Abstract
Differentiation of impaired gait seen in idiopathic normal pressure hydrocephalus (iNPH) from parkinsonian gait is sometimes a great challenge and important for future medication in the clinical setting. To investigate dopaminergic contribution to its pathophysiology, two aspects of the transsynaptic dopamine functions in the striatal region in eight iNPH patients naïve to dopaminergic drugs were examined using positron emission tomography with a presynaptic marker [11C]CFT ([11C]2-β-carbomethoxy-3β-(4-fluorophenyl) tropane) that binds to dopamine transporter and a postsynaptic marker [11C]raclopride that binds to D2 receptor. Quantitative values of binding potentials (BPs) for [11C]CFT and [11C]raclopride were compared between patients and eight age-matched healthy subjects. The BPs and magnetic resonance imaging-based morphometric measures in iNPH were used for correlation analyses between the magnitude of binding of these in vivo markers and clinical severity of the patients. Analysis of variance showed significant reduction in [11C]raclopride binding in the putamen and nucleus accumbens (P < 0.05, corrected for multiple comparison) and unchanged striatal [11C]CFT binding in iNPH. The dorsal putamen [11C]raclopride binding correlated negatively with gait severity (r = 0.720, P < 0.05), and the nucleus accumbens [11C]raclopride binding correlated positively with emotional recognition score (r = 0.727, P < 0.05) in the disease group. No significant relationship was observed between BPs and morphometric measures. The current result of the postsynaptic D2 receptor reduction along with preserved presynaptic activity in the nigrostriatal dopaminergic system reflects a pathophysiology of iNPH. Postsynaptic D2 receptor hypoactivity in the dorsal putamen may predict the severity of gait impairment in iNPH.
Introduction
Clinical features of normal pressure hydrocephalus (NPH) are gait disturbance, progressive dementia, and urinary incontinence, known as a clinical triad (Hakim and Adams, 1965). The NPH symptoms are thought to be a clinical entity that can be treated by surgical interventions such as shunting (Adams et al, 1965). The possible treatment of this disease led us to explore a surrogate marker for NPH that enables proper diagnosis at an early stage. Clinically, gait disturbance is likely the first sign and important symptom in NPH (Fisher, 1982). However, the hypokinetic type of gait disturbance is often seen in other neurologic diseases such as Parkinson's disease (PD) and dementia with extrapyramidal symptoms. Differentiating NPH from PD may be possible by observing improvement of gait velocity after withdrawal of cerebral spinal fluid (CSF) (Sudarsky and Simon, 1987) or after
Combination of a presynaptic dopamine transporter radiotracer, [11C]2-β-carbomethoxy-3β-(4-fluorophenyl) tropane ([11C]CFT), and a predominantly postsynaptic D2 receptor radiotracer, [11C]raclopride, in PET studies allows us to evaluate presynaptic and postsynaptic dopaminergic activities in the basal ganglia (Farde et al, 1986; Ouchi et al, 1999a). The in vivo findings characteristic of early PD were an asymmetric pattern of tracer binding, that is, decreased binding of presynaptic radiotracer and normal or increased binding of postsynaptic radiotracer (Ouchi et al, 1999a), and differences in regional susceptibility (Ouchi et al, 1999b) in line with the pathologic finding (Fearnley and Lees, 1991; Kish et al, 1988). Likewise, it can be speculated that there will be an uneven pattern of tracers' accumulation in the dopaminergic projection areas in idiopathic communicating NPH, because morphological change of the midbrain might be related to severity of gait impairment in the disease (Lee et al, 2005).
The purpose of this study was to measure quantitatively the binding potentials (BPs) of [11C]CFT and [11C]raclopride in drug-naive patients with idiopathic normal pressure hydrocephalus (iNPH) on the same day, because any delay in the measurement of the two tracers could devalue the accurate relationship of the viability of presynaptic and postsynaptic dopaminergic neurons that might be affected by increased intra-parenchymal fluid of CSF (Owler et al, 2004) in the living brain.
Materials and methods
Subjects
Eight patients with iNPH who were all naïve to dopaminergic drugs (five men, three women: mean age 72.4 years ± 4.0 s.d. (range 64 to 77)), and eight age-matched healthy subjects (six men, two women, mean age 66.9 years ± 4.5 s.d. (range 62 to 73)) participated in the current study. Diagnosis of iNPH was based on the clinical and imaging features: gait disturbance, cognitive deterioration with more or less urinary incontinence, normal lumber CSF pressure <20 cm H2O (Adams et al, 1965; Hakim and Adams, 1965), and enlargement of the brain ventricles with some degree of deep white matter hyperintensities and reduction of the cortical sulcal space in the superior convexity on the coronal view of magnetic resonance imaging (MRI) (Kitagaki et al, 1998). The morphological MRI and neurologic and blood tests excluded a possibility of secondary NPH and other neurologic diseases such as progressive supranuclear palsy or corticobasal degeneration. In the present study, however, half of the patients were on medication for internal diseases (Table 1). Patient No. 3 had a small meningioma in the cerebellum, which was considered irrelevant to the NPH etiology. As shown in Table 1, no patients suffered from extrapyramidal symptoms such as rigidity or tremor but freezing, magnetic gait, cognitive decline, and some degree of difficulty in urination were observed. A CSF tap test (Wikkelso et al, 1986) after PET measurement was performed to support the diagnosis and introduction of shunt surgery. All medicines for their accompanying diseases were temporarily ceased 12 h before the PET measurement. All subjects were free from regular use of neuroleptic and hypnotic drugs. The current study was approved by the local Ethics Committee of the Hamamatsu Medical Center, and written informed consent was obtained from all participants after full explanation of the nature of the present study.
Clinical characteristics of idiopathic normal pressure hydrocephalus patients
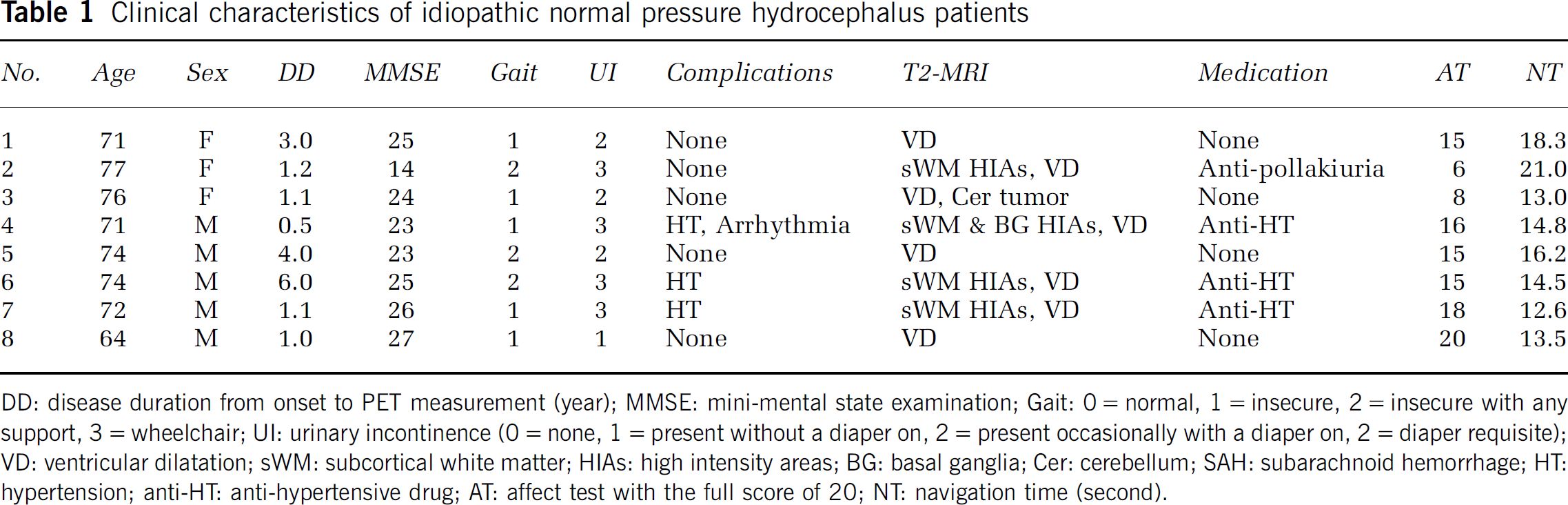
DD: disease duration from onset to PET measurement (year); MMSE: mini-mental state examination; Gait: 0 = normal, 1 = insecure, 2 = insecure with any support, 3 = wheelchair; UI: urinary incontinence (0 = none, 1 = present without a diaper on, 2 = present occasionally with a diaper on, 2 = diaper requisite); VD: ventricular dilatation; sWM: subcortical white matter; HIAs: high intensity areas; BG: basal ganglia; Cer: cerebellum; SAH: subarachnoid hemorrhage; HT: hypertension; anti-HT: anti-hypertensive drug; AT: affect test with the full score of 20; NT: navigation time (second).
Psychological and Behavioral Assessments
The tests consisted of a general cognition and memory test (Mini-Mental State Examination: full score = 30); an affect test in which subjects evaluate facial expressions on different cards by choosing appropriate answers from the following basic affects: happiness, sadness, surprise, disgust, anger, fear (full score = 20), and a walking test (the time required for navigating a 5-m-long path on a flat corridor both ways). Our preliminary examination showed that 11 healthy subjects (mean age = 50.4 years) scored more than 28 on the MMSE, 20 in the affect test, and took < 10 secs in the back-and-forth walk (data not shown).
Magnetic Resonance Imaging and Positron Emission Tomography Procedures
We performed three-dimensional MRI for all participants just before the PET measurement using a static magnet (0.3 T MRP7000AD, Hitachi, Tokyo, Japan) with three-dimensional mode sampling to determine the areas of the midbrain and the striatal nuclei for setting the regions of interest (ROIs) and ventricles for morphometric analysis. The MRI measures and a mobile PET gantry allowed us to generate PET images parallel to the intercommisural (ACPC) line without reslicing during image reconstruction; using this approach, we were able to allocate ROIs on the target regions of original PET images (Ouchi et al, 1999b).
For PET scans, we used a high-resolution brain-purpose SHR12000 (Hamamatsu Photonics KK, Hamamatsu, Japan) tomograph (intrinsic resolution, 2.9 × 2.9 × 3.4 full-width half-maximum, 47 slices, 163 mm axial field of view). After setting the scanner's gantry parallel to the AC-PC line by tilting it, a 10-mins transmission scan for attenuation correction was performed using a 68Ge/68Ga source with the subject's head fixed by a radiosurgery purpose thermoplastic facemask, which enabled the head to be fixed to the same place between the first and second PET measurements among the same subjects. In the [11C]CFT PET study, we performed serial scans (time frames: 4 × 30, 20 × 60, 14 × 300 secs) and periodical arterial blood sampling for 92 mins after injecting intravenously a 350-MBq dose of [11C]CFT at a slow bolus taking 1 min (Ouchi et al, 1999b). To determine unchanged radioligand and radioactive metabolites, additional arterial blood samples were drawn at 1, 5, 20, 30, and 45 mins after the tracer injection and analyzed using thin-layer chromatography and a storage-phosphor-screen bioimaging analyzer (BAS-1500, Fuji Film, Tokyo, Japan). The free metabolite-corrected plasma activities were fitted to a sum of three exponentials by the nonlinear least-squares method with the nonweighted Gauss—Newton algorithm. In the [11C]raclopride study, PET measurement was performed after a time interval of 3 h to allow for the decay of radioactivity. Each participant with the subject's head fixated at the same position as in the [11C]CFT study underwent dynamic PET scans (4 × 30, 20 × 60, and 8 × 300 secs) for 62 mins after a slow bolus injection of a 300-MBq dose of [11C]raclopride with periodical arterial blood sampling for metabolite correction (Ouchi et al, 2002).
Data Analysis
Because various types of structural alterations including ventricular enlargement existed in this iNPH group, we first made a morphometric study for morphology of the midbrain and lateral ventricle. The ventricular dilatation was evaluated by Evans index larger than 0.3 (the ratio of the longest distance between the frontal horns of the lateral ventricles to the longest diameter of the right-to-left side of the brain) (Synek et al, 1976). We also measured the maximum oblique diameter of the midbrain from the aqueduct via the substantia nigra to the edge of cerebral peduncle and the maximum interpeduncular distance at the level of the substantia nigra and red nucleus (Doraiswamy et al, 1992; Ouchi et al, 2005), and the size of frontal horn of the lateral ventricle (O'Hayon et al, 1998) on the MR images. All morphometric values except for Evans index were expressed as ratios (percentage) to the maximal diameter between the frontal and occipital poles along the intercommissural line.
Then, multiple irregular ROIs (40 to 200 mm2) were drawn bilaterally over the nucleus accumbens, ventromedial striatum (head of the caudate), the inferolateral (ventral putamen) and superodorsal parts (dorsal putamen) of the striatum, and the cerebellum on the MR images (Mai et al, 1997). These ROIs were then transferred onto the corresponding dynamic [11C]CFT and [11C]raclopride images with 6.8-mm slice-thickness data generated after adding two consecutive slices using image-processing software (Dr View, Asahi Kasei Co., Tokyo, Japan) on a SUN workstation (Hypersparc ss-20, SUN Microsystems, San Diego, CA, USA) (Ouchi et al, 1999b). A brain morphological change because of ventricular enlargement may cause an error of parameter estimation due to partial volume effect. However, this method of determining the ROIs based on individual MRI minimized the error and the pitfalls of applying a standardized normal brain template to the anatomically affected NPH brain (Owler et al, 2004; Giovacchini et al, 2005). Indeed, we performed a morphometric analysis for the ROI volumes to test any difference in volume because of the presence of structural atrophy in iNPH. A simple t-test revealed no significant difference in volume between the two groups (see Table 3). The values of bilateral ROIs for the cerebellum in the disease group and for all the regions examined in the control group were averaged for further analysis.
MRI-based linear measurements (percentage)
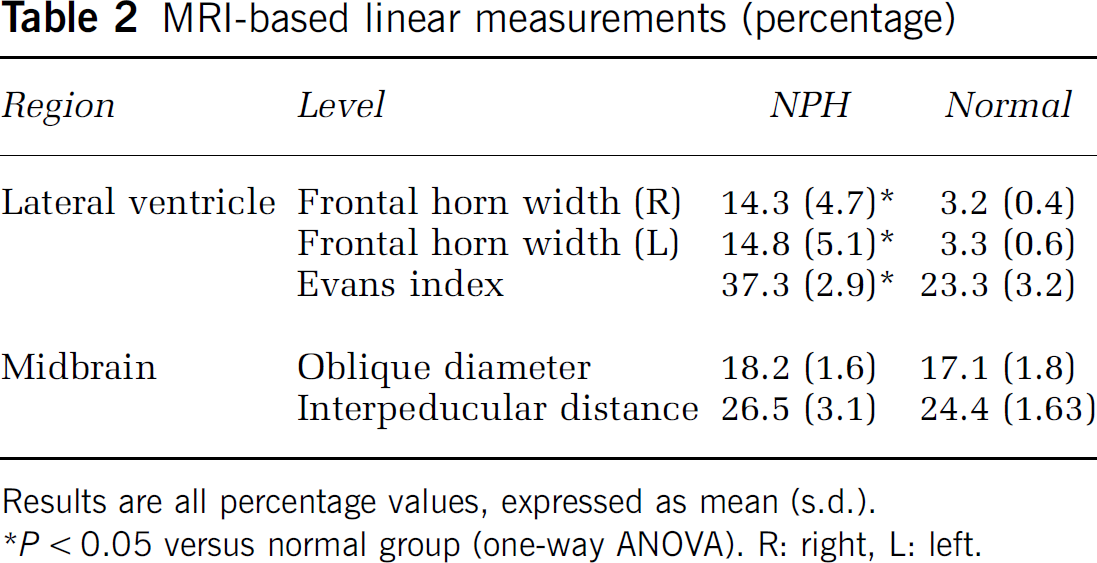
Results are all percentage values, expressed as mean (s.d.).
P < 0.05 versus normal group (one-way ANOVA). R: right, L left.
Levels of binding potential for [11C]CFT and [11C]raclopride and ROI volume
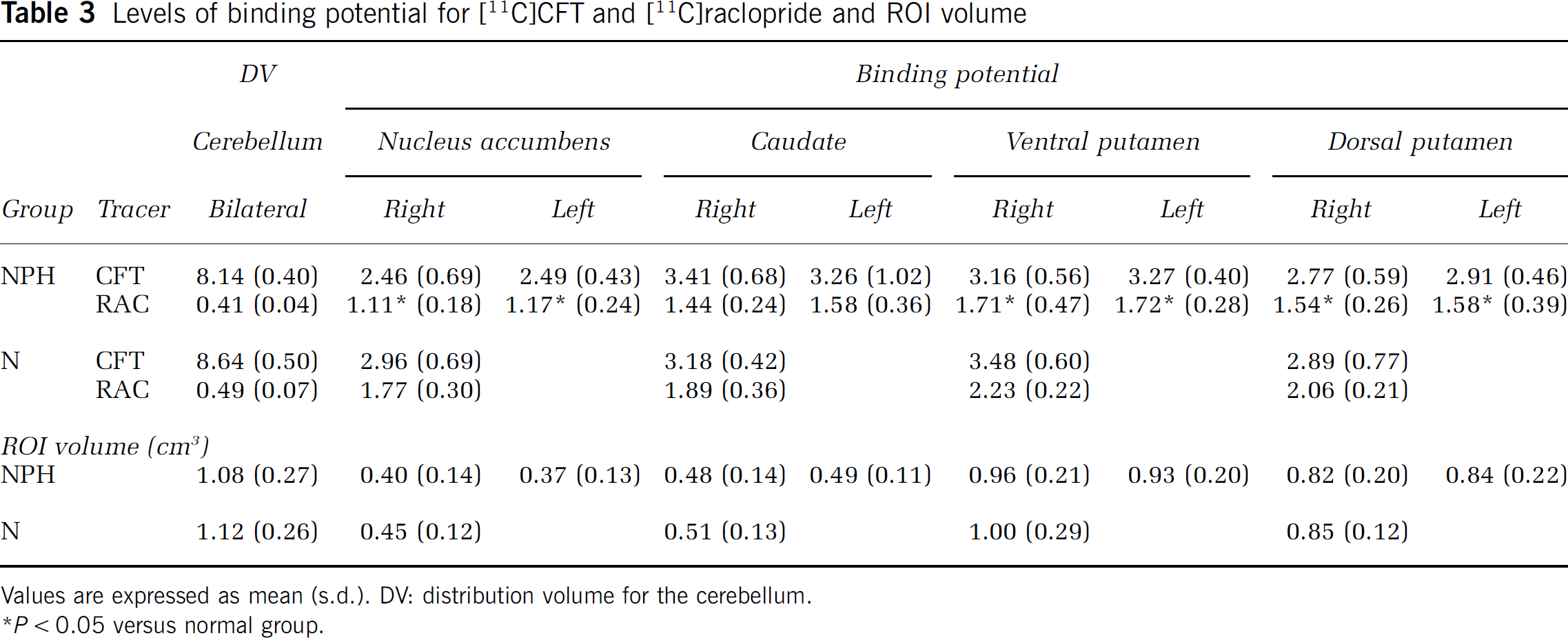
Values are expressed as mean (s.d.). DV: distribution volume for the cerebellum.
P < 0.05 versus normal group.
Because, as described in the PET procedure, participants were scanned in the same position between the two PET measurements, the same ROIs could be placed on both [11C]CFT and [11C]raclopride parametric images. This approach allowed us to examine both aspects of dopaminergic functions presynaptically and postsynaptically in vivo. The BP, Bmax′/kd (or k3/k4) for [11C]CFT was estimated based on the three-compartment model by fitting artery and tissue TACs for blood-brain barrier transport rates (K1), the free plus nonspecific distribution volume (DV)_ (K1/k2), and the binding and dissociation rate constants (k3 and k4 respectively), as described elsewhere (Ouchi et al, 1999a). In addition, the BP for [11C]raclopride was estimated using the following equation and the nonlinear least-squares fitting method; Bmax′/kd = (target tissue Vd)/(cerebellum Vd)–1, where each Vd (DV) was obtained by the Logan graphical method (Logan et al, 1994; Ouchi et al, 2002). Although age-dependent decreases were reported in the binding for dopamine transporter and receptors (Antonini et al, 1993), we did not correct the BPs estimated for aging, because there was not a significant difference in age between groups in the present study (P > 0.05, χ2 test).
Statistics
For comparisons of [11C]CFT and [11C]raclopride binding levels between groups, two-way analysis of variance (ANOVA) was first performed to evaluate the levels about one inter-subject factor and the intra-subject factor (i.e., the hemispheric side of the basal ganglia nucleus) for evaluating the interhemispheric effect to exclude a possible entry of early parkinsonism in the present group. It was found that there was no significant interaction in the two-way ANOVA between the hemispheric side and types of group (P > 0.1), and, therefore, all estimates were separately evaluated by one-way ANOVA in either region with Bonferroni's test for the correction of multiple comparisons. Statistical significance was given as P < 0.05 because the post hoc multiple comparisons were performed in these analyses. The one-way ANOVA was also performed when analyzing the morphometric data. The multiple regression analyses between regional [11C]CFT binding and [11C]raclopride binding, and between the measures of the lateral ventricle or midbrain and the tracers' binding were performed within each group. Spearman's rank correlation analysis was used to compare psycho-behavioral scores with BPs of the two tracers in each region. The significance level was given as a P-value less than 0.05.
Results
Morphometric Evaluation of the Ventricle and Midbrain Sizes in the Idiopathic Normal Pressure Hydrocephalus Group
As shown in Table 2, the width of the lateral ventricle at the caudate head was significantly greater in the iNPH group, as expected. However, the measured distance of the midbrain in the iNPH group was found to be the same as that in the normal group (Table 2) albeit with a tendency to have a wider angle between the bilateral peduncles.
The Levels of [11C]2-β-carbomethoxy-3β-(4-fluorophenyl) tropane and [11C]raclopride Binding Potentials
One-way ANOVA showed that the levels of BP for [11C]raclopride were significantly lower in the nucleus accumbens (–34%) and putamen (–25%) in the iNPH group than in the healthy group (P < 0.05). The BP level in the dorsal putamen decreased more than that in the ventral putamen. The statistics showed a tendency of reduction in the level of [11C]raclopride BP in the caudate (–18%) in the iNPH group compared with the normal counterpart (P = 0.09). In contrast, the magnitude of [11C]CFT BP (–5% in average) was not significantly different between the two groups (Table 3). No differences in DV of [11C]CFT and [11C]raclopride were found between the two groups. There was no significant regional difference in magnitude of [11C]raclopride BP among the dopamine projection areas in the iNPH group.
Correlation Between [11C]2-β-carbomethoxy-3β-(4-fluorophenyl) tropane and [11C]raclopride Binding Potentials
Regression analyses showed a significantly positive correlation between [11C]CFT and [11C]raclopride binding in the dorsal putamen of the healthy group (Figure 1A, y = 0.22x + 1.40, r2 = 0.59) and a negative correlation in the dorsal putamen of the iNPH group (Figure 1B, y = −0.43x + 2.78, r2 = 0.51). In the ventral putamen, there was a tendency of positive correlation in healthy subjects and negative correlation in iNPH patients.
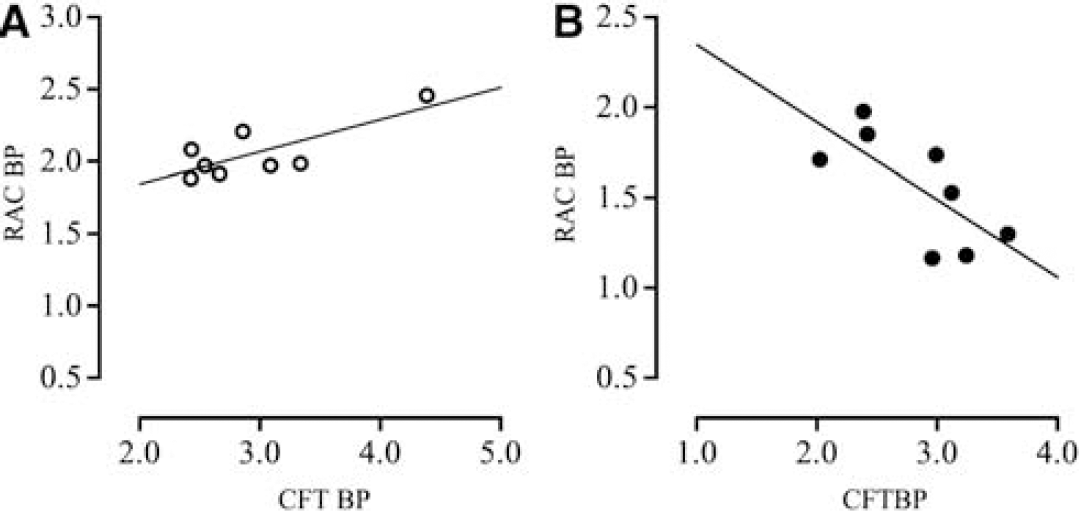
Correlations between levels of [11C]CFT and [11C]raclopride BPs in the dorsal putamen in the healthy group (
Comparison Between Psycho-Behavioral Scores and Binding Levels of two Tracers in the Nucleus Accumbens and Dorsal Putamen in Idiopathic Normal Pressure Hydrocephalus Patients
Correlation analyses showed a significantly positive correlation between [11C]raclopride binding in the nucleus accumbens and emotion score (Figure 2A, y = 0.03x + 0.70, r2 = 0.52) and a negative correlation between [11C]raclopride binding in the dorsal putamen and navigation time (Figure 2B, y = −0.08x + 2.84, r2 = 0.53). However, there was no significant correlation between [11C]CFT binding and these psycho-behavioral parameters. Figure 3 shows one example of this inverse association.
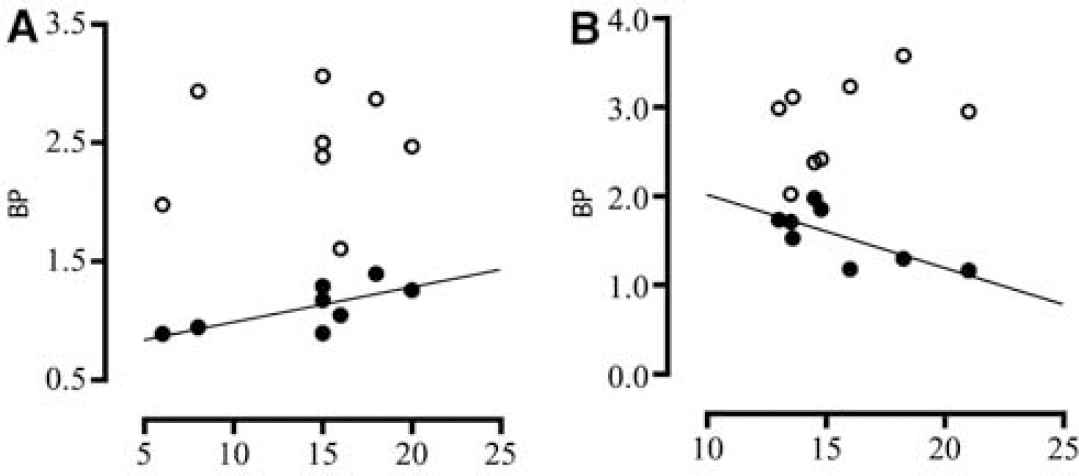
Correlations of [11C]CFT (open circle) and [11C]raclopride (closed circle) BPs in the nucleus accumbens with emotional scores (abscissa) (
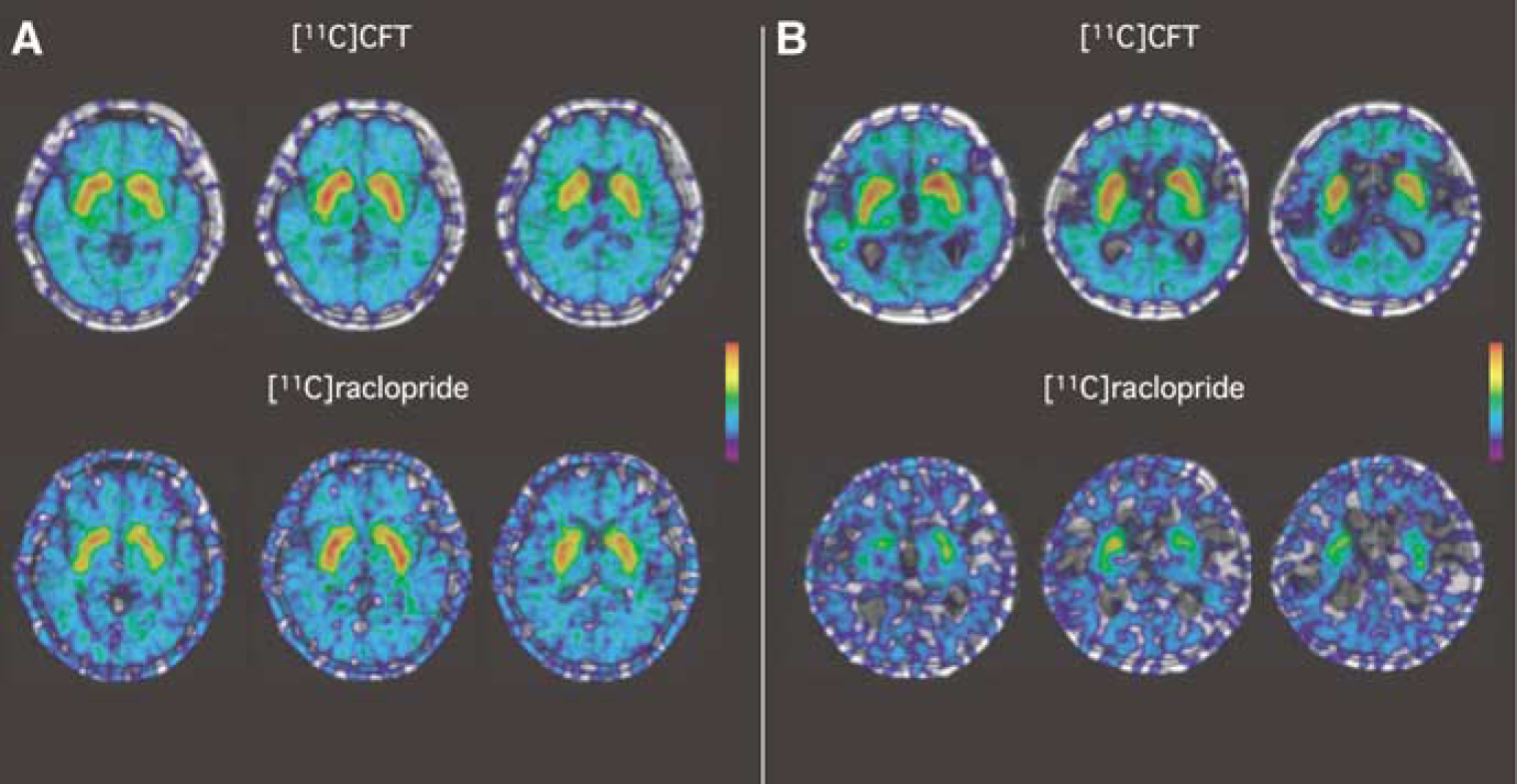
Magnetic resonance imaging—positron emissiontomography fusion parametric images of [11C]CFT and [11C]raclopride binding normalized to the cerebellum in a 68-year-old healthy subject (
Discussion
The present results for the first time show an asymmetric change in BPs of presynaptic and postsynaptic radiotracers and the clinical-pathophysiologic relevance of postsynaptic dopaminergic hypoactivity in the dorsal putamen to the severity of gait impairment in iNPH. This asymmetric pattern of tracers' binding is remarkably different from the opposite direction of the asymmetric change in PD; reduction in binding of the presynaptic marker and a tendency of upregulation of postsynaptic maker (Heiss and Herholz, 2006; Ouchi et al, 1999a), and parallel changes in those two tracers in normal aging and sporadic parkin-linked parkinsonism (Scherfler et al, 2004). This suggests that alteration in striatal postsynaptic dopaminergic function and preservation of nigral dopamine function may reflect a pathophysiology of iNPH.
The present normal finding of [11C]CFT binding along with reduction in striatal [11C]raclopride binding in iNPH patients indicates that the entity of iNPH resides chiefly in dysfunction in the dopamine projection area involved in the basal ganglia—cortical circuit. The previous reports, albeit in the experimental setting, showing that there was not significant reduction either in dopamine level (Del Bigio and Vriend, 1998) or dopaminergic nigral neurons (Ishizaki et al, 2000) in rats 4 weeks after induction of hydrocephalus support the present finding of preserved [11C]CFT binding in iNPH patients. As for reduction in [11C]raclopride binding, two explanations may be possible at the moment, downregulation and loss of the D2 receptors. The level of [11C]raclopride binding at baseline varies in the pathophysiologic status of the disease or the amount of drug interaction on the D2 receptor. This is because in PD, upregulation of [11C]raclopride binding to the receptor shown at an early stage of PD declines with the disease severity increasing (Antonini et al, 1995) and with dopamine agonists administered (Heiss and Herholz, 2006). Since all iNPH patients in the current study were naïve to drugs known to affect the D2 receptor (Table 1), it is confirmed that the present reduction in [11C]raclopride binding reflects a pathophysiologic change characteristic of the disease. Although there is no histochemical study on alterations in D2 receptor in the striatum in chronic hydrocephalus, the presence of axonal injury in the white matter (Del Bigio et al, 2003) and a decrease in the number of cholinergic neurons (Ishizaki et al, 2000) in the rat neostriatum in the subacute hydrocephalic condition can exrapolate that loss of D2 receptors might be responsible for the reduction of [11C]raclopride binding in the present study. This speculation is in line with the result that D2 receptor-lacking knockout mice exhibited a phenotype similar to the extrapyramidal symptoms of PD (Baik et al, 1995). Because D2 receptor mediates motor information from the cortex (Calabresi et al, 1997), either downregulation or loss of D2 receptors can impair the corticostriatal neuronal transmission in iNPH.
It is interesting that [11C]raclopride binding in the caudate located closer to the enlarged ventricle was not significantly decreased than that in the dorsal putamen and nucleus accumbens in the present study. This may implicate that the vicinity to the dilated ventricle is not an important factor for the influence of [11C]raclopride binding in iNPH. The caudate nucleus is principally connected to the frontal eye field (Gerardin et al, 2003). Thus, these findings support the rarity of disturbance in saccadic eye movement in iNPH patients clinically. In contrast, the dorsal part of the putamen chiefly received the neuronal input from the sensorimotor cortex (Kunzle, 1975). A recent functional MRI study has shown that the foot area is somatotopically located in the dorsal putamen (Gerardin et al, 2003). Our previous PET study with [11C]raclopride showed that the foot motor execution increased dopamine release in this dorsal putamen (Ouchi et al, 2002). These lines of evidence support the present finding that [11C]raclopride binding in the dorsal putamen significantly correlated with gait performance (navigation time) in iNPH patients because the connection between the sensorimotor cortex and the corresponding putaminal region might be disrupted anatomically and functionally. In addition, significant association of mood with nucleus accumbens [11C]raclopride binding in our iNPH patients might be explained the same way because the prefrontal cortical input in the nucleus accumbens is modulated selectively by phasic dopamine release through the D2 receptor in regulation of motivation processing (Goto and Grace, 2005). These findings suggest that dopaminergic derangement in the ‘motor' putamen and ‘motivational' nucleus accumbens might be characteristic of iNPH pathophysiology.
Morphological alteration in the brain is an important sign to be considered for clinical diagnosis of iNPH. In the present study, in addition to significant dilatation of lateral ventricles, there was a tendency of flattening of the midbrain anteroposteriorly in the iNPH group, that is, a longer interpeduncular diameter with a wider angle between the bilateral crus cerebri on the horizontal MRI plane. This tendency was in line with a shorter anteroposterior diameter measured on the sagittal MR plane (Lee et al, 2005), in which the severity of gait disturbance was reported to correlate negatively with the midbrain diameter. In view of the dopaminergic activity, however, we did not see either significant reduction in the presynaptic ([11C]CFT) marker binding or significant correlation between the marker binding and behavioral measures in the present study. Unlike the finding in PD showing that the midbrain pedunculopontine nucleus, known as a lower locomotion center, is degenerated (Pahapill and Lozano, 2000), there is no such evidence in iNPH. Furthermore, the average disease duration in our iNPH patients is found to be relatively short and gait disturbance milder, indicating that the cellular activities of dopamine neurons in the midbrain can be spared at an early stage of iNPH despite ensuing deformity of the midbrain structure.
In summary, we showed significant reduction in the postsynaptic D2 receptor binding and preservation of the presynaptic dopamine transporter binding in the striatum of iNPH patients. The reduction of D2 receptor binding in the dorsal putamen (striatal foot area) may be pathophyiologically important for progressive gait deterioration in the disease. Alterations in D2 receptor control in the corticostriatal system may contribute to the clinical manifestations reminiscent of parkinsonism seen in iNPH.
Footnotes
Acknowledgements
The authors thank Mr Yutaka Naito (Nippon Environment Research KK) and Mr Masami Futatsubashi (Hamamatsu Photonics KK) for their contribution to tracer synthesis, and other members of our staff for their help with scanning.
