Abstract
White matter (WM) injury after bilateral common carotid artery occlusion (BCAO) in rat is associated with disruption of the blood—brain barrier (BBB) by matrix metalloproteinases (MMPs). We hypothesized that WM injury as seen on magnetic resonance imaging (MRI) would correlate with regions of increased MMP activity. MRI was performed 3 days after BCAO surgery in rats. Apparent diffusion coefficients (ADC) were calculated and vascular permeability was quantified by the multiple-time graphical analysis (MTGA) method, using gadolinium-diethylenetriamine pentaacid (Gd-DTPA). After MRI, one group of animals had BBB permeability measured in the WM with 14C-sucrose, and another had Evans blue (EB) injected for fluorescent microscopy for MMP-2, MMP-9, tight junction proteins (TJPs), and
Introduction
Vascular cognitive impairment is an important cause of dementia in patients with cerebrovascular disease and impacts the course of Alzheimer's disease (Bowler, 2007). A major type of vascular cognitive impairment involves disease of the small blood vessels from hypertension and diabetes, leading to hypoperfusion of the deep white matter (WM) with subsequent demyelination (O'Brien et al, 2003). Hypoxic/ischemic injury to the deep WM is frequently observed on magnetic resonance imaging (MRI) in elderly patients and has been associated with increased risk of stroke and vascular cognitive impairment (Vermeer et al, 2003). An animal model has been developed that involves permanent bilateral carotid artery occlusion (BCAO) in the rat, which causes hypoxic hypoperfusion of the WM regions (Wakita et al, 1994). Disruption of the blood vessels in the poorly perfused deep WM with vasogenic edema has been proposed as a major contributing factor in WM injury (Feigin and Popoff, 1963; Wardlaw et al, 2003).
Rats with permanent BCAO have hypoxic hypoperfusion injuries that produce extensive damage to the WM of the corpus callosum (CC), optic tracts, and hippocampus (Farkas et al, 2007). Damage to the WM becomes evident after 3 days of BCAO and continues to progress over several months (Schmidt-Kastner et al, 2005). Demyelination, loss of oligodendrocytes, and expression of matrix metalloproteinases (MMPs) are observed in regions of reactive astrocytes and activated microglia (Ihara et al, 2001; Ueno et al, 2002). Recently, the expression of MMP-2 (gelatinase A or 72-kDa type IV collagenase) has been associated with opening of the blood—brain barrier (BBB) in a mouse model of hypoxic hypoperfusion. Mice lacking the
Most studies of BBB function have required tissue sampling without information regarding the regions of injury. A method that is noninvasive and would allow quantification of ongoing tissue injury would be important for long-term pathobiology and treatment studies. MRI is a noninvasive method that can be used to identify increased tissue water, which can be quantified as an increase in the apparent diffusion coefficient (ADC). In addition, quantification of vascular permeability can be performed with MRI, using a time-dependent graphical analysis method, initially developed for use with autoradiography (Patlak et al, 1983), and recently adapted to quantify permeability measurements with gadolinium-diethylenetriamine pentaacid (Gd-DTPA)-enhanced MRI multiple-time graphical analysis (MTGA) plot (Ewing et al, 2003). MRI also allows localization of the site of abnormal permeability, which can be used to guide tissue sampling to understand the underlying mechanisms. Therefore, we tested the hypothesis that hypoxic hypoperfusion leads to an increase in vascular permeability, resulting in increased ADC with Gd-DTPA leakage on MRI, and that regions of ADC increase would correspond to EB leakage and MMP activity. To determine the type of injury to the BBB, we measured vascular permeability in rats with BCAO, using three methods with different sized molecules; the MTGA method with the small Gd-DTPA molecule, brain uptake of 14C-sucrose, and EB fluorescent microscopy. We used MRI to correlate the sites of WM injury with gelatinase activity on
We report that sites of increased ADC corresponded with extravasation of EB and colocalized with gelatinase activity from MMP-2 and MMP-9 in both the anterior CC and external capsule. Paradoxically, there was no increase in permeability coefficients as measured with the Gd-DTPA or 14C-sucrose. Furthermore, TJPs appeared to be intact in vessels showing EB extravasation. We propose that BBB damage because of activity of the MMPs occurs in rats with hypoxic hypoperfusion secondary to BCAO. Our results show that ADC can be used as a noninvasive biomarker to identify regions of BBB injury in animals with chronic hypoxia.
Methods
The BCAO Model
The University of New Mexico Animal Care and Use Committee approved the animal protocol. Twenty-six adult, male Wistar rats, weighing 280 to 320 g, were used in the study of which 22 completed the protocol. They were anesthetized with 2% isoflurane in 70% N2O and 30% O2. After a midline incision, both common carotid arteries were dissected free and doubly ligated with a 3 to 0 silk suture. The BCAO model was used to induce a moderate reduction of cerebral blood flow to the brain, causing global cerebral hypoperfusion. After the procedure, all animals appeared to be normal without any visual evidence of grossly abnormal behavior.
MRI Technique
At 3 days post surgery, rats were transported to the MRI room, placed in a dedicated rat holder and moved to the isocenter of the magnet before the imaging session. For the MRI study, the animals were divided into two groups: the first group consisted of 11 sham-operated rats that were used as controls. The second group consisted of 11 rats that underwent BCAO surgery. All 22 rats underwent the MRI study. Animals were anesthetized using isoflurane gas (induction dosage 2% to 3%; maintenance dosage 1.5% to 2%) and real time monitoring of physiologic parameters (heart rate and respiratory rate) was performed during the entire duration of the study. A PE-50 polyethylene catheter was placed into the femoral vein of each animal for injection of Gd-DTPA.
Magnetic resonance imaging was performed on a 4.7T Biospec dedicated research magnetic resonance scanner (Bruker Biospin, Billerica, MA, USA), equipped with 500mT/m (rise time 80 to 120 µs) gradient set (for performing small animal imaging) and a small bore linear radio frequency coil (inner diameter 72 mm). The details of the scan parameters used in this protocol have been described and are given here in brief (Sood et al, 2008). Initial localizer images were acquired using the following parameters: 2D fast low-angle shot, repetition time/echo time 10/3 ms, matrix 256×128, field of view 6.4 cm, 1 slice per orientation. After the localizer images were acquired, T2-weighted (T2W) and diffusion-weighted (DW) images was performed with the following parameters; T2W-2D rapid acquisition with relaxation enhancement, repetition time/echo time 4,000/65 ms, field of view 3.2×3.2 cm, slice thickness 2 mm, slice gap 1 mm, number of slices 2, matrix 256×128, number of averages 5, receiver bandwidth 100 kHz; DWI—2D DW rapid acquisition with relaxation enhancement, repetition time/echo time 2,000/31.2 ms, field of view 3.2×3.2 cm, slice thickness 2 mm, slice gap 1 mm, number of slices 2, matrix 64×64, number of averages 5, receiver bandwidth 100 kHz,
The magnetic resonance protocol for acquiring data for the MTGA method was then implemented. In this acquisition, a reference baseline acquisition using the fast T1 mapping protocol was obtained. 0.2 mmol/kg of Gd-DTPA (Magnevist; Bayer Healthcare, Montville, NJ, USA; molecular weight 938 Da) was injected as a bolus into the femoral vein by the in-dwelling catheter, followed by imaging with rapid T1 mapping protocol for a total time of 45 mins, resulting in a total of 14 time points. The following optimized MRI parameters were used for this protocol: axial plane, 2D inversion recovery-spin echo-echoplanar, repetition time/echo time 8 secs/19.4 ms, field of view 4.0×4.0 cm, slice thickness 2 mm, slice gap 1 mm, number of slices 2, matrix 64×64, number of averages 2, receiver bandwidth 250 kHz. Nonslice selective magnetization inversion was performed using a hyperbolic secant (sech) radio frequency pulse with pulse width 4 ms. The T1 mapping specific parameters for this protocol were; time for inversion (TI) TI = (100 + 600×
The acquired data were transferred to a dedicated computer workstation for post processing. Post processing of the raw data involved generating ADC maps from DW images, T1 maps from the raw data, reconstruction of permeability coefficient maps, and construction of the MTGA plots. The ADC maps were generated, and the natural logarithms of the images were averaged to form the resultant image. Using a linear least-squares regression on a pixel-by-pixel basis, the resultant image and the natural logarithm of the reference T2W image (
Data processing was performed using in-house software written in 64-bit MATLAB (MathWorks, Natick, MA, USA) and implemented on a 64-bit processor (AMD64) workstation running Red Hat Enterprise Linux v3 (64 bit). Image analysis was performed using ImageJ (NIH, Bethesda, MD, USA) and MRVision (Winchester, MA, USA) software.
Image analysis of BBB permeability data was performed using the methods described (Sood et al, 2008). Damage to the WM was observed as regions with high signal on ADC maps, with the regions localized to anatomic WM (CC) on the T2W images. ADC values were obtained by drawing the region of interest on ADC maps around the lesion on the ipsilateral and contralateral WM. ADC values were recorded for bilateral WM regions for all the slices and a scatter plot was drawn for the ADC changes seen on DW images. ADC data were read into a spreadsheet and statistical data analysis of the ADC was performed with an analysis of variance, using Prism 5.0 (GraphPad Software, San Diego, CA, USA).
EB Fluorescent Microscopy of Brain Tissue
After the MRI study, the animals in the BCAO and control group were moved back to the laboratory for further histochemical studies and analysis. In the laboratory, 2% EB was injected in BCAO (
In Situ Zymography
Sections from animals injected with EB as described above were used for
Brain Uptake Index Measurement Using 14C-Sucrose Technique
Brain uptake of sucrose was measured in rat brains that had undergone BCAO (
Immunohistochemistry
Fluorescent staining of MMP-2 (1:300; Chemicon International Inc., Temecula, CA, USA), MMP-9 (1:300; Chemicon International Inc.), claudin-5 (1 µg/mL, Zymed Laboratories, South San Francisco, CA, USA), and occludin (1:500; Zymed Laboratories) were performed in fresh sections. Sections with EB were fixed in 4% paraformaldehyde in phosphate-buffered saline for 30 mins and then rinsed in phosphate-buffered saline. Primary antibodies were incubated for two nights at 4°C and slides were incubated for 90 mins at room temperature with secondary antibodies conjugated with fluorescein isothiocynate (green). Slides were photographed with a fluorescent microscope (Olympus Optical Co., Ltd.). Sections incubated in the absence of the primary antibody or with rabbit immunoglobulin Gs were not immunoreactive.
Results
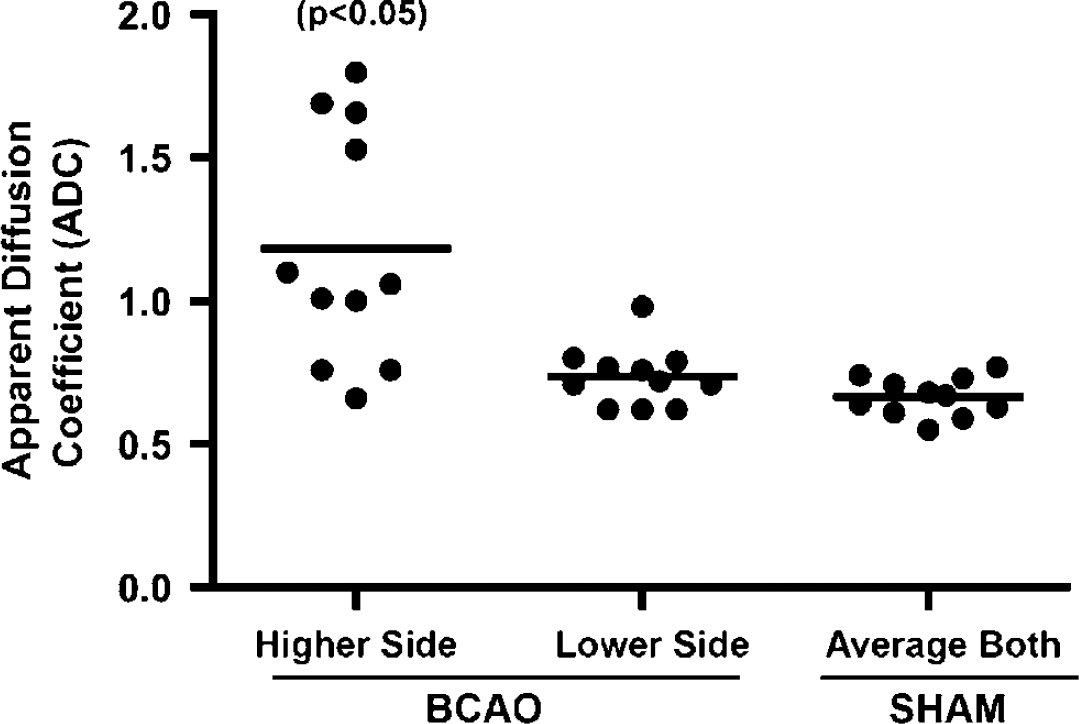
There was no WM damage seen in the 11 control rats on T2W images and the ADC values in the control rats were found to be in the normal range. One rat in the BCAO group did not survive the MRI study and hence was not included in the analysis. Increased ADC was found in the BCAO group (Figure 1). There were asymmetric increases in the ADC; higher ADC values were always found on one side except for one animal where the ADC was significantly greater on both sides. When the side with the increased ADC was used for statistical comparisons with an average value from both control sides (Figure 2), a significant difference in ADC values in the WM was observed between the sham-operated control group (
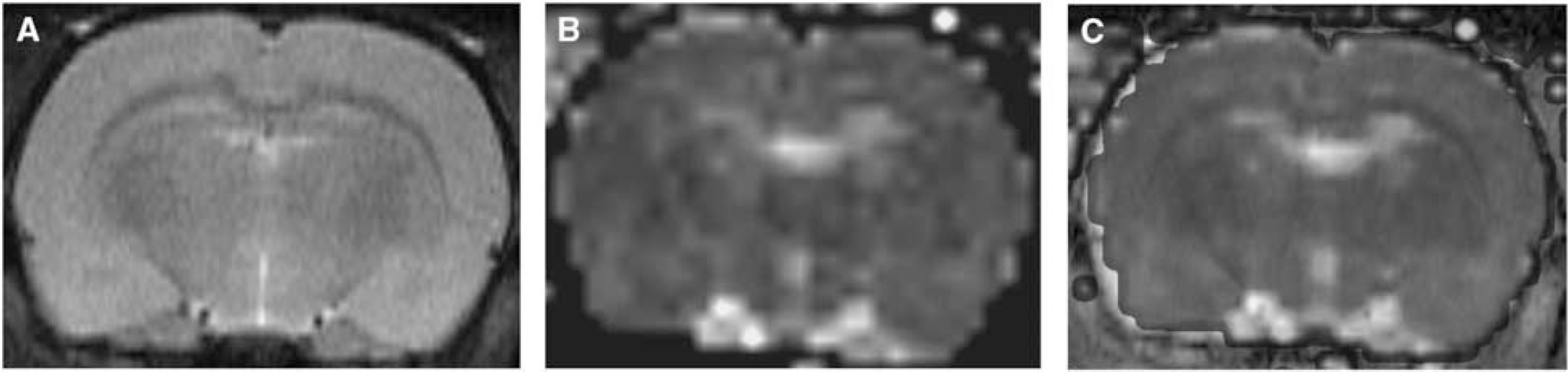
(
A graph of apparent diffusion coefficient (ADC) values in 11 rats with permanent BCAO for 3 days. In the BCAO group, the decrease side refers to ADC values that were comparatively lower than those found on the contralateral side (the higher side) and in some cases were in the normal ADC range. In seven rats, the ADC was higher on one side than in the other, whereas in one it was higher than controls on both sides. Three BCAO rats had normal ADC values on both sides. The sham ADC values were consistently in the range for healthy white matter. There was a significant difference between the high side compared with the lower side and the sham. There was no difference observed between the lower side and the controls.
We were unable to show changes in vascular permeability with either the MTGA or 14C-sucrose methods. Permeability coefficient values in the WM region obtained from the MTGA permeability color maps were found to be in the normal range (0 to 1×10−3 mL/g mins) in control and BCAO groups. Similarly, the results from the 14C-sucrose brain uptake permeability calculations failed to show an increase in the sucrose space in the regions of the WM after BCAO (data not shown).
The sham-operated animals did not show EB extravasation or gelatinase activity (Figure 3A). Three days after BCAO, fluorescent microscopy showed EB extravasation around blood vessels in the WM that corresponded with the sites of increased ADC on the MRI (Figure 3A).
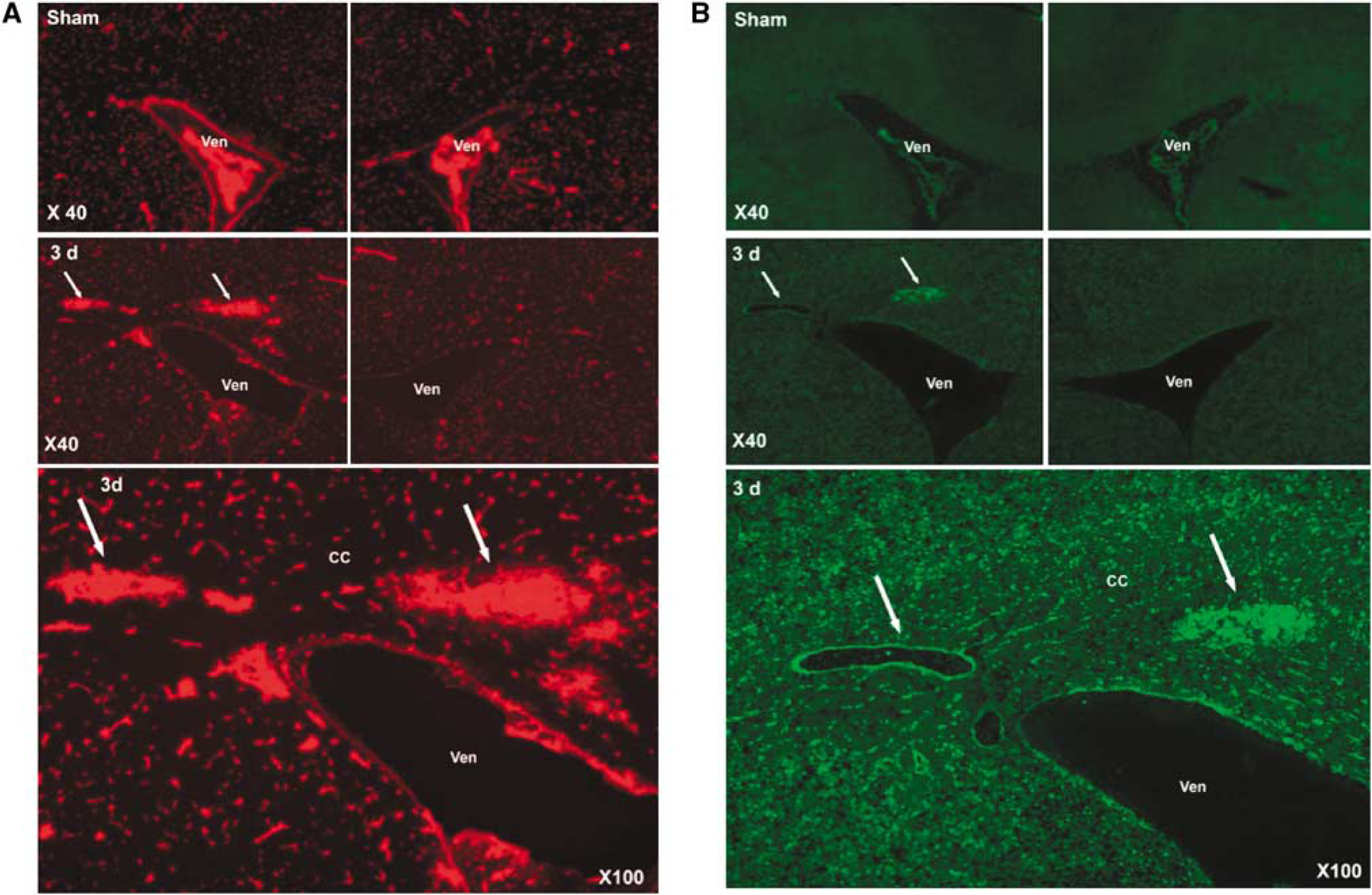
(
In addition to the CC in the periventricular regions, WM near the hippocampus showed increased ADC (Figure 4A). Sections from regions that corresponded to the increased ADC signal showed EB extravasation, which was restricted primarily to the WM (Figures 4B and 4C).
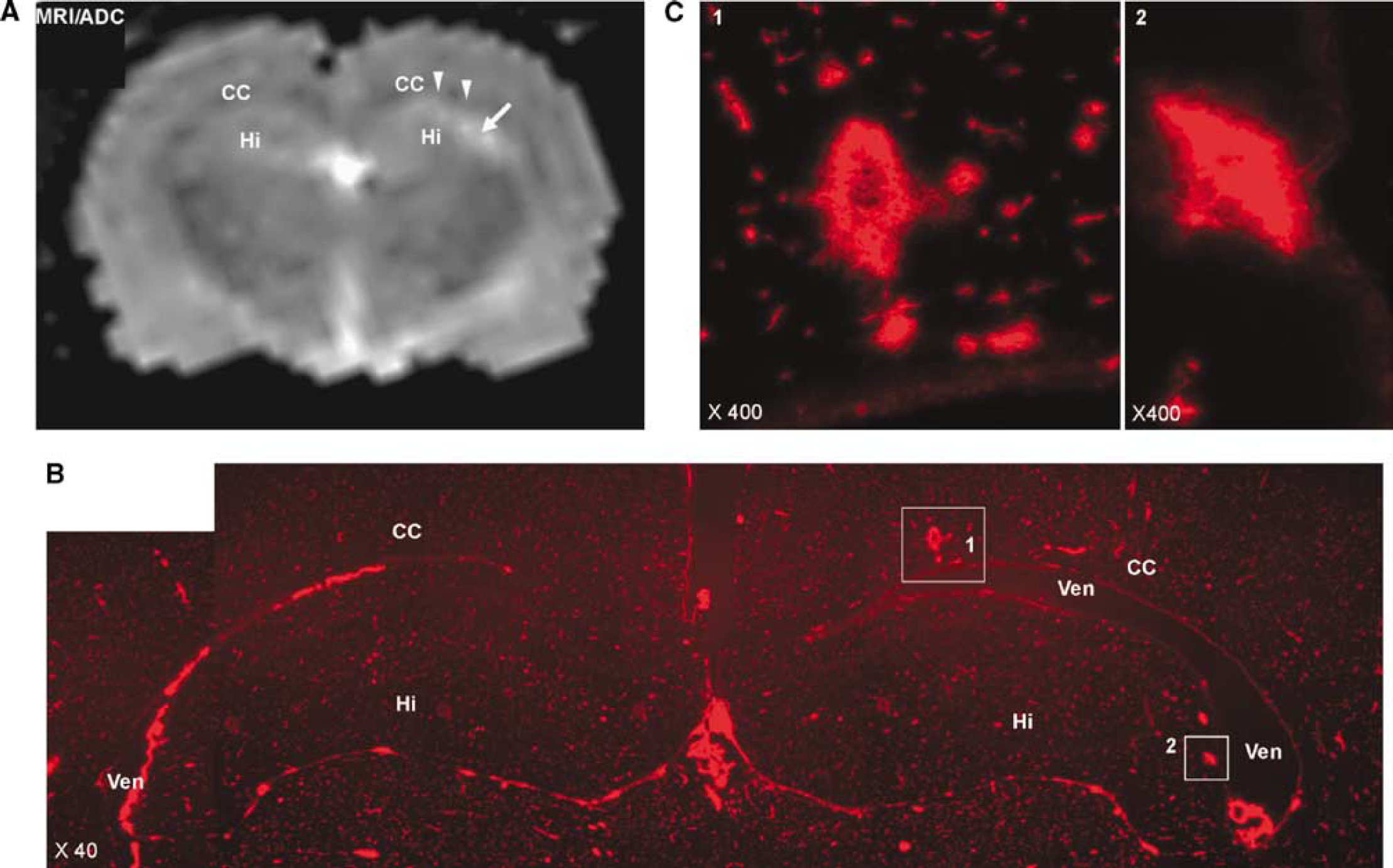
(
Tight junction proteins, occludin, and claudin-5, were intact in the CC regions that showed EB extravasation (Figure 5). In spite of the intact TJPs, there was focal EB leakage around the blood vessels, suggesting a breach of the vascular integrity (Figure 5).
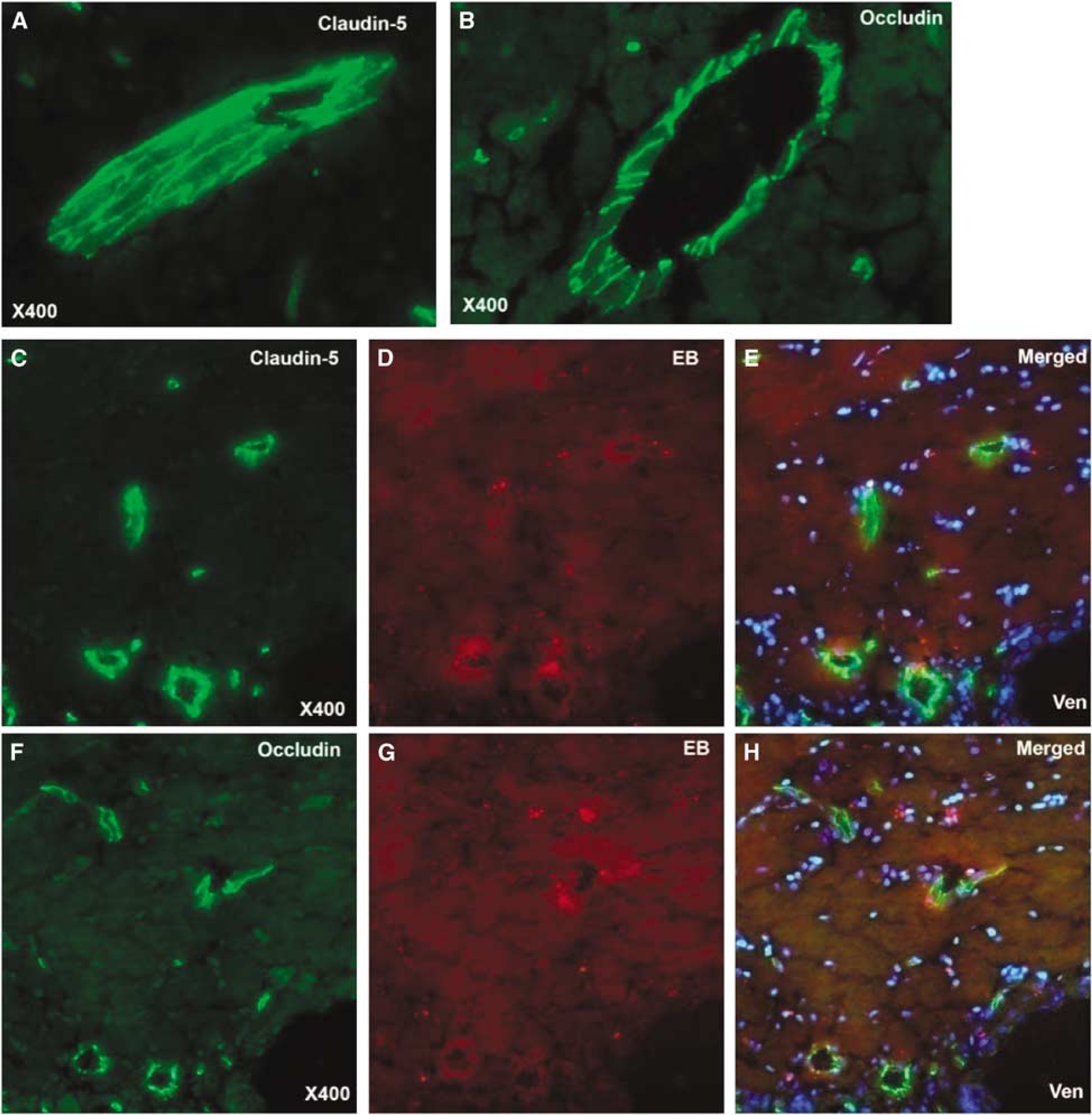
Micrographs showing immunohistochemistry (green) of claudin-5 and occludin in injured corpus callosum (CC) after 3 days of BCAO. (
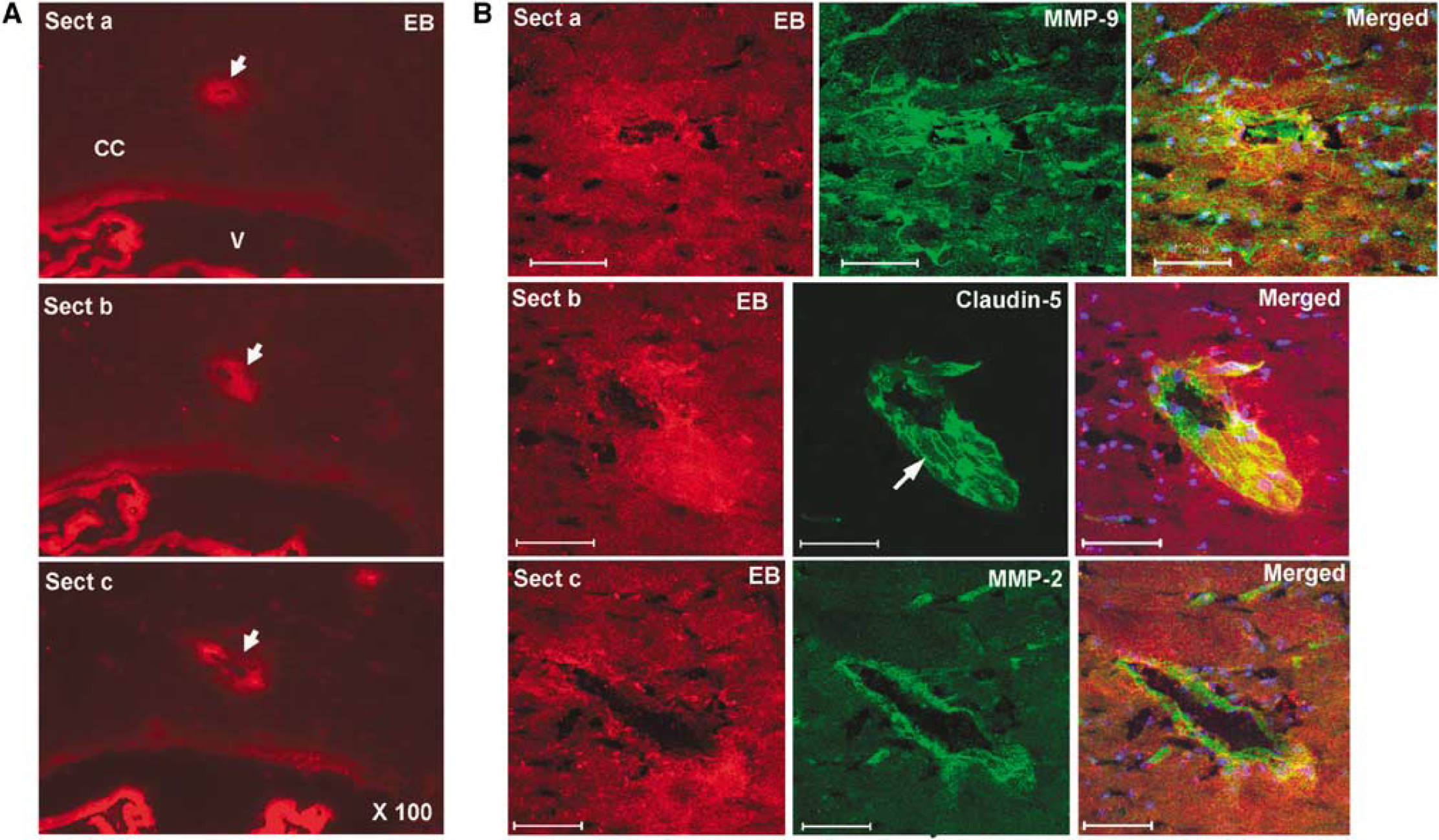
Immunohistochemistry for MMPs and claudin-5 in CC where EB showed BBB leakage after 3 days of BCAO. (
Discussion
Hypoxic hypoperfusion secondary to permanent BCAO in rats produces extensive changes in the WM, which appear by 3 days and progress for months. Expression of MMPs, which are increased in WM, have been associated with BBB breakdown and demyelination. We found that MRI identified regions in WM with increased signal on ADC maps; the increased ADC corresponded with leakage of EB. There was marked asymmetry in ADC signals between the two hemispheres with one side being significantly more affected than the other in all but one of the rats. Surprisingly, neither Gd-enhanced MRI, using the MTGA method, nor radiolabeled sucrose showed leakage in the WM in the same regions where EB fluorescence was observed microscopically. Our results show an asymmetric opening of the BBB in rats with permanent BCAO that was detected with MRI ADC maps and EB extravastion, but was not seen with 14C-sucrose or with the MTGA method.
Using the abnormalities seen on MRI to guide the immunohistochemistry, we found that the regions with higher ADC values had extravasation of EB from blood vessels that colocalized with gelatinase activity on
We found that sites of increased ADC in the WM corresponded with EB leakage and increased MMP expression. Others have shown that MMPs are increased in WM in rats with BCAO, and that MMP-2 knockout mice have reduced BBB damage as shown by EB leakage (Ihara et al, 2001; Nakaji et al, 2006). The MMPs found in the WM could have a dual role in disrupting the BBB and breakdown myelin. Matrix metalloproteinases disrupt the basal lamina and degrade TJPs after reperfusion in focal ischemia because of middle cerebral artery occlusion (Yang et al, 2007). Myelin proteins are degraded by MMP (Chandler et al, 1995). Human autopsy studies in patients diagnosed with vascular dementia showed expression of MMP-2 in reactive astrocytes and MMP-3 in macrophages around blood vessels in regions of demyelination (Rosenberg et al, 2001). Vasogenic edema has been proposed as an etiologic factor in the damage to the WM, but further studies will be needed to link the increases in ADC with the mechanism of WM damage (Starr et al, 2003; Wardlaw et al, 2003).
We measured BBB permeability with MRI by the graphical method originally designed for use with autoradiography (Patlak et al, 1983). This method has been used successfully to quantify BBB permeability after middle cerebral artery occlusion in rats (Ewing et al, 2003). We found in the BCAO model that the MTGA method failed to show an increase in permeability in the regions that had increased ADC. Furthermore, using the standard 14C-sucrose brain uptake method, we were unable to show increased sucrose leakage. Finally, the proteins that form tight junctions in the endothelial cells, claudin-5, and occludin, which are disrupted early in reperfusion injury after middle cerebral artery occlusion were unaffected. This suggests that EB, which is bound to albumin, managed to escape the blood vessel by a route other than through the tight junctions, but that the smaller, Gd-DTPA and sucrose molecules were restricted. Our observation is in agreement with findings from other studies that have shown a selective increase in permeability of blood proteins, such as albumin, during the development of vasogenic edema under conditions of hypoxia (Plateel et al, 1997). This suggests that EB-albumin may be crossing the BBB by a transendothelial rather than a paracellular route. Further study at the electron microscopic level would be needed to confirm this physiologic observation. Although the reason for this anomalous behavior is unclear from our data, there are possible explanations. One possibility is that gelatinases act on basal lamina before disruption of the more deeply buried TJPs. Loosening the basal lamina could have an effect on the transendothelial movement of proteins. Another possible explanation is that hypoxia leads to oxidative stress, which activates MMPs through protein tyrosine kinase signaling. This could cause the phosphorylation of TJPs, affecting their function without degrading the proteins (Haorah et al, 2007). Alteration of BBB structural and functional integrity because of tyrosine phosphorylation of TJP could affect the molecular ‘fencing’ effect of occludin and claudin-5 and alter the interaction with the intracellular anchoring proteins, ZO-1, -2, and -3 (Hawkins and Davis, 2005). However, answers to these questions will have to await further studies.
In conclusion, we have shown that ADC is a sensitive neuroimaging biomarker for the identification of the sites of putative vasogenic edema. Using MRI to localize the injury, we found MMP activity around blood vessels with spotty EB leakage; both MMP-2 and MMP-9 were seen in the region of the leaky blood vessels. Finally, we have observed extravasation of the large albumin-bound EB molecule despite intact tight junctions, suggesting movement of the large EB-albumin complex through vessels. Our results show that long-term studies of drug treatments to block the damage to the WM will be greatly enhanced by MRI, reducing the need for large groups of animals by guiding tissue sampling. Considering the increasing importance of vascular cognitive impairment as a cause of age-related, adult-onset dementia, use of MRI will greatly enhance preclinical studies to test potential therapies.
Contributions to study
RS and YY made equal contributions to the study. RS and ST did imaging and MTGA studies. YY, ECJ, EW, JT did the biochemical and histologic studies. EYE and EW did the animal surgery and contributed to imaging. GAR conceived and coordinated the study. All authors contributed to the writing of the article. Confocal images were generated in the University of New Mexico Cancer Center Fluorescence Microscopy Facility, supported as detailed on the web page: http://hsc.unm.edu/crtc/microscopy.
