Abstract
In the present study, we studied the effect of bacteremia on cerebral blood flow (CBF) autoregulation in a rat model of pneumococcal bacteremia and meningitis. Anesthetized rats were divided into five groups (A to E) and inoculated with pneumococci intravenously and normal saline intracisternally (group A, N = 10); saline intravenously and pneumococci intracisternally (group B, N = 10); pneumococci intravenously and pneumococci intracisternally (group C, N = 5); saline intravenously, antipneumococcal antibody intravenously (to prevent bacteremia), and pneumococci intracisternally (group D, N = 10); or saline intravenously and saline intracisternally (group E, N = 10), respectively. Positive cultures occurred in the blood for all rats in groups A, B, and C, and in the cerebrospinal fluid for all rats in groups D and E. Twenty-four hours after inoculation, CBF was measured with laser-Doppler ultrasound during incremental reductions in cerebral perfusion pressure (CPP) by controlled hemorrhage. Autoregulation was preserved in all rats without meningitis (groups A and E) and was lost in 24 of 25 meningitis rats (groups B, C, and D) (P <0.01). In group A, the lower limit was higher than that of group E (P <0.05). The slope of the CBF/CPP regression line differed between the meningitis groups (P < 0.001), being steeper for group B than groups C and D, with no difference between these two groups. The results suggest that pneumococcal bacteremia in rats triggers cerebral vasodilation, which right shifts the lower limit of, but does not entirely abolish, CBF autoregulation in the absence of meningitis, and which may further aggravate the vasoparalysis induced by concomitant pneumococcal meningitis.
Introduction
Sepsis affects 240 of 100,000 inhabitants annually in the United States, with a case fatality ratio that ranges from 15 to 70%, depending on the presence and severity of organ dysfunction (Martin et al, 2003). Up to 70% of patients with sepsis are reported to have central nervous system dysfunction (Bolton et al, 1993; Young et al, 1990). One potential cause for the neurological dysfunction in patients with the sepsis syndrome is abnormalities in cerebral blood flow (CBF) secondary to systemic flow disturbances (Hariri et al, 1993). Sepsis influences systemic hemodynamics, triggering vasodilation, flow redistribution, and in some cases hypotension (Wheeler and Bernard, 1999); however, few and conflicting studies have addressed the influence of sepsis on CBF. Thus, human sepsis studies found an unchanged (Pollard et al, 1997) and a reduced CBF (Bowton et al, 1989; Maekawa et al, 1991), whereas experimental studies have found increased (Hariri et al, 1993) or decreased CBF (Ekstrom-Jodal et al, 1982b; Parker and Emerson, 1977; Weiner, 1970). One potential reason for these conflicting findings may be fluctuations in CBF between studies due to a disrupted CBF autoregulation, the mechanism that preserves CBF in the face of changes in mean arterial pressure within certain limits (Lassen, 1959). However, even fewer studies have addressed this issue, although one clinical study reported an intact CBF autoregulation in hemodynamically stable patients with sepsis (Matta and Stow, 1996).
We have previously found loss of CBF autoregulation from 12 h onwards after inoculation (Pedersen et al, 2007). The objective of the present study was to investigate the effect of pneumococcal bacteremia on CBF autoregulation in rats with or without concomitant pneumococcal meningitis. To this end, we studied CBF autoregulation in rats with pneumococcal bacteremia after intravenous inoculation; in rats with pneumococcal meningitis after intrathecal inoculation and concomitant bacteremia either induced by intravenous inoculation or as a spontaneous event secondary to the intrathecal inoculation; in rats that were treated with antipneumococcal antibody to rid coexisting bacteremia; in uninfected control rats. We hypothesized that autoregulation would be preserved in rats with bacteremia alone but would be lost in rats with meningitis independent of whether bacteremia was present or not.
Materials and methods
Bacterial Strain
Streptococcus pneumoniae serotype 3 (strain 68034; State Serum Institute, SSI, Copenhagen, Denmark) was used for the experiments. Before experiments, bacteria were passed through rats to ensure virulence. A culture from rat cerebrospinal fluid (CSF) was obtained and stored at −80°C. For experiments, the pneumococci were thawed and grown on 5% blood agar plates (SSI) for 18 h at 35°C, diluted in cold sterile isotonic saline to an optic density of 0.3 (546 nm, Sherwood Colorimeter 254), and thereafter diluted 10-fold to achieve a final concentration of 1 × 107 colony-forming units (CFU)/mL, as confirmed by quantitative cultures.
Antipneumococcal Antibody
A purified polyclonal rabbit antipneumococcal capsular serotype 3 antiserum (Pneumosera, SSI, Copenhagen, Denmark, Ref. 16746, Lot. no. C313C1) was diluted 1:1 in phosphate-buffered saline for a final concentration of 5.9 mg/mL.
Experimental Pneumococcal Bacteremia and Meningitis
The study was based on a previously described model of pneumococcal meningitis in rats (Brandt et al, 2004). All animal experiments were carried out in accordance with the European Communities Council Resolves of 24th November 1986 (86/609/EEC). The experimental protocol was approved by the Danish State Research Inspectorate (J. no. 2002/561-527). Young adult male Wistar rats (Harlan, The Netherlands), weighing 250 to 350 g, were used for the experiments. Normal day/night cycles were provided for the rats with food and water ad libitum. Twelve hours after inoculation, 3 mL of saline was administered subcutaneously to the rats.
The rats were divided into the following groups A: primary bacteremia (N = 10); B: meningitis and secondary bacteremia (N = 10); C: meningitis and primary bacteremia (N = 5); D: meningitis without bacteremia (N = 10); E: control (N = 10).
Inoculation
On the day of inoculation, the rats were anesthetized with a subcutaneous injection of a mixture consisting of fentanyl (0.315 mg/mL) and fluanisone (10 mg/mL) (Hypnorm, VetaPharma Ltd., Leeds, UK), and midazolam (5 mg/mL) (Midazolam, Hameln Pharmaceuticals gmbh, Hameln, Germany), diluted 1:1:2 in sterile water, at a dose of 1.3 mL/kg of body weight.
Induction of Primary Bacteremia (Groups A and C)
Rats were inoculated with an intravenous injection of pneumococcal suspension (3.5 mL/kg body weight). The remaining groups (B, D, and E) were inoculated intravenously with an identical volume of normal saline.
Induction of Meningitis (Groups B, C, and D)
The rats were inoculated with an intracisternal injection of pneumococcal suspension (30 μL) using a 25-gauge butterfly needle. The remaining groups (A and E) were inoculated intracisternally with an identical volume of normal saline.
Meningitis without Bacteremia (Group D)
The rats were injected intravenously with antipneumococcal AB (0.7 mL/kg body weight) at the time of bacterial inoculation.
Clinical Assessment
The rats were assessed clinically at 24 h after inoculation. Severity of illness was classified by a clinical score (0 to 4) as follows: 0 = no signs of clinical illness; 1 = affected activity; 2 = without activity, but able to turn if laid on the back (intact righting reflex); 3 = without activity, but not able to turn if put on the back (loss of righting reflex); 4 = death or lethargic, lying spontaneously on the side. All the rats were weighed immediately before inoculation as well as before preparation for autoregulation studies.
Surgical Procedures
At 24 h after inoculation, the rats were anesthetized by induction with isoflurane (Forane© vapor; Dräger, Lübeck, Germany), after which anesthesia was maintained with isoflurane in a mixture of 30% 02 and 70% N20. The inspired concentration of isoflurane was 5% during induction, 2.5% during surgical procedures, and 1.7% thereafter. The rats were tracheotomized and artificially ventilated on a small animal respirator (Harvard Apparatus Ltd., Kent, UK). End tidal carbon dioxide (CO2) was monitored continuously (Brüel & Kjær, Copenhagen, Denmark) and adjusted to a level of 5 to 6 kPa by varying the tidal volume. The femoral veins and arteries were cannulated bilaterally for continuous blood pressure measurement (Type 8011/8021/8031; Simonsen & Weel, Herlev, Denmark), blood sampling, and administration of blood (to avoid hypovolemia). The rats were placed on a heating pad (HB 101/2 Pan Lab, Barcelona, Spain), and rectal temperature was kept at 37°C. The head was immobilized in a stereotactic frame (David Kopf Instruments, Germany), and the parietal bone was exposed bilaterally. A 0.7 mm burr hole was drilled in the right parietal bone, and a catheter was placed for measurement of intracranial pressure (ICP) (Simonsen & Weel, Herlev, Denmark). Using a surgical microscope, the left parietal bone was thinned over an area of 3 × 2 mm, and a 1-mm laser-Doppler probe (probe 407; Perimed, Stockholm, Sweden) was placed on the thinned bone over an area of the cerebrum without any major blood vessels. This probe was used for measurement of CBF at a wavelength of 780 nm. After surgical procedures, the rats were rested for 30 mins before CBF measurements. The duration from induction of anesthesia to commencement of measurements was approximately 2 h. At the end of measurements, 10 μL of CSF was sampled intracisternally by using a 21-gauge butterfly needle and 300 μL of peripheral blood, which after the rats were killed by decapitation.
Autoregulation
After a stable baseline was established, mean arterial pressure (MAP) was reduced in decrements of 5 mm Hg to the lowest possible level by controlled hemorrhage. Simultaneous values of CBF, as measured by laser-Doppler flowmetry, MAP, and ICP were collected at a sampling rate of 35 secs both at baseline and throughout the experiment, which lasted approximately 1.5 h.
Blood Analysis
Arterial blood from the femoral artery was analyzed for arterial pH (pHa), oxygen tension (PaO2), arterial CO2 tension (PaCO2), oxygen saturation (SaO2), whole-blood lactate and glucose, and plasma sodium (ABL 605; Radiometer, Copenhagen, Denmark). Values of pHa, PaO2, and PaCO2 were corrected for temperature.
Microbiological Analysis
Bacterial load in the CSF and blood were determined by plating 10-fold serial dilutions of 10 μL of CSF. Blood was plated undiluted 40 μL and as 100-fold dilution. Cultures were grown on 5% blood agar plates (SSI) for 18 h at a temperature of 35°C, and the number of CFU was counted.
Autoregulation Analysis
Values of laser-Doppler blood flow (arbitrary units) were normalized, with the baseline level equaling 100. Autoregulation curves in individual rats were plotted, with cerebral perfusion pressure (CPP, calculated as MAP-ICP) on the X axis and laser-Doppler flow values on the Y axis. Because manual identification of the exact value of the limits of autoregulation is difficult and subjected to bias (Schmidt et al, 1990), the lower limit was calculated by computer software as described previously (Pedersen et al, 2003; Tonnesen et al, 2005). Briefly, the program uses the corresponding values of CPP and laser-Doppler flow in each individual rat to fit a pair of regression lines. This fitting is done in a repetitive manner, starting at the baseline CPP and continuing at 1 mm Hg decrements to the lowest measured value. The pair of regression lines consists of a line with a positive slope through the points below the given CPP value and a horizontal line through the points above that CPP. Finally, the corresponding sum of squares is calculated. The pair of regression lines yielding the least sum of squares was defined as the autoregulation curve, and the CPP corresponding to their intersection was defined as the lower limit of autoregulation.
For each individual rat, autoregulation was recorded as present or absent according to the following criteria:
The range of CPP values was at least 40 mm Hg. The sum of squares obtained by the two calculated regression lines was lower than that of a single linear regression line fitted to all pressure flow data of that animal. The CPP value of the lower limit of autoregulation identified by the software was physiologically acceptable; it was at least 10 mm Hg higher than the lowest CPP measured in the lower limit experiment; was given with a standard error of less than 25% of the value itself.
If these criteria were fulfilled, autoregulation was classified as preserved. If the criteria were not fulfilled, a single linear regression was identified, and the slope of the line was calculated as percent decrease in CBF per mm Hg decrease in CPP. Cerebral blood flow autoregulation was considered to be intact if this slope was equal to or less than 0.33%/mm Hg decrease in CPP, and considered to be lost if it was greater than this value.
After classifying CBF autoregulation curves into normal and abnormal ones, all data were pooled for each of the groups, and mean CBF% autoregulation curves and limits were calculated again using the aforementioned criteria.
Statistics
Pooled data of autoregulation limits are presented as mean ±s.e.; the remaining data are presented as medians and interquartile ranges. The Kruskal—Wallis and Mann—Whitney U-tests were used for comparison between groups. Fisher's exact test was used for comparison of categorical data. Linear regression was used to assess the CBF—CPP relationship in animals with lost autoregulation; regression lines were compared using analysis of variance followed by Bonferroni-corrected t-tests to pinpoint significant differences. P <0.05 was considered statistically significant. Statistical analyses were performed using the Statistical Package for Social Sciences (SPSS, version 11.5; Chicago, IL, USA).
Results
Fifty-five rats were prepared for the study. Of the 15 rats exposed to primary bacteremia (group A), five rats died, leaving 10 to be included in the study. None died in the meningitis group (group B), whereas 5 of the 10 rats exposed to both primary bacteremia and meningitis (group C) died. By contrast, no rats died in the antibody treated (group D) and control (group E) groups. Thus, a total of 45 rats were included in the study of CBF autoregulation.
Autoregulation studies were commenced at a median of 25 h (25 to 26) after inoculation and lasted 2.1 h (2.0 to 2.2), without differences between groups.
Clinical Presentation
Clinical signs of infection were observed in all surviving rats before the study of autoregulation, 24 h after inoculation with S. pneumoniae. In the primary bacteremia group (group A), five rats had a score of 1 and five rats a score of 2. In meningitis groups (B, C, and D), nine rats had a score of 2 and 16 rats a score of 3, without differences between groups (B, C, and D). Control rats (group E) exhibited no signs of disease, as indicated by a clinical score of 0 in all animals. The clinical score was higher in each of the groups A, B, C, and D than in the group E (P < 0.001 for all comparisons).
The weight did not differ between groups before inoculation. At the start of autoregulation studies, the rats with bacteremia alone (group A) weighed 257 g (239 to 285); group B, 259 g (251 to 283); group C, 225 g (222 to 248); group D, 236 g (214 to 256); control rats (group E), 319 g (269 to 342). Groups A, B, C, and D weighed less than group E (P < 0.05). The weight loss was larger in group A than in controls (group E; P = 0.011), and in each of the groups B, C, and D than in control rats (group E; P < 0.001 for all comparisons; Table 1). The weight loss in group A was less than in either of the meningitis groups (B, C, and D; P < 0.001 for all comparisons).
Microbial, clinical, and physiological variables
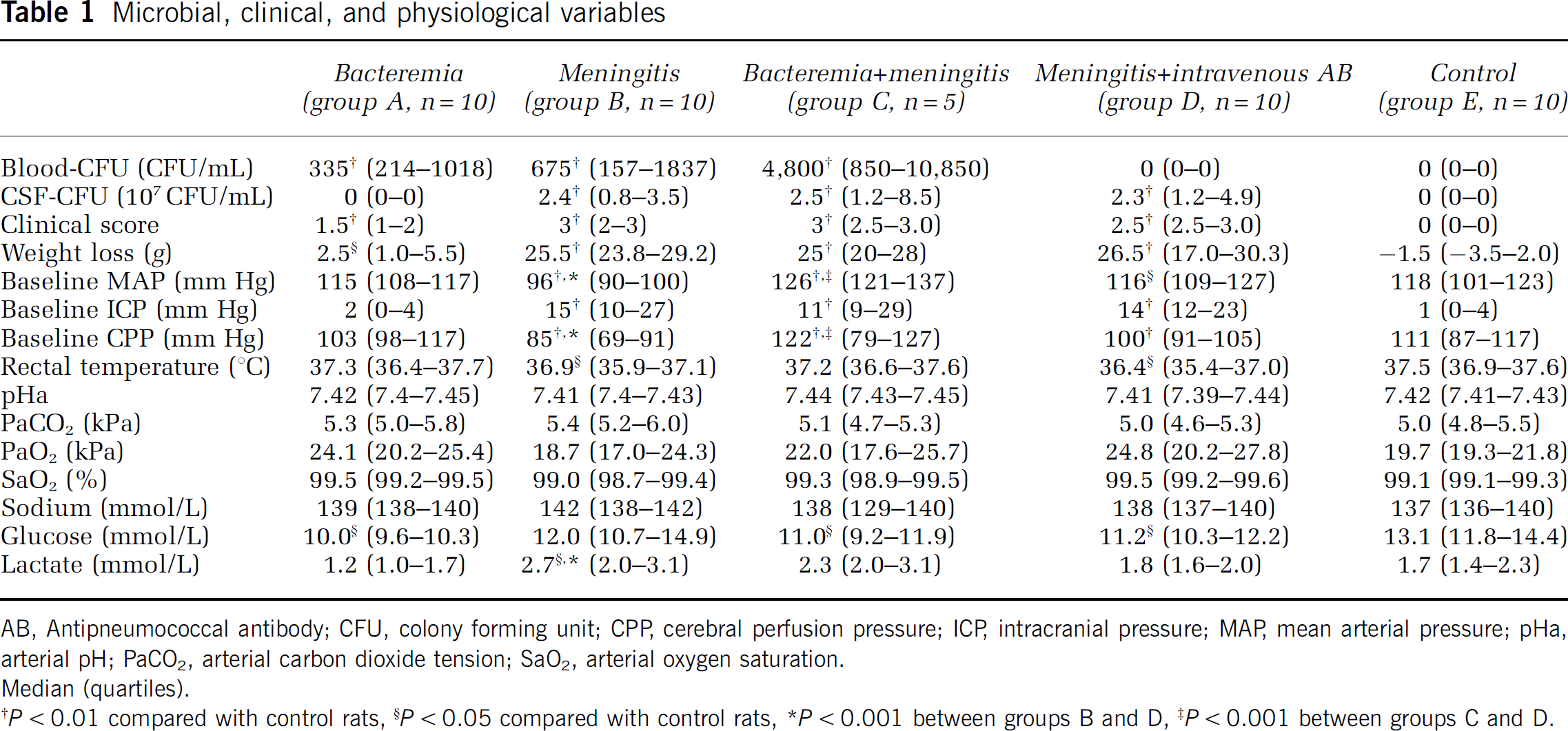
AB, Antipneumococcal antibody; CFU, colony forming unit; CPP, cerebral perfusion pressure; ICP, intracranial pressure; MAP, mean arterial pressure; pHa, arterial pH; PaCO2, arterial carbon dioxide tension; SaO2, arterial oxygen saturation.
Median (quartiles).
P < 0.01 compared with control rats
P < 0.05 compared with control rats
P < 0.001 between groups B and D
P < 0.001 between groups C and D.
Microbiology
The rats were inoculated with 3.75 × 107 CFU/mL (3.25 to 4.25). All blood cultures of rats with meningitis without bacteremia (group D) and control rats (group E) groups as well as all CSF cultures in the primary bacteremia (group A) and control rats (group E) groups were negative (Table 1). The bacterial concentrations in the blood for groups A, B, and C did not differ between groups (P = 0.303). Similarly, CSF bacterial concentrations did not differ between groups B, C, and D (P = 0.173).
Temperature and Blood Gas Analyses
Temperature and blood gas data are presented in Table 1. Despite the attempt to control temperature, control rats had slightly, but significantly, higher rectal temperatures than rats with meningitis and either secondary (group B; P <0.01) or primary bacteremia (group C; P <0.01). Lactate was significantly higher in untreated meningitis rats (group B) than in control animals (P <0.05). No differences were found between groups with regard to pHa, PaCO2, PaO2, SaO2, and plasma sodium.
Intracranial Pressure
At 24 h after inoculation, ICP differed between groups (P < 0.001; Table 1), with no differences between the three meningitis groups (P = 0.810), but with higher values in each of the meningitis groups (B, C, and D) than in the control group (group E; P <0.01). There was no difference in ICP between primary bacteremia (group A) and the control rats (group E; P = 0.853).
Mean Arterial and Cerebral Perfusion Pressures
The baseline MAP values differed between groups (P <0.001; Table 1). Thus, compared with control rats (group E), both untreated (group B; P < 0.01) and the rats with meningitis without bacteremia (group D; P <0.05) had lower MAP, whereas MAP in rats with meningitis and primary bacteremia (group C; P < 0.01) was higher. In rats with primary bacteremia alone (group A), MAP did not differ from control rats.
Accordingly, baseline CPP values did not differ between rats with primary bacteremia alone (group A) and the control rats (group E), whereas CPP in meningitis rats with secondary bacteremia (group B) and rats with meningitis without bacteremia (group D) was lower than that of control rats (group E; P <0.01 for both comparisons), whereas CPP was higher in rats with meningitis and primary bacteremia (group C) than in control rats (group E; P <0.01).
For both MAP and CPP, the values differed between the meningitis groups (B, C, and D). Thus, baseline values of group C were higher than those of group D, which were higher than those of group B (P <0.001 for all comparisons).
Lower Limit of Cerebral Blood Flow Autoregulation
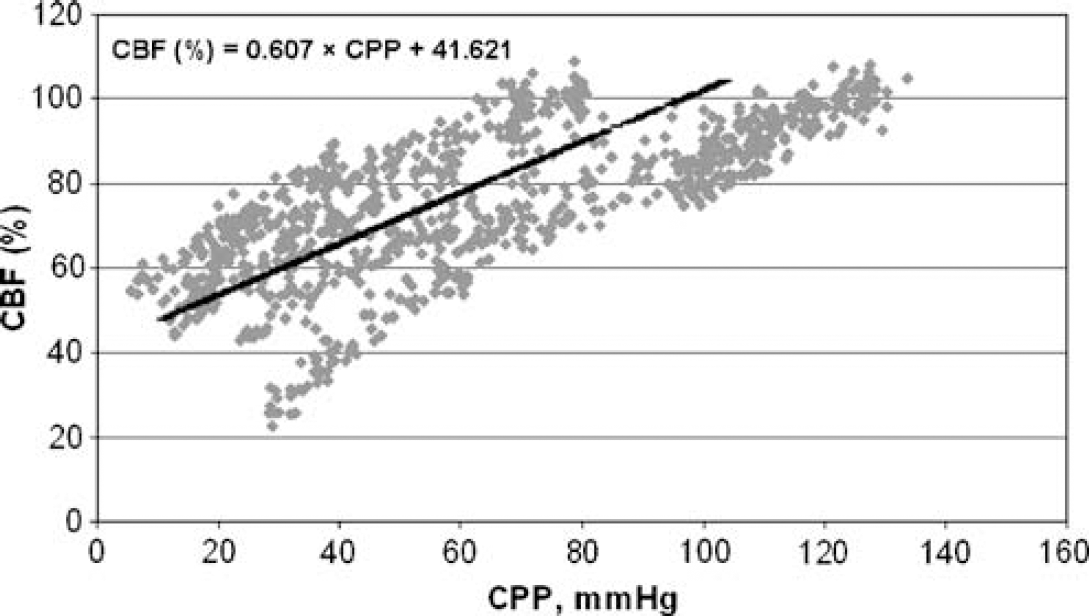
Autoregulation was present in 10 of 10 rats with primary bacteremia (group A; Figure 1); in none of 10 rats with meningitis and secondary bacteremia (group B; Figure 2); in none of five rats with meningitis and primary bacteremia (group C; Figure 3); in 1 of 10 rats with meningitis without bacteremia (group D; Figure 4); in 10 of 10 control rats (group E; Figure 5). Thus, more rats with meningitis (24 of 25) than control (0 of 10) rats exhibited loss of autoregulation (Fisher's exact test; P < 0.01), whereas rats with bacteremia alone did not exhibit loss of autoregulation.
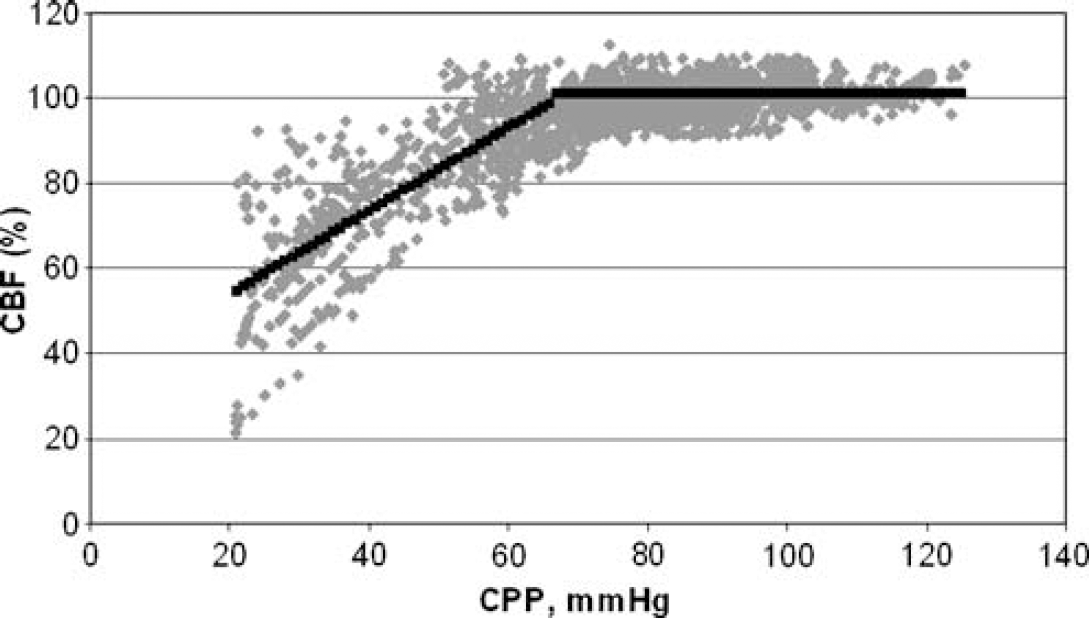
Primary bacteremia (group A, N = 10). Autoregulation preserved in all rats. For the group, the lower limit of autoregulation is 67 ± 1.5 mm Hg (s.e.).
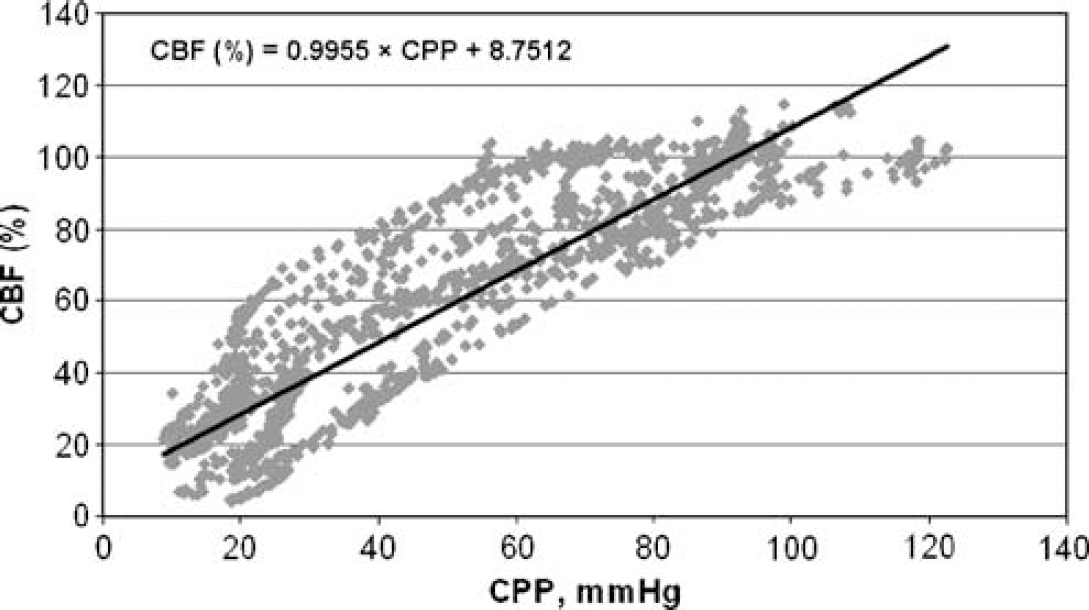
Meningitis and secondary bacteremia (group B, N =10). Autoregulation lost in all rats (P <0.01, compared with control).
Meningitis and primary bacteremia (group C, N = 5). Autoregulation lost in all rats (P <0.01, compared with control).
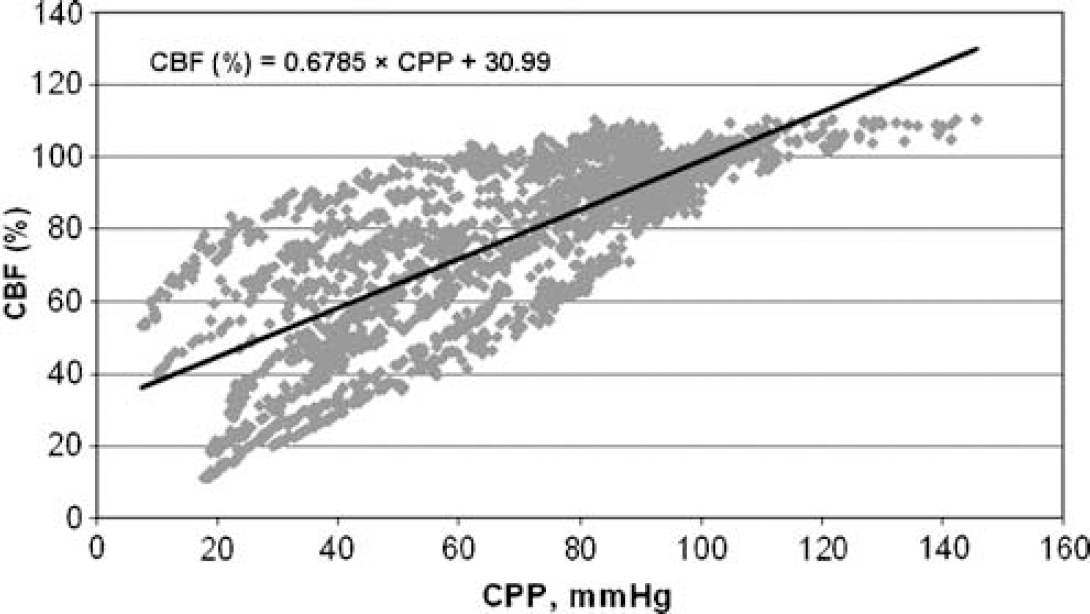
Meningitis without bacteremia (group D, N =10). Autoregulation lost in nine rats (P <0.01, compared with control).
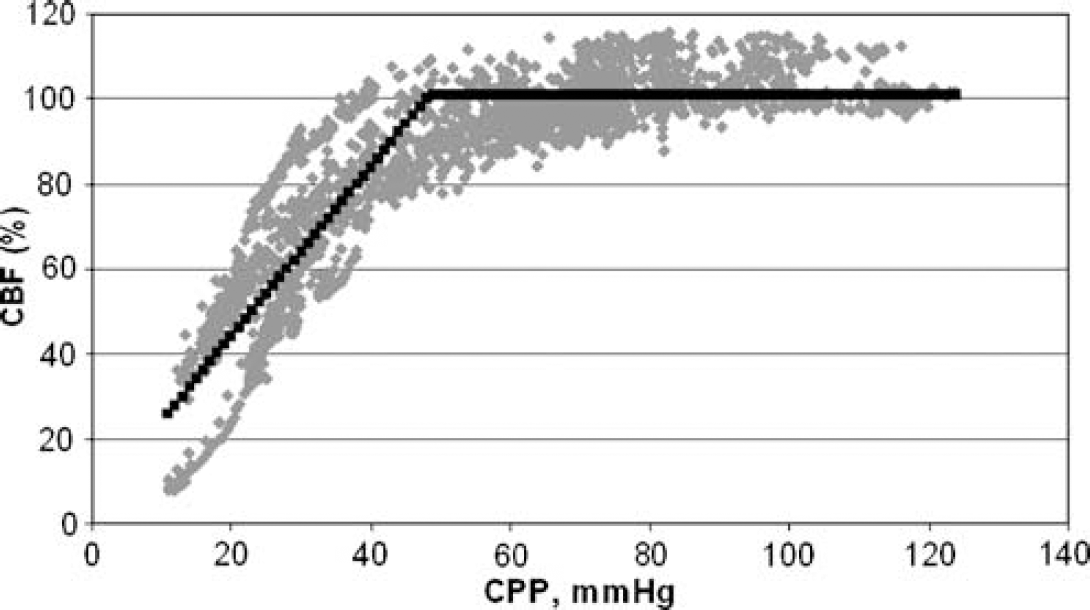
Control (group E, N = 10). Autoregulation preserved in all rats. For the group, the lower limit of autoregulation is 49 ±1.1 mm Hg (s.e.).
In rats with primary bacteremia (group A), the lower limit of CBF autoregulation of the pooled data was identified at a CPP of 67 ± 1.5 mm Hg, which was higher than the limit identified for the controls (group E; CPP: 49 ± 1.1 mm Hg, P <0.05). For meningitis rats (groups B, C, and D) there was no autoregulation, and therefore no lower limits were identified, except for one rat with meningitis without bacteremia (i.e., in group D). This rat exhibited preservation of CBF autoregulation with a lower limit at a CPP of 54 ± 0.8 mm Hg.
Slopes of Regression Lines
Regression lines for the CBF/CPP relationship were calculated for the three groups exhibiting loss of autoregulation, that is the three meningitis groups (B, C, and D). To assess the effect of bacteremia on the relationship between CBF and CPP, a pooled regression line was calculated for each of the three groups of rats with meningitis. The equations of the regression lines for individual groups are given in the respective figures. Analysis of variance showed a significant difference between the slopes of these three lines. According to the post hoc t-test, the slope of the regression line was significantly steeper in rats with meningitis and secondary bacteremia (group B; Figure 2) than in rats with meningitis and primary bacteremia (group C; Figure 3) as well as rats with meningitis without bacteremia (group D; Figure 4; P < 0.001 for both comparisons); slopes did not differ between the latter two groups (P = 0.33).
Discussion
The findings of the present study indicate a slight but distinct vasodilatory action of pneumococcal bacteremia on cerebral vessels in rats. Two lines of evidence support this notion. First, rats exposed to pneumococcal bacteremia without meningitis exhibited preservation of CBF autoregulation, although with a lower limit that was shifted to the right when compared with uninfected control rats. Second, when autoregulation was lost during meningitis, the regression line for the CBF/CPP relationship was steeper in rats with secondary bacteremia, than in those in which bacteremia had been prevented by antibody treatment.
We used continuous laser-Doppler flowmetry during induced blood pressure changes to evaluate the performance of static CBF autoregulation. Laser-Doppler flowmetry does not provide an absolute measure of CBF, but it has previously been used to study relative changes in cortical CBF during meningitis in the rats (Pedersen et al, 2007; Pfister et al, 1990), and the method estimates the limits of autoregulation correctly when validated against the 133Xe clearance method (Tonnesen et al, 2005). Even though absolute CBF values would have aided in the interpretation of the present data, transcranial Doppler with the measurement of relative changes in CBF, but without provision of absolute CBF values, have been used previously for evaluation of cerebrovascular tone in sepsis (Matta and Stow, 1996).
Twenty-five hours after inoculation, the bacterial count in the blood was similar among groups and comparable to that of other studies using an equivalent experimental model of S. pneumoniae meningitis (Koedel et al, 2004); also, the bacterial count in the CSF was similar in the rats inoculated with S. pneumoniae intracisternally. Nonetheless, the mortality differed markedly between groups after inoculation. Thus, 50% of rats inoculated intracisternally as well as intravenously and 33% of those with primary bacteremia alone succumbed, whereas all animals survived in the remaining groups, including rats with secondary bacteremia after intracisternal inoculation. Perhaps most surprising was the high mortality among rats with meningitis and primary bacteremia (due to intravenous inoculation), given that rats with secondary bacteremia (due to spread from the meninges) all survived and that blood bacterial counts were similar among survivors in the two groups. As a possible explanation, rats with primary bacteremia conceivably had a longer duration of intravenous infection than rats with secondary bacteremia (Ostergaard et al, 2006). This may have led to more severe sepsis in the former group; alternatively, the presence of bacteria in both the bloodstream and the intrathecal space may have led to more widespread blood—brain barrier damage than that observed during meningitis alone (Quagliarello et al, 1986, 1991), with more severe cerebral edema ensuing. We did not measure other markers of disease severity, such as systemic cytokine levels or CSF white blood cell count, but the ICP did not differ between the different meningitis groups. However, the potential selection bias in groups A and C remains an important caveat when interpreting the present data.
The increase in ICP during meningitis was similar whether bacteremia was present or not, whereas bacteremia alone was not associated with an ICP increase. To our knowledge, this is the first study measuring ICP in rats during pure bacteremia. In pigs, unchanged (Karakoulas et al, 2006) and increased (Hariri et al, 1993) ICP has been reported after endotoxin injection; in the latter study, an increase in cerebral blood volume was found to be the probable explanation. Moreover, a study in dogs (Ekstrom-Jodal et al, 1982b) observed a similar increase in ICP after an initial decrease in ICP on endotoxin injection (Ekstrom-Jodal et al, 1982b). Thus, the development of intracranial hypertension may depend on the species studied as well as the dose of endotoxin or bacteria.
Although the baseline MAP differed among groups, it was within normal limits for all groups at the start of autoregulation studies. Rats with meningitis and secondary bacteremia (group B), as well as those with meningitis without bacteremia (group D), had lower MAP than controls, whereas rats with primary bacteremia without (group A) and with meningitis (group C) had a higher MAP, that is the groups with the highest disease severity exhibited the highest MAP. We suggest that this was due to activation of the sympathetic nervous system due to extreme physiologic stress. In group C, a Cushing response may have contributed, that is a reflex increase of MAP to an increased ICP to maintain the CPP (Schmidt et al, 2005).
Hydration status is of importance during experimental pneumococcal meningitis; dehydration may reduce intravascular volume (Tureen et al, 1992). For technical reasons, we did not measure hemoglobin or hematocrit, which would have provided an indirect measure of intravascular volume status. However, although a reduction in hematocrit or hemoglobin increases CBF (Henriksen et al, 1981; Paulson et al, 1973) and vice versa, this would not affect the integrity of CBF autoregulation. The autoregulation studies in themselves were performed by withdrawal of whole blood, which would not affect hemoglobin concentration or the hematocrit in the short term; even if the animals had been studied for longer than a few hours, the compensatory mechanism would have been that of fluid recruitment from the extra- to the intravascular compartment, resulting in a slow decline in hemoglobin. This would have tended to steadily increase CBF and reduce the autoregulation slope, that is counteract the findings of impaired autoregulation. Finally, although meningitis rats had lower MAP values than rats without meningitis, all groups had baseline values of MAP and CPP that were located well above the normal lower limit of CBF autoregulation, ensuring the validity of the model for autoregulation testing.
Although a lower limit of autoregulation was detected in rats with primary bacteremia, indicating that an autoregulatory plateau was maintained, the CPP value of that lower limit was increased, compared with control rats. This suggests that the presence of bacteremia induced a cerebral arteriolar dilation, which impaired the capacity of vessels to dilate further in response to a reduced perfusion pressure. As a physiological corollary, hypercapnia leads to both cerebral arteriolar dilatation (Lassen, 1959) and an increase in the lower limit of autoregulation (Haggendal and Johansson, 1965). Alternatively, an increase in sympathetic nervous activity, leading to constriction of major cerebral arteries, might have increased the lower limit of autoregulation (Edvinsson et al, 1985). However, the extent of sympathetic activation in the group of rats with primary bacteremia alone was probably limited, given the fact that baseline MAP was not increased over that of control rats.
Although CBF autoregulation was lost in all groups of meningitis rats, the slopes of the CBF-CPP relationship were significantly different between groups, indicating that cerebrovascular resistance differed as well. Thus, the group of rats that was inoculated intracisternally and was allowed to develop secondary bacteremia had a steeper slope than the group of rats that was infected intracisternally but in which secondary bacteremia was abolished by specific antibody treatment. This means that the increase in CBF for any given increase in CPP was higher in bacteremic than in nonbacteremic meningitis rats, that is cerebrovascular resistance was lower in the former group than in the latter. The results of rats that were inoculated both intracisternally and intravenously at the same time (meningitis with primary bacteremia) are in apparent disagreement with this hypothesis: The CBF/CPP slope of these rats was significantly lower than that of rats with meningitis and secondary bacteremia and did not differ from that of rats with non-bacteremic meningitis. However, because of a much higher mortality, the rats that remained for analysis constituted a highly selected group with a small sample size. Thus, it remains a possibility that the selection favored rats with less disease severity and less affection of the cerebrovascular circulation. Hence, the results of this group should be interpreted with considerable caution.
The impairment of autoregulation in rats during the early phase of bacterial meningitis may be explained by cerebral arteriolar dilatation (Paul et al, 1997; Pfister et al, 1995). Processes at the level of the vasculature, including activation and disruption of endothelial cells, are initiated by pneumococcal cell wall components such as pneumococcal surface adhesin A, cholin-binding protein A, capsular polysaccharide, and the pneumolysin located in the cytoplasm of the pneumococcus; however, the precise mechanism is poorly understood (Cockeran et al, 2002; Tuomanen et al, 1985). Arteriolar dilatation during meningitis has been suggested to be mediated by oxygen-free radicals (McKnight et al, 1992), cerebral interstitial acidosis (Andersen et al, 1989), or nitric oxide (Koedel et al, 1995). An alternative pathway could include the astrocytic Ca2+-triggered vasodilation and increase of CBF via the cyclooxygenase pathway of arachidonic acid metabolism (Paul et al, 2000; Takano et al, 2006). Nitric oxide production is markedly upregulated and plays a major role in systemic vasodilation and shock during sepsis (Petros et al, 1994; Watson et al, 2004); however, whether cerebral vasodilation occurs as well, and whether nitric oxide plays a role in this regard, is controversial. Animal studies appear to indicate that CBF is reduced but that autoregulation is preserved. Endotoxin administration increased the cerebrovascular resistance and reduced CBF in dogs (Ekstrom-Jodal et al, 1982a). In a fluid-resuscitated pig model of endotoxic shock, CBF and MAP were both reduced after endotoxin administration. An increase in MAP from 50 to 75 mm Hg by dopamine, phenylephrine, or norepinephrine failed to increase global CBF (Breslow et al, 1987), although cerebellar blood flow did increase (Miller et al, 1987); moreover, when MAP was reduced to 50 mm Hg by superimposed hemorrhage during continued sympathomimetic infusion, CBF did not change significantly (Miller et al, 1987). Thus, in this model, CBF autoregulation appeared to be intact even at relatively low MAP values. In a sheep model of sepsis, treatment with NG-monomethyl-
In conclusion, rats inoculated intravenously with pneumococci exhibited an increase in the lower limit of autoregulation compared with healthy control rats. Moreover, rats inoculated intracisternally to develop pneumococcal meningitis developed loss of autoregulation with a slope of the CBF/CPP curve that was steeper if secondary bacteremia was present than if this bacteremia was cleared by antibody treatment. These results indirectly suggest a cerebral vasodilatory action of bacteremia.
