Abstract
CADASIL (Cerebral Autosomal Dominant Arteriopathy with Subcortical Infarcts and Leukoencephalopathy) is caused by mutations in the Notch3 receptor expressed at the surface of vascular smooth muscle cells. The functional consequences of the disease at the peripheral microcirculation level are incompletely elucidated. In this study, we aimed to assess, in vivo, the endothelium-dependent and independent vasodilation of the skin microvasculature in CADASIL patients. Twenty-three affected subjects were compared with 23 gender and age-matched controls. The brachial artery endothelium-dependent and endothelium-independent vasodilation were assessed after forearm cuff occlusion and nitroglycerin administration. Skin vasoreactivity to transcutaneous administration of acetylcholine and sodium nitroprussiate, and after postocclusive hyperemia were measured by Laser Doppler flowmetry. The maximum changes in the diameter of the brachial artery after the cuff release or after nitroglycerin administration did not differ between patients and controls. With iontopheresis, only the peak value of the dose response was found decreased in normocholesterolemic patients after nitroprussiate administration. The postocclusive test revealed a large increase of the time to peak value and whole duration of the hyperemic response in CADASIL patients. The results of this study show that the skin vasoreactivity is altered in CADASIL. Particularly, the kinetics of reactive hyperemia after cuff occlusion is dramatically changed with a lengthened and delayed response. This characteristic pattern may be related to the specific ultrastructural modifications related to Notch3 gene mutations involving smooth muscle cells in the microvasculature.
Introduction
CADASIL (Cerebral Autosomal Dominant Arteriopathy with Subcortical Infarcts and Leukoencephalopathy), the most frequent inherited small-artery disease characterized by recurrent ischemic stroke and dementia, is caused by highly stereotyped mutations in the Notch3 receptor (Joutel et al, 1996, 1997) expressed at the surface of vascular smooth muscle cells (VSMC) (Joutel et al, 2000). In CADASIL patients, an accumulation of the Notch3 protein is detected at the plasma membrane of VSMC, presumably related to altered clearance of the receptor (Joutel et al, 2001; Joutel and Tournier-Lasserve 2002). In the vessel wall, the disease is characterized by a degeneration of VSMC and the presence of a granular osmiophilic material within the basement membrane (Joutel et al, 2000; Kalimo et al, 2002; Ruchoux et al, 1995).
The functional consequences of these vascular wall modifications are incompletely elucidated. At the cerebral level, they are presumably responsible for a decrease in basal perfusion and reduction of hemodynamic reserve (Chabriat et al, 2000; Pfefferkorn et al, 2001). In vitro, abnormal vasoconstrictor responses to angiotensin II and noradrenaline have been detected in arteries of CADASIL patients (Hussain et al, 2004). Moreover, recent investigations of the vasomotor function of tail arteries from transgenic mice expressing a mutant Notch3 in VSMC showed that flow-induced dilation was decreased while pressure-induced myogenic tone was increased (Dubroca et al, 2005). These results suggest that vascular mechanotransduction is mainly impaired in the presence of mutations of the NOTCH3 gene (Dubroca et al, 2005). In vivo, vasoreactivity has not been investigated at the peripheral level in CADASIL patients.
In the present study, the vasoreactivity of skin vessels was compared between CADASIL patients and healthy controls.
Subjects and methods
Patients
Twenty-three CADASIL patients (12 males, 11 females, mean age: 53.8±10.7 years (range: 39 to 75) were included in the study based on the following criteria: (1) genetic test showing a characteristic mutation in the Notch3 gene; (2) no history of cardiovascular disease, hypertension or diabetes mellitus; (3) no history of heavy smoking or current use of tobacco; and (4) no medication use likely to interfere with vasoreactivity.
As it was extremely difficult to recruit CADASIL patients without hypercholesterolemia in addition to the other selection criteria, hypercholesterolemia was not used as an exclusion criteria in this group. However, low-density lipoprotien (LDL) cholesterol level was considered in the statistical analysis by splitting the patients into two subgroups: (1) patients with treated or untreated hypercholesterolemia (hypercholesterolemic or HC) or (2) patients without hypercholesterolemia (normocholesterolemic or NC); LDL cholesterol < 4.2 mmol/L. Patients treated for hypercholesterolemia with normal LDL cholesterol values as defined above were included in the HC group.
Magnetic resonance imaging (MRI) was performed at the time of the study using identical sequences in each subject on a 1.5 T MR scanner. The severity of MRI white-matter lesions on FLAIR images was assessed in four grades, as previously reported (Chabriat et al, 1998).
Controls
A group of 23 gender- and age-matched healthy subjects (12 males, 11 females, mean age: 55.5±10.2 years, range: 40 to 74) were selected based on the following criteria: (1) no history of cardiovascular or cerebrovascular disease, hypertension or diabetes mellitus; (2) no history of hypercholesterolemia and LDL cholesterol < 4.2 mmol/L at the time of the study; (3) no history of heavy smoking or current use of tobacco; (4) no medication susceptible to interfere with vasoreactivity; and (5) normal neurological and clinical examination.
The study protocol was approved by an independent regional ethic committee (CCPPRB/40-01/Pitie-Salpetriere).
Experimental Protocol
Preparation: All measures were obtained as previously recommended (Bircher et al, 1994; Fullerton et al, 2002) in a quiet room and after a 30 mins period for acclimatization.
Hemodynamic parameters
(1) Vasodilation of the brachial artery: (a) Endothelial-dependent flow-mediated vasodilation of the brachial artery: the left brachial artery was imaged in the longitudinal plane using a two-dimensional ultrasound system (7.5 MHz probe) with simultaneous electrocardiogram (ECG) recording. As recommended (Corretti et al, 2002), a segment with anterior and posterior intimal interfaces between the lumen and vessel wall was selected for continuous imaging. A baseline rest image was acquired and the pulsed Doppler blood flow velocity was measured from the midartery sample volume. Thereafter, a blood pressure cuff was placed on the forearm and was inflated up to 50 mm Hg above the systolic pressure for 4 mins. Changes in diameter of the brachial artery were then calculated non-continuously at 30 secs and 90 secs after the cuff deflation. The maximum change in diameter at these different times was used for analysis.
(b) Endothelium-independent vasodilation with nitroglycerin of the brachial artery: After a 10 min period of rest, the endothelium-independent vasodilation of the brachial artery was assessed by measurements of the maximum change of diameter, on a longitudinal segment of the brachial artery above the antecubital fossa, at 3 and 5 mins after sublingual nitroglycerin administration.
(2) Cutaneous blood flow measurement: The skin blood flow was assessed using a laser doppler flowmeter (Periflux system 4100, Perimed, Jarfalla, Sweden). A probe was attached to the finger by a double-sided adhesive for the emission of the red light laser (632 nm; power 1 mW; beam diameter 1 mm).
(a) Iontophoresis with acetylcholine and sodium nitroprusside: iontophoresis was used to deliver charged vasoactive agents into the skin by applying a small electric current. A drug delivery electrode chamber was fixed to the laser probe on the dorsal surface of the third finger of one hand. A small hole allowed the passage of the laser light through the chamber. As recommended (Fullerton et al, 2002), the drug delivery electrode was filled with 180 ml of acetylcholine (ACh) at 1% or sodium nitroprusside (SNP) also at 1%. Both were diluted in NaCl 0.9% and prepared immediately before use. After recording of the baseline flow, each drug was delivered one after the other on the dorsum of the right hand (second finger for ACh, third finger for SNP) using a cathodal (ACh) or an anodal (SNP) stimulus. Four increasing stages were recorded during the delivery of each drug: 150 μA during 10 secs, 200 μA during 10 secs, 200 μA during 20 secs and 200 μA during 60 secs were delivered with a 360 secs interval between each single dose. As previously recommended (Bircher et al, 1994; Ramsay et al, 2002), a low resistance vehicle and low current were used to prevent artifacts induced by hyperemia. The microcirculation hemodynamic response was expressed by using two parameters at each stage and for each drug application: (1) the peak value measured as the maximum blood flow minus the basal flow value; and (2) the area under the curve measured on a 6-mins-length at each iontophoretic stage.
(b) Postocclusive hyperemia test: The postocclusive reactive hyperemia test was assessed by laser Doppler flowmetry after a 4-mins forearm cuff occlusion. Arterial occlusion was obtained by inflating a cuff 50 mm Hg above the systolic blood pressure of the subject. Immediately after deflation, the blood flow was recorded until recovery of the baseline flow value. Five parameters were analysed: the peak flow value assessed as the maximum flow value minus the basal flow value (maximum FV–basal FV), the time to peak value (TTP) calculated from the deflation to the maximum flow value, the time of half decrease (HDT) from the deflation to the return to 50% of the peak value (basal flow value + (maximum FV–basal FV)/2) and the whole duration of the hyperemic response (WRD) calculated from the deflation to a flow value of 25% of the peak value (basal flow value + (maximum FV–basal FV)/4). The area under curve (AUC) was also calculated during the whole duration of the response.
(3) Statistical analysis: Analysis of variance or Student's t-tests were used for comparison between CADASIL patients and controls. The data collected during the iontophoresis experiment were analyzed using two-way ANOVA for repeated measurements. All parametric tests were made after check of the Gaussian distribution of the data or after their logarithmic transformation when necessary (in this case, geometric mean was used to summarize the data). When no normalizing transformation was adequate, Wilcoxon tests were used for comparisons between patients and controls. The nonparametric Spearman correlation test was used to test correlations between hemodynamic data and clinical scores (Rankin, NIHSS and MMS). Values of P < 0.05 were considered statistically significant. The statistical analysis was performed using the Statview statistical software (Abacus Inc., USA).
Results
Clinical Data
In the 23 CADASIL patients, rankin scores ranged from 0 to 4 (0 to 1 in 18 patients, 2 to 3 in two patients, 4 in three patients). MMSE score ranged from 9 to 30, five patients were demented with an MMSE score < 20. All patients had varying degrees of hyperintense white-matter lesions on FLAIR images (scores B to D). On T1-weighted images, all patients except one (an asymptomatic 41-year-old) had evidence of small deep infarctions.
The mean values of systolic and diastolic blood pressure, body mass index, serum levels of LDL cholesterol, homocysteine, fasting glucose, hemoglobin and counts of platelets and leukocytes did not differ between CADASIL patients and their gender- and age-matched controls.
Among the 23 CADASIL patients, nine were included in the HC group at the time of the study. These HC patients were older, had a significantly lower MMSE score and higher diastolic blood pressure than the normorcholesterolemic patients (Table 1). Nine patients in the HC group and 10 in the NC group were treated by aspirin.
Clinical data in NC and HC patients
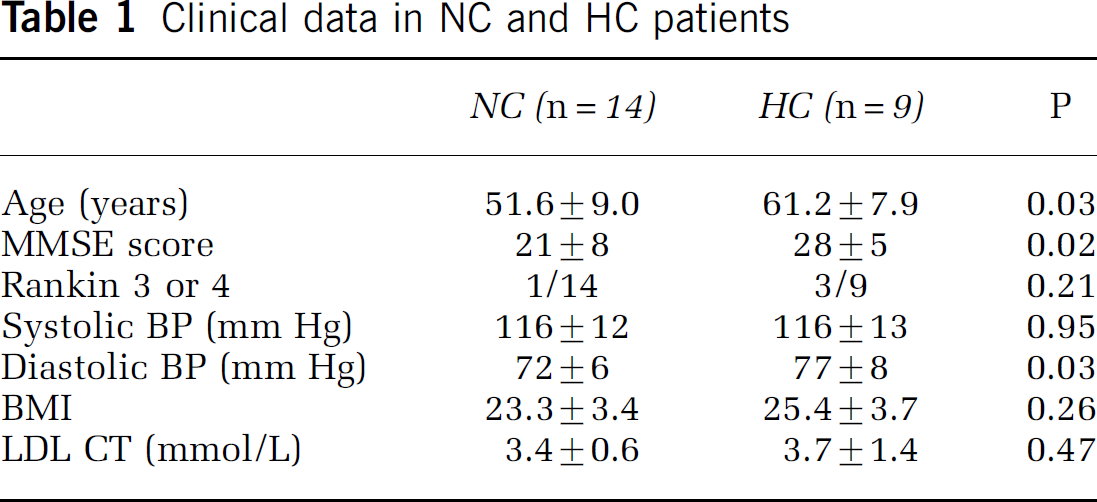
(1) Vasodilation of the brachial artery (Table 2): (a) Endothelial-dependent flow-mediated vasodilation of the brachial artery: at baseline, the brachial artery diameter did not differ between CADASIL patients and controls (3.4±0.69 versus 3.4±0.46 mm, P =0.81). The maximum changes in the diameter of the brachial artery after the cuff release did not differ between CADASIL patients and controls. In addition, no significant difference was detected when HC and NC patients were compared separately to their matched-controls.
Vasodilation of the brachial artery (maximum changes in diameter): reactive hyperemia after the cuff release (RHE) or after nitroglycerin administration (TNT)
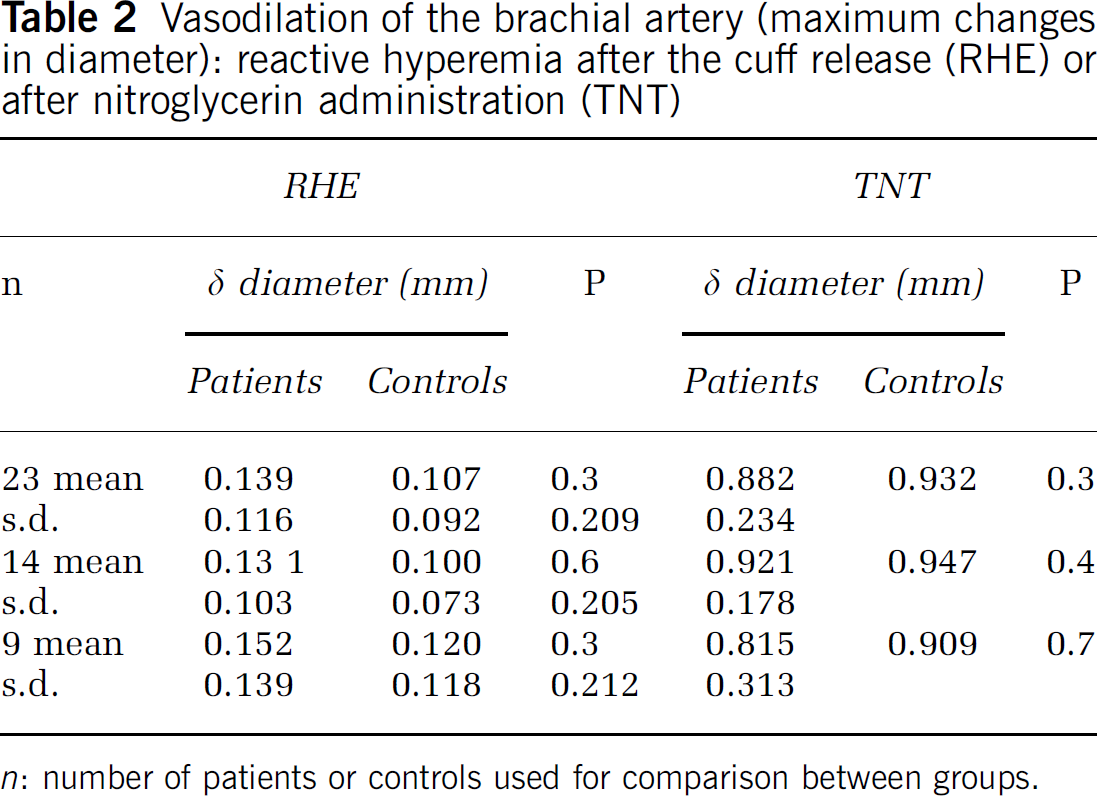
n: number of patients or controls used for comparison between groups.
(b) Endothelium-independent vasodilation of the brachial artery with nitroglycerin: similarly, between CADASIL patients and controls, there was no significant difference in the change in diameter measured after nitroglycerin administration. This was also true when the two subgroups of HC and NC patients were separately compared with their matched controls.
(2) Cutaneous blood flow measurements: cutaneous blood flow (CuBF) measurements are presented Figures 1 and 2.
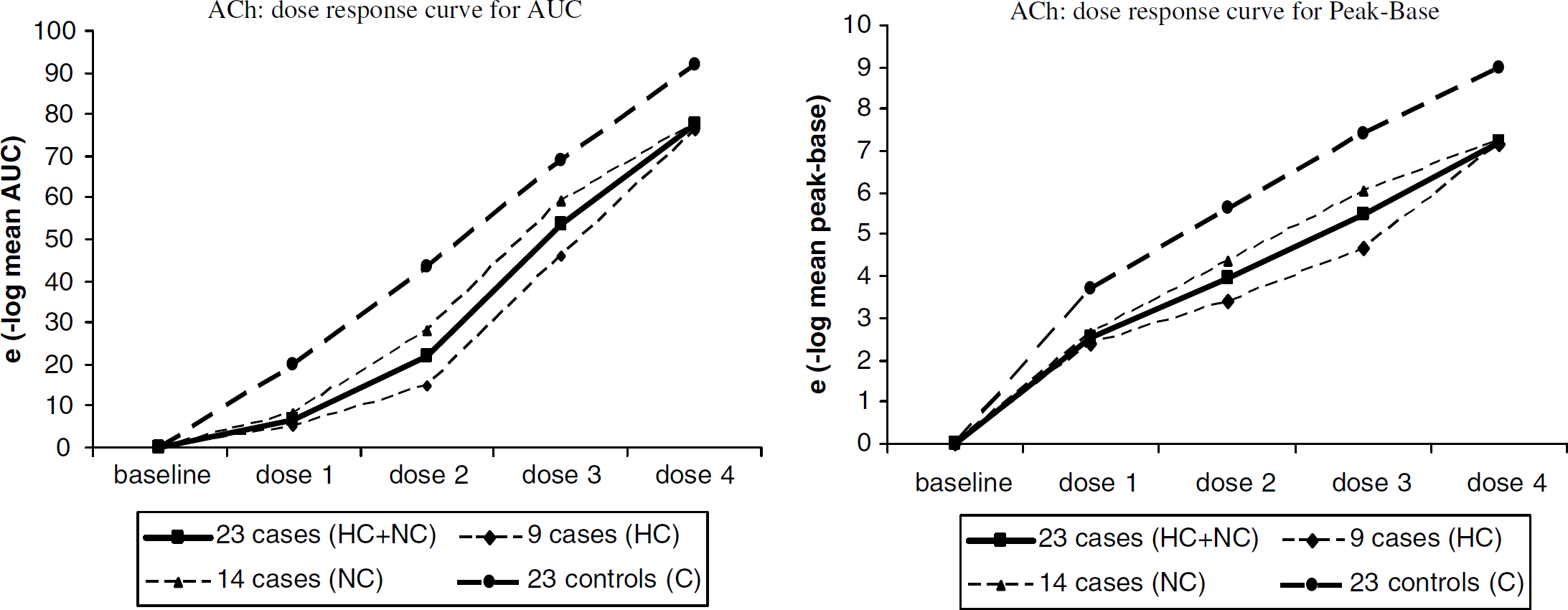
Cutaneous blood flow and endothelium dependent response to ACh. The modifications of the AUC and of the peak of flow after ACh administration obtained at different dosages are shown. A significant decrease of AUC and of the maximum peak value was detected in the whole group of patients. When the groups were considered separately, the decrease appeared significant for HC patients but did not reach statistical significance for NC patients. The dose–response curve significantly differed only between the whole group of patient and controls, and between HC patients and controls.
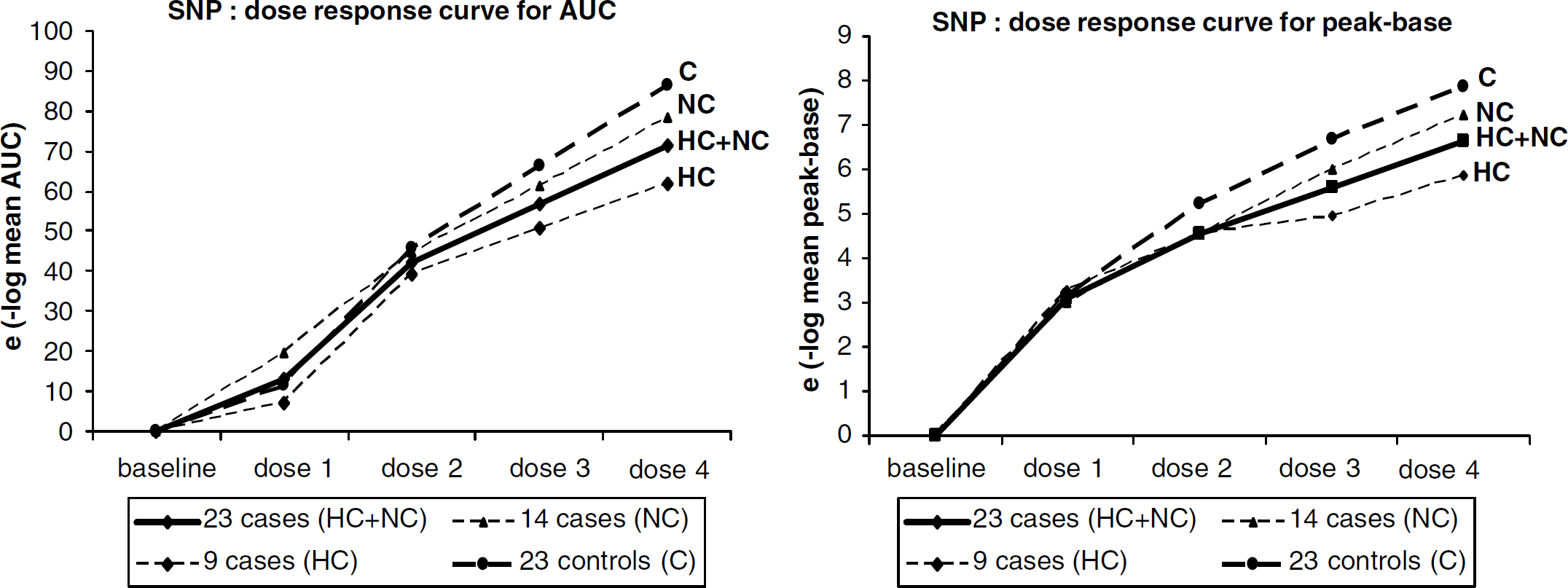
Cutaneous blood flow and endothelium independent response to SNP (dose–response curve to incremential SNP administration). The modifications of the AUC and of the peak of flow after SNP administration at different dosages are shown. After SNP administration, CuBF was increased over the baseline both in patients and controls; this was detected in the whole group of patients as well as in HC and NC patients (P-values < 0.0001). The dose effect of SNP did not differ between patients and controls for both parameters, in the whole group of patients, in HC as well as in NC patients.
(a) Iontophoresis with acetylcholine: the results expressed as geometric mean value (elog mean value) are presented in Figure 1. The baseline CuBF did not differ between patients and controls. The iontophoresis of ACh significantly increased the CuBF over baseline both in CADASIL patients and in controls. This effect was highly significant in the whole group of patients but also in NC as well as in HC patients (all P-values < 0.0001). A significant decrease of AUC and of the maximum peak value was detected in the whole group of patients (P-values respectively 0.03 and 0.004). This decrease was significant for HC patients (AUC: P= 0.012; maximum peak value: P= 0.015) but not for NC patients (all P-values >0.1). Moreover, the dose response significantly differed between the whole group of patients and controls for the AUC (P =0.03). This was also observed for HC patients (P =0.03). In contrast, the dose response did not significantly differ between NC patients and controls (P-values > 0.5).
(b) Iontophoresis with sodium nitroprusside: the results expressed as geometric mean value (elog mean value) are presented in Figure 2. The baseline CuBF did not differ between patients and controls before the application of SNP. After SNP administration, CuBF was increased over the baseline both in patients and controls; this was detected in the whole group of patients as well as in HC and NC patients (P-values < 0.0001). No significant difference was detected between the 23 patients and controls for both the AUC and peak value. Only a significant difference was detected for the peak value (P =0.04) but not for AUC (P =0.71) between NC patients and controls. The dose effect of SNP did not differ between patients and controls for both parameters, in the whole group of patients, in HC as well as in NC patients.
(c) Postocclusive hyperemic test: the results obtained in one patient and one control are illustrated in Figure 3. Results in the entire group of patients are expressed as median values (non-Gaussian distribution) in Figure 4. At baseline, the CuBF did not differ between patients and controls. The transient arterial occlusion significantly in-creased the CuBF over baseline both in CADASIL patients and controls. Both the peak value of the post-occlusive hyperemic flow and the AUC did not differ between the two groups. In contrast, the TTP (29 secs [8 to 67] versus 19 secs [6 to 51]; P =0.02), and the whole duration time (WDT) of the hyperemic response (103 secs [47 to 253] versus 61 secs [8 to 140]; P =0.005) were much larger in CADASIL patients than in controls. There was a trend towards elevated half decrease time (HDT) (38 secs [10 to 98] versus 15 secs [3 to 86]; P =0.09) in patients compared with controls. Similar results were obtained when only NC patients were compared with age- and gender-matched controls (TTP: median value [min–max] 30 secs [8 to 67] versus 19 secs [7 to 51] secs; P =0.01; WTD: median value [min-max] 95sec [76 to 160] versus 52 secs [27 to 120], P =0.015; HDT median value [min–max]: 39 secs [14 to 98] versus 12 secs [3 to 86] P =0.07).
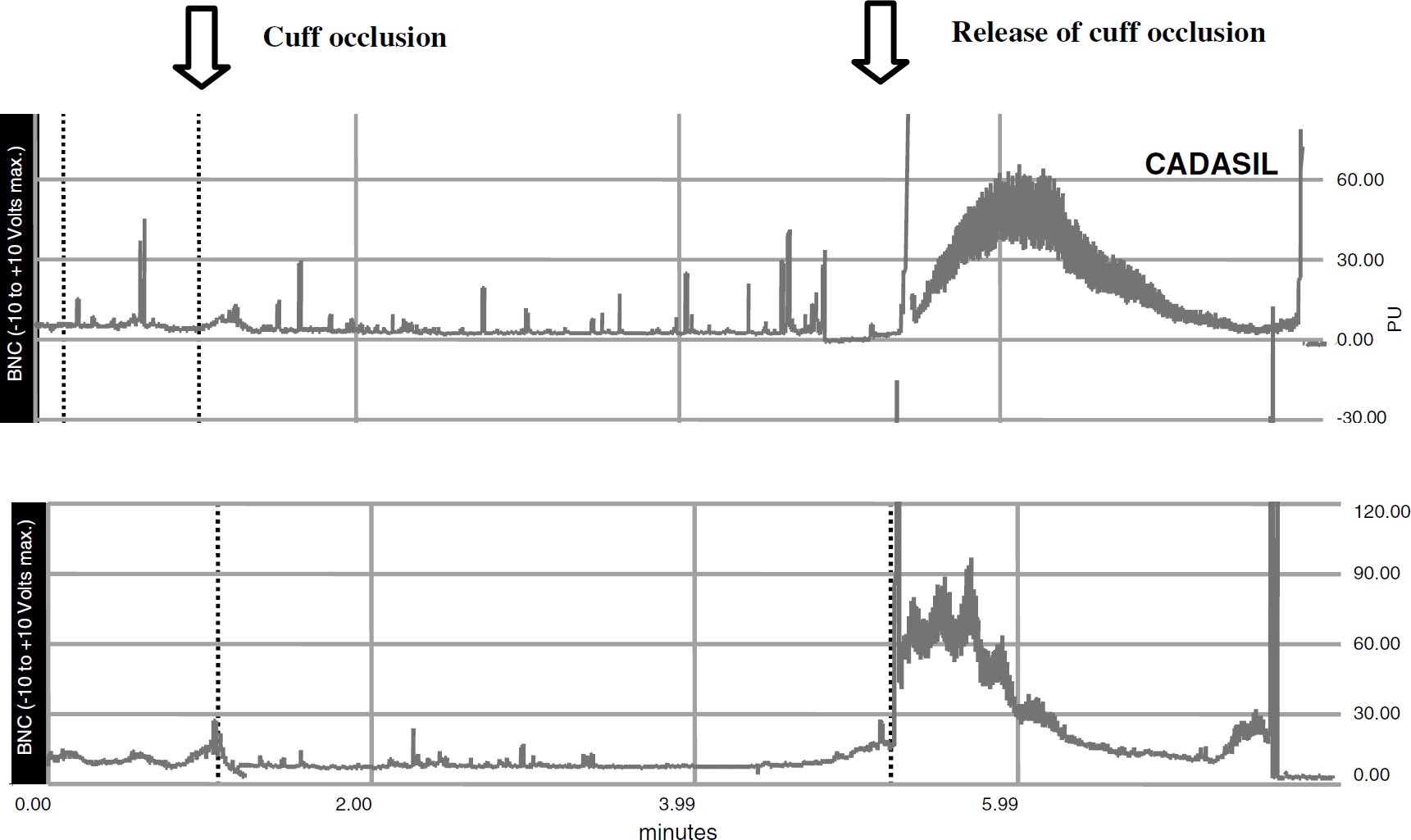
Curves of cutaneous blood flow obtained during reactive hyperemia in a CADASIL patient (upper curve) and an agematched control (lower curve). The following parameters were obtained in one patient: peak flow value = 45PU, AUC = 2609, WTD = 91 secs, TTP = 44 secs, HTD = 26 secs. Identical parameters were obtained in the matched control: peak flow value = 34PU, AUC = 1954, WTD = 45 secs, TTP = 27 secs, HTD = 23 secs. Note the characteristic larger WTD and TTP in the CADASIL patient.
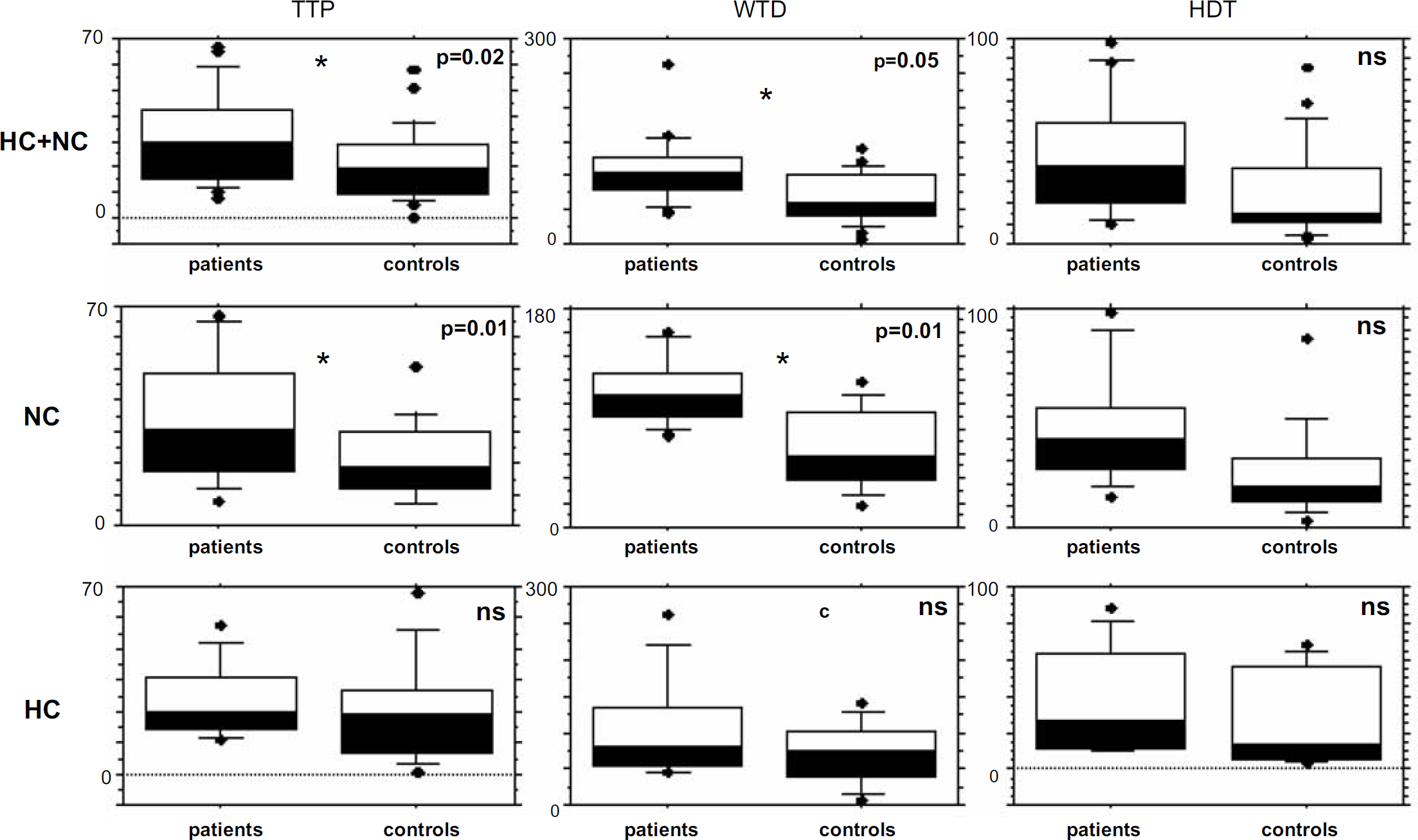
Analysis of the cutaneous hyperhemic response after transient brachial occlusion. The results showed a significant increase in the WTD and time to the peak in CADASIL patients compared with controls, whereas amplitude parameters (peak-base and AUC) were similar between the two groups. The results remain significant when HC patients were excluded from the analysis. (TTP: time in seconds calculated from the deflation to the maximum flow value of the hyperhemic response, WTD: whole time duration of the response from the deflation to a flow value of 25% of the peak value (basal flow value + (maximum FV–basal FV)/4), HDT: half decrease time of the response calculated from the deflation to a return to 50% of the peak value (basal flow value + (maximum FV–basal FV)/2)).
Discussion
To our knowledge, this study provides the first evidence of in vivo alterations of skin microvascular reactivity in CADASIL patients.
The most important difference between CADASIL patients and healthy subjects was found in the CuBF curves during reactive hyperemia. While both the amplitude of the response and AUC did not differ between patients and controls (suggesting a complete restoration of the CuBF after transient ischemia), both the TTP and the whole duration of the hyperemic response were significantly increased in CADASIL patients. This increase was significant in the whole group as well as in the subgroup of NC patients. Together, these results suggest that dilation of the microvasculature (capillaries and arteries of diameter < 200 μm) is of normal amplitude after transient ischemia but dramatically lengthened in the presence of the CADASIL-related angiopathy.
Reactive hyperemia is an adaptive response to ischemia involving three types of mechanisms (Monsuez, 2001): (1) a myogenic response secondary to the stretch and transmural pressure modifications; (2) a metabolic response related to adenosine, PiO2, PiCO 2 or local pH modifications; and (3) a shear stress response related to prostacycline, nitric oxide, and potassium channels activation. Previous studies have shown that a selective blockade of the metabolic response or of shear stress molecules can alter the amplitude but do not modify the kinetics of the hyperemic response (Bijlstra et al, 1996; Joannides et al, 1995). Modifications of the myogenic response are presumably responsible for the characteristic profile of the hyperemic response observed in CADASIL.
Using iontophoresis, we observed that the endothelium dependant response to ACh was of normal amplitude in NC CADASIL patients. This response has been previously shown to be mainly mediated by prostanoids (Noon et al, 1998; Vicaut et al, 1994). There was no difference between NC CADASIL patients and controls, despite the fact that most of our patients were treated with aspirin, which could potentially alter this response (Abou-Elenin et al, 2002; Berghoff et al, 2002). The reduced response detected in our HC patients is consistent with what has been previously reported in healthy subjects with increased serum cholesterol (Chowienczyk et al, 1992). Indeed, increased total cholesterol levels have been implicated in early endothelial dysfunction (Chowienczyk et al, 1992).
Conversely, the application of SNP (a direct donor of nitric oxide to VSMC) resulted in a lower peak flow in NC CADASIL patients although the AUC was not significantly changed. This also suggests different kinetics of vasodilation in CADASIL patients. Ex vivo, Hussain et al (2004) previously detected a normal response to direct NO donors of gluteal arteries obtained from CADASIL patients. However, they did not analyze the kinetic profile of the vasoactive response at a given dose, and the different environment, transmural pressure and parietal constraints were not comparable. The results obtained with ACh and SNP iontophoresis further support that the kinetics (but not the amplitude) of VSMC relaxation is impaired in CADASIL after direct or endothelium-mediated stimulation.
These findings are substantially different from modifications of the skin vasoreactivity related to hypertension (Prewitt, 2002; Taddei et al, 1999). Several authors have previously reported that the endothelial-dependent response to ACh is dramatically altered (reduced peak and AUC values) while the SNP response is preserved at high blood pressure (Prewitt 2002). Furthermore, while reactive hyperemia is also altered in hypertension, with reduction of both peak and AUC, there is no change of the kinetic parameters of the response (Dakak et al, 1998). Thus, although hypertension is a major risk factor for small artery diseases, the corresponding patterns of the skin microvascular reactivity are distinct from CADASIL. The present results are consistent with the relative preservation of endothelial cells and primary involvement of VSMC in CADASIL-related angiopathy. Conversely, in hypertension-related vasculopathy, endothelial cells are the primary targets and their functions may be altered long before any structural changes in VSMC.
In this study, we did not find any significant alterations of the brachial artery vasoreactive responses after cuff occlusion or nitroglycerin administration in CADASIL patients. The interpretation of these negative results is difficult. The results may possibly be related to the distribution of ultrastructural vascular wall changes seen in the disease; middle-sized arteries being less severely affected than the microvasculature. However, we cannot exclude insufficient statistical power to detect some subtle differences between the two groups due to the relatively limited size of samples.
In conclusion, the present study demonstrates that significant alterations in the skin vasoreactivity are present in CADASIL. While the amplitude of the endothelium and non-endothelium dependent vasodilation appears either relatively preserved or linked to associated hypercholesterolemia, the kinetics of skin vasodilation is considerably modified. This novel pattern presumably results from the characteristic Notch3-related structural modifications of microvascular smooth muscle cells detected in this disorder. Additional studies in larger populations and performed in a prospective way may help to understand the clinical value of this peripheral marker and its potential value for future therapeutic evaluation.
Footnotes
Acknowledgements
The authors thank Jocelyne Ruffie, Solange Hello, Richard Sciascia, Maria Giquel, Stephanie Leclerc for technical help.
