Abstract
Delivery of therapeutic or diagnostic agents to the brain is majorly hindered by the blood–brain barrier (BBB). Recently, many studies have demonstrated local and transient disruption of the BBB using low power ultrasound sonication combined with intravascular microbubbles. However, BBB opening and closure mechanisms are poorly understood, especially the maximum gap that may be safely generated between endothelial cells and the duration of opening of the BBB. Here, we studied BBB opening and closure under magnetic resonance (MR) guidance in a rat model. First, MR contrast agents (CA) of different hydrodynamic diameters (1 to 65 nm) were employed to estimate the largest molecular size permissible across the cerebral tissues. Second, to estimate the duration of the BBB opening, the CA were injected at various times post-BBB disruption (12 minutes to 24 hours). A
Introduction
Cerebral tissues are isolated from circulating blood by the blood–brain barrier (BBB) (Rubin and Staddon, 1999). This physiological barrier consists of a lining of tightly packed vascular endothelial cells, different from the peripheral blood vessels (Hawkins and Davis, 2005). The tight junctions between these cells restrict the diffusion of microscopic objects (e.g., bacteria) and of large, hydrophilic molecules (>400 Da) from blood to brain parenchyma, while allowing the passage of small, hydrophobic molecules such as O2, CO2, proteins, and metabolites (Pardridge, 2005). Because of the neuroprotective nature of the BBB, delivery of potentially important diagnostic and therapeutic agents is a major challenge in the treatment of most brain disorders. Strategies to design specific drugs targeted to the brain involve finding an efficient mode of drug delivery across the BBB.
Recently, it has been demonstrated that the use of low power focused ultrasound combined with a systemic injection of lipid- (or polymer-) shelled microbubbles enables a noninvasive, local and transient disruption of the BBB (Hynynen et al, 2001). Many studies were then carried out to (1) establish optimal ultrasound parameters that permit adequate tissular penetration without causing tissue damage (Choi et al, 2006; Hynynen et al, 2005; O’Reilly et al, 2010, 2011a, 2011b; Sheikov et al, 2008), (2) quantify permeability of the disrupted brain tissue (Vlachos et al, 2010, 2011), and (3) evaluate responses to treatments of particular brain disorders including tumors (Chen et al, 2010; Liu et al, 2010; Treat et al, 2008) and Alzheimer's disease (Jordao et al, 2010; Raymond et al, 2008). Most of these studies used magnetic resonance contrast agents (MR-CA) for monitoring the processes.
Despite a rapidly growing number of studies, the mechanism of ultrasound-induced BBB opening is understood only poorly. In particular, the maximum space that can be safely generated (ensuring reversibility) between endothelial cells, and the duration for which this opening lasts, have not been measured. The knowledge of these parameters is crucial for the current development of brain targeted nanoparticles. Functionalized MR-CA for instance, are available in a wide range of hydrodynamic diameters: gadolinium (Gd) chelates, a few nm (Wadghiri et al, 2003), iron nanoparticles, 25 to 100 nm (Mendonca Dias and Lauterbur, 1986; Renshaw et al, 1986), gadolinium-based emulsions or liposomes, 200 to 300 nm (Devoisselle et al, 1988), and have very different vascular remanences, ranging from a few minutes to tens of hours. An estimate of the time window during which a molecule of a given size may be delivered across the BBB would be useful to monitor the amount of drug released to cerebral tissue and to adjust the dosage. For example, it has been shown that 3 to 8 nm wide particles are able to cross BBB 20 minutes after its ultrasound-induced disruption, but not particles of size 50 nm (Choi et al, 2010). Additionally, as the brain is unprotected while the BBB is open, it is important to know the time-to-recovery to limit any possible brain damages induced by tissular penetration of pathogenic agents.
The quantification of the amount of MR-CA crossing the BBB requires development of molecular imaging sequences via imaging techniques that are sensitive (high-field magnetic resonance imaging (MRI)) and provide outputs that can be correlated with the MR-CA concentration. Contrast agent quantification is one of the principal challenges for MR molecular imaging. Unlike other techniques such as fluorescence or nuclear imaging where the received signal comes directly from the CA, MR signal comes from the surrounding water protons and is therefore indirectly related to the presence of an exogenous probe. To quantify the CA concentration using MRI, it is necessary to model its interaction with the surrounding water molecules. Paramagnetic complexes interact with the surrounding water protons by decreasing their relaxation times (Swift and Connick, 1962). Contrast agent concentration can be linked to the
In this study, a calibrated BBB opening procedure was performed in healthy young adult rats under MR guidance. Five MR-CA were used (three paramagnetic and two superparamagnetic) with different hydrodynamic diameters (from 1 to 65 nm) to investigate the maximum permissible size of the gaps induced in the endothelial wall under safe conditions. Then, using the
Materials and methods
Animal Preparation
All experiments were performed in accordance with the recommendations of the European Community (86/609/EEC) and the French legislation (decree no. 87/848) for use and care of laboratory animals. This study has been approved by the Comité d’ÉThique en Expérimentation Animale du Commissariat à l’Énergie Atomique et aux énergies alternatives Direction des Sciences du Vivant Ile de France (CETEA CEA DSV IdF) under protocol ID 12_009. A total of 47 Sprague Dawley male rats (80 to 100 g, Janvier, Le Genest-Saint-Isle, France) were used. Their head was shaved to ensure proper coupling of the ultrasound beam to the brain. Rats were anesthetized with 1.5% isoflurane in a mixture of air and oxygen, and placed in a cradle in prone position. A catheter (25 G needle) was positioned in the caudal vein to inject microbubbles and MR-CA from outside the scanner with minimal dead volume. Ten percent heparin was added to all injected solutions to avoid clot formation in the catheter. Body temperature and respiration rate were continuously monitored during the experiments.
Magnetic Resonance Contrast Agents
Magnetic resonance contrast agents of different hydrodynamic diameters were provided by Guerbet Research (Roissy-Charles de Gaulle, France). Main characteristics and injected doses of these nanoparticles are summarized in Table 1. After each MR-CA injection, the bolus was flushed by injection of 100
Principal features of the paramagnetic and superparamagnetic nanoparticle contrast agents (CA) used in this study
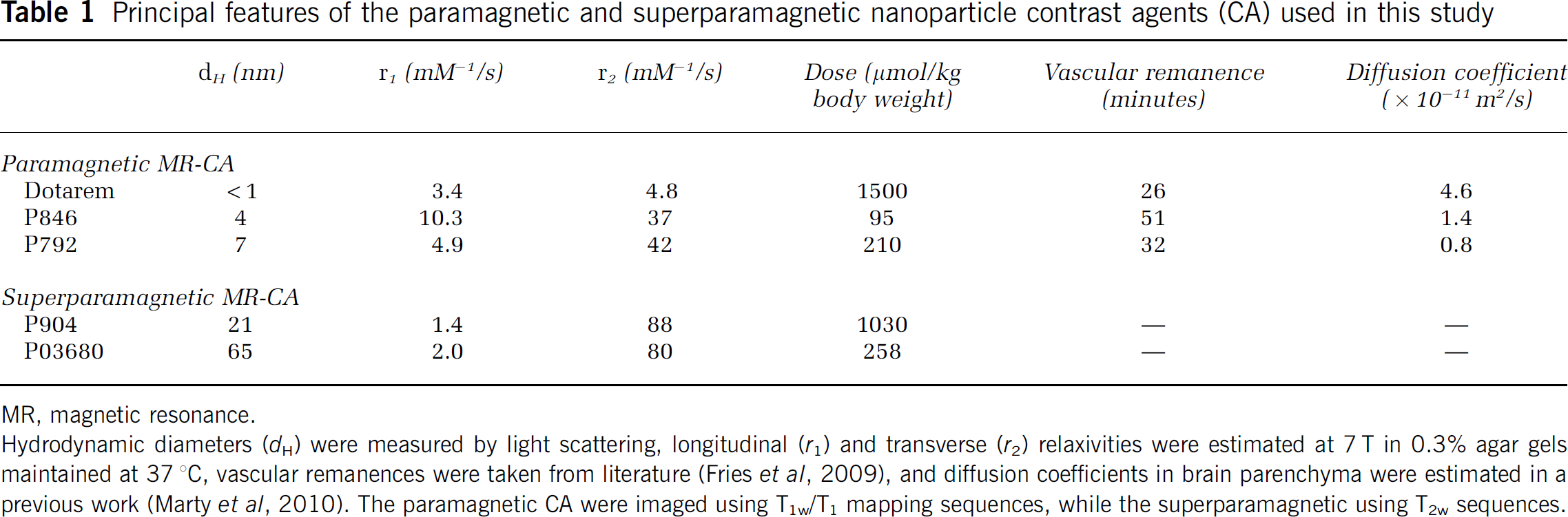
MR, magnetic resonance.
Hydrodynamic diameters (
Ultrasound Equipment
A MR-compatible focalized transducer (central frequency 1.5 MHz, diameter 30 mm, focal depth 20 mm, Imasonic, Voray sur I’Ognon, France) driven by a programmable function generator was used to produce ultrasound waves. It was coupled to the rat skull using a latex balloon filled with deionized and degassed water. Electrical power sent to the transducer was monitored during the BBB opening session. Main characteristics of the transducer (focal point size 0.6 × 0.6 × 3 mm3, transcranial acoustic transmission factor 52%±5%) were estimated in a previous study (Larrat et al, 2010).
Blood–Brain Barrier Opening
Blood–brain barrier disruption was performed during MR imaging session thanks to a dedicated holder maintaining the ultrasound transducer above rat head. Its position was monitored using an ARFI (acoustic radiation force imaging) sequence and the right thalamus in the brain was chosen as a target for BBB disruption. Sonovue microbubbles (Bracco, Milan, Italy) were administrated via a bolus (1.5 × 108 bubbles/mL, 200
Magnetic Resonance Imaging Acquisitions
Magnetic resonance imaging was performed on a 7 T/90 mm Pharmascan scanner (Bruker, Ettlingen, Germany). A saddle coil was specially designed in-house for excitation and signal reception. The geometry and size of the resonator (diameter 30 mm) were optimized to maximize the overall signal-to-noise ratio over the whole brain while allowing the ultrasound beam to propagate from the transducer to the rat brain.
A multislice spin-echo sequence was modified to include additional motion-sensitizing gradients, and synchronized to ultrasonic bursts so that the phase signal was proportional to local acoustic intensity (Larrat et al, 2010; McDannold and Maier, 2008). This ARFI sequence was acquired with the following parameters: TE (echo time)/TR (repetition time)=40/1,700 milliseconds,
A high spatial resolution
To measure concentration of Gd-chelates, a
Data Analysis
Data were analyzed using dedicated codes written in Matlab software (MathWorks, Natick, MA, USA). Maps of acoustic pressure index (
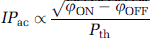
where
To generate
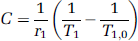
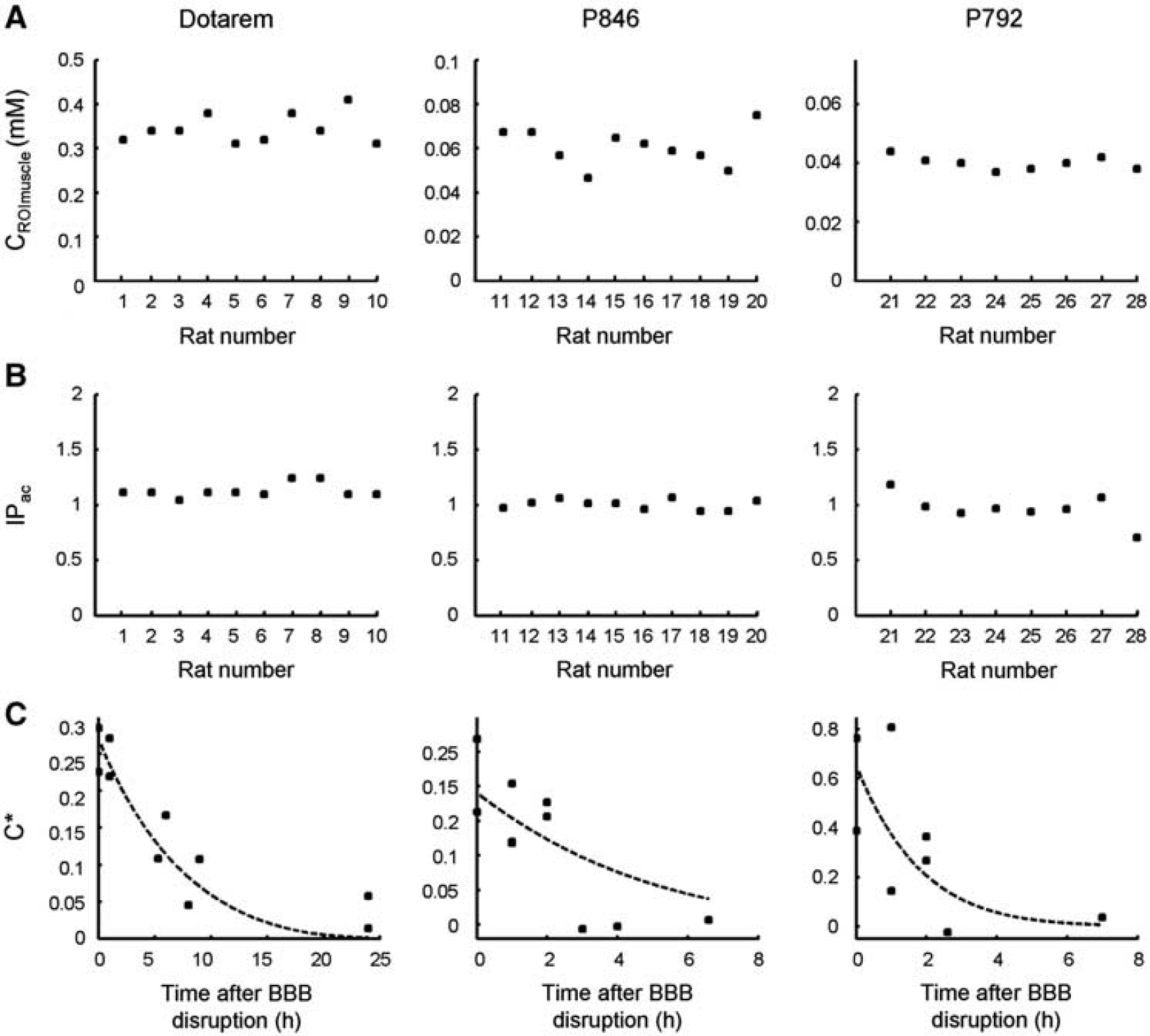
Signals from different manually drawn ROIs (regions of interest) were analyzed. A ROIspot of 0.6 × 0.6 × 3 mm3 corresponding to the transducer focal point size was drawn in the right thalamus. To analyze the effect of BBB disruption, a similar ROIcontra was taken in the corresponding contralateral (left thalamus) region of the brain. This enabled correction for the residual vascular concentration of MR-CA during imaging. Another control ROI was drawn in an extracerebral region in the cheek muscles (ROImuscle). For each rat, a corrected MR-CA concentration at the focal point (
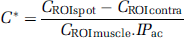
This processing step was mandatory to ensure the correction of the bias introduced by unequal injected volumes (unequal plasmatic concentrations) and the bias introduced by variations in acoustic wave penetration through the skull. These corrections allowed comparing MR-CA concentrations among animals.
Results
Low Power, Pulsed Ultrasound for Localized Disruption of Blood–Brain Barrier
In a rat model, BBB was opened transiently with the use of ultrasound, immediately followed by intravenous injection of Dotarem (Gd-chelate) and measurement of the amount of the MR-CA delivered across the BBB (Figure 1). Before the ultrasound sonication, ARFI was performed to obtain the acoustic pressure map in the brain (Figure 1A). This sequence was used to verify that the ultrasound beam was focused at the desired point in the right thalamus, and to estimate the acoustic pressure at this focal point before microbubbles injection, ensuring that the acoustic pressure remains below a predetermined safety threshold (Chopra et al, 2010; Larrat et al, 2011). Once the transducer was set at the desired position, Sonovue microbubbles were injected intravenously via a catheter positioned in the caudal vein, and the BBB was disrupted by pulsed sonication for 1 minute. Dotarem was then injected using the same catheter. To measure the concentration of Dotarem delivered,
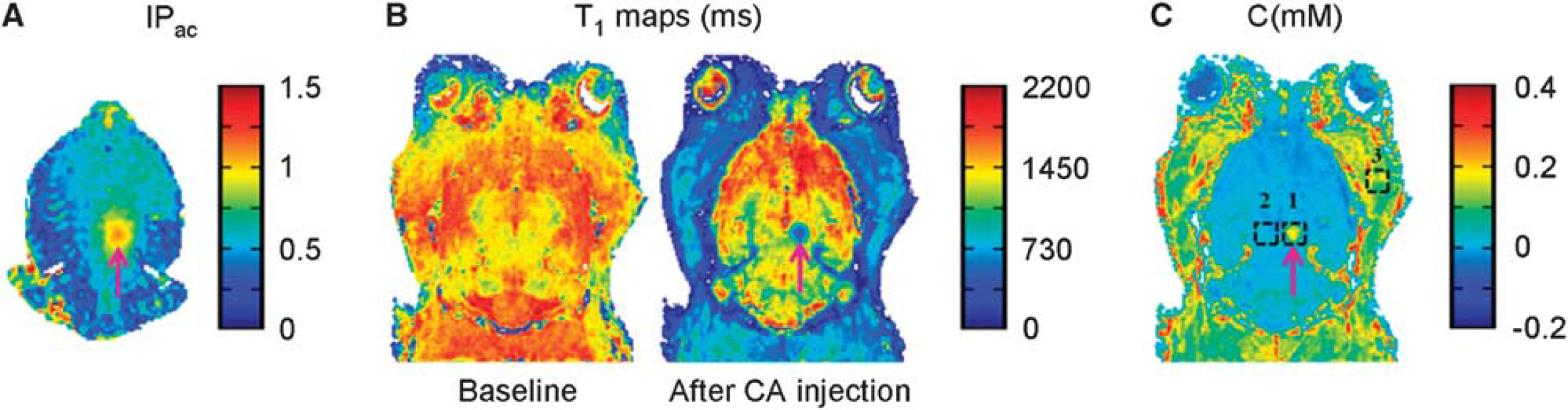
Different steps of magnetic resonance imaging (MRI)-guided ultrasound-induced disruption of blood–brain barrier (BBB). BBB disruption was achieved by intravenous injection of microbubbles via a catheter in the tail vein, in conjunction with transcranial ultrasound sonication. This was followed by injection of MRI contrast agent (CA) via the same catheter. The entire procedure was performed inside a 7 T MRI scanner. (
Maximum Gap Between Endothelial Cells After Reversible Ultrasound-Induced Disruption of the Blood–Brain Barrier
To determine the size of gaps generated after ultrasound-induced disruption of the BBB, we chose five different MR-CA of varying hydrodynamic diameters (Table 1) and verified their passage across BBB at the ultrasound focal point. The CA were intravenously injected (
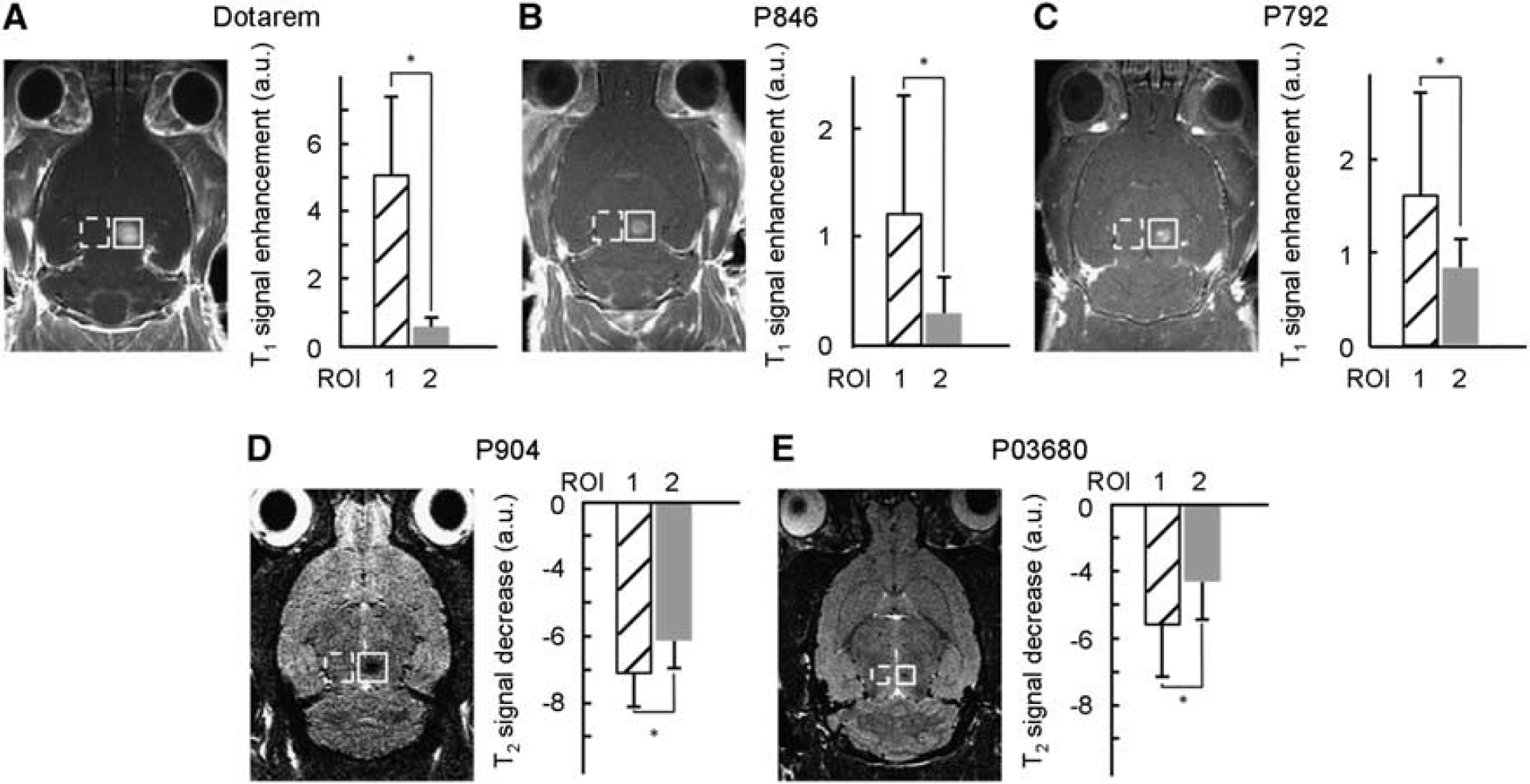
Ultrasound-induced localized delivery in rat brain of magnetic resonance imaging (MRI) contrast agents (CA) with different hydrodynamic diameters. Paramagnetic CA (Dotarem, P846 and P792) or ultrasmall superparamagnetic iron oxide (USPIO) CA (P904 and P03680) were injected in rats immediately after ultrasound-induced blood–brain barrier (BBB) disruption, and the signal intensities were measured within 5 minutes at the ultrasound focal point.
Quantification of Magnetic Resonance Contrast Agent Concentration
Rats were injected with Dotarem at increasing doses (375, 750, and 1,500
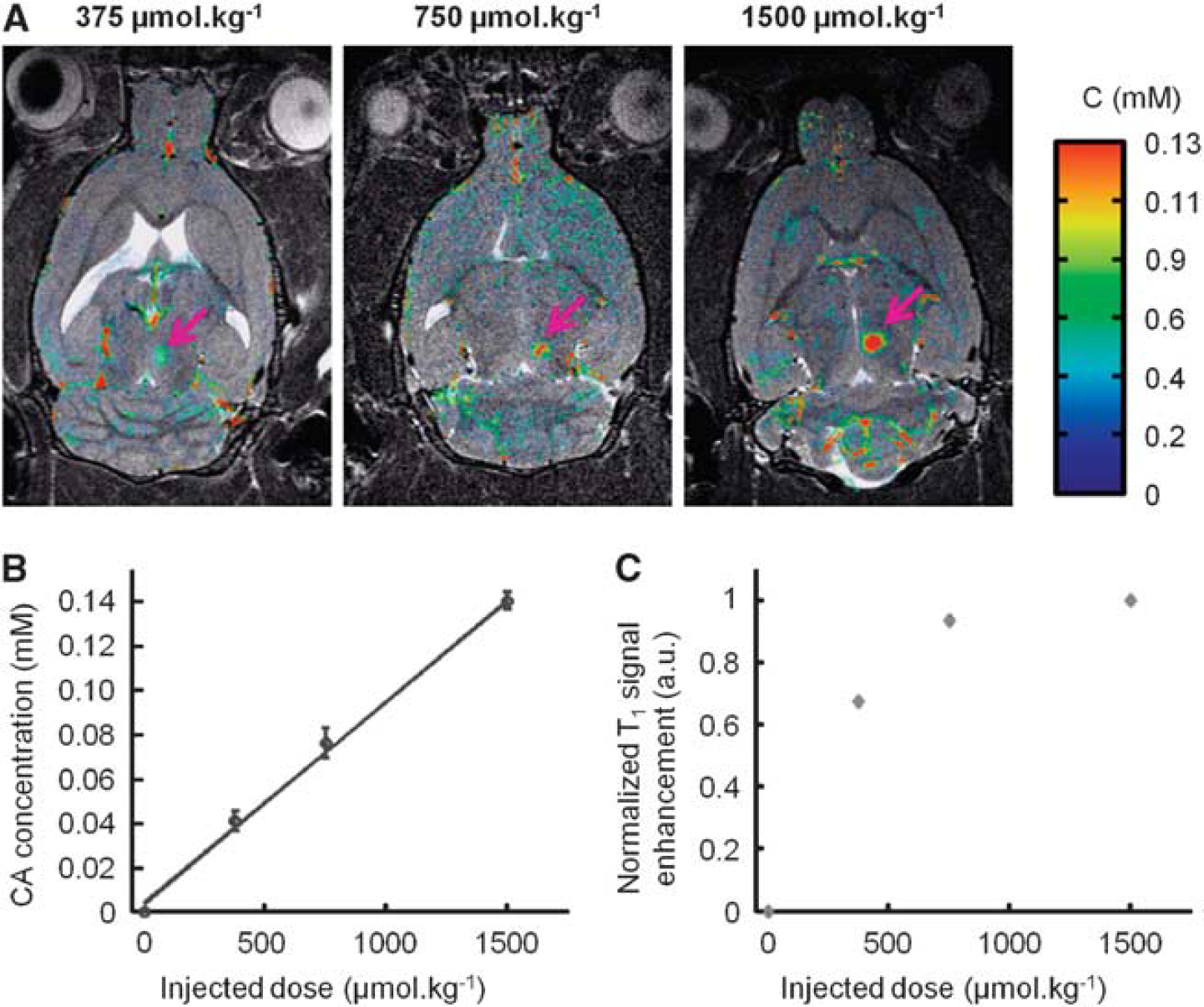
Comparison of
Quantitative Study of Blood–Brain Barrier Closure Dynamics
The BBB closure dynamics was assessed using paramagnetic CA of different molecular sizes, administrated at different times after ultrasound-induced disruption. Rats were injected with the following CA: Dotarem (
Blood–brain barrier (BBB) closure dynamics after ultrasound-induced disruption for magnetic resonance contrast agent (MR-CA) of different molecular sizes. Paramagnetic CA were injected at different times (0 to 24 hours) after BBB disruption. (
Corrected CA concentrations (
These findings confirmed that the duration of BBB crossing decreases when increasing CA hydrodynamic diameter. Second, it appears from the shape of the time-dependent concentration curve (Figure 4C) that the BBB permeability decreased at a faster rate initially and at a lesser rate later on.
A Model for Blood–Brain Barrier Closure Dynamics
Based on the experimental observations, the BBB closure dynamics was modeled as follows. First, it was assumed that each individual gap generated in the BBB by ultrasound returns to its equilibrium position (i.e., closed) as a harmonic oscillator damped by fluid friction. The gap diameter as a function of time was therefore expressed as
Next, the initial size distribution of gaps was assumed to be a hemi-Gaussian function centered on 0 with a standard deviation
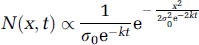
For each time
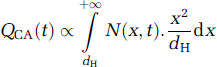
The CA diffusion in the brain outside of the BBB disrupted region is also neglected since the CA tissular concentration is quantified within a few minutes after CA intravenous injection. A previous study from our team (Marty et al, 2010) showed that the Gd-chelates used in this study do not diffuse fast enough in the brain parenchyma to make this effect significant: for example, the diffusion coefficient of Dotarem (1 nm particle) was estimated around 46
Integration of equation (5) gives:
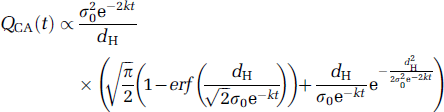
All experimental data (
The half closure time (
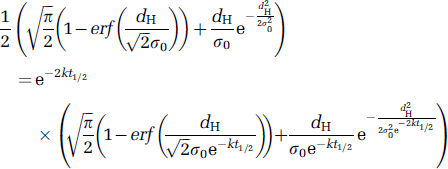
From the fitted parameters (
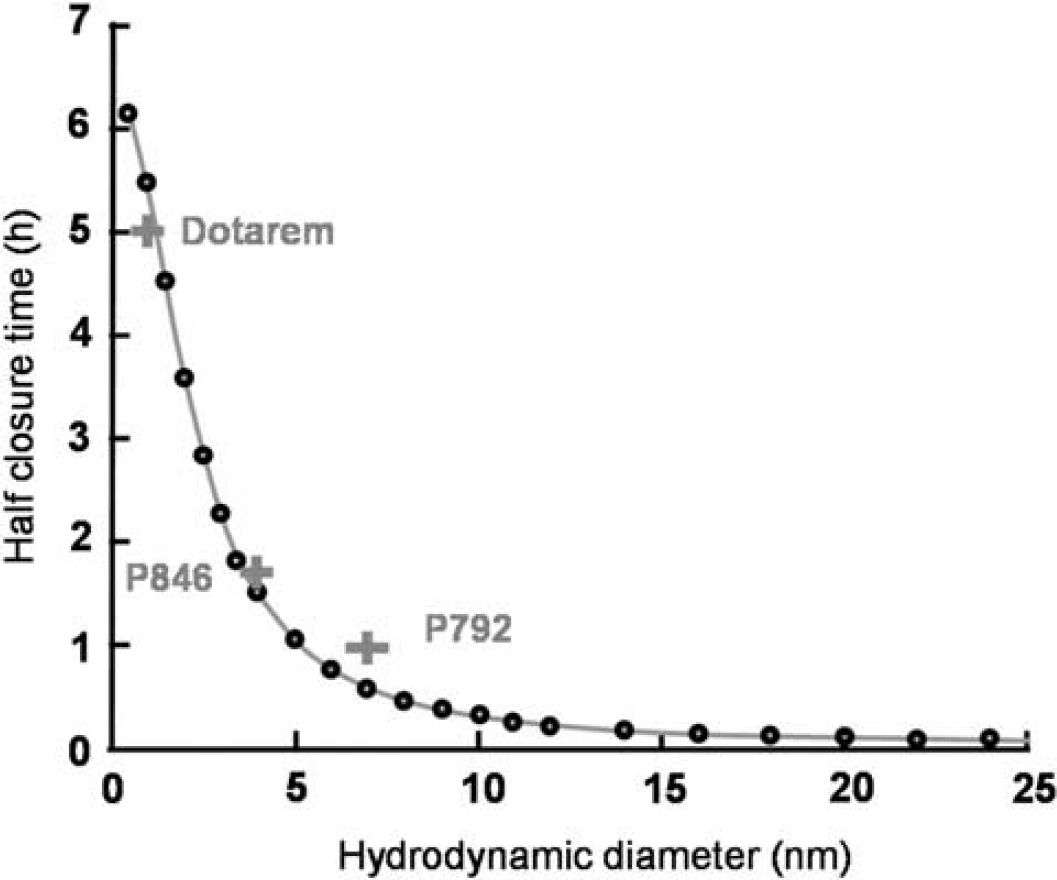
Prediction of the time window of blood–brain barrier (BBB) passage for a given nanoparticle size. Based on the theoretical model proposed here, half closure time,
For larger molecules, half closure times were greatly reduced (around 3 minutes for a 25-nm particle and only few seconds for a 65-nm particle). These results match the qualitative results obtained with USPIOs (Figure 2). Therefore, to deliver a significant amount of larger molecular size CA across the BBB, their intravenous circulation should coincide with the end of ultrasound sonication.
Lastly, it appears that the following analytical formula fits reasonably well the numerical solution of equation (7), enabling to directly estimate half closure time as a function of hydrodynamic diameter:
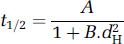
with
Discussion
Ultrasound-Induced Blood–Brain Barrier Disruption
Drug delivery to the brain remains a major challenge for the treatment of most brain diseases. Although pathologies such as cancer degrade BBB integrity (Liu et al, 2012), it was proven that endothelial wall permeability is still poorer in these regions than in vessels of other organs. Recently, ultrasound-induced BBB disruption was demonstrated to be an effective method of significantly increasing permeability of the blood–tumor barrier in a controlled manner (Chen et al, 2010). Another study reported that vessel wall permeability after ultrasound-induced BBB disruption was comparable to permeability values for unprotected organs (Vlachos et al, 2010). Our study further demonstrates that ultrasound combined with intravenous injection of microbubbles is a reliable, reproducible and noninvasive technique to deliver nanoparticles with hydrodynamic diameters up to 65 nm. Moreover, in our protocol, no edema or hemorrhages were detected on
Magnetic Resonance Guidance
The ultrasound-induced BBB disruption described here was performed entirely under high-field MRI guidance. This is a challenge as it requires integrating a stereotactic frame, a MR-compatible ultrasound transducer adapted to the geometry of the rodent head and a dedicated RF (radiofrequency) coil into the 8.5-cm bore of a preclinical MRI scanner. The optimized RF coil allowed the quantification of Gd MR-CA at a micromolar range, with sufficient signal homogeneity over the whole brain and the ability for the ultrasound beam to propagate through the coil.
The anesthetized animal was placed inside the MRI scanner just before the start of the BBB disruption procedure till recovery. Thus, it was possible to obtain an acoustic intensity map before BBB disruption. This ensured a proper positioning of the ultrasound focal spot in the right thalamus and a precise
Maximum Gap Obtained Between Endothelial Cells
Contrast agents of increasing hydrodynamic sizes were tested, and the maximum gap width generated between endothelial cells after BBB disruption was estimated with our acoustic parameters. Gaps slightly above 65 nm could be generated safely, and the BBB recovered full integrity within 24 hours. The mechanical index of our setup was 0.37 which is below the standard value proposed by other studies for reversible BBB opening (McDannold et al, 2008). This result is particularly interesting from the view of development of targeted CA. Using a similar sonoporation protocol, it would be possible to deliver nanoparticles such as USPIOs, functionalized to target tissular biomarkers. In contrast, larger objects like emulsions or liposomes that typically have hydrodynamic diameters >100 nm, would be difficult to deliver to the brain using this technique, assuming a purely paracellular pathway. In general, an estimate of the maximum possible gap diameter would be helpful when the technique is applied to deliver therapeutic molecules. Several therapeutic drugs could benefit from this information: for instance, chemotherapeutic drugs such as Avastin (
The maximum gap diameter measured in this study is probably dependent of the sonication parameters, in particular ultrasound frequency, pulse duration and duty cycle, total sonication time, and size distribution of injected microbubbles (Sonovue has a broad distribution between 1 and 8
Blood–Brain Barrier Closure Dynamics
In this study, the concentration of paramagnetic CA delivered at the ultrasound focal point was quantitatively measured, based on which, the duration of passage across the BBB was estimated for molecules of different sizes. This duration decreased rapidly as a function of hydrodynamic diameter of the CA. For instance, small molecules (around 1 nm) continued to cross the BBB for >10 hours, whereas large iron oxide particles (around 25 nm) had a time window of only a few minutes. Therefore, to maximize the amount of extravasation in the brain, large molecules should be injected right after or even during ultrasound sonication.
The results presented here also suggest a closure of the BBB at a progressively decreasing rate. We present a simple, damped, elastic model assuming an exponential decay of individual gap sizes to semi-quantitatively describe the observed dynamics of BBB closure. The experimental data were fed in the model to derive important physiological parameters, such as the typical closure time constant
This model assumes a paracellular passage of injected molecules with an inflow limited by the size of the molecule relative to the size of the pores. Our results support the notion that most of the crossing happens between endothelial cells rather than through them. This reinforces the hypothesis that during ultrasound-induced BBB opening, sonicated microbubbles exert a mechanical stress on endothelial cells, which makes them contract on themselves and loosen the junctions between them creating gaps (Vykhodtseva et al, 2008).
Limitations of the Study
The proposed BBB closure model has certain limitations. It does not take into account potential increased permeability of endothelial cell membranes (transcellular BBB crossing). It does not distinguish between hydrophilic and lipophilic molecules although it is known that lipophilic agents present an increased ability to cross the BBB via transcytosis (Pardridge, 2005). This concurrent release pathway could explain the observed differences between experimental data of P846 and P792 and predictions from the theoretical model.
The three Gd-chelates compared in this study have similar but not equal vascular remanences (Table 1). The tissular concentrations were measured within 12.5 minutes after CA injection, which is not a negligible time delay as the plasmatic half-life of the CA are between 26 and 51 minutes (Fries et al, 2009). Although during data processing the differences in overall injected plasmatic concentration were corrected for each CA (equation (3)), temporal variations of vascular concentrations during the
Conclusion
In this study, we developed a complete methodology for noninvasive, reversible and controlled ultrasound-induced BBB disruption under MR guidance that permitted precise quantification of the amount of MR-CA delivered to the brain parenchyma in a rat model. The two key parameters in designing drug delivery across the BBB, namely, the largest deliverable molecular size of the agent, and the time window for delivery of an agent of a given molecular size, were estimated for the first time.
Footnotes
Acknowledgements
The authors thank M Leopoldie for providing animal care, B Bresson for RF coil support, and Dr A Alahari for editing this manuscript.
Disclosure/conflict of interest
The authors declare no conflict of interest.
