion
Reperfusion of ischemic tissue causes enhanced production of free radicals (Chan et al, 1984), release of proteases (Gidday et al, 2005) and a biphasic opening of the BBB (Kuroiwa et al, 1985). The hemorrhagic conversion occurs in approximately60% of patients after an embolic stroke (Hornig et al, 1993). The use of rtPA carries the risk of increasing the incidence of symptomatic hemorrhage 10 times. Leakage of rtPA across a damaged BBB is potentially toxic to the brain cells (Kaur et al, 2004). There is sufficient evidence to support the fact that activation of MMPs by rtPA plays a significant role in causing hemorrhage and that synthetic MMP inhibitors prevent the BBB opening and protect the brain from rtPA toxicity. In this study, we have investigated the ability of an hydroxymate MMP inhibitor, BB-1101, to block BBB leakage and its effect on stroke lesion size using MRI. The multiple graphical analysis method implemented using MRI is based on the model of unidirectional tracer kinetics in one compartment and provides a noninvasive approach to investigate the BBB-blocking properties of novel pharmaceutical agents without killing the animal. This in turn enables subsequent estimation of stroke lesion size at 48 h after MCAO and evaluation of neurologic function.
The permeability coefficient maps were reconstructed from Kalman filtered data. Digital Kalman filtering (Kalman, 1960) is a robust adaptive filtering technique that optimally estimates, in real time, the state of a system (true MR signal) based on its noisy outputs. The technical details of the filtering technique and its application to improving the reliability of permeability coefficient estimates is described elsewhere (Taheri and Sood, 2006). As a result, application of this filtering technique allowed reliable estimation of permeability maps from data that has low signal-to-noise ratio, a drawback of fast MR data acquisition techniques such as IR-SE-EPI. In the treated group, the permeability coefficient values on the ischemic side (0.77 ± 0.31 × 10−3mLg/min) and the nonischemic side (0.31 ± 0.21 × 10−3 mLg/min) were well within the range for permeability coefficient values in healthy tissue (0 to 1 × 10−3 mL g/min), suggesting that BB-1101 was successful in reducing BBB permeability on the ischemic side. On comparing permeability coefficient values for ipsilateral side, a 75% reduction in mean permeability coefficient values has been observed in treated group compared with the control group. The decrease in permeability coefficient values owing to BB-1101 treatment was found to be statistically significant (P < 0.05). The MRI provided both a permeability coefficient and a spatial map of the permeability changes. This suggests that BB-1101 may be working to reduce BBB leakage in the region of maximal ischemic injury and confirms both the utility of the Patlak plot graphical method for measuring drug effects and the earlier findings by us (Rosenberg et al, 1998), using an invasive BBB permeability measurement technique, that BB-1101 is effective in blocking the BBB leakage.
The lesion size at 48 h was not significantly different between the treated and control groups. The lack of effect of BB-1101 on lesion size at 48 h is consistent with earlier study showing that BB-1101 did not affect permeability at 48 h (Rosenberg et al, 1998). Interestingly, when an MMP-9-neutralizing monoclonal antibody was administered systemically (Romanic et al, 1998), animals exhibited significantly reduced infarct size (a 30% reduction was observed compared with non-immune antibody controls; P < 0.05). However, this difference between the antibody and the synthetic inhibitor is difficult to explain and warrants further investigation.
Apparent diffusion coefficient maps obtained from whole rat brain were used for performing lesion size estimation, as they provide a reliable way of measuring damaged tissue due to ischemia compared with signal intensity-based methods. In this study, ischemic tissue was defined as region with ADC values less than 80% of the normal values. Published research suggests that using 80% ADC threshold at early MR imaging has the strongest correlation with histopathological analysis and is the only independent significant variable affecting irreversible infarct volume (Wang et al, 2006a). Additionally, ADC threshold set at approximately 80% of the normal ADC has been found to correlate well with the loss of ATP and breakdown of energy metabolism in animal studies of focal cerebral ischemia during ischemia and reperfusion (Olah et al, 2000). At 3 h after MCAO, BB-1101 did not have an effect on the ADC values in the treated groups, suggesting that the drug may not have a neuroprotective effect. Regional cerebral blood flow studies were not done in this work and may have potentially provided helpful insight into the neuroprotective effect of BB-1101. In this study, the absolute ADC values obtained for healthy rat brain tissue are higher than those found in the literature (ADC values for healthy rat brain tissue are in the range 0.6 to 0.7 × 10−3 mm2/sec) (Does and Gore, 2000). One possible explanation for this finding would be motion-induced artefact during data acquisition.
The results of the behavior study in the treated group showed a significant increase in neurologic deficit at 3 and 4-week period, suggesting the detrimental effects of BB-1101 on neurologic function. In the control group, the deficit appeared to decrease over the 4-week period of evaluation. The lack of long-term benefit from a broad-spectrum inhibitor was seen in a recent study that showed the inhibition of MMP blocks angiogenesis and neurogenesis, which interfered with recovery (Zhao et al, 2006). We removed one high permeability coefficient value in a BB-1101-treated animal because it was an extreme outlier. The high permeability value was observed due to the leakage of Gd-DTPA into the cerebrospinal fluid space resulting in very high and inaccurate permeability estimates.
In conclusion, BB-1101, an MMP inhibitor, appears to be a promising candidate for BBB blockage during the early phase of BBB disruption. The drug did not have any effect on stroke lesion size at 48 h after MCAO and had significant adverse effects on neurologic function in rats at 3 and 4 weeks after MCAO. Future studies will need to define the optimal timing of delivery of a MMP inhibitor so that the BBB damage is blocked and that there is no interference with recovery.
