Abstract
The glutamate—glutamine cycle is thought to be of paramount importance in the mature brain; however, its significance is likely to vary with regional differences in distance between astrocyte and synapse. The present study is aimed at evaluating the role of this cycle in cultures of cerebellar neurons, mainly consisting of glutamatergic granule cells. Cells were incubated in medium containing [U-13C]glutamate or [U-13C]glutamine in the presence and absence of unlabeled glutamine and glutamate, respectively. Cell extracts and media were analyzed using high-performance liquid chromatography (HPLC) and gas chromatography combined with mass spectrometry (GC/MS). Both [U-13C]glutamate and [U-13C]glutamine were shown to be excellent precursors for synthesis of neuroactive amino acids and tricarboxylic acid (TCA) cycle intermediates. Labeling from [U-13C]glutamate was higher than that from [U-13C]glutamine in all metabolites measured. The presence of [U-13C]glutamate plus unlabeled glutamine in the experimental medium led to labeling very similar to that from [U-13C]glutamate alone. However, incubation in medium containing [U-13C]glutamine in the presence of unlabeled glutamate almost abolished labeling of metabolites. Thus, it could be shown that glutamate is the preferred substrate for intermediary metabolism in cerebellar neurons. Label distribution indicating TCA cycle activity showed more prominent cycling from [U-13C]glutamine than from [U-13C]glutamate. Labeling of succinate was lower than that of the other TCA cycle intermediates, indicating an active role of the γ-amino butyric acid shunt in these cultures. It can be concluded that the cerebellar neurons rely more on reuptake of glutamate than supply of glutamine from astrocytes for glutamate homeostasis.
Introduction
Glutamate is a multipurpose amino acid in the mature central nervous system. It is not only the major excitatory neurotransmitter, in addition it takes part in transamination and thus nitrogen homeostasis and is the precursor for other important molecules, including the main inhibitory neurotransmitter, γ-amino butyric acid (GABA). Although glutamate is ubiquitous in all parts of the central nervous system and present in large amounts in the brain, it is of critical importance that brain glutamate homeostasis is strictly controlled. The extracellular concentration of glutamate needs to be kept low, both to increase the signal-to-noise ratio in binding of transmitter substance in the synaptic cleft and to prevent excitotoxicity caused by excessive excitation of glutamate receptors and subsequent cell injury or death (for references, see Daikhin and Yudkoff, 2000). Rapid transport of glutamate from the synaptic cleft is performed through several types of specific transporter proteins, and uptake into astrocytes surrounding the synapse is believed to be more important than reuptake into the presynaptic neuron (Schousboe et al, 1977; Danbolt, 2001). Thus neurons experience a net loss of glutamate, which must be replenished by astrocytes because of the lack of the anaplerotic enzyme pyruvate carboxylase in neurons (Shank et al, 1985). This constitutes the basis for the pathway known as the glutamate—glutamine cycle (Berl and Clarke, 1983) in which neurotransmitter released from neurons is taken up by surrounding astrocytes, where it is converted to glutamine by the glial-specific enzyme glutamine synthetase (Norenberg and Martinez-Hernandez, 1979). Glutamine is not neuroactive and can move in the extracellular space without interfering with receptors, and is the most abundant amino acid in blood and cerebral spinal fluid with a concentration of ~0.5 mmol/L (Grill et al, 1992; White et al, 2004). Glutamine uptake into neurons is mediated by different general amino-acid transporters (Su et al, 1997; Dolinska et al, 2004). In neurons, glutamine can be converted into glutamate by the enzyme phosphate activated glutaminase and act as precursor for restoring the neurotransmitter pool, completing the glutamate—glutamine cycle. Indeed, several studies have confirmed the importance of glutamine as precursor for neurotransmitter glutamate (for a review, see Peng et al, 1993). As described, the cycle necessitates extensive interaction between neurons and astrocytes. This will, however, vary because of differences in the number of glial cells per neuron and also in respect to how closely the astrocytes envelop the synapses. Thus, it can be expected that the importance of the glutamate—glutamine cycle varies with location in the brain.
The present study is aimed at evaluating the role of the glutamate—glutamine cycle in cultured cerebellar neurons. It has been shown that after 7 days in vitro, these mainly glutamatergic cultures express glutamate and glutamine metabolizing enzymes analogous to the brain in vivo (Drejer et al, 1985). The cells were incubated in medium containing [U-13C]glutamate or [U-13C]glutamine in the presence and absence of unlabeled glutamine and glutamate, respectively. High-performance liquid chromatography (HPLC) and gas chromatography/mass spectrometry (GC/MS) analysis of cell extracts and media revealed that both [U-13C]glutamate and [U-13C]glutamine were excellent precursors for synthesis of neuroactive amino acids and tricarboxylic acid (TCA) cycle intermediates in these cultures. However, glutamate was shown to be the preferred substrate.
Materials and methods
Materials
NMRI mice were obtained from Taconic M&B (Copenhagen, Denmark). Plastic tissue culture dishes were purchased from Nunc A/S (Roskilde, Denmark) and fetal calf serum from Seralab Ltd. (Sussex, UK). Culture medium, glutamate receptor antagonists DNQX (6,7-dinitroquinoxa-line-2,3-dione) and D-AP5 (D-2-amino-5-phosphonopentanoic acid) were from Sigma Chemical Co. (St Louis, MO, USA). [U-13C]Glutamate and [U-13C]glutamine were from Cambridge Isotope Laboratories (Woburn, MA, USA), and the GC/MS derivatization reagent MTBSTFA (N-methyl-N-(tert-butyldimethylsilyl)trifluoroacetamide) +1% t-BDMS-Cl (tert-butyldimethylchlorosilane) was purchased from Regis Technologies, Inc. (Morton Grove, IL, USA). All other chemicals were of the purest grade available from regular commercial sources.
Cell Cultures
Cerebellar neurons were isolated and cultured from 7-day-old mice as described by Schousboe et al (1989). Briefly, tissue was trypsinized followed by trituration in a DNase solution containing a trypsin inhibitor from soybeans. Cells were suspended (2.5 × 106 cells/ml) in a modified Dulbecco's minimum essential medium containing 24.5 mmol/L KCl, 31 mmol/L glucose, 7 μmol/L p-aminobenzoic acid, 0.05 mmol/L kainic acid, and 10% (v/v) fetal calf serum, and seeded in poly-
Experiments Using [U-13C]Glutamate and [U-13C]Glutamine for Gas Chromatography/Mass Spectrometry Analysis
The culture medium was removed from 7-day-old cultures and the cells were incubated for 2 h at 37°C in 2 ml serum-free experimental medium (prepared without glutamine) containing 3 mmol/L glucose and either no additions (for HPLC analysis), [U-13C]glutamate (0.25 mmol/L), [U-13C] glutamine (0.50 mmol/L), [U-13C]glutamate (0.25 mmol/L) plus unlabelled glutamine (0.50 mmol/L) or [U-13C] glutamine (0.50 mmol/L) plus unlabelled glutamate (0.25 mmol/L). To avoid toxic effects of glutamate during the incubation period, two glutamate receptor antagonists DNQX (25 μmol/L), a selective antagonist at the α-amino-3-hydroxy-5-methyl-isoxazole-4-propionic acid (AMPA) and kainate receptor subtypes, and D-AP5 (100 μmol/L), an N-methyl-
High-Performance Liquid Chromatography
Amino acids in cell extracts and experimental media were quantified by HPLC on a Hewlett Packard 1100 system (Agilent Technologies, Palo Alto, CA, USA). The amino acids were precolumn derivatized with o-phthaldialdehyde (Geddes and Wood, 1984) and subsequently separated on a ZORBAX SB-C18 (4.6 × 250 mm, 5 μm) column from Agilent using a phosphate buffer (50 mmol/L, pH = 5.9) and a solution of methanol (98.75%) and tetrahydrofuran (1.25%) as eluents. The separated amino acids were detected with fluorescence and compared with a standard curve derived from standard solutions of amino acids run after every 12 samples.
Gas Chromatography/Mass Spectrometry
Lyophilized cell extracts were redissolved in HCL (10 mmol/L), adjusted to pH < 2 with 6 mol/L HCL, and dried under atmospheric air. The amino acids were extracted into an organic phase of ethanol and benzene and dried again under atmospheric air before derivatization with MTBSTFA in the presence of 1% t-BDMS-Cl (Mawhinney et al, 1986). The samples were analyzed on a Hewlett Packard 5890 Series II gas chromatograph linked to a Hewlett Packard 5972 Series mass spectrometer.
Data Analysis
Peaks from MS spectra were integrated, and atom percent excess (13C) of glutamate, glutamine, GABA, succinate, malate, aspartate, and citrate was determined after calibration using unlabeled standard solutions (Biemann, 1962). Results from HPLC quantification of amino acids in cell extracts were combined with values of atom percent excess obtained from GC/MS to give nmol/mg protein of different 13C labeled isotopomers of glutamate, aspartate, and GABA. Consumption of [U-13C]glutamate and [U-13C]glutamine was calculated by subtracting the remaining amounts of the two amino acids measured in the experimental medium from the amounts added followed by correction for the amount of cellular protein in the culture. Results are presented as means ± s.d. Differences between groups were analyzed statistically using one-way analysis of variance followed by the least significant difference post hoc test, and P < 0.05 was considered statistically significant.
Results
Cerebellar neurons were incubated in medium with different additions: no addition (control group), [U-13C]glutamate, [U-13C]glutamine, [U-13C]glutamate plus glutamine, and [U-13C]glutamine plus glutamate (see Materials and methods). Since HPLC analysis does not distinguish between isotopomers, the latter two groups were combined in Table 1, which shows the cellular content of selected amino acids in cerebellar neurons and the consumption of glutamate and glutamine. Compared with control, adding glutamate to the experimental medium led to increased intracellular levels of aspartate, glutamate, glutamine, and GABA. Cultures incubated with glutamine contained increased levels of GABA corresponding to the increase seen when glutamate was added. Aspartate and glutamate also increased as a response to glutamine addition, but not to the same extent as when glutamate was added. As expected, intracellular glutamine concentration also increased when glutamine was added to the medium. When both glutamate and glutamine were added to the experimental medium, aspartate, glutamate, and glutamine levels were increased more than when the two amino acids were added individually, whereas GABA content was increased to the same extent as when glutamate and glutamine were added alone. Quantification of glutamate and glutamine in the experimental media showed that glutamate consumption was much higher than glutamine consumption despite the medium concentration of glutamine being twice that of glutamate. Glutamate consumption was unaffected by the presence of glutamine, whereas the consumption of glutamine was reduced by nearly 50% when glutamate was added to the medium.
Cellular content of selected amino acids (nmol/mg protein) in extracts of cultured cerebellar neurons and consumption of glutamate and glutamine (μmol/mg protein/2 h) from the experimental medium
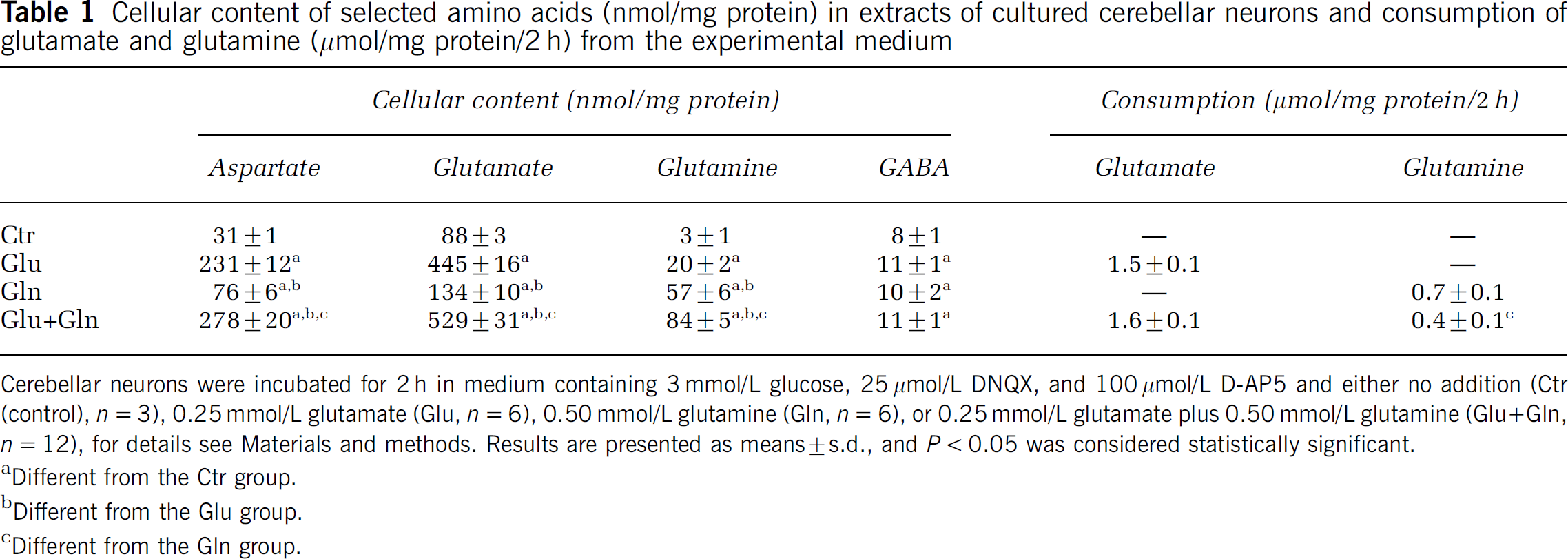
Cerebellar neurons were incubated for 2 h in medium containing 3 mmol/L glucose, 25 μmol/L DNQX, and 100 μmol/L D-AP5 and either no addition (Ctr (control), n = 3), 0.25 mmol/L glutamate (Glu, n = 6), 0.50 mmol/L glutamine (Gln, n = 6), or 0.25 mmol/L glutamate plus 0.50 mmol/L glutamine (Glu+Gln, n = 12), for details see Materials and methods. Results are presented as means ± s.d., and P < 0.05 was considered statistically significant.
Different from the Ctr group.
Different from the Glu group.
Different from the Gln group.
[U-13C]Glutamate or [U-13C]glutamine from the experimental medium enters neurons through specific transporter proteins. Once inside the cells, [U-13C]glutamine can be converted to [U-13C]glutamate, which can be decarboxylated to uniformly labeled GABA in GABAergic neurons, or in all cells, be turned into α-[U-13C]ketoglutarate and be metabolized in the TCA cycle for energy production and metabolite synthesis. The 13C label from the precursor's carbon skeleton will in the latter case be distributed among the TCA cycle metabolites and GABA as illustrated in Figure 1.
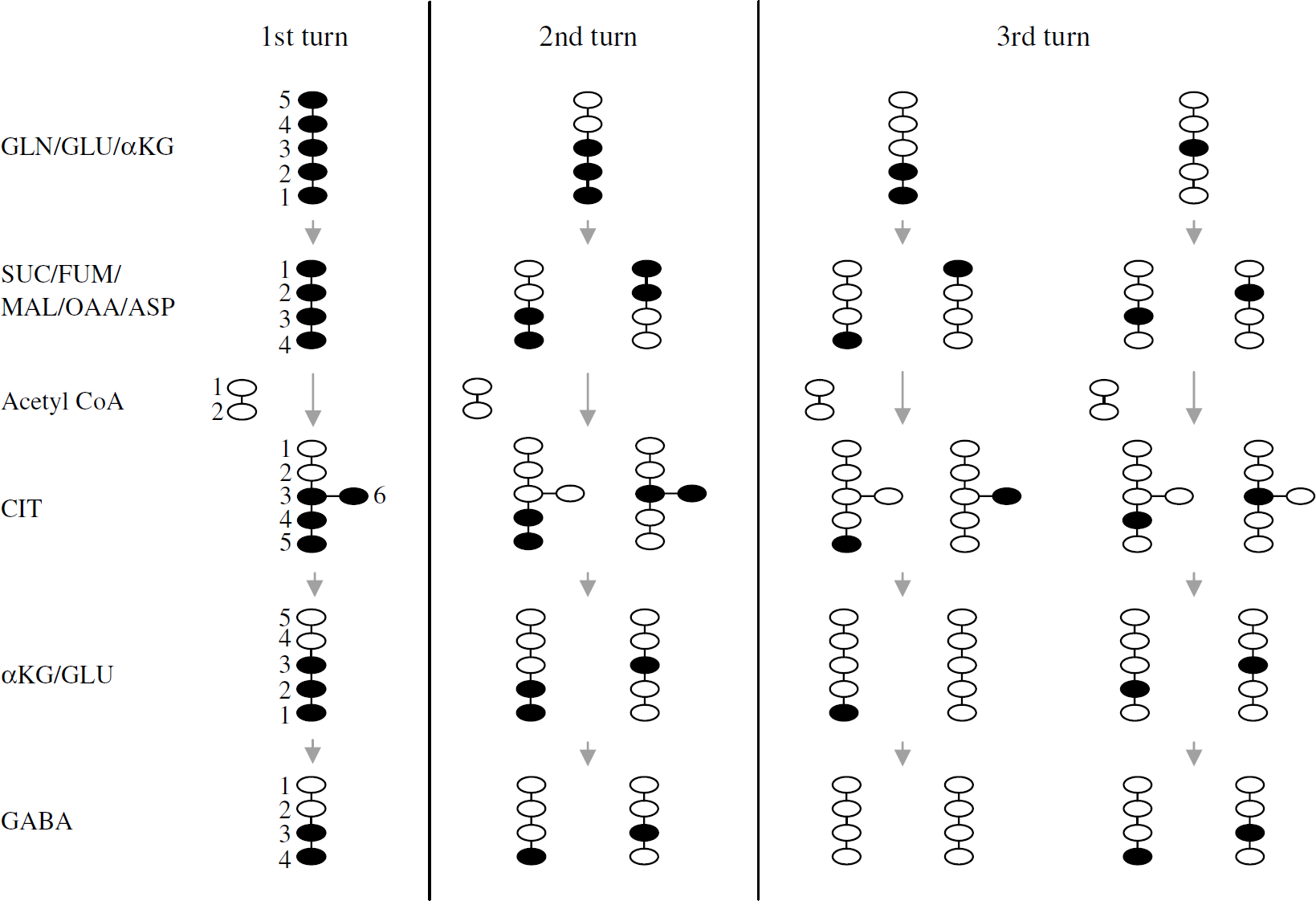
Schematic representation of possible isotopomers of various metabolites arising from [U-13C]glutamate or [U-13C]glutamine via the three first turns in the TCA cycle in neurons: • represents 13C and ○ represents 12C atoms; GLN: glutamine; GLU: glutamate; αKG: α-ketoglutarate; SUC: succinate; FUM: fumarate; MAL: malate; OAA: oxaloacetate; ASP: aspartate; CIT: citrate; GABA: γ-amino butyric acid.
The labeling of glutamate, GABA, succinate, malate, aspartate, and citrate as detected by GC/MS is presented in Figure 2 as atom percent excess (% labeling). When [U-13C]glutamate is turned into α-[U-13C]ketoglutarate and enters the TCA cycle, [1,2,3-13C]glutamate (M + 3) is formed after one turn. This isotopomer results from condensation of uniformly labeled oxaloacetate (OAA) with unlabeled acetyl-CoA forming [3,4,5,6-13C]citrate (Figure 1), which can be turned into [1,2,3,6-13C]iso-citrate, α-[1,2,3-13C]ketoglutarate and finally into [1,2,3-13C]glutamate. After another turn in the TCA cycle, half of the glutamate formed will be [1,2-13C]glutamate (M + 2) and half [3-13C]glutamate (M + 1). After a third turn, 25% of glutamate will be unlabeled, whereas 75% will be labeled either in the 1, 2, or 3 position (M + 1) (Figure 1).
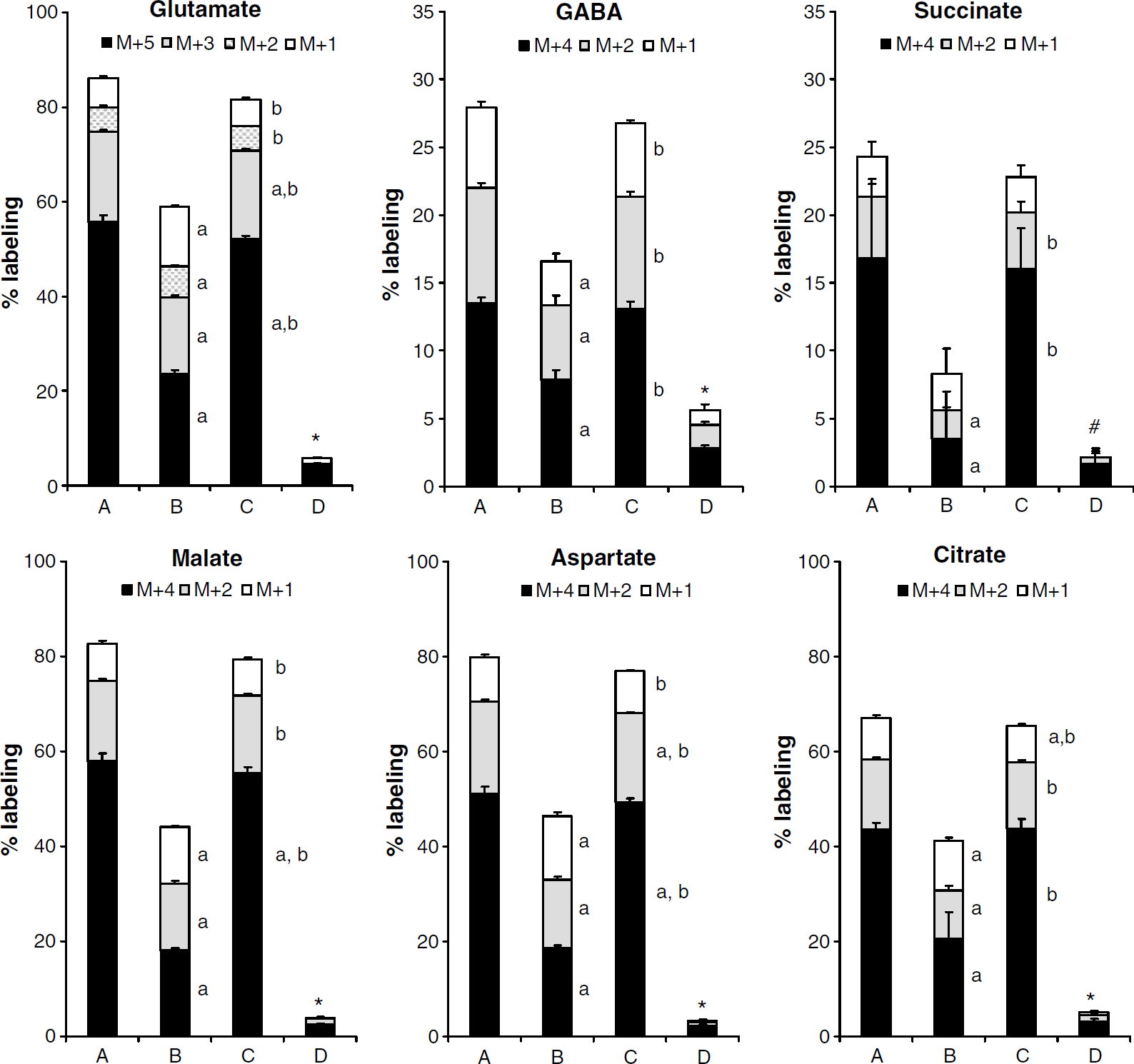
Percent labeling in glutamate, GABA, succinate, malate, aspartate and citrate as detected by GC/MS in cell extracts of cultured cerebellar neurons after incubation with [U-13C]glutamate (column A), [U-13C]glutamine (column B), [U-13C]glutamate plus unlabeled glutamine (column C) or [U-13C]glutamine plus unlabeled glutamate (column D), for details see Materials and methods. Results are presented as means + s.d. in atom percent excess, and P < 0.05 was considered statistically significant. *All masses are different from the corresponding masses in the other groups. #M + 4 is different from the groups incubated with [U-13C]glutamate with and without glutamine, the other masses are different from all other groups. aDifferent from the corresponding mass in the A group; bdifferent from the corresponding mass in the B group; cdifferent from the corresponding mass in the C group.
From [U-13C]glutamate entering the TCA cycle via α-[U-13C]ketoglutarate, uniformly labeled succinate, malate, and OAA are formed (Figure 1), and OAA can be transaminated into aspartate. All of these compounds have four C atoms, and their [U-13C] isoforms have the mass M + 4 and are presented in Figure 2. [U-13C]OAA can, as already mentioned, condense with unlabeled acetyl-CoA to form [3,4,5,6-13C]citrate with six carbon atoms, four of which are 13C (M + 4). Hence, the mass M + 4 represents the first turn in the TCA cycle for all these compounds. In the next turn, they will all contain two labeled C atoms and appear as M + 2, and in the third turn, they will contain one labeled C atom (M + 1) (Figure 1).
As shown in Figure 2, labeling from [U-13C]glutamate (column A) and [U-13C]glutamine (column B) was substantial, with glutamate giving the highest percent labeling in all metabolites analyzed. When [U-13C]glutamate and unlabeled glutamine were added to the experimental medium (column C), percent label decreased only slightly compared with when [U-13C]glutamate was added alone (column A). However, when [U-13C]glutamine and unlabeled glutamate were added to the medium, labeling was almost abolished for all metabolites (column D). Figure 2 shows that labeling of glutamate and malate, aspartate and citrate was high (>65% from [U-13C]glutamate and 40% to 60% from [U-13C]glutamine (in the absence of glutamate)), whereas that of succinate was much lower (< 30% labeling from glutamate and < 10% from glutamine). Although the percent labeling in succinate was lower, the pattern was the same as for the other TCA metabolites.
GABA can be formed from glutamate in GABA ergic neurons, which constitute about 6% of cerebellar neuronal cultures (Sonnewald et al, 2004). GABA formed directly from [U-13C]glutamate is uniformly labeled (M + 4). From [1,2,3-13C]glutamate (after one turn in the TCA cycle), [3,4-13C]GABA is formed (M + 2). Another turn in the TCA cycle for the glutamate carbon skeleton and subsequent formation of GABA will result in labeling in one C atom in GABA (M + 1), from both [1,2-13C] and [3-13C]glutamate, [4-13C] and [3-13C]GABA, respectively. The same two isotopomers are also formed after three turns in the TCA cycle (Figure 1). Figure 2 shows that the labeling of GABA analyzed by GC/MS was lower than that of glutamate, malate, aspartate, and citrate. Total labeling from [U-13C]glutamate was ~30% regardless of the presence of unlabeled glutamine. Significantly less (17%) GABA was labeled from [U-13C]glutamine alone, and this was further reduced in the presence of unlabeled glutamate (6%).
Intracellular amounts of glutamate, aspartate, and GABA as quantified by HPLC are shown in Figure 3. The amounts of glutamate and aspartate varied considerably with addition of glutamate or glutamine in the experimental medium, but followed the same pattern, whereas GABA concentration was independent of glutamate and glutamine content in medium. Results from mass spectrometry provide information about percent labeling, which combined with information about amounts of metabolites gives quantitative data. Amounts of different 13C labeled isotopomers of glutamate, aspartate, and GABA from the first two turns in the TCA cycle are shown in Figure 3. The intracellular amount of uniformly labeled glutamate was 248 nmol/mg protein after incubation with [U-13C]glutamate. This was reduced to 32 and 17 nmol/mg protein when glutamine was the labeled precursor, in the absence and presence of unlabeled glutamate, respectively. When labeled glutamate was added in the presence of unlabeled glutamine, the amount of intracellular uniformly labeled glutamate was, however, increased to 279 nmol/mg protein. The same trend was seen in [U-13C]aspartate, although the amounts were ~50% of those of [U-13C]glutamate. These two excitatory amino acids also showed similarities in the next turn in the TCA cycle. GABA had a much lower labeling from both labeled precursors than glutamate and aspartate, labeling from [U-13C]glutamine being lower than that from [U-13C]glutamate.
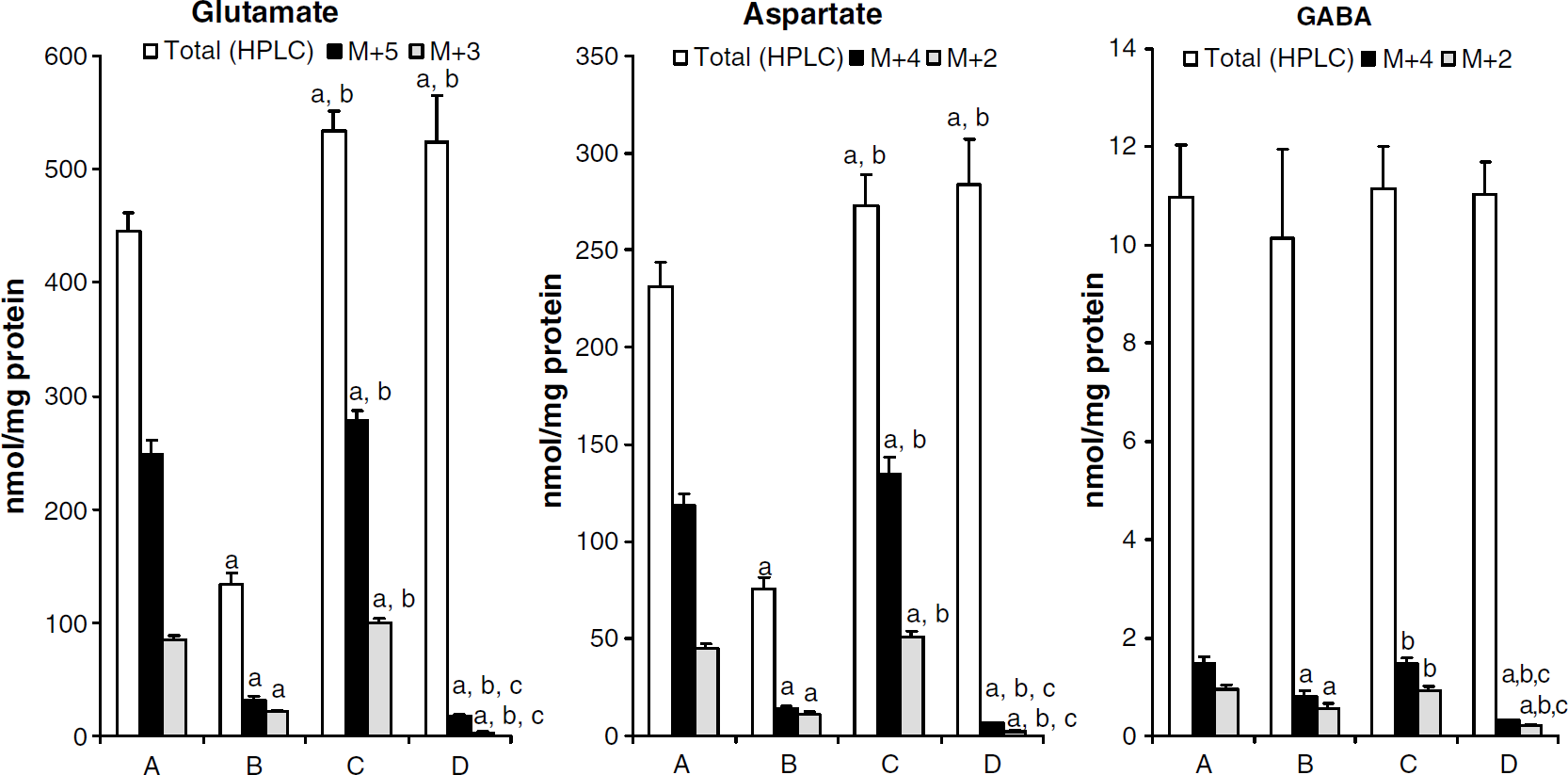
The amounts of glutamate, aspartate, and GABA (white bars), the amounts of M + 5 ([U-13C]glutamate), M + 4 ([U-13C]aspartate, [U-13C]GABA) (black bars) and the amounts of M + 3 ([1,2,3-13C]glutamate), M + 2 ([1,2-13C] and [3,4-13C]aspartate) and M + 2 ([3,4-13C]GABA) (grey bars) in cell extracts of cultured cerebellar neurons after incubation with [U-13C]glutamate (column A), [U-13C]glutamine (column B), [U-13C]glutamate plus unlabeled glutamine (column C), or [U-13C]glutamine plus unlabeled glutamate (column D), for details see Materials and methods. Results are presented as means + s.d. in nmol/mg protein, and P < 0.05 was considered statistically significant. aDifferent from the corresponding metabolite in the A group; bdifferent from the corresponding metabolite in the B group; cdifferent from the corresponding metabolite in the C group.
Discussion
Metabolism of glutamate and glutamine in the brain is closely connected via the glutamate—glutamine cycle, in which neurotransmitter glutamate taken up from the synaptic cleft by astrocytes is converted to glutamine and transported back to neurons as a precursor for the neurotransmitter pool (Berl and Clarke, 1983). Similar mechanisms operate in the glutamate—glutamine—GABA cycle (van den Berg and Garfinkel, 1971; Sonnewald et al, 1993b). However, reuptake into the presynaptic neuron is believed to be the preferred mechanism for removal of GABA from the synapse (Schousboe, 2003). To which extent glutamine and thus this cycle is important for cultured cerebellar neurons has been explored in the present study by comparing [U-13C]glutamate and [U-13C]glutamine as substrates for intermediary metabolism. In our model, addition of glutamine to the neuronal cultures mimics astrocyte—neuronal interactions in the brain, with the advantage of isolating neuronal from glial metabolism and thus making interpretation of results unambiguous.
[U-13C]Glutamate and [U-13C]Glutamine Metabolism
Glutamine has been shown to be an excellent precursor for the neurotransmitters glutamate and GABA, both in cultured cerebellar neurons (Waagepetersen et al, 2005), cortical neurons (Westergaard et al, 1995), and in freshly isolated cortical synaptosomes from rat brain (Yudkoff et al, 1989; Sonnewald and McKenna, 2002). Label from [U-13C]glutamine was also found in glutamate and GABA in the present study. Moreover, labeling of TCA cycle intermediates and aspartate was detected, showing that the carbon skeleton of [U-13C]glutamine entered the TCA cycle. The presence of isotopomers from subsequent turns also showed that the carbon skeleton stayed in the TCA cycle. This confirms that the glutamate—glutamine cycle does not operate in a stochiometric fashion (McKenna et al, 1993, 1994; Sonnewald et al, 1993a) and that glutamine is readily oxidized by cerebellar cultures for energy.
Exogenous glutamate has also been used to label neuronal metabolites. Westergaard et al (1995) showed that in cultured cortical neurons, which are predominantly GABAergic, incubation with [U-13C]glutamate gave high enrichment in aspartate in addition to labeling of GABA. Incubating cerebellar neurons in medium containing [U-13C]glutamate in the present study led not only to uniformly labeled intracellular aspartate and glutamate, but to some extent, the carbon skeleton also stayed in the TCA cycle for several turns. This is in accordance with similar studies of cerebellar neurons analyzed with magnetic resonance spectroscopy (Sonnewald et al, 1996; Santos et al, 2006). Using the more sensitive method mass spectrometry, extensive labeling of TCA cycle intermediates and GABA was also detected in the present study. Labeling of GABA in cerebellar cultures has earlier been shown by Qu et al (2000). The present study confirms that [U-13C]glutamate serves as an excellent precursor for intermediary metabolism in cultured cerebellar neurons.
As shown in the previous paragraphs, both [U-13C]glutamine and [U-13C]glutamate are well suited as precursors for neurotransmitter formation and substrates for neuronal intermediary metabolism. However, in the present study [U-13C]glutamate gave a higher percent labeling than [U-13C]glutamine in all metabolites, including GABA. Sonnewald and McKenna (2002) found that [U-13C]glutamate was superior in labeling of aspartate, whereas GABA labeling was only observed from [U-13C]glutamine, and not from [U-13C]glutamate when unlabeled glutamine was present, in cortical synaptosomes. Although [U-13C]glutamate was a better precursor for GABA than [U-13C]glutamine in the present study, glutamine was a relatively better precursor for GABA than for the other metabolites investigated, indicating that GABA synthesis occurs in a separate compartment (see below) consistent with findings in cortical synaptosomes (Sonnewald and McKenna, 2002). Westergaard et al (1995) showed that in primary cultures of cortical neurons labeling of aspartate, GABA, and [1,2,3-13C]glutamate was very similar from [U-13C]glutamine and [U-13C]glutamate. Thus, not surprisingly, GABAergic neurons in the cerebellum appear to have a different substrate preference for GABA synthesis than GABAergic neurons in cerebral cortex. In cultured cortical neurons, TCA cycle metabolism of the carbon skeleton subsequently used in formation of GABA was more pronounced from [U-13C]glutamine than from [U-13C]glutamate (Westergaard et al, 1995). This was also observed in the present study in cultured cerebellar neurons. With [U-13C]glutamate as the precursor, approximately 60% of the labeled succinate, malate, aspartate, and citrate was uniformly labeled and thus derived from the first turn, ~ 20% was from the second and ~10% from the third turn in the TCA cycle. When [U-13C]glutamine was present in the experimental medium, these numbers were ~40%, ~30%, and ~25%, respectively, and similar results were seen in GABA. Thus, cycling from glutamine was more prominent than that from glutamate. The fact that mitochondrial metabolism of the carbon skeleton of endogenous glutamate derived from glutamine and glutamate taken up into the cells differed could be due to differences in the distribution of the enzyme phosphate activated glutaminase catalyzing the formation of glutamate from glutamine. This compartmentation could perhaps be related to intracellular mitochondrial heterogeneity (Westergaard et al, 1995; Sonnewald et al, 1998). Interestingly, label distribution indicating TCA cycling of the carbon skeleton subsequently converted into GABA was similar whether [U-13C]glutamate or [U-13C]glutamine was the precursor present in the experimental medium, showing intercellular compartmentation in addition to the intracellular compartmentation mentioned above.
To evaluate the preference for glutamate and glutamine as substrates for intermediary metabolism in cultured brain cells or synaptosomes, both substrates have to be present in the medium simultaneously. When Sonnewald and McKenna (2002) incubated synaptosomes with [U-13C]glutamate in the presence of unlabeled glutamine, the carbon skeleton from [U-13C]glutamate entered the TCA cycle and labeled aspartate and [1,2,3-13C]glutamate, but not GABA. However, label from [U-13C]glutamine in the presence of unlabeled glutamate was incorporated into GABA, but not aspartate. This is consistent with what has been reported in cortex for the two precursors applied separately (Westergaard et al, 1995), showing that the compartmentation is maintained under more physiological conditions with both substrates present (Sonnewald and McKenna, 2002). The importance of glutamine as a precursor and thus the importance of the glutamate—glutamine—GABA cycle for GABA synthesis is surprising since reuptake of GABA into the presynapse is believed to be prominent (Schousboe, 2003). Neurotransmitter glutamate, however, is thought to be removed from the synaptic cleft mainly by uptake into astrocytes (Schousboe et al, 1977; Danbolt, 2001). On the basis of this, it can be assumed that the importance of the glutamate—glutamine cycle should be greater for the synthesis of neurotransmitter glutamate than for GABA. However, results from the present study suggest otherwise. Consumption of glutamate in the cerebellar neurons was twice that of glutamine when given alone and together with glutamine, even though the glutamine concentration in the medium was twice as high as that of glutamate. Most importantly, glutamine consumption was reduced by nearly 50% in the presence of glutamate compared with when glutamate was not present in the experimental medium. Thus, glutamate could substitute for glutamine, but the reverse was not the case. Labeling of intracellular metabolites was only slightly reduced when neurons were incubated in medium containing [U-13C]glutamate in the presence of unlabeled glutamine compared with that in the absence of glutamine. Surprisingly, when unlabeled glutamate was present together with [U-13C]glutamine, labeling was almost abolished in all metabolites measured. The carbon skeleton of [U-13C]glutamate and [U-13C]glutamine will, in the TCA cycle, not only be distributed into the metabolites mentioned above, but also be converted to 13CO2, which is not detected by the experimental setup used in the present study. As mentioned above, the carbon skeleton of glutamine stayed in the TCA cycle longer than that of glutamate and thus it is likely that a higher percentage of glutamine compared with that of glutamate was converted to 13CO2.
Considering the results presented, it can be postulated that the glutamate—glutamine cycle is of less importance for neurons in the cerebellum than what has been described for cerebral cortical neurons. This is further supported by the fact that granule neurons are by far the most abundant cells in the rat cerebellum (265 × 106) and outnumber glia (2.2 × 106) in the granule layer significantly (Korbo et al, 1993). A similar quantitative neuronal dominance has been reported in human cerebellum (Andersen et al, 1992). Thus, in the cerebellar cortex, glial processes do not envelop all synapses. Indeed, it has been shown that around synapses between the parallel fibers, the axons of granule neurons, and interneuron dendrites, astrocyte processes are lacking (Danbolt, 2001). Therefore, the distance from the synapse to the nearest glial cell is quite large and, in addition, the relative number of glutamate transporters on these astrocytes is low (Danbolt, 2001). However, it has been shown that in the cerebellar granule cell layer, the density of mRNA for a neuronal glutamate transporter was very high (Velaz-Faircloth et al, 1996). Considering the relatively large distance between synaptic regions of cerebellar granule cells and astrocytes and the high level of glutamate transporters on these neurons, it can be argued that reuptake of glutamate must be important for granule neurons. This has indeed been shown by Waagepetersen et al (2005), who reported that the intracellular pool of glutamate in cerebellar neurons was dependent on reuptake of extracellular glutamate.
The ‘Partial Tricarboxylic Acid Cycle‘
Battaglioli and Martin (1990) showed that in synaptosomes, aspartate synthesis was strongly stimulated by glutamate and glutamine, but the stimulation by glutamate was greatest. Similarly, in the present study, an increase in intracellular glutamate concentration was always accompanied by a corresponding increase in that of aspartate. The intracellular glutamate concentration was highest in the cells incubated with both glutamate and glutamine, followed by those incubated with glutamate alone, and thereafter cells incubated with glutamine, which also led to an increase compared with control. The coupling between glutamate and aspartate can be explained by the fact that cerebellar neurons have a high activity of glutamate dehydrogenase and aspartate amino transferase (Drejer et al, 1985; Westergaard et al, 1991; Zaganas et al, 2001). Thus, entry of glutamate via α-ketoglutarate into the TCA cycle and conversion of oxaloacetate to aspartate is very efficient in the cerebellum and energy is obtained from this so-called ‘partial TCA cycle’ (Hertz et al, 1991; Sonnewald and McKenna, 2002).
GABA Shunt in Cerebellar Neurons
The cerebellar neuronal cultures consist primarily of glutamatergic granule cells with a minor contribution of GABAergic stellate and basket neurons, that is, GABAergic as well as glutamatergic characteristics are expressed in these cultures (Pearce et al, 1981; Hertz et al, 1985; Hertz and Schousboe, 1987; Drejer and Schousboe, 1989; Kovacs et al, 2003). Thus, cultures of dissociated cerebellum constitute an excellent model system for the in vivo situation, in which the association of GABAergic with glutamatergic neurons can be investigated. The major enzyme responsible for GABA synthesis in brain, glutamate decarboxylase, was shown to be present in 6% of cultured cerebellar neurons (Sonnewald et al, 2004) indicating the presence of two distinct cell types and thus, at least two cellular compartments. In the present study, the amount of intracellular GABA in the cultures was increased to the same extent by all experimental conditions, compared with control cultures. This is in contrast to the results obtained for aspartate, which increased with increasing glutamate concentration, as mentioned above, showing cellular compartmentation. It has been shown that the glutamate decarboxylase positive neurons, which constitute the GABAergic compartment, produce GABA during the whole culture period and that GABA is distributed throughout the whole culture (Sonnewald et al, 2006). In the present study, no labeled substrates were present in the medium during the 7-day culture period, thus GABA produced during this time was unlabeled. When medium containing [U-13C]glutamate or [U-13C]glutamine was added to the cultures for the 2-h incubation period, labeled GABA could be formed only in the small number of GABAergic cells. Hence, most GABA in the cultures in general, and in the cerebellar granule neurons in particular, was unlabeled at the time of culture extraction.
GABA can be catabolized via the GABA shunt, in which the carbon skeleton of GABA enters the TCA cycle after conversion to succinate. Interestingly, percent labeling of succinate was very similar to that of GABA and much lower than that of the other TCA cycle intermediates measured. The number of GABAergic neurons in the cultures is small, and thus it seems unlikely that the succinate content in these neurons could account for the dilution of labeling in the total succinate pool. It can be assumed that catabolism of GABA and entry of the carbon skeleton of GABA into the TCA cycle through the GABA shunt is taking place in the glutamatergic cells from unlabeled GABA. Hence, a possible function of GABA in the glutamatergic neurons is the ability to produce a separate succinate pool for potential energy production in the TCA cycle, suggesting a special role for the GABA shunt in the cerebellum.
Conclusion
Through the results presented, it could be shown that glutamate is preferred over glutamine as a substrate for intermediary metabolism in cultured cerebellar neurons. It can be concluded that these neurons rely more on reuptake of glutamate than supply of glutamine from astrocytes for glutamate homeostasis.
Footnotes
Acknowledgements
We thank Bente Urfjell and Lars Evje for excellent technical assistance.
