Abstract
Female animals are protected from ischemic brain damage relative to age-matched males, in part through protection provided by endogenous estradiol. In brain, estradiol is produced from testosterone by cytochrome P450 aromatase (cyp 19), a steroid synthetic enzyme present in astrocytes. We tested the hypothesis that astrocytes derived from neonatal female rat brain are less susceptible than male cells to oxygen–glucose deprivation (OGD), and that this endogenous protection is associated with enhanced aromatase activity. Primary cultured cortical astrocytes were prepared from male and female rat pups separately and grown to confluence in estrogen-free media. Cell death in response to OGD, alone or in combination with hydrogen peroxide, lipopolysaccharides, interleukin-1β, tissue necrosis factor-α, or nitric oxide (NO) donor diethylenetriamine/nitric oxide adduct (DETA/NO) was quantified as the ratio of propidium iodide to calcein AM-positive cells. Aromatase activity and cyp19 mRNA and protein levels were measured in cultures from each sex. Female astrocytes are more resistant to OGD and oxidant cell death induced by H2O2, but sustain greater cell death when inflammatory mediators are combined with OGD compared with OGD alone. Media transfer from female to male cells conferred protection against OGD-induced cell death. Aromatase activity and expression is greater in female than in male astrocytes. The aromatase inhibitor, Arimidex (100 nmol/L), abolishes sex differences in OGD-induced cell death, whereas treatment with 17β-estradiol (10 nmol/L) protects cells of either sex. We conclude that astrocytes isolated from neonatal cortex exhibit marked sex differences in sensitivity to OGD, in part because of enhanced aromatization and estradiol formation in female cells.
Keywords
Introduction
Women in their reproductive years enjoy protection from ischemic stroke relative to age-matched men (Thom, 1993; Sarti et al, 2000). Such sex differences have been historically attributed to the protective effect of female reproductive steroids. Female rats or mice from inbred and outbred strains experience smaller tissue damage for an equivalent insult from focal or global cerebral ischemia (Hall et al, 1991; Carswell et al, 1999; Alkayed et al, 2000), as well as improved functional outcome (Li et al, 2004), and the protection is partially because of the action of 17β-estradiol (Simpkins et al, 1997). However, sexual dimorphism in human stroke risk persists well beyond the menopause (Sacco et al, 1998). Furthermore, female sex is associated with favorable outcome from ischemic stroke in children (Fullerton et al, 2003) and after brain injury in newborns (Lauterbach et al, 2001; Donders and Hoffman, 2002; Ingemarsson, 2003), suggesting that ovarian estradiol may not fully account for the female phenotype.
In brain, estradiol can be synthesized locally from testosterone via the P450 aromatase. Mice with targeted deletion of P450 aromatase (encoded by cyp19) sustain increased brain damage after middle cerebral artery occlusion as compared with wild-type littermates or to ovariectomized females (McCullough et al, 2003). Accordingly, brain aromatase and local estradiol production may be important to ischemic pathophysiology. Consistent with this hypothesis, aromatase activity and expression are induced in brain after injury in astrocytes (Garcia-Segura et al, 1999; Azcoitia et al, 2003). More recently, Carswell et al (2005) showed that aromatase expression is also induced in astrocytes after experimental stroke. The purpose of the present study was to examine differential sensitivity of male and female astrocytes to oxygen-glucose deprivation (OGD) and to other cytotoxic challenges such as lipopolysaccharides (LPS), cytokines interleukin-1β (IL-1β) and tissue necrosis factor (TNF-α), nitric oxide (NO), and hydrogen peroxide. We hypothesized that female cells in culture would be relatively insensitive to OGD and oxidant stress, by a mechanism involving the P450 aromatase.
Materials and methods
The study was conducted in accordance with the National Institutes of Health guidelines for care and use of animals in research, and protocols were approved by the institutional IACUC.
Sex Determination
Astrocytes were cultured from 1- to 3-day-old male and female Sprague–Dawley pups separately (dams from Charles River, Wilmington, MA, USA). Male and female rat pups were distinguished by a larger genital papilla and longer ano-genital distance in male than female pups. Sex was confirmed by inspecting internal organs and gonads after laporatomy, for example, uterine horns in females and ductus deferens in males, and by multiplex polymerase chain reaction (PCR) using male-specific marker SRY (sex determination region on the Y chromosome, responsible for testes formation) and the universal marker myogenin (Myog), which is expressed in both males and females, as described previously (McClive and Sinclair, 2001).
Cell Culture
Primary cultured cortical astrocytes were prepared, as described previously (Liu and Alkayed, 2005). Cell suspension was diluted with a feeding media consisting of 10% charcoal-stripped, estrogen-free fetal bovine serum (HyClone, Logan, UT, USA) in phenol red-free Dulbecco's modified Eagles's medium (DMEM) (Invitrogen, Grand Island, NY, USA) with 1% penicillin-streptomycin. Cells were seeded in 100-mm dishes and 75-cm2 flasks for aromatase expression and activity studies, and in 24-well plates for cell death assay, and incubated at 37°C in a 95%/5% mixture of atmospheric air and CO2. Feeding media were changed after 2 days and subsequently twice a week. Confluent monolayers (10 to 14 days in vitro (div)) of primary cortical cultures consisting of 98.4% ± 0.5% glial fibrillary acidic protein-positive cells were used, as described previously (Liu and Alkayed, 2005).
Oxygen-Glucose Deprivation
At 10 to 14 div, the feeding media were replaced with oxygen-depleted, glucose-, serum-, and phenol red-free DMEM, and cultures were incubated in a hypoxic chamber perfused with 94% N2/5% CO2/1% O2 for 6 h. Immediately after OGD, the medium was replaced with fresh feeding media containing glucose, and cells were returned to normoxia for 24 h. Cell death was estimated by the ratio of propidium iodide (PI, dead)- to calcein AM (live)-positive cells, as described previously (Liu and Alkayed, 2005). In separate experiments, the following reagents were added 30 mins before OGD, and concentrations were maintained during OGD and re-oxygenation: 17β-estradiol (10 nmol/L), LPS (25 μg/ml), IL-1β (15 ng/ml), and TNF-α (20 ng/ml), hydrogen peroxide (H2O2, 50 μmol/L), NO donor diethylenetriamine/nitric oxide adduct (DETA/NO, 500 μmol/L), and Arimidex (anastrozole, 100 nmol/L).
Aromatase Activity Assay
Cells were rinsed with ice-cold PBS, scraped, andcentrifuged at 4°C for 8 mins at 5,000 r.p.m. Pellets were homogenized in 30 vol of phosphate buffer (10 mmol/L KH2PO4, 100 mmol/L KCI, 1 mmol/L ethylenediaminetetraacetic acid, and 1 mmol/L dithiothreitol, pH 7.4). Aliquots of the homogenates (100 μl) were incubated for 1 h at 37°C with 0.3 μmol/L [1β-3H]androstenedione in the presence of NADPH-generating system. The 3H2O generated during incubation was purified through charcoal and Dowex minicolumns and quantified by liquid scintillation spectrophotometry, as described previously (Roselli and Resko, 1993). Results are expressed as fmol 3H2O produced/h/mg protein.
Western Blot Analysis
Protein was prepared as described previously (Liu and Alkayed, 2005). Microsomal proteins (100 μg) were boiled for 5 mins before loading on 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis. Samples were transferred to PVDF membrane, blocked for 2 h in TBS containing 5% nonfat dry milk and 0.05% Tween 20 at room temperature, and incubated overnight at 4°C with one of two anti-aromatase antibodies: a commercially available monocolonal antibody that recognizes human aromatase amino-acid sequence 376–390 (Serotec, Raleigh, NC, USA; 1:5,000) or a polyclonal anti-peptide antibody developed by one of the authors (Dr Roselli) in chicken against aromatase amino-acid sequence 98–115. Antibody solution was incubated in TBST containing 5% normal sheep serum, followed by incubation with peroxidase-coupled goat anti-mouse or goat anti-chicken antibody (1:5,000) for 1 h at room temperature. The signal was developed by enhanced chemiluminescence and visualized on a Kodak Gel Imaging Station. Blots were stripped and re-probed with anti-β-actin antibody (1:2,500) as an internal control for loading.
RNA Preparation and Reverse Transcriptase-Polymerase Chain Reaction
Astrocytes were rinsed with ice-cold PBS, scraped, and transferred to RNAse-free tubes for extraction of total RNA using the RNA kit (RNeasy, Qiagen, Valencia, CA, USA). RNA was reverse-transcribed with SuperScript™ III First-Strand Synthesis System (Invitrogen), and first-strand cDNA was amplified by PCR using the following cyp19 aromatase primers: TGGAC GTGGT GACCC TCATG (sense) and CCACC AGAAT TTCGA TCTCGT (antisense) (GenBank # M33986). The size of PCR product was 224 bp, and β-actin was measured as an internal control. The PCR reaction was performed in a mixture containing 2 μl first-strand cDNA, 0.4 μmol/L each of primers, 1.5 mmol/L MgCI2, 0.2 mmol/L dNTP mixture, and 1.25 U Taq polymerase (Invitrogen) in a total of 50 μl of 20 mmol/L Tris-HCI (pH 8.4). The template was denatured by incubation at 94°C for 1 mins, and cDNA was amplified over 35 cycles of 94°C for 40 secs, 55°C for 30 secs, and 72°C for 45 secs. The reaction was terminated by an additional incubation of 5 mins at 72°C. Product was resolved by electrophoresis in 2% agarose gel and visualized by ethidium bromide staining.
Statistical Analysis
Brains from male and female pups were pooled into separate groups. Cells prepared from each pool constitutes one experiment (n = 1). The number of experiments (n) for any sex refers to number of cultures prepared from different litters. Differences between male and female astrocytes were examined using Student's t-test. The effect of Arimidex or vehicle on male and female astrocytes, and the effect of cytokines on male and females were determined by ANOVA with post hoc Newman–Keuls. Values are presented as mean + s.e.m. with statistical significance at P < 0.05.
Results
Visual sex identification was highly reliable (100% accuracy, n = 6), as determined by subsequent PCR confirmation of sexual genotype. Figure 1 illustrates the method of PCR-based sexing of male (M) and female (F) cultures. When subjected to 6-h OGD, F astrocytes were more resistant to OGD than male cells (Figure 2A). Cell death at 24 h was 19% ± 4% (n = 13) in F, as compared with 41%± 5% (n =7) in M (Figure 2B) (P < 0.05). To determine if sex-specific sensitivity is unique to OGD, we compared responses to other cytotoxic agents. F cells were more resilient to oxidant stress induced by H2O2 exposure (27%± 6% (n = 4) versus 51%± 7% (n = 4) in M, P < 0.05). No cell death was observed in either sex at 24 h after exposure to NO donor DETA/NO.
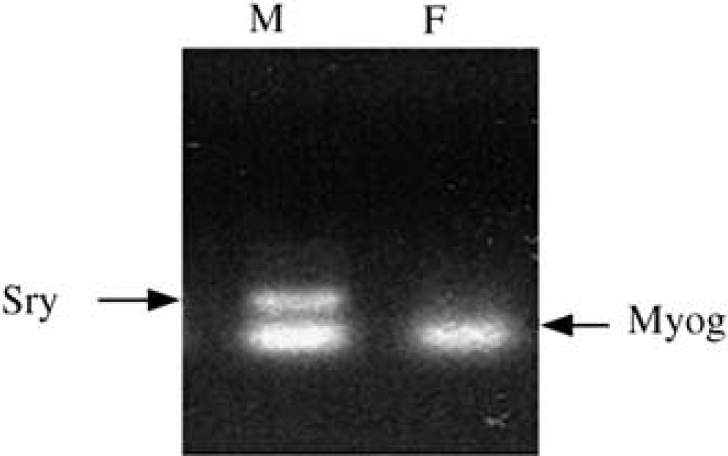
Sex in culture was determined by PCR using male-specific SRY and universal marker myogenin (Myog).
We observed no cell death in response to LPS, TNF-α, or IL-1β under baseline conditions. However, as illustrated in Figure 2C, LPS, IL-1β, and TNF-α exacerbated OGD-induced cell death in F (from 19% ± 4% in OGD alone to 33% ± 5% (n = 7, P < 0.05), 32% ± 4% (n = 5, P < 0.05), and 31% +4% (n = 5, P < 0.05) when OGD was combined with LPS, IL-1β, and TNF-α, respectively). In contrast, these agents did not alter OGD-induced cell death in M (41%± 5% (n = 7) in OGD alone versus 40% ±6% (n =7, P > 0.05), 40% ± 6% (n = 5, P > 0.05), and 34% ± 5% (n = 5, P > 0.05) in OGD plus LPS, IL-1β, and TNF-α, respectively). There were no differences between male and female astrocytes when inflammatory mediators were applied in combination with OGD.
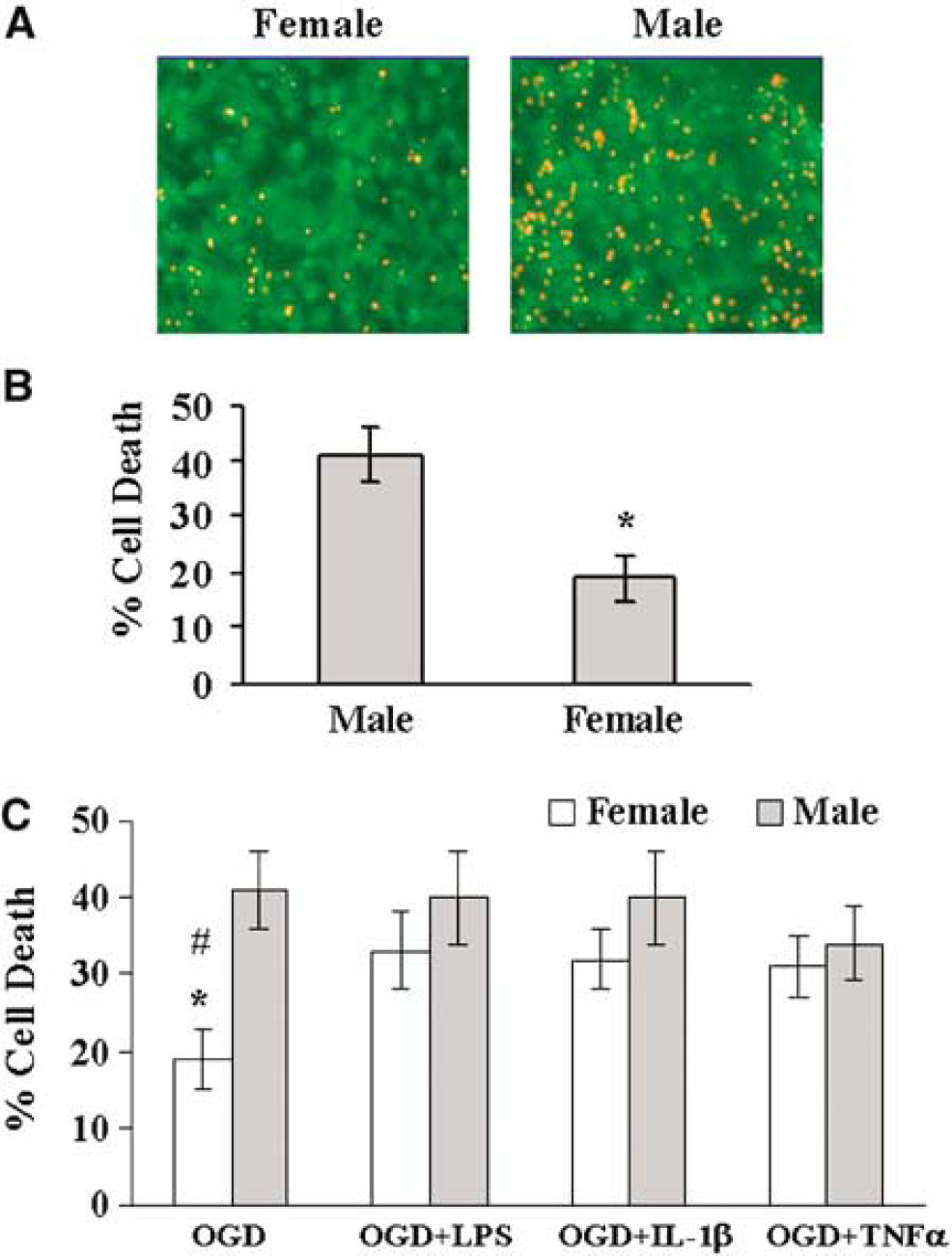
(
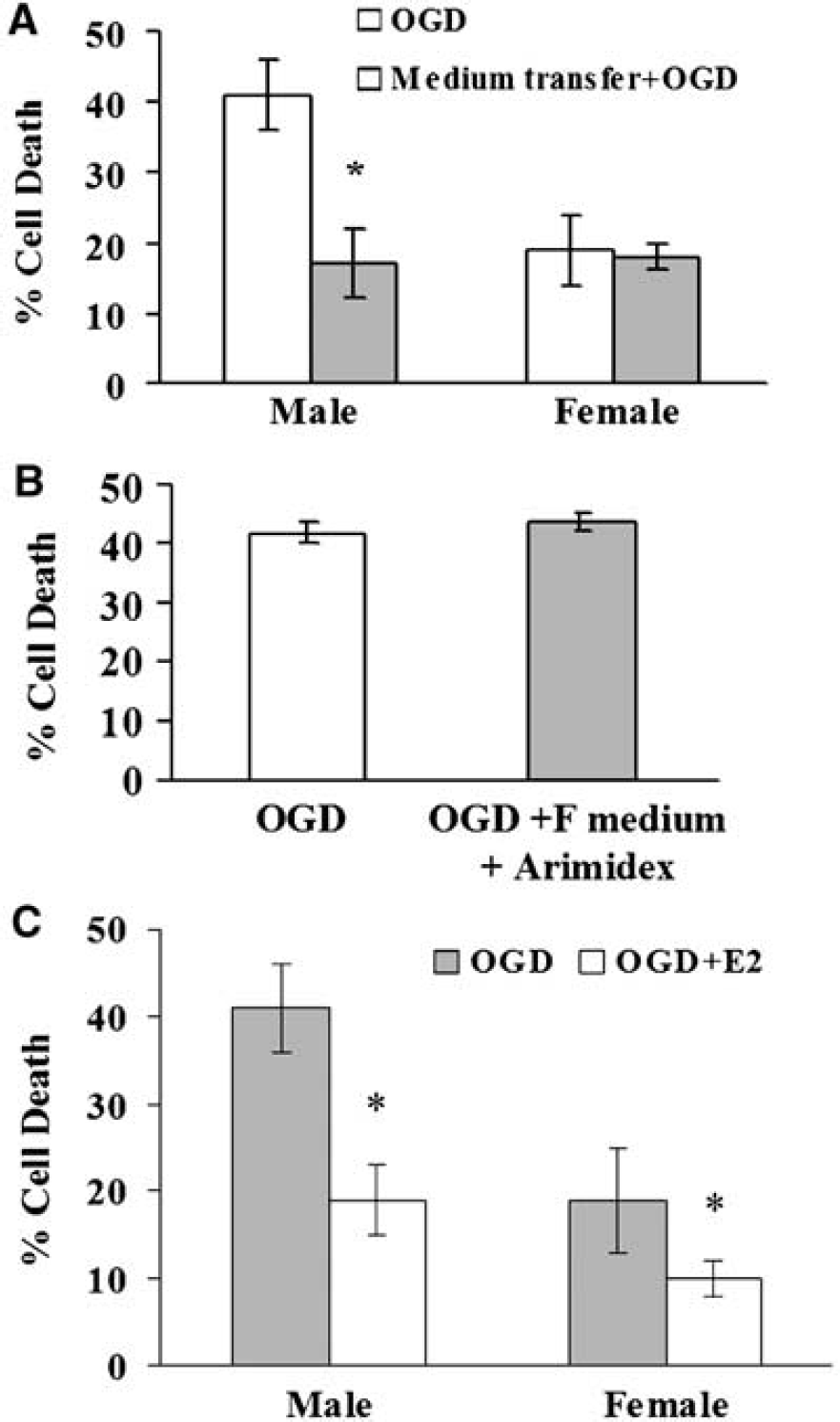
To determine if these responses were related to differences in P450 aromatase activity or expression, we compared cyp19 aromatase mRNA, protein, and enzymatic activity in both sexes (Figures 3A and 3B). In Figure 3C, Western blot analysis using a monoclonal antibody shows that P450 aromatase protein is higher in F than M astrocytes (2.3 ± 0.1-fold, n = 6 per group, P < 0.05). Similar results (2.6 ± 0.1-fold higher in females compared with males, n = 6, P < 0.05) were obtained using a polyclonal antibody that recognizes a different epitope. In addition, aromatase activity was higher in F cells (25 ± 5 fmol 3H2O/mg/h, n = 11 versus 13 ± 2 fmol 3H2O/mg/h, n = 15 in M, P < 0.05, Figure 4A). To determine if differences in aromatase activity mediate the sex-specific response to OGD, we treated cultures with the aromatase inhibitor, Arimidex. Although the inhibitor had no effect in M, it markedly increased death in F and abolished sex-specific responses to OGD (40% ± 2%, n = 10 in F versus 43%± 1%, n = 10 in M, P > 0.05; Figure 4B).
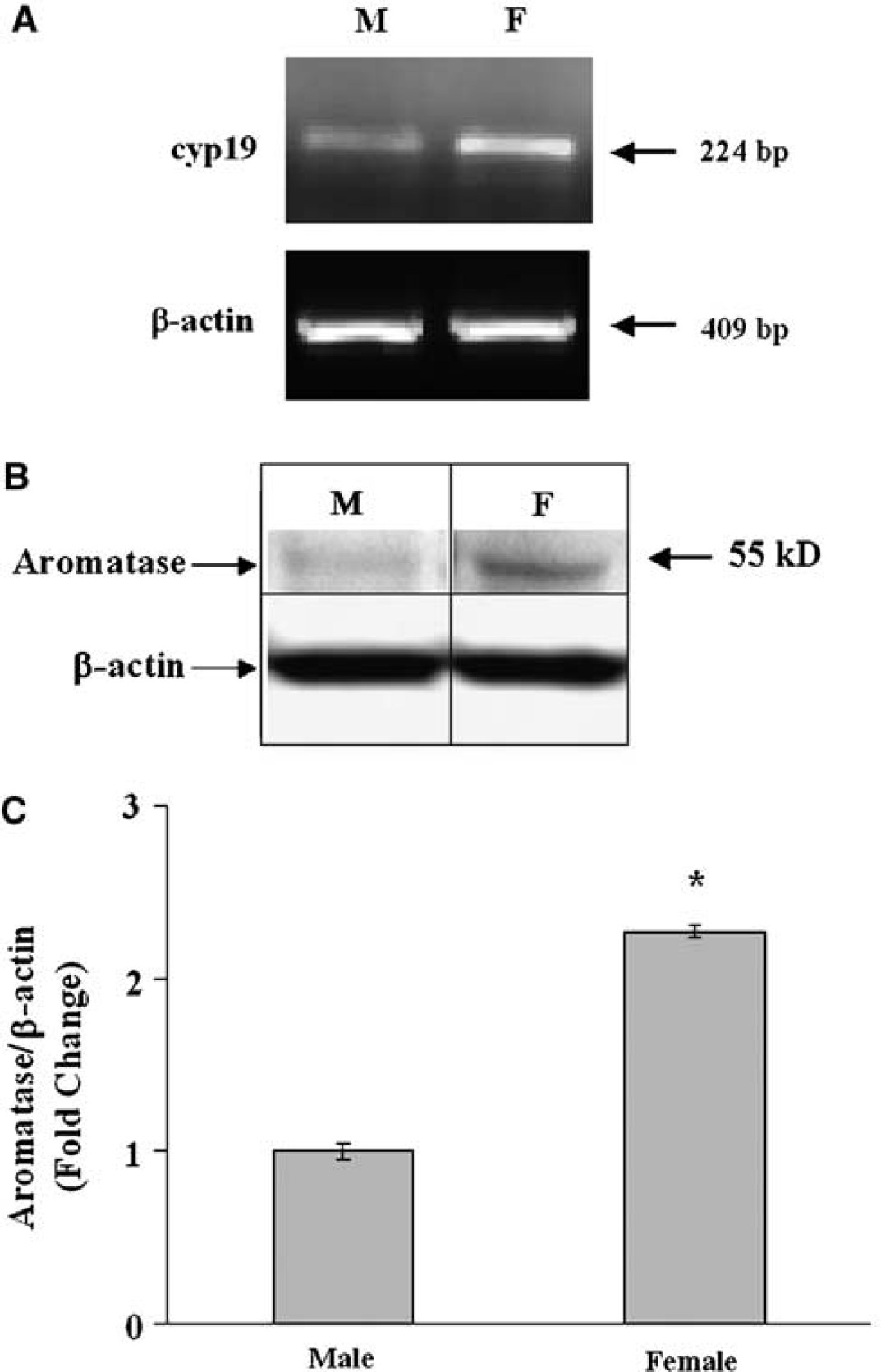
Higher P450 aromatase mRNA and protein in F than M astrocytes, as determined by RT-PCR for cyp19 mRNA (
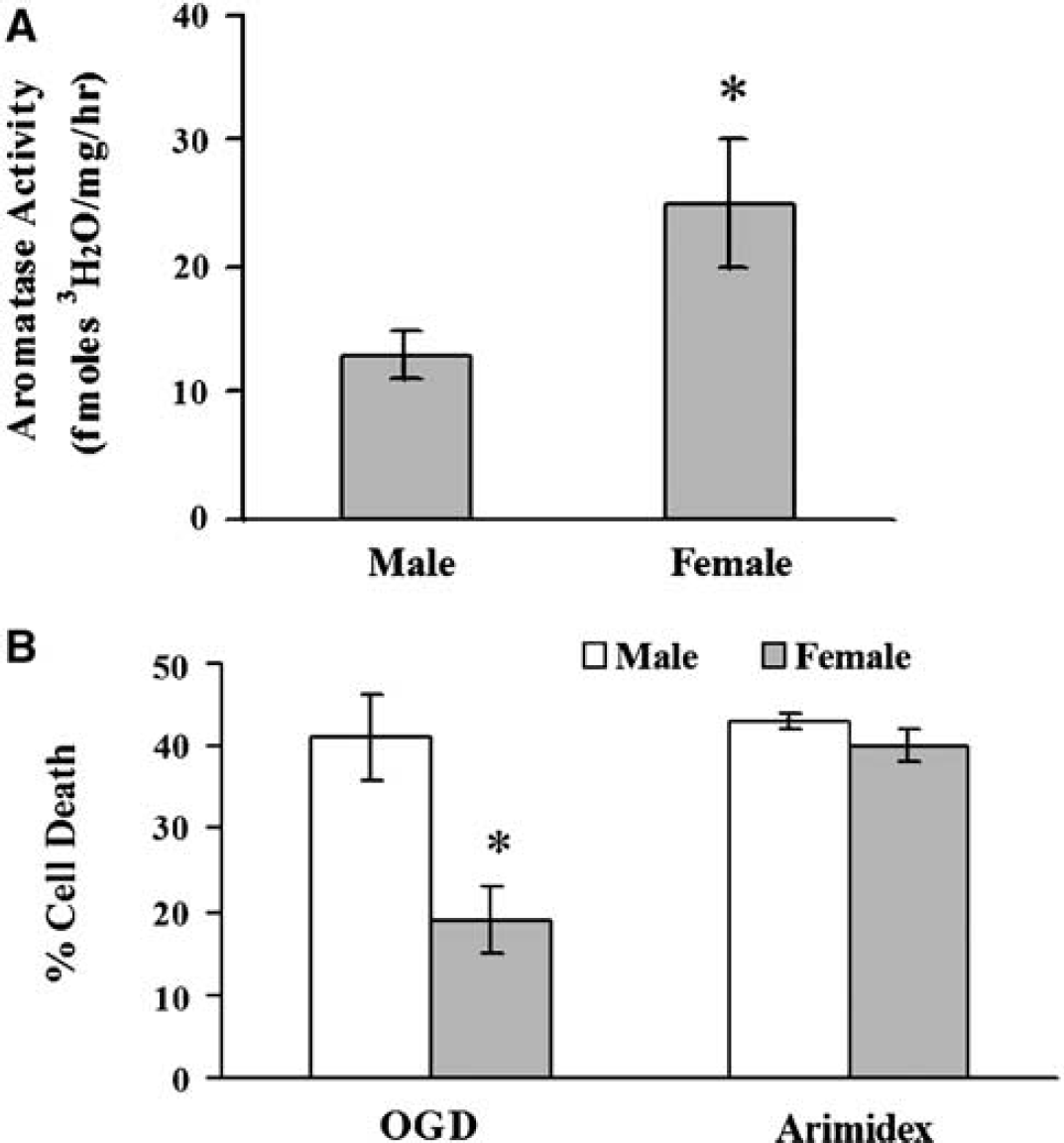
(
We reasoned that if estradiol is released into the media, astrocyte-conditioned media from female cultures would protect male cultures exposed to OGD. Media from F conferred protection against OGD-induced cell death in M (from 41%± 5% (n = 7) to 17% ± 5% (n = 6), P < 0.05), whereas transfer from M to F did not alter outcome (19%±4%, n = 13 compared with 18%± 6%, n = 6, respectively, P > 0.05) (Figure 5A). However, when pretreated with Arimidex, conditioned medium from F astrocytes was unable to protect M astrocyte (44%± 5% (n = 5), P > 0.05; Figure 5B). Direct application of aromatase product 17β-estradiol reduced cell death in both sexes (Figure 5C).
(
Discussion
There are three important findings in this study. First, female astrocytes are more resistant than their male counterparts to OGD and oxidant cell death induced by H2O2. This resilience appears to be specific to injury mechanism. Second, female astrocytes express higher cyp19 mRNA and protein and exhibit higher P450 aromatase activity than male cells under basal conditions. Further, inhibition of the enzyme's function blunts female resistance to OGD. Third, female cells release a protective factor(s) that is transferable to male cells. The aromatase product, estradiol, is one potential candidate, as exogenous estradiol application protects against OGD-induced cell death in both sexes. We conclude that there are sex-specific astrocytic responses to injury, in part because of a higher aromatase expression and activity in female cells.
This is the first demonstration of sex-specific astrocyte cell death in vitro. Our observations, however, are in agreement with previous reports in neurons showing that survival over time is greater in female than male cultures (Zhang et al, 2003). Furthermore, neuronal death after exposure to various cytotoxins is sex-specific, male cells being more sensitive to nitrosative stress and excitotoxicity than female neurons (Du et al, 2004). This is also in agreement with an earlier report showing that female dopaminergic neurons are more tolerant to dopamine-induced cell death compared with male neurons (Lieb et al, 1995). In contrast, female neurons were more susceptible to apoptotic cell death induced by etoposide and staurosporine (Du et al, 2004). Our results in astrocytes show that sex differences in ischemic cell death extend from the intact animal to the cellular level. These experiments were conducted in steroid-free media, indicating that these sex differences are either independent of sex steroids or that they arose as a result of exposure to sex steroids in the fetal and perinatal periods.
Initial sexual differentiation in brain has been attributed to the expression of male sex-determining gene sry located on the Y chromosome, which then initiates testicular development, androgen production, and masculinization from an otherwise female default sex (Arnold et al, 2003). After the onset of puberty, the adult brain is exposed to sex hormones, without doubt shaping sensitivity and response to cerebrovascular insult. However, the differences in cell survival reported here arise in male and female neonatal tissue and suggest that these cells diverge in phenotype early in life and in brain areas specialized for reproductive function. Further work is needed to determine if sex-specific sensitivity to ischemia is conferred by fetal and perinatal exposure to sex steroids, or this is a steroid-independent property of XX versus XY chromosomal content. Although female astrocytes are resistant to OGD and to oxidant stress, suggesting higher antioxidant capacity, they appear to be more susceptible to inflammatory stimuli when these stimuli are combined with OGD. As shown by Falsig et al (2004), cytokines do not induce cell death in astrocytes on their own; however, they induce a switch in resting astrocytes to a reactive phenotype, rendering them more susceptible to cell death-inducing stimuli. It is, therefore, possible that this change in phenotype occurs more readily in female compared with male astrocytes, suggesting that inflammatory mechanisms may be more important contributors to ischemic cell death in female than male brain. However, it is also possible that the apparent insensitivity of male cells to inflammatory cytokines is simply because death rate in male astrocytes has reached its maximum. The brain's inflammatory response to ischemia has both beneficial and detrimental roles (Wang and Feuerstein, 2004), depending on its magnitude, cell type, timing, as well as ratios and distribution of various inflammatory molecules (Zheng and Yenari, 2004). Similarly, NO is toxic to neurons, but it may play a protective role in astrocytes, as shown by Almeida et al (2004). This may explain why NO donors had no effect on astrocytes death in our study.
The protection enjoyed by female astrocytes against OGD-induced cell death appears to be related, in part, to higher expression of P450 aromatase relative to the male. In support of this notion, pharmacological inhibition of P450 aromatase with Arimidex abolished the sex difference in astrocyte cell death and eliminated the ability of female astrocytes to protect male astrocytes. Surprisingly, Arimidex did not increase cell death in male astrocytes. There are at least two possibilities for why aromatase inhibition did not increase cell death in male astrocytes. First, because the levels of aromatase activity and expression are much lower in male compared with female astrocytes, it is possible that aromatase plays less of a role in protecting male astrocytes from OGD than female cells. Therefore, aromatase inhibition may have less of an impact on cell death in male than female astrocytes. Second, the decreased level of aromatase in male cells may render it less sensitive to pharmacological inhibition, so that it may require a higher dose of Arimidex to achieve significant inhibition in male compared with female astrocytes.
The expression of P450 aromatase in rodent and bird brain under baseline conditions is confined to neuronal cell bodies and processes. However, in a recent study by Yague et al (2006), it was shown that P450 aromatase is expressed in human brain in subpopulations of pyramidal neurons and astrocytes. Furthermore, as shown by Azcoitia et al (2003) and Saldanha et al (2005), P450 aromatase is induced in rat and zebra finch brain after injury in astrocytes. Finally, our study and that of others (Zwain and Yen, 1999; Schlinger et al, 1994) show that the P450 aromatase is expressed in astrocytes in culture without apparent injury. The expression of P450 aromatase in cultured astrocytes may indicate that astrocytes in culture exhibit signs characteristic of a reactive phenotype, which may be because of some degree of cellular stress under culture conditions. In vivo, Garcia-Segura et al (1999) showed that P450 aromatase is induced in brain in both male and female rats and mice after kainate injury. However, in a semiquantitative analysis to estimate the number of immunoreactive glial cells in a limited number of rats and mice, the authors in that study did not observe a sex difference in the number of distribution of imunoreactive glial cells.
We hypothesize that heightened aromatization capability in female astrocytes yields high 17β-estradiol production, which is subsequently secreted into the culture media. We did not directly quantify estradiol secretion into the media, because concentrations are below levels detectable by radioimmunoassay. However, exogenous estradiol protected both male and female cells. This finding is in agreement with previous reports that estradiol protects neurons in vitro from excitotoxins (Singer et al, 1996; McCullough and Hurn, 2003).
Sex differences in aromatase expression and regulation have been previously reported in brain regions known to be sexually dimorphic in function (Roselli and Resko, 1993). However, in contrast to astrocytes, aromatase levels in the medial preoptic nucleus and other sexually dimorphic nuclei are higher in adult males than females (Lavaque et al, 2006; Peterson et al, 2005). Similarly, aromatase expression and activity are higher in male than in female cultured rat hypothalamic neurons (Negri-Cesi et al, 2001). Aromatase expression and activity have also been detected in brain regions other than sexually dimorphic hypothalamic regions, including cerebral cortex (Lephart et al, 2001; Stoffel-Wagner, 2003). Aromatase activity is controlled at the cellular level by increasing or decreasing mRNA expression (Simpson et al, 2002). In brain, transcriptional control is through a tissue-specific promoter (Simpson, 2003) and can be influenced by a variety of hormones, transcription factors, and signaling molecules (Bulun et al, 2003).
Lastly, the concept that astrocyte cell survival is linked to aromatase activity is consistent with previous observations that the enzyme is induced in astrocytes in situ after brain injury in vivo and results in reduced tissue damage (Azcoitia et al, 2003). There is increasing recognition that astrocytes play an important role in brain function and disease, and that they provide trophic support to neurons and protect neurons from ischemic and other injuries (Giffard and Swanson, 2005) through multiple mechanisms, including extracellular glutamate uptake, buffering oxygen-free radicals, and, according to our study, release of neuroprotectants such as estradiol. Therefore, if female astrocytes are more resistant to ischemic injury and produce more estradiol after injury than male astrocytes, then that would translate to higher overall resistance and better neuronal survival in female than male brain.
We show here that aromatase is differentially expressed in male and female astrocytes, and may underlie sex-specific sensitivity to ischemic and oxidant injury. These data and that of others in neurons (Lieb et al, 1995; Du et al, 2004) emphasize that males and females may have inherently different susceptibility to cerebral ischemia. Recent studies also suggest that fundamental mechanisms of brain injury and response to therapy may also be different between male and females. For example, the N-methyl-D-aspartate receptor antagonist AP-5 rescues female, but not male, neurons from dopamine-induced cell death (Lieb et al, 1995). Nitric oxide and poly-ADP ribose polymerase are neurotoxic in males, but appear to be protective in females (McCullough et al, 2005). Similarly, programmed cell death (PCD) proceeds in male neurons via apoptosis-inducing factor, whereas in female cells, PCD predominantly proceeds via cytochrome c-dependent pathways (Du et al, 2004).
In conclusion, astrocytes isolated from neonatal cortex exhibit marked sex differences in sensitivity to OGD and oxidant injury, in part because of enhanced aromatization capacity in female cells. These findings underscore the importance of studying male and female cells separately to better understand sex-specific molecular death and survival pathways. The net result of this strategy may be more effective intervention strategies for clinical stroke.
