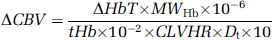Abstract
Near-infrared spectroscopy combined with partial jugular venous occlusion (JVO) offers promise for determining cerebral venous saturation (CSvO2) in sick preterm infants, but has not been validated in the newborn brain or under conditions of hypoxaemia. We assessed the accuracy of the CSvO2 estimate using cerebral venous oxygen saturation in superior sagittal sinus blood (SSSO2) as the ‘gold standard’. Comparisons were made in seven newborn lambs over a wide range of arterial oxygen saturations (SaO2) of 20% to 100%. Overall, median (range) CSvO2 was 49.8% (10.6% to 88.5%), whereas SSSO2 was 45.5% (4.3% to 76.6%); Bland—Altman analysis revealed a mean difference (CSvO2—SSSO2) of 5.1% and limits of agreement of ±27.4%. The change in cerebral blood volume (ΔCBV) induced by JVO increased with SaO2 (P <0.05). In addition, the strength of the correlation of CSvO2 with SSSO2 progressively improved with increasing change in total haemoglobin concentration (ΔHbT) induced by JVO. With Bland—Altman analysis repeated for data with ΔHbT >30 μmol cm, the mean difference (CSvO2—SSSO2) decreased to 2.4% with limits of agreement of ±18.8%. We conclude that the accuracy of estimating CSvO2 varies with the ΔCBV induced by JVO. Potential differences of optical properties between the head of the lamb and the human infant suggest that caution be exercised in directly applying these data to the human newborn. Nevertheless, this critical aspect of the JVO technique needs to be taken into consideration in developing an accurate measurement for sick preterm human infants.
Keywords
Introduction
Abnormal cerebral haemodynamics and oxygen delivery are major aetiologic factors for cerebral injury of sick preterm infants (Meek et al, 1999b; Tsuji et al, 2000), and new methods for assessment of cerebral circulation and oxygenation are needed to aid their clinical management and improve their outcomes. Continuous wave near-infrared spectroscopy (NIRS) has been used for several assessments of cerebral circulatory function in preterm infants including cerebral blood flow (CBF), cerebral blood volume (CBV), and cerebral autoregulation (Hintz, 2001; Owen-Reece et al, 1999; Volpe, 2001c). Despite its promise, the difficulty of applying continuous wave NIRS to determine CBF and CBV renders it of limited use for routine bedside investigations. For example, CBF and CBV measurements require manipulation of arterial oxygenation, and cannot be performed in babies with severe respiratory disease or in babies who are not oxygen-dependent. However, several other NIRS-based assessments of cerebrovascular oxygenation offer great clinical promise. Estimation of cerebral venous saturation (CSvO2), a measure which is sensitive to cerebral oxygen delivery, can be obtained using NIRS by inducing small changes in CBV by head-tilting (Skov et al, 1993; Wyatt et al, 1986), jugular venous occlusion (JVO) (Yoxall et al, 1995), and making use of ventilator-induced changes in CBV (Wolf et al, 1997).
Among these methods, JVO used in combination with NIRS (Yoxall et al, 1995) is a simple and easily applicable technique. The JVO technique has been applied in several studies of term (Buchvald et al, 1999) and preterm infants (Kissack et al, 2004a, b, 2005; Victor et al, 2006; Wardle et al, 2000; Yoxall and Weindling, 1998), after it was given limited validation in children (Yoxall et al, 1995). In this single validation study (Yoxall et al, 1995), CSvO2 was contrasted with jugular venous oxygen saturation obtained during cardiac catheterization. However, as jugular venous blood includes contributions from extracerebral tissue, it may not accurately represent cerebral venous blood.
The lamb has been widely used in NIRS studies as a surrogate model for the human newborn brain (Volpe, 2001a), and NIRS-derived values of CBV (mL/100 g tissue) in the lamb are essentially identical to those in the human neonate (Barfield et al, 1999; Wyatt et al, 1990) and identical to the value derived from the gold-standard radiolabel technique (Cr51 red blood cells, Barfield et al, 1999). Using the newborn lamb model, we aimed to validate the estimate of CSvO2 obtained using NIRS in combination with JVO. Validation in a newborn model is needed as critical features of cerebral oxygen metabolism change significantly between birth and childhood. In the newborn, CBF and cerebral oxygen delivery are relatively low, whereas cerebral metabolic rate for oxygen is high, (Jones et al, 1982; Richardson et al, 1989) and CSvO2 is likely to be lower than the mature brain. Possibly, estimation of low venous saturation levels by the NIRS-JVO technique may be problematic in the immature brain, and the applicability of the technique at this age requires evaluation.
Our validation compared CSvO2 estimates with cerebral venous oxygen saturations measured in blood samples taken from the superior sagittal sinus (SSSO2) as the ‘gold standard’. In addition, we assessed the correspondence of CSvO2 to SSSO2 over a range of hypoxaemic conditions that predispose to cerebral injury.
Methods
Animal Preparation
Seven newborn lambs of Merino-Border Leicester cross were studied at 1 to 3 days after birth. All procedures were performed in accordance with guidelines established by the National Health and Medical Research Council of Australia, and were approved by the Ethics in Animal Experimentation Committee of Monash University. A nonocclusive intravenous catheter (Intracath 19 GA; Becton Dickinson, Sandy, UT, USA) was inserted into the left jugular vein of the newborn lamb for administration of maintenance fluid and anaesthetic medication. Newborn lambs were intubated and ventilated under general anaesthesia (100 mg/kg α-chloralose and 5 mg/kg ketamine hydrochloride for induction, followed by 25 to 50 mg/kg/h α-chloralose for maintenance). An intravenous catheter (Insyte-N 24 G, Becton Dickinson) filled with a saline-heparin solution (50 IU/mL) was inserted in the right axillary artery to enable monitoring of arterial pressure and extraction of arterial blood samples. For cerebral venous blood sampling, a similar catheter (Insyte-N 24 G, Becton Dickinson) was inserted into the superior sagittal sinus through a small hole (2 mm in diameter) drilled in the skull along the sagittal suture just anterior to the lamboid suture.
Physiological Measurements
Beat-to-beat arterial oxygen saturation (SpO2) was measured with a pulse oximeter placed on the lamb's tongue (Nellcor 200; Nellcor Inc., Pleasanton, CA, USA). Arterial blood pressure was measured with a calibrated strain-gauge pressure transducer (Cobe CDX III; Cobe Laboratories, Lakewood, CO, USA) connected to a bridge amplifier (Quad Bridge Amp; ADInstruments, Sydney, Australia). Heart rate was derived from the arterial pressure pulse (Chart v4.2, ADInstruments). The signals were sampled at 400 Hz and displayed throughout the study (PowerLab/16R SP, ADInstruments).
Near-infrared Spectroscopy Measurement of Cerebral Venous Saturation
Optodes (NIRO-500; Hamamatsu Photonics K.K., Hamamatsu City, Japan) were positioned 2.5 cm apart on the right parietal-occipital area, and covered with a lightproof dressing. NIRS measurements of changes in concentrations (μmol cm) of oxyhaemoglobin (μHbO), deoxyhaemoglobin (ΔHb), and total haemoglobin (ΔHbT = ΔHbO + ΔHb) were recorded at 0.5 sec intervals. Partial jugular venous occlusions (JVOs) were performed by investigator FW by applying gentle pressure to the lamb's neck over the right jugular vein for 20 secs. Occlusions induced a small and temporary increase in HbO and HbT (Figure 1A) measured by NIRS. Cerebral venous saturation was calculated off-line using Microsoft Excel from the NIRS data obtained over the first 5 secs of the rise in HbT and HbO (Yoxall et al, 1995) using the relationship CSvO2 = AHbO/ΔHbT.
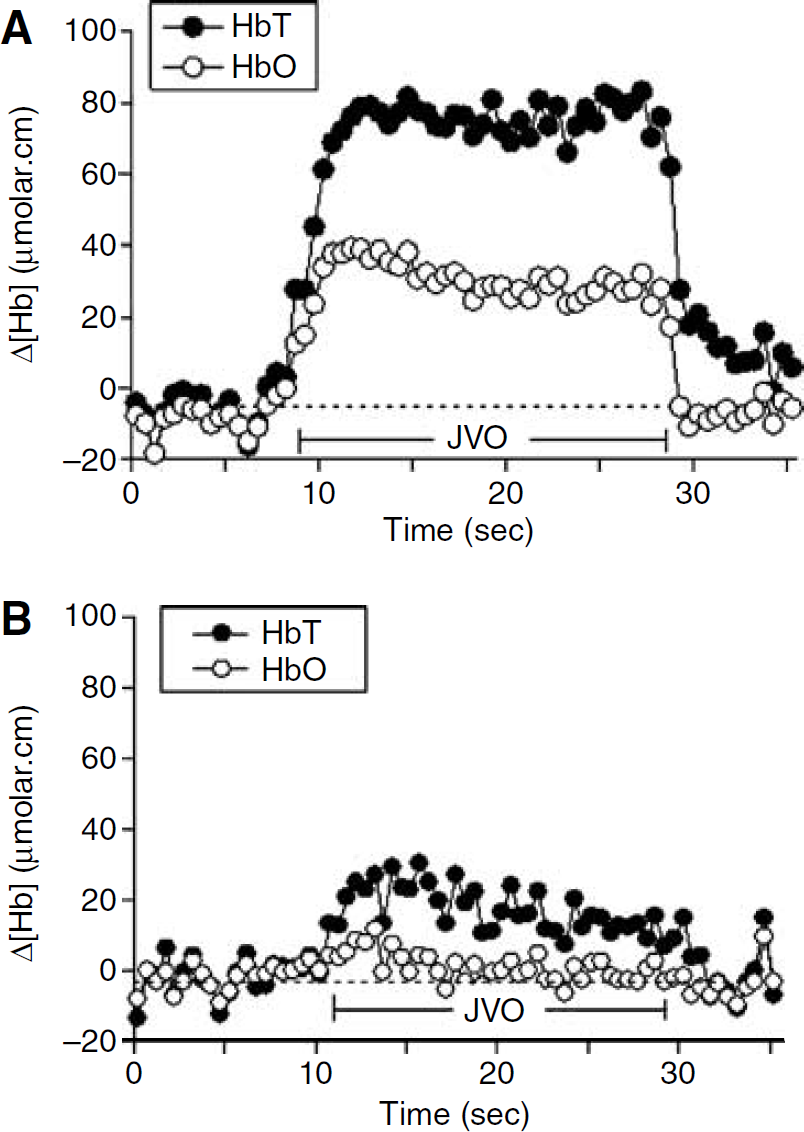
Changes in cerebral HbT and HbO induced using JVO in (
Data were accepted as suitable for the calculation of CSvO2 if there was a stable baseline before the occlusion, a rise in the concentration of both HbT and HbO during the occlusion, and a return to the baseline after release of the occlusion.
Blood Gas Analysis
After each JVO, blood samples of 500 μL were collected from superior sagittal sinus and the axillary artery in heparinised plastic syringes and analysed immediately for HbO saturation (SSSO2 and arterial oxygen saturation (SaO2), respectively; OSM2 Hemoximeter; Radiometer, Copenhagen, Denmark).
Induced Hypoxia
JVO was performed at least three times while SaO2 was ≥95%, then over a range of arterial hypoxaemia (SaO2 of 20% to 94%) induced by ventilating the lamb using 10% to 40% inspired oxygen, balance nitrogen. Arterial oxygen saturation was reduced by ∼10% at each step, monitored with pulse oximetry measurements (SpO2) and confirmed with arterial blood gas analysis. After SpO2 was held stable for 15 mins at each step, JVO was performed, and blood was sampled from the superior sagittal sinus for measurement of SSSO2 (Hemoximeter OSM2, Radiometer).
Change in Cerebral Blood Volume during Jugular Venous Occlusion
A differential pathlength factor of 4.99 was used for conversion of NIRS measurements into μmol/L (Delpy et al, 1988; Duncan et al, 1995). Change in CBV (mL/100 g) was calculated from NIRS measurement of the maximum ΔHbT (μmol/L) during the JVO, using the following formula (Elwell, 1995):
where MWHb = molecular weight of haemoglobin = 64,500, tHb = concentration of haemoglobin in large vessels in g/100 mL, CLVHR = cerebral to large vessel haemotocrit ratio = 0.69, and Dt = brain tissue density in g/mL = 1.05.
Statistics
Measurements of CSvO2 by NIRS-JVO were compared with the corresponding SSSO2 measurements using Pearson linear regression analysis (SigmaStat; SPSS Inc., Chicago, IL, USA). To examine the accuracy of the NIRS-JVO technique with respect to the extent of ΔCSV induced by JVO, the coefficient of determination (R2) was calculated for different levels of ΔHbT. An analysis of agreement of CSvO2 and SSSO2 was based on the method described by Bland and Altman (1986). Measurements of ΔCRV and ΔHbT induced by JVO during normoxaemia and hypoxaemia were compared using the Mann—Whitney U test. In all statistical tests, P <0.05 was considered significant.
Results
Near-infrared spectroscopy data collected during 95 JVOs met selection criteria for the calculation of CSvO2. Fifty-two occlusions were performed when the lambs were normoxaemic with SaO2 ≥95%, whereas 43 occlusions were performed during induced hypoxemia when SaO2 was 20% to 94%. Over all tests, the median (range) of CSvO2 was 49.8% (10.6% to 88.5%), whereas SSSO2 was 45.5% (4.3% to 76.6%). The mean difference was 5.1% and limits of agreement (2 standard deviations) were ± 27.4% (Bland and Altman, 1986).
The NIRS-determined magnitudes of ΔHbT and ΔHbO generated using JVO during hypoxia tend to be smaller than those that could be generated during normoxaemia (Figure 1). Similarly, estimates of ΔCRV generated by JVO during hypoxia was smaller than that during normoxaemia (0.52 (0.37 to 0.58) mL/100 g versus 0.58 (0.45 to 0.68) mL/100 g, P <0.05).
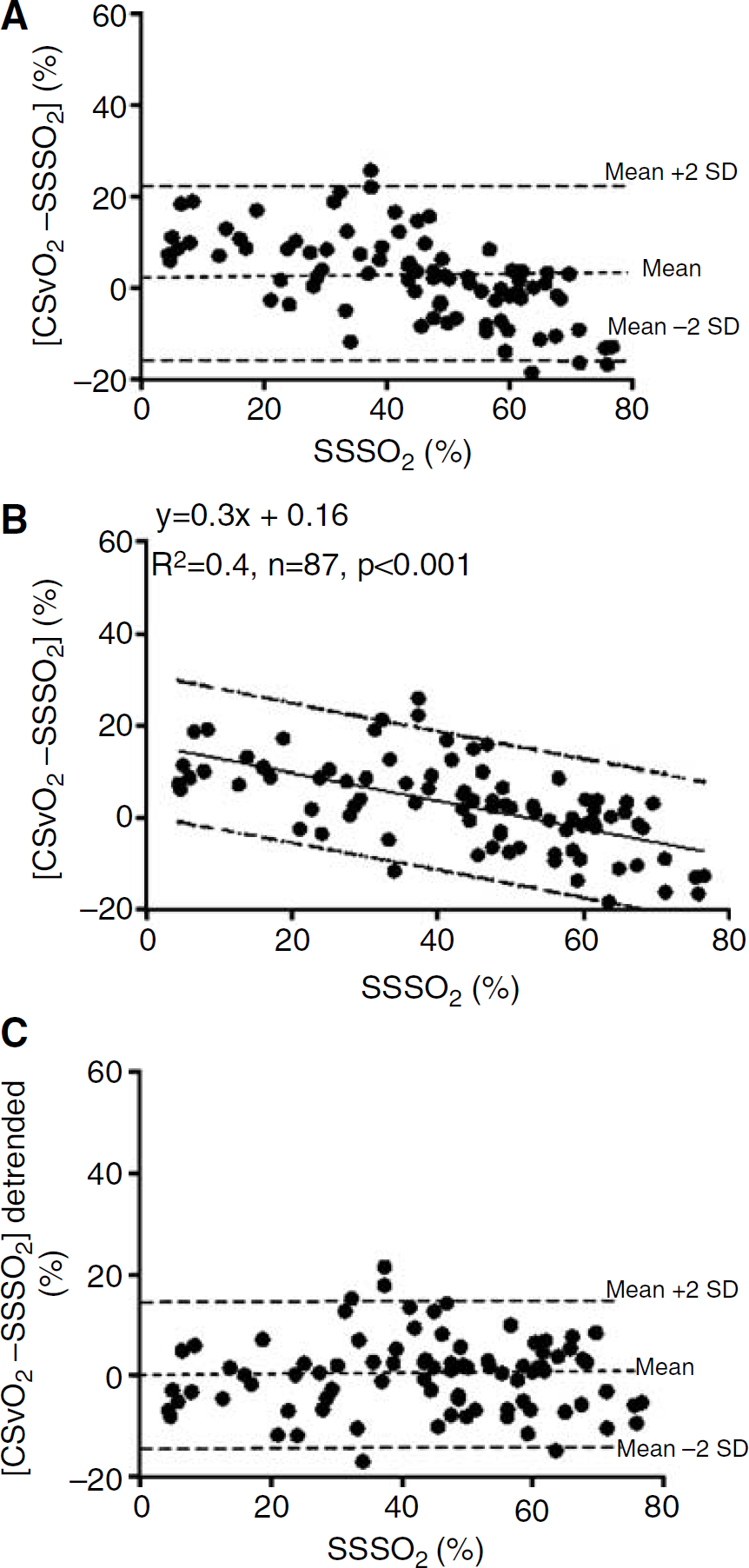
Values of R2 for the regression of CSvO2 on SSSO2 were dependent on the extent of ΔHbT (μmol cm) during JVO (Figure 2). R2 equalled 0.51 when all data were included in the analysis, increased as data with smaller ΔHbT were excluded, and then plateaued for ΔHbT more than 30 μmol cm. Figure 3 illustrates the significant correlation between CSvO2 and SSSO2 (R2 = 0.76, P <0.0001) for the 87 occlusions with ΔHbT > 30 μmol cm.
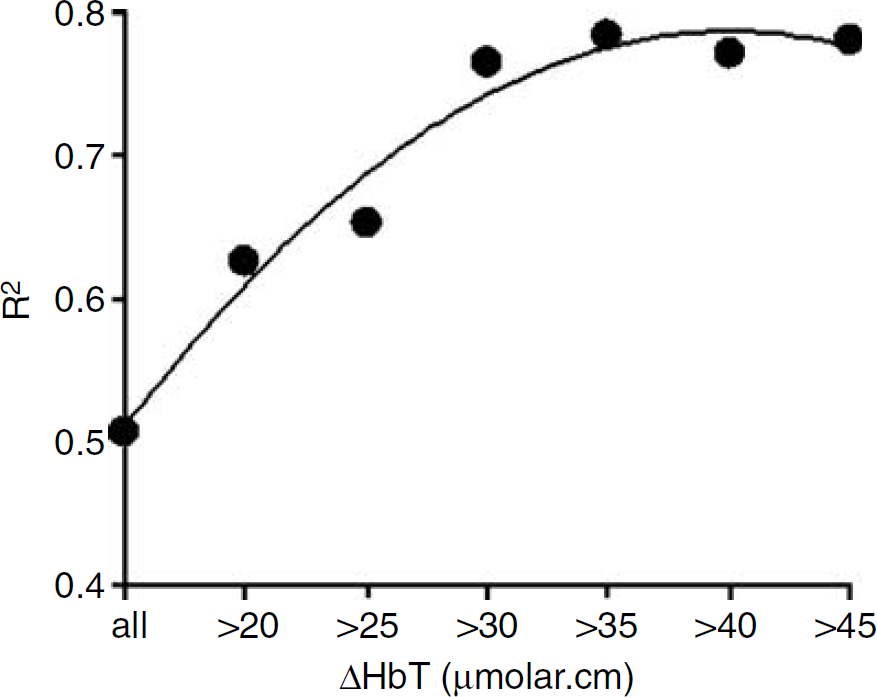
The degree of correlation between CSvO2 on SSSO2 was affected by the degree of ΔHbT induced by JVO. Stronger correlation (greater R2) was obtained with larger ΔHbT, with a plateau reached for ΔHbT >30 μmol cm.
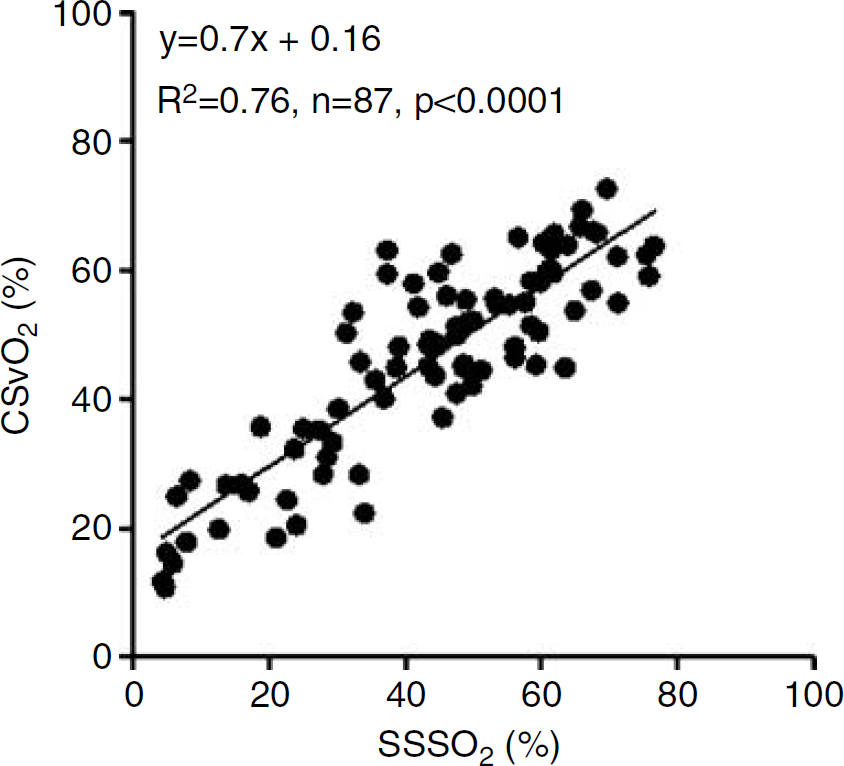
Correlation between CSvO2 and SSSO2 for JVO-induced ΔHbT >30 μmol cm.
The Bland-Altman analysis of CSvO2 versus SSSO2 for data with ΔHbT > 30 μmol cm is shown in Figure 4A. The mean difference (CSvO2—SSSO2) was 2.4% with limits of agreement (2 standard deviations) of ±18.8%. The CSvO2—SSSO2 difference varied with the level of SSSO2, with CSvO2 slightly higher than SSSO2 at low values of oxygen saturation, and slightly lower at high values of oxygen saturation (Figure 4B). The mean difference (limits of agreement) was 9.6% (−5.5% to 24.7%) at SSSO2 of 20%, 3.6% (−11.4% to 18.6%) at SSSO2 of 40%, and −2.4% (−17.5% to 12.6%) at SSSO2 of 60%. Figure 4C represents CSvO2 data detrended by subtracting values calculated using the regression equation of Figure 4B. After this correction, the limits of agreement around the mean difference of 0 were ±14.8.
Discussion
Near-infrared spectroscopy in combination with partial JVO has been used to estimate CSvO2 in human subjects in several studies, principally premature newborns who are at risk of cerebral hypoxia (Kissack et al, 2004a, b , 2005; Wardle et al, 2000; Yoxall and Weindling, 1998). Despite its wide application, the prior validation of the method consisted of a single study in children where CSvO2 was compared with oxygen saturation of jugular venous blood (Yoxall et al, 1995). Our study is the first validation of the technique against oxygen saturation in the cerebral venous outflow. Our principal finding is the confirmation that CSvO2 corresponds closely to sagittal sinus saturation (SSSO2) provided that the magnitude of ΔHbT induced by JVO is adequate.
Our data clearly show that correlation between CSvO2 and SSSO2 progressively improved with increases in ΔHbT, until the increase in ΔHbT reached ∼30 μmol cm (Figure 2). With this degree of HbT increment, the agreement of CSvO2 with SSSO2 approximated the limits of agreement reported in the previous validation study (Yoxall et al, 1995). At lower ΔHbT, the relatively greater noise-to-signal ratio in the NIRS measurements, evident in Figure 1B, may cause less precision in the CSvO2 estimate and poorer agreement with SSSO2.
During conditions of severe hypoxaemia, the estimated increment in CBV with the JVO manoeuvre was smaller than that during normoxaemia. This may be explained by the nonlinear form of the volume-pressure relationship for the cerebral vascular compartment (Folkow and Neil, 1971). Under conditions of cerebral hypoxaemia, the cerebral circulation would be widely dilated and the cerebral venous compartment likely to be highly distended (Bennet et al, 1998; Lassen, 1966; Livera et al, 1991). When highly distended, the compliance of veins would be low and JVO would induce little increment in CBV. Small CBV increments may result in small ΔHbT, especially if the blood haematocrit is low. As our findings suggest, ΔHbT <30 μmol cm may compromise the applicability of the NIRS-JVO technique.
It is unlikely that infants being ventilated would have cerebral hypoxaemia to the extent induced in our experimental manipulation. However, hypercapnia (Wyatt et al, 1991), or hypotension (Ferrari et al, 1992), or vasoparalysis due to hypoxicischaemic injury (Meek et al, 1999a; Wyatt et al, 1990), all common conditions in sick preterm infants, could increase baseline CBV and limit the CBV increment produced by JVO. In published studies in sick preterm infants (Kissack et al, 2004a, b , 2005; Wardle et al, 2000; Yoxall and Weindling, 1998), the change of CBV or HbT induced by JVO was not reported, so the accuracy of previous CSvO2 estimates is uncertain. The estimated increase in CBV in the validation study (Yoxall et al, 1995) was in the order of 0.25 to 0.33 mL/100 g, equivalent to ΔHbT of 42 to 55 μmol cm assuming a haemoglobin concentration of 12 g/100mL. On the basis of our findings, this ΔHbT should have ensured accurate estimate of CSvO2 in that study. The safety of manipulating the cerebral circulation in infants at risk for intracranial haemorrhage merits careful consideration (Volpe, 2001b). Spontaneous fluctuations in CBV similar in magnitude to those induced by JVOs were observed in ventilated and nonventilated preterm infants (Yoxall et al, 1995), suggesting that an adequate CBV can be safely incremented using JVO without risk of intracranial haemorrhage.
We have identified a systematic variation in the difference between CSvO2 and SSSO2 that is related to the level of cerebral venous oxygen saturation (Figure 4B). CSvO2 slightly overestimated SSSO2 at low oxygen saturations (<20%), and underestimated SSSO2 at high oxygen saturations (>70%). We propose that this finding arises from inhomogeneity of blood flow and oxygenation within the region of tissue illuminated by NIRS, and their variation with arterial oxygen saturation. At extremely low levels of oxygen saturation, overestimation of SSSO2 by the NIRS-JVO technique may be explained by recruitment of vessels that are upstream of the venous compartment and that contain blood of relatively higher oxygen saturation. Wide vasodilatation and high CBF, which can be anticipated under the conditions of severe hypoxaemia, may amplify the increment in intravascular pressure arising from JVO. This, combined with lower compliance of the widely dilated venous compartment, may result in pressure arising from JVO distending capillary or small arterial vessels in addition to cerebral veins. With inclusion of ΔHbO and ΔHbT arising from the upstream compartment, CSvO2 estimate would exceed SSSO2. At the high end of cerebral oxygenation, underestimation of CSvO2 by the NIRS-JVO measurement (Figure 4B) may reflect greater contribution of white matter to the NIRS signal. As previously shown, CBF in most regions of the newborn lamb's brain decreases with increase in arterial oxygenation, except in the white matter (Szymonowicz et al, 1988). Should higher cerebral oxygenation reduce CBV in the cortex, the vasculature of the white matter may become more prominently represented in the NIRS measurement. As the white matter is mostly drained by the deep cerebral venous system, venous saturation of the sagittal sinus blood which principally drains the cortex (Grant et al, 1995) may not fully represent the venous oxygenation detected by the NIRS.
Importantly, we have validated CSvO2 estimate against cerebral venous oxygen saturation, using SSSO2 as the gold standard. When corrected for the small systematic variation across the SSSO2 range, the limits of agreement improved to ±14.6% (Figure 4C), almost identical to that reported previously (Yoxall et al, 1995). The close correspondence in the previous validation study (Yoxall et al, 1995) may be explained by comparable contributions of extracerebral tissue to both the jugular venous blood and to the NIRS measurement. In our study, close agreement may have occurred because extracerebral contamination of the NIRS measurement was limited. The overlying skin and skull tissue of the newborn lamb is thin (<5mm), whereas the light penetration by NIRS is approximately 1.6 cm with the interoptode distance of 2.5 cm (Brown et al, 2002).
Whether these results can be transferred directly to the human newborn is uncertain, as differences in geometry of the head of the human infant and lamb may lead to optical property differences affecting NIRS measurements. The propagation of light by NIRS in heterogeneous tissue is theoretically complex and is affected by the geometry and thickness of the layered structures such as skin, skull, cerebrospinal fluid, and grey and white matter (Boas et al, 2001; Okada and Delpy, 2003a, b ). Hence, the possibility exists for differences in NIRS measurements, and in CSvO2 estimates between the two species. Moreover, optical properties (e.g., light scattering and absorption coefficients) have not been studied in the lamb brain, and at comparable gestational age, the lamb brain is more mature in the degree of myelination (Mallard et al, 1998; Raju, 1992). However, gross cortical anatomy is similar, as cortical thickness is 1.66 mm in the term lamb, compares closely with human infant values of 1.4 mm at 30 weeks gestation and 2 mm at term (Marin-Padilla, 1970; Rees et al, 1988). In any event, in practical terms the effect of superficial tissues on NIR light propagation is low in the neonatal head compared with the adult, because of the low scattering coefficient of the neonatal brain, the thin cerebrospinal fluid layer, and the small cranial diameter (Fukui et al, 2003; Okada, 2000).
A major technical advantage of measuring CSvO2 with the NIRS-JVO method is that, being a ratio of two concentrations, the measurement is free of the potential errors introduced by uncertainty of the differential pathlength factor (Owen-Reece et al, 1999). A practical advantage of the NIRS-JVO method is the simplicity and ease of application. By estimating venous oxygen saturation, the measurement offers important information on the balance of cerebral oxygen transport and oxygen utilisation, in addition to the cerebral haemodynamic parameters that can be measured by NIRS (Edwards et al, 1988; Wyatt et al, 1990, 1986). Our validation lends further support to its application in sick preterm infants.
Conclusion
Near-infrared spectroscopy in combination with JVO produces reliable estimates of CSvO2, provided there is adequate change in cerebral venous blood volume induced by the JVO manoeuvre. Potential differences of optical properties between the head of the lamb and the human infant suggest that caution be exercised in directly applying these data to the human newborn. Nevertheless, this critical aspect of the NIRS-JVO technique needs to be taken into consideration in clinical applications.
Footnotes
Acknowledgements
We acknowledge financial support from the National Health and Medical Research Council of Australia.