Abstract
Decreased cerebral blood flow (CBF) in hydrocephalus is believed to be related to increased intracranial pressure (ICP), vascular compression as the result of enlarged ventricles, or impaired metabolic activity. Little attention has been given to the relationship between cardiac function and systemic blood flow in chronic hydrocephalus (CH). Using an experimental model of chronic obstructive hydrocephalus developed in our laboratory, we investigated the relationship between the duration and severity of hydrocephalus and cardiac output (CO), CBF, myocardial tissue perfusion (MTP), and peripheral blood flow (PBF). Blood flow measures were obtained using the microsphere injection method under controlled hemodynamic conditions in experimental CH (n = 23) and surgical control (n = 8) canines at baseline and at 2, 4, 8, 12, and 16 weeks. Cardiac output measures were made using the Swan–Ganz thermodilution method. Intracranial compliance (ICC) via cerebrospinal fluid (CSF) bolus removal and infusion, and oxygen delivery in CSF and prefrontal cortex (PFC) were also investigated. We observed an initial surgical effect relating to 30% CO reduction and ∼50%> decrease in CBF, MTP, and PBF in both groups 2 weeks postoperatively, which recovered in control animals but continued to decline further in CH animals at 16 weeks. Cerebral blood flow, which was positively correlated with CO (P = 0.028), showed no significant relationship with either CSF volume or pressure. Decreased CBF correlated with oxygen deprivation in PFC (P = 0.006). Cardiac output was inversely related with ventriculomegaly (P = 0.019), but did not correlate with ICP. Decreased CO corresponded to increased ICC, as measured by CSF infusion (P = 0.04). Our results suggest that CH may have more of an influence on CO and CBF in the chronic stage than in the early condition, which was dominated by surgical effect. The cause of this late deterioration of cardiac function in hydrocephalus is uncertain, but may reflect cardiac regulation secondary to physiologic response or brain injury. The relationship between cardiac function and CBF should be considered in the pathophysiology and clinical treatment of CH.
Introduction
Chronic hydrocephalus (CH), characterized by enlarged cerebral cerebrospinal fluid (CSF) ventricles with or without increased intracranial pressure (ICP), has often been associated with a global reduction in cerebral blood flow (CBF) (Graff-Radford et al, 1987; Mamo et al, 1987; Larsson et al, 1994; Tanaka et al, 1997; Chang et al, 1999; Mori et al, 2002; Klinge et al, 2002a, b ; Mataro et al, 2003; Owler et al, 2004; Momjian et al, 2004) and a disruption of cerebral autoregulation (Czosnyka et al, 2002, 2003; Minhas et al, 2004). The reduction of CBF seen in CH is believed to be caused by increased CSF pressure and ventricular volume (VV) (Wozniak et al, 1975; Mabe et al, 1990; Nakada et al, 1992; Goh and Minns, 1995), which results in cortical compression, and blood vessel and white matter fiber stretching (Del Bigio, 1993; Edwards et al, 2004). These structural brain changes leading to decreased CBF may be directly responsible for ischemic conditions in CH, and have been shown to be reversible with CSF shunt treatment (Del Bigio and Bruni, 1988a, b ; Del Bigio, 1989; Fukuhara et al, 2001; Luciano et al, 2001). While in acute hydrocephalus increased ICP, decreased cerebral perfusion pressure, and blood vessel compression are most likely the primary determinants of decreased CBF, in CH these may not play as significant a role. Other factors that affect CBF, such as changes in metabolic activity, systemic blood flow, and autoregulation, may play a more significant role in the chronic condition.
Using an experimental model of chronic obstructive hydrocephalus developed previously in our laboratory (Johnson et al, 1999; Fukuhara et al, 2001; Luciano et al, 2001), we investigated the relationship between the duration and severity of CH and cerebral and systemic blood flow, cardiac function, tissue oxygen delivery in the prefrontal cortex (PFC), and intracranial compliance (ICC). We hypothesize that in CH, cardiac and systemic factors are the primary determinants leading to decreased CBF.
Materials and Methods
Animals
Thirty-one (n = 31) young adult, male hounds (canis familiaris), approximately 8 to 9 months of age, weighing 22 to 30 kg were used in this study. Animals were divided into two groups: obstructive CH (N = 23) and surgical controls (SC, N =8). Animals were obtained from licensed suppliers and quarantined for a minimum of 7 days before entering into the study. All animals were maintained in the Cleveland Clinic Foundation fully accredited Animal Care Facility in accordance with Public Health Service (PHS) policy and the Health Research Extension Act (PL99-158) under the rules and regulations of the Guide for the Care and Use of Laboratory Animals.
Surgery
The surgical procedure to induce chronic obstructive hydrocephalus in an experimental animal was previously developed and investigated in our laboratory (Johnson et al, 1999; Fukuhara et al, 2001; Luciano et al, 2001). In brief, each animal received a thoracotomy for cannulation of the left atrium, and a suboccipital craniectomy for the induction/sham -induction of hydrocephalus under general anesthesia (1.0% to 1.5% isoflurane) and sterile conditions. Presurgical medications included Dilantin (5 mg/kg, intravenous) to prevent postoperative seizures, dexamethasone (0.25 to 1.0 mg/kg, intravenous) to reduce inflammation, glycopyrrolate (0.01 mg/kg, intravenous) to reduce respiratory secretions, and gentamicin (3 mg/kg, intravenous) and cefazolin (1 g, subcutaneous) to prevent infection.
Left Atrium Catheterization and Microsphere Injection
In each animal, the left atrium was cannulated (Access Technologies, Skokie, IL, USA; #CNC-7IS) and a subcutaneous portal system (Access Technologies, #CP2) was implanted for CBF microsphere technique. Hemodynamic parameters were monitored via femoral artery (pH = 7.4; PaCO2 (35 to 45 mmHg); PaO2 (150 to 300 mmHg); O2 saturation (95% to 100%)). Approximately 10 million (4 mL) microspheres (BioPal™, Medford, MA, USA) were injected into the left atrial (LA) portal system, while a reference blood sample was collected from the femoral artery as a comparative baseline blood flow measure. Randomly chosen, colored microspheres were selected for different time points (i.e., baseline, 2, 4, 8, 12, and 16 weeks) to measure CBF change over time.
Induction of Chronic Obstructive Hydrocephalus
In brief, animals were placed in a prone position in a stereotaxic headframe, and a sub occipital craniectomy was performed under sterile conditions to access the CSF fourth ventricular space. A flexible, silicon catheter (1.5 mm outer diameter (OD)) was then inserted into the fourth ventricle and approximately 0.4 to 0.6 cm3 of cyanoacrylic gel was injected. Surgical control animals were generated using procedures identical to those described for experimental CH animals, with the only exception being the substitution of saline instead of the gel.
Postoperative Care
Immediately postoperatively, all animals (i.e., CH and SC) received Dexamethasone (0.5 mg/kg, four times daily (q.i.d.), intravenous), Gentamicin (3 mg/kg, q.i.d., intravenous) and Cefazolin (1 g, q.i.d., subcutaneous). The animals also received Tylenol in combination with Butorphanol (0.2 mg/kg, intravenous), buprenorphine (0.02 mg/kg, intravenous), or buprenex (0.01 to 0.03 mg/ kg i.v., or subcutaneous (s.q.) thrice daily (t.i.d.)) for the management of pain. Oral antibiotics (cephalexin, 1 g, twice daily (b.i.d.), per os (p.o.)) were administered to prevent infection for 5 days postoperatively. Routine postoperative care was provided for pain, hydration, and infection, and included regular examination of vitals, pupillary reflex, and leg responsiveness. Dextrose feeding (intravenous) was initiated in two animals in which prolonged food/water abstinence occurred. In CH animals, medical treatment for increased ICP included mannitol (1.5 mg/kg) and decadron (0.5 mg/kg). To control for drug treatment differences, all animals received the same dose of mannitol b.i.d. during the immediate postoperative period in CH (48 h) and SC (24h), and discontinued when the animal recovered well enough to return to normal housing. Similarly, intravenous fluids were limited to a rate of 25 mL/h until animals (CH and SC) were able to drink independently. Body temperature, respiratory rate, heart rate (HR), urine production, activity, fluid intake, and intravenous fluid intake were recorded every hour.
Magnetic Resonance Imaging and Volumetric Analysis
Magnetic resonance images were collected before CH induction (i.e., baseline), magnetic resonance imaging (MRI) surgery, and again at killing (2, 4, 8, 12, or 16 weeks) for CH and SC groups. The MRI at baseline and killing were used to evaluate the anatomical severity and progression of hydrocephalus. 3D magnetic resonance images were acquired using a 1.5-T Siemens Vision Magnetom and used for volumetric analysis. Separate measures for brain and VV were obtained by manually tracing their contours on approximately 60 to 80 sections in the coronal plane of 1 mm thickness from MR digital images using a commercially available image analysis system (Microbrightfield™, Colchester, VT, USA) (Figure 1).
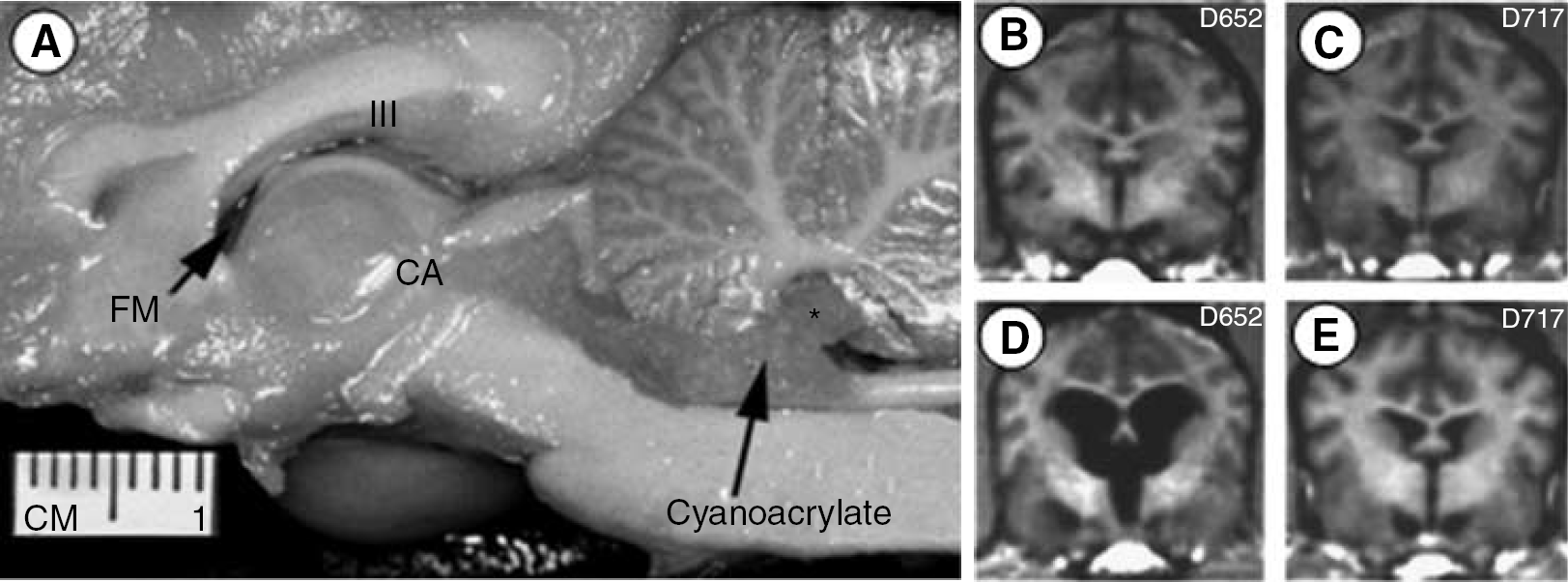
Gross hemi-section photograph in the sagittal plane (A) showing our model for chronic obstructive hydrocephalus (reprint modification from J. Neuroscience Methods, 1999) using cyanoacrylic injection into the fourth ventricle (*); confirmed on MRI for pre- and postsurgical induction of CH (
Cardiac Output
Cardiac function measures were obtained via the thermodilution method by pulmonary artery (Swan-Ganz) catheterization. Measures included cardiac output (CO) (L/min), ejections fraction (EF, %), end diastolic volume (EDV), HR (b.p.m.), aortic arterial pressure (AoP, mm Hg), pulmonary arterial pressure (PAP, mm Hg), central venous pressure (CVP, mm Hg), and wedge pressure (W, mm Hg). Cardiac output measures were obtained at baseline and before killing (2, 4, 8, 12, and 16 weeks).
Microsphere and Blood Flow Studies
Stable isotope labeled microspheres (BioPALTM, BioPhysics Assay Laboratory, Inc.). were injected directly into the arterial blood circulation via LA access port to determine regional blood flow. At killing, tissue samples (∼0.5g) were collected from the brain, heart, and peripheral organs. Brain tissue samples were obtained from eight regions: anterior and posterior medial frontal cortex, occipital cortex, superior temporal lobe, inferior frontal cortex, dorsolateral PFC, caudate nucleus, and cerebellar lobules I to VII from the right hemisphere in each animal. Cardiac samples included tissue collected from 13 regions of the left and right atria, and left and right ventricles. Peripheral organ tissue collection included samples from the liver, lung, kidney, and spleen. Tissue and blood samples were stored at 4°C until processed by BioPhysics Assay Labs (Worcester, MA, USA).
Intraoperative Intracranial Pressure and Oxygen (O2) Measurement
Intraoperative ICP and O2 measures were obtained for both CH and SC animals at baseline and before killing as described previously (Fukuhara et al, 2001). Hemodynamics conditions were mechanically controlled via ventilative volume and frequency. Under general anesthesia and sterile conditions, two small (5 mm) burr hole craniectomies were performed using stereotaxic coordinates to: (1) allow access to the CSF lateral ventricle and monitoring of CSF O2 saturation (O2-CSF) (Licox CC1-SB; Integra, Plainsboro, NJ, USA), and (2) monitor ICP (Camino, #110-4BT; Integra NeuroSciences) and tissue O2 saturation (O2-TISSUE) in the cortex. Intracranial pressure and O2 measurements were obtained online and in real time with a digital acquisition system and software (LICOX DAQ ver2.03.07, GMS-Integra, Germany). Intracranial pressure measurements were obtained continuously at normal ventilation, hyperventilation, and during CSF removal and infusion. Intracranial compliance was measured using a modification of the Masserman technique. A bolus of CSF (0.2 cm3) was withdrawn via the ventricular access catheter. After equilibrium was re-established, a bolus of CSF (0.2 cm3) was infused. Intracranial compliance was then calculated as the change in CSF volume divided by the change in ICP. Separate values were calculated for ICC removal (ICCr) and infusion (ICCi).
Killing and Tissue Preparation
For killing, animals were deeply anesthetized with sodium pentobarbital in combination with inhaled isoflorane and perfused with 4.0% paraformaldehyde (PFA) through the LA portal system. Brains were removed and postfixed for 24 h in 4% PFA for study of gross pathology, frozen sectioning, and routine histology (hematoxyline and eosin (H&E), cresyl violet). All CBF tissue samples were consistently obtained from the right hemisphere, while the left hemisphere was cut at 40 μm in the coronal plane and either stored long term or subsequently use for routine immunohistochemical analyses. In addition, ∼ 0.5 g brain tissue samples (described in the section Microsphere and Blood flow Studies) were collected for microsphere CBF analyses.
Statistical Analysis
The results of this study are expressed as mean ± the standard error of the mean. Statistical comparisons were made across six different time points (baseline, 2, 4, 8, 12, and 16 + weeks) using repeated-measures analysis of variance (ANOVA) and the Student Neuman-Keuls test. For analysis between CH and SC groups, two-tailed unpaired t-tests were used. Correlations between CSF volume and pressure, CBF, CO, oxygen delivery, and ICC were also made among time points and between groups. Multiple linear regression analysis was performed using CBF as the outcome (dependent) measure and CO, ventricular CSF volume and ICP, and time as independent variables. Statistical significance was accepted at the probability level less than 0.05.
Results
Confirmation of Chronic Obstructive Hydrocephalus
Overall, there was no incidence of mortality or morbidity in any animal for either the CH or SC groups. Animals recovered from anesthesia without complication and began eating and drinking within 36 h. In four CH animals, postoperative care was extended and assistance in feeding was necessary. Most CH animals exhibited signs of lethargy, motor weakness, anorexia, and ataxia in the first 7 days immediately after surgery. Vomiting often accompanied neurologic signs (i.e., pupillary dilation and reflex impairment) reflecting increased ICP in CH animals. These neurologic signs were observed to be transient and resolved within the first 1 to 2 days, with the exception of a few animals which were successfully managed with mannitol to reduce the ICP. In both the CH and SC groups, animals showed a similar reduction in body weight 2 to 4 weeks postoperatively, which returned to normal baseline levels after 12 weeks (Figure 2C). Statistically, there was no significant difference in weight across time points for either CH or SC group.
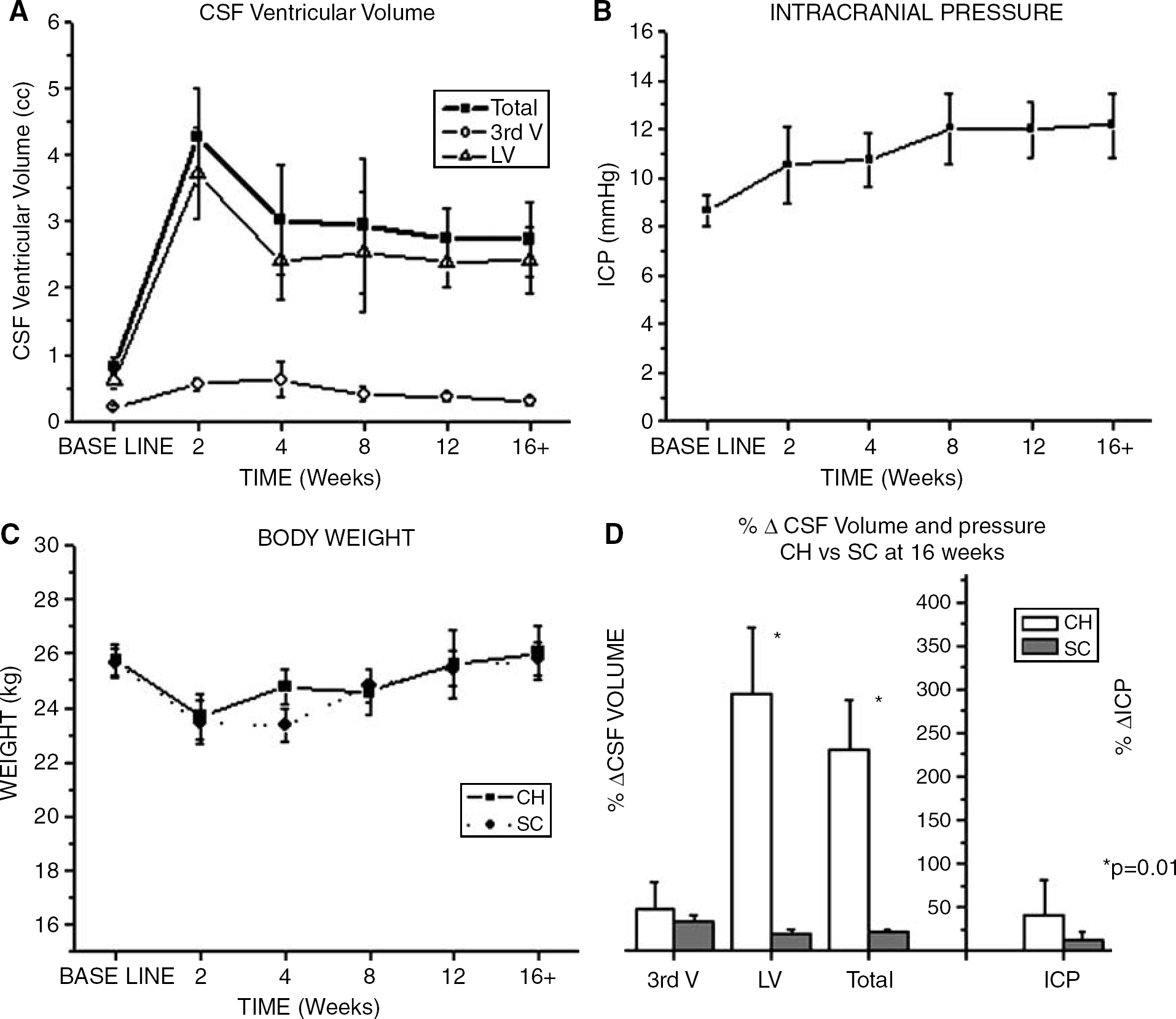
Average CSF volume in the lateral ventricles and third ventricle for the CH group (
Cerebrospinal Fluid Volume: Changes in CSF volume that were quantitated from MRI are graphically illustrated in Figure 2A for animals undergoing CH induction surgery. Overall, baseline CSF volume for both the third ventricle (0.04. to 0.45 cm3, mean 0.21cm3) and the lateral ventricle (0.05 to 1.75 cm3, mean 0.61 cm3) increased significantly at 2 weeks after CH induction surgery (P < 0.01), then decreased slightly, but remained significantly higher than baseline from 4 to 16 + weeks. The increase in CSF volume was shown to be greatest in the lateral ventricles (three- to sixfold) compared with the third ventricle (two- to threefold). Direct comparison of the percent change (%Δ) in CSF VV from baseline to 16 weeks was significantly greater in CH (232% ± 55%) than SC (21% ± 5%) (P < 0.01; Figure 2D).
Intracranial Pressure: Baseline ICP, which ranged from 4.5 to 16.0 mm Hg, mean 8.65 mmHg (Figure 2B), gradually increased, but was not significantly different at any time points after CH induction (ANOVA, P = 0.057). In the majority of CH animals, the average increase in ICP ranged from 21% (2 weeks) to 41% (16 + weeks) from baseline. There was no significant difference in the percent change (%Δ) of CSF pressure from baseline to 16 weeks between CH and SC groups (Figure 2D).
Gross Morphology and Histologic Analysis: Gross morphologic changes were observed in animals that were induced with CH. In animals with moderate to severe hydrocephalus (i.e., ventriculomegally), the brains were shown to have ventricular distension, sulcal widening, gyral flattening, cortical compression, and distortion. Immediately after termination of each animal, autopsy revealed no evidence of intracerebral or intraventricular bleeding, which corroborates with MRI. Histologic analysis using routine cresylate violet staining to identify architectonic boundaries and structures showed no signs of intracerebral bleeding or anomalous pathologic condition.
Cerebral Blood Flow
Overall, baseline CBF ranged from 0.441 mL/ming. (anterior cingulated gyrus) to 1.023 mL/ming (caudate nucleus), with an average of 0.59 mL/ming (Table 1). In general, anterior cortical regions had a lower CBF than posterior areas and deep structures. In addition, cortical regions that have almost exclusively a primary blood supply such as anterior cingulated gyrus (anterior cerebral artery (ACA)), superior temporal cortex (middle cerebral artery (MCA)), and occipital cortex (posterior cerebral artery (PCA)) had a lower baseline CBF compared with watershed areas or regions that have an anastamosis of two or more blood vessels, including cerebellum (SCA, PCA), dorsolateral PFC (MCA, ACA), and inferior frontal cortex (ACA, MCA).
Cerebral blood flow for hydrocephalus and control animals obtained from eight different brain regions: anterior and posterior cingulated gyrus, inferior and dorsolateral prefrontal cortex, superior temporal cortex, occipital cortex, cerebellum, and caudate nucleus
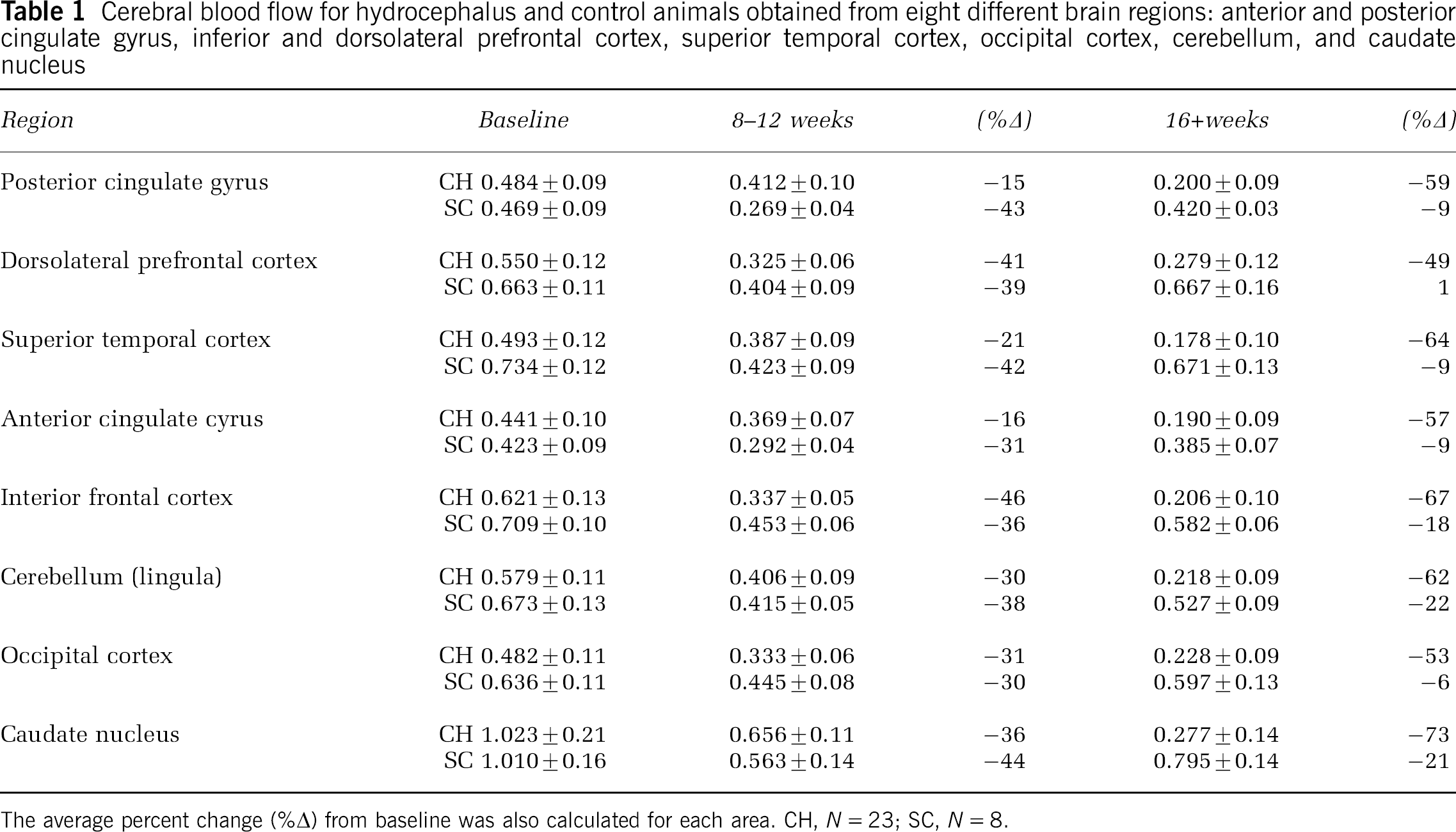
The average percent change (%Δ) from basline was also calculated for each area. CH, N = 23; SC, N = 8.
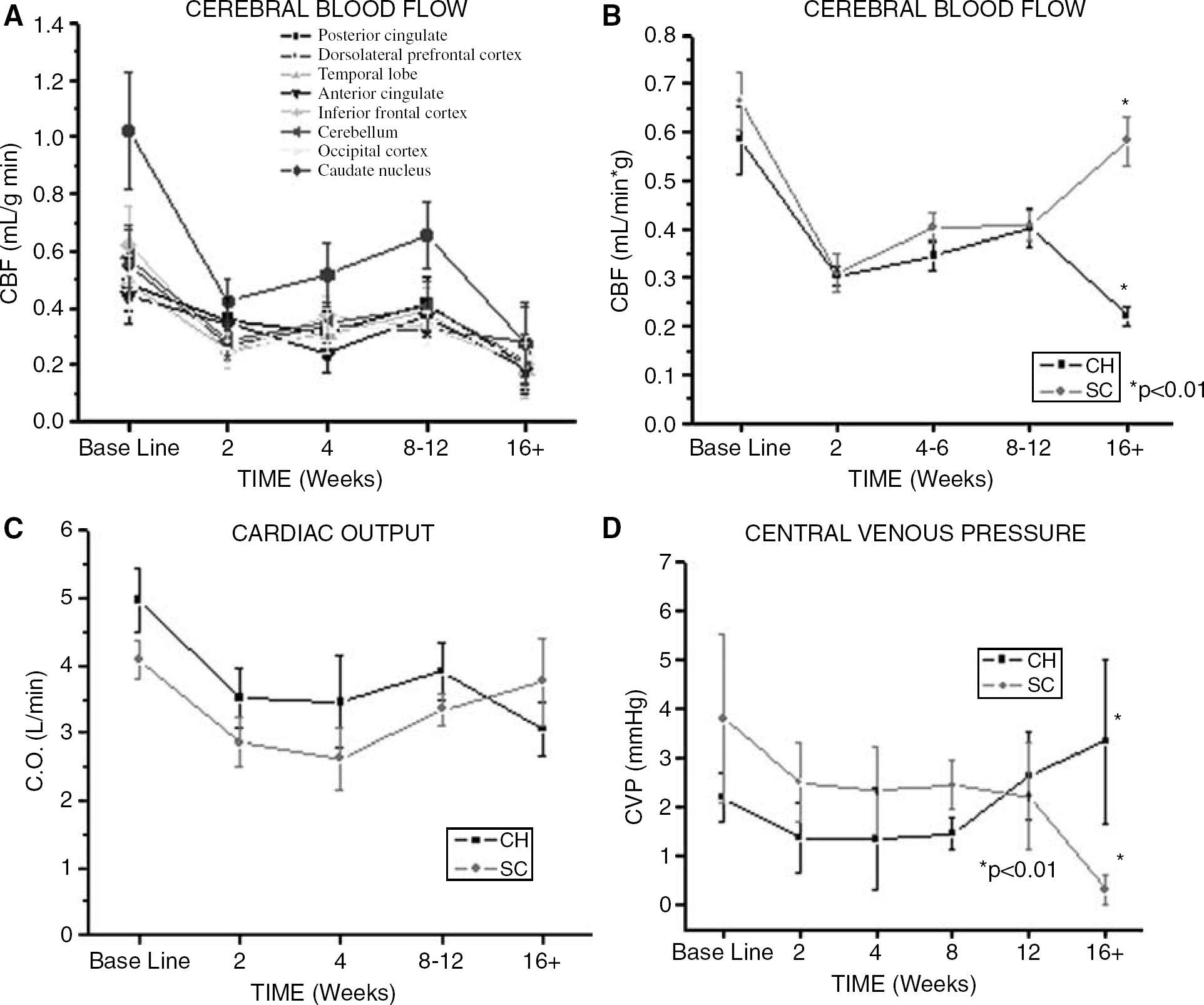
After CH induction surgery, changes in CBF for all eight areas investigated showed a consistent pattern (Figure 3A). Overall, CBF decreased significantly at 2 weeks (P < 0.05), followed by a subsequent and relative improvement or recovery from 4 to 12 weeks, and finally a decrease again at the latest time point (16 + weeks). After 2 weeks of CH, CBF decreased by 22% to 60%, with the greatest effect observed in the inferior frontal cortex (−60%) and caudate nucleus (−59%) compared with least in the anterior and posterior cingulate cortices (−22% and −26%, respectively). After 16 weeks of CH, CBF decreased by 49% to 73% compared with baseline CBF levels in which the inferior frontal and caudate nucleus remained most affected.
Individual CBF for eight different brain regions for CH group (
The pattern of CBF for CH compared with SC was similar early after surgical CH induction (Figure 3B). CBF was shown to significantly decrease in both CH and SC groups after 2 weeks, and remained similar, while slightly increasing through 8 weeks postoperatively. However, at 12 and 16 + weeks postoperative, CBF in SC animals was significantly higher than CH animals (P < 0.01).
Myocardial Tissue Perfusion
Myocardial tissue perfusion (MTP) obtained from 13 unique cardiac samples at baseline ranged from 0.269 mL/ming. (right atrium) to 1.87 mL/ming (posterior papillary muscle), with an average of 1.31 mL/min g. In general, the left ventricle had the greatest MTP, followed by the right ventricle, while the left and right atria had the least MTP.
An average MTP for all 13 areas investigated was calculated and used in the direct comparison between CH and SC groups (Table 2, Figure 4A). At 2 weeks postoperative, MTP in both the CH and SC groups decreased significantly (P < 0.05). From week 4 through week 12, the average MTP for CH and SC groups was similar. At 16 + weeks postoperation, the average MTP for the CH group was significantly less than baseline and earlier time points (P < 0.01), and was significantly less than the SC group (P < 0.05). By comparison, the average MTP for the SC group was not significantly different between baseline and the last time point (16+ weeks).
Average CBF, MTP, and PBF for CH and SC groups for baseline 2, 4–6, 8–12, and 16 weeks, and the percent change (%Δ) from baseline
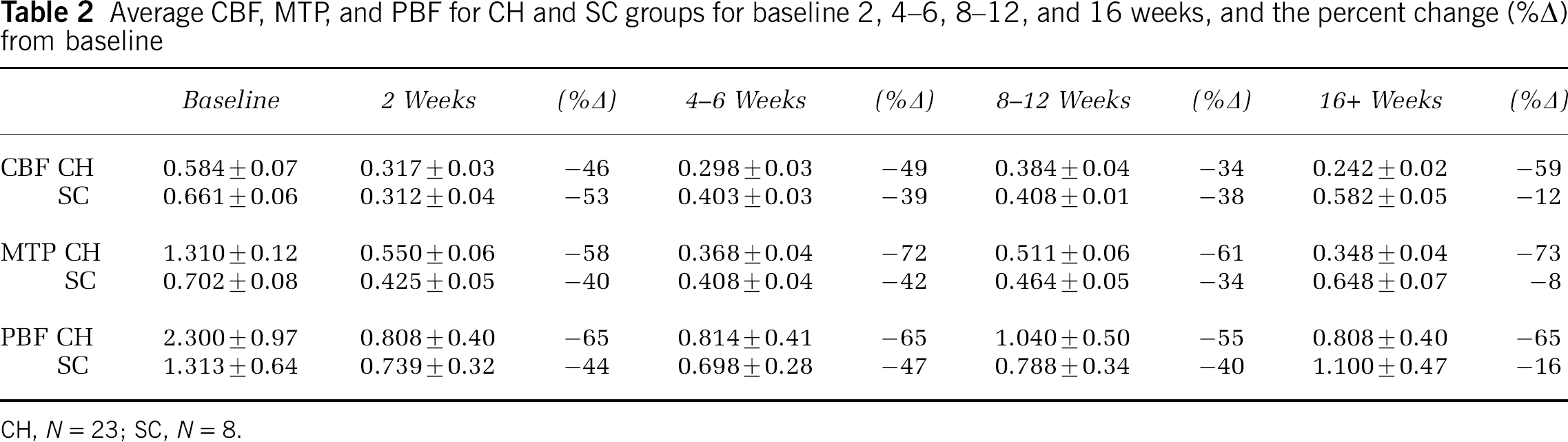
CH, N = 23; SC, N = 8.
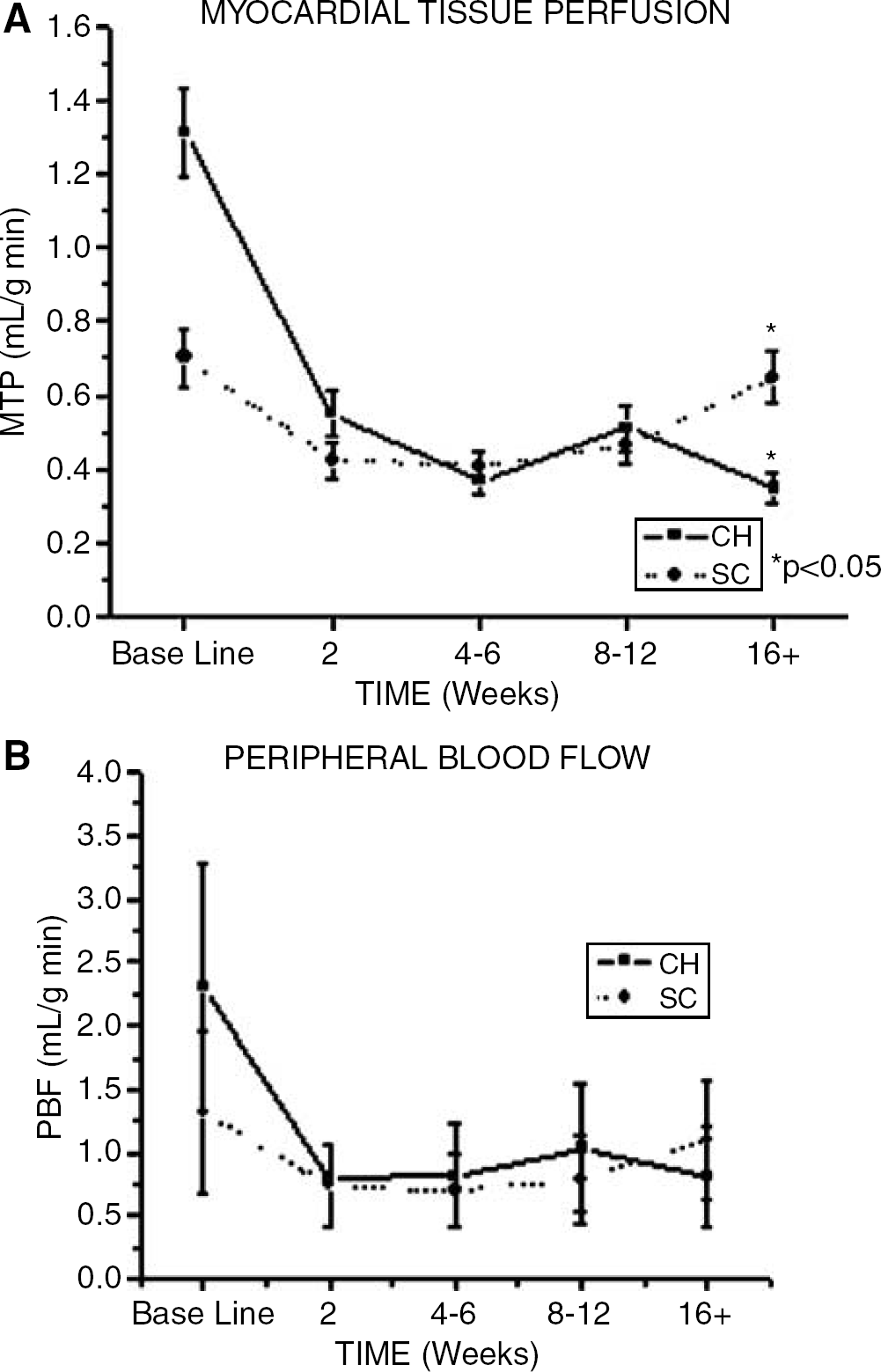
Average MTP calculated from 13 different cardiac regions (A), and the average PBF calculated from kidney, liver, lung, and spleen (B), comparing CH and SC groups at baseline, 2, 4 to 6, 8 to 12, and 16 + weeks postoperatively (CH, N = 23; SC, N = 8). *Significance was determined at the P < 0.05 level.
Peripheral Blood Flow
Overall baseline peripheral blood flow (PBF) varied widely, ranging from 0.216 mL/ming. (liver), 1.14 mL/ming (spleen), 3.53 mL/ming (lung), and 4.31 mL/min g (kidney). Except for the kidney which showed a significant decrease in blood flow 2 weeks postoperatively (P < 0.05, data not shown), PBF for liver, lung and spleen did not differ across time or between CH and SC groups. Peripheral blood flow for liver, kidney, lung and spleen was combined to calculate the average PBF and compared between CH and SC groups (Table 2, Figure 4B). By comparison, the average PBF between CH and SC groups was not significantly different at any time point investigated.
Cardiac Output
Overall, baseline CO ranged from 2.65. to 8.00 L/min with an average of 4.97 L/min for CH and 4.10 L/min for SC (Table 3). Figure 3C illustrates the differences in CO over time for CH and SC groups. In general, CO in both the CH and SC groups significantly decreased from baseline to 2 weeks (P < 0.05). From 2 weeks through 12 weeks, CO for CH and SC groups was similar. At 16 + weeks, CO for the CH group was significantly less than baseline (P < 0.05), compared with no significant difference for the SC group (Figure 3C).
Average CO, intracranial compliance via ICCr and ICCi methods, and CVP for CH and SC for baseline 2, 4–6, 8–12, and 16 weeks, and the percent change (%Δ) from baseline
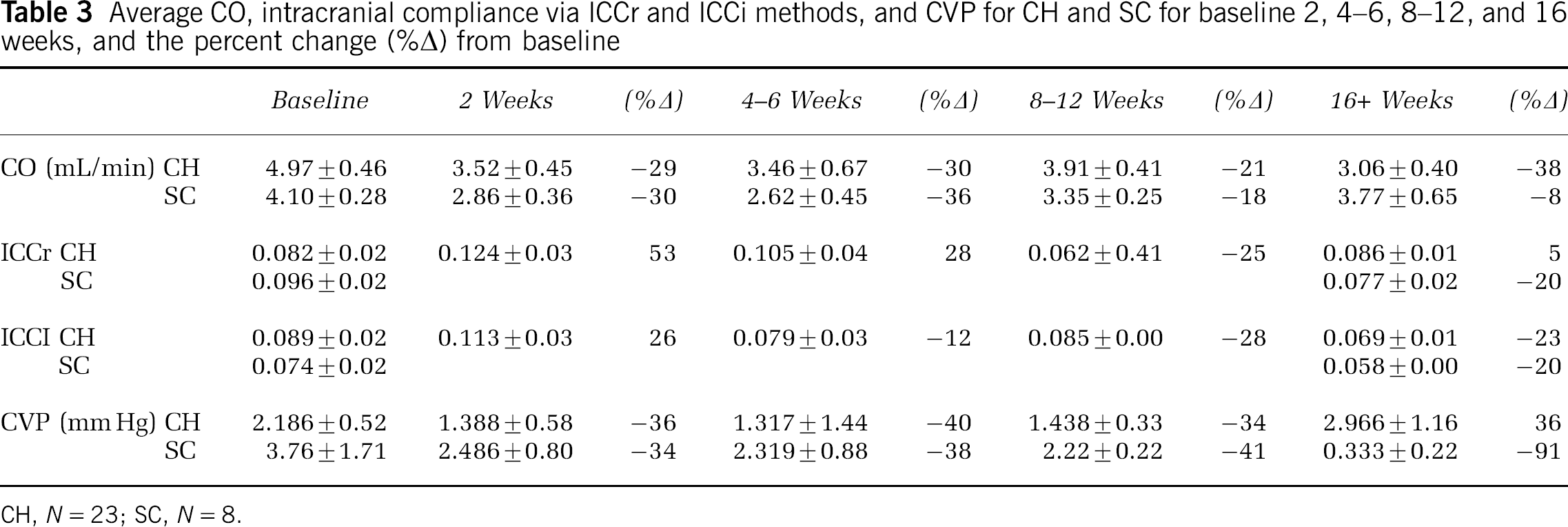
CH, N = 23; SC, N = 8.
Central Venous Pressure
In addition to CO, CVP has been an additional indicator of cardiac function. Central venous pressure, obtained during CO measures, was similar from baseline through 8 weeks for both the CH and SC groups (Figure 3D). Then at 12 weeks, CVP increased for the CH group and decreased for the control group. Central venous pressure for the control group was significantly lower than baseline (P < 0.01) and the CH group (P < 0.01) after 16 weeks (Figure 3D).
Intracranial Compliance
Intracranial compliance (ΔV/ΔP) was obtained via bolus CSF removal (ICCr) and infusion (ICCi) intraoperatively for CH and SC groups (Table 3, Figure 5). Overall, the patterns of ICCr and ICCi were similar. There was no significant difference in ICCr and ICCi across time or between groups. Both measures showed a nonsignificant increase from baseline to 2 weeks postoperatively. Increased ICCr was directly correlated with an increase in CO (P = 0.04, R = 0.39).
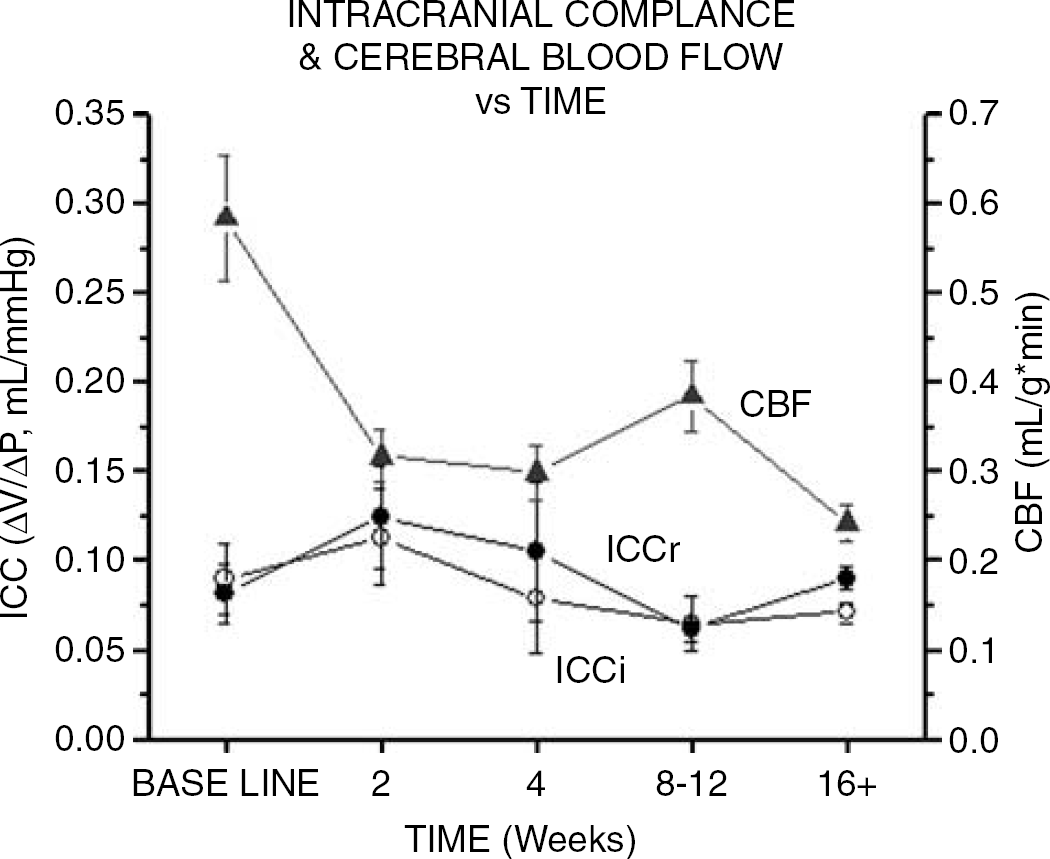
Intracranial compliance measured via bolus ICCr (closed circle) and infusion (ICCi; open circle) methods for CH compared with CBF (closed triangle, blue line) at baseline, 2, 4 to 6, 8 to 12, and 16 + weeks postoperatively (CH, N = 23, SC, N = 8). Decrease in CBF corresponds with increase in ICC at 2 weeks, inversely compared with an increase in CBF and decrease in ICC at 8 to 12 weeks. No significant difference was observed between ICCr and ICCi at any time period.
Relationship between Chronic Hydrocephalus and Cerebral Blood Flow, Cardiac Output, O2 and Intracranial Compliance
Cardiac output was shown to be directly correlated with CBF averaged from eight brain regions (P =0.028, R = 0.389) (Figure 6A), and inversely related to the degree of CH CSF VV (P = 0.019, R = −0.389) in the CH group (Figure 6B). Cerebral blood flow did not correlate with changes in CSF volume or ICP for either CH or SC groups (F =0.33, F = 0.21, respectively), except for CSF volume in CH of the caudate nucleus (P = 0.043; R = −0.307; Figure 6D). In addition, CBF obtained for dorsolateral PFC (DLPFC) was directly correlated with O2-TISSUE measures obtained from the same area (P = 0.006, R = 0.450) (Figure 6C). Multiple regression analysis showed a significant relationship between CBF and CO, CSF VV and ICP, and CH duration (P = 0.05). Figure 7 shows the inter-relationship between CBF, CO, O2 (PFC), and CSF ventriculomegaly.
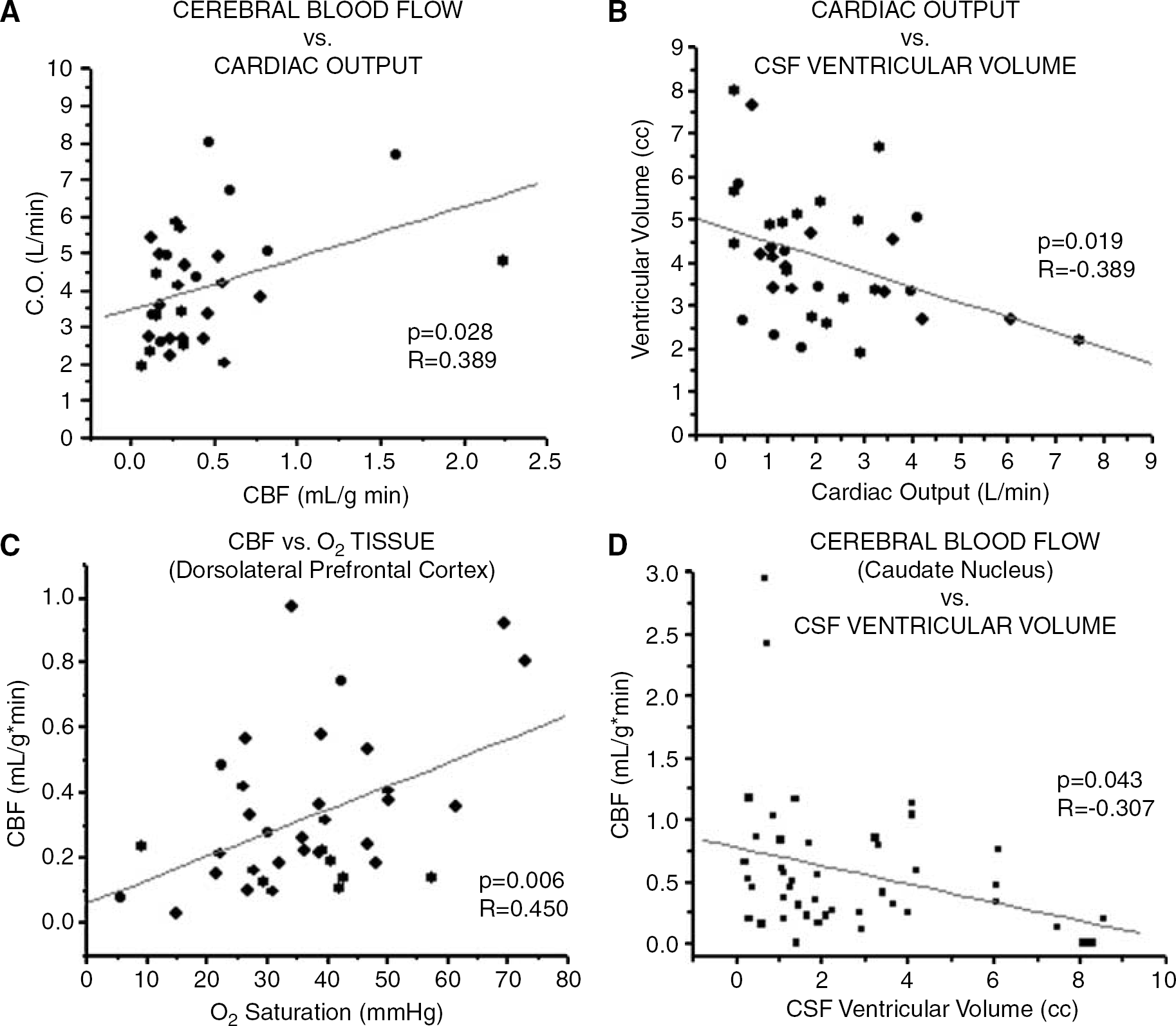
Scatterplots showing significant correlation between (
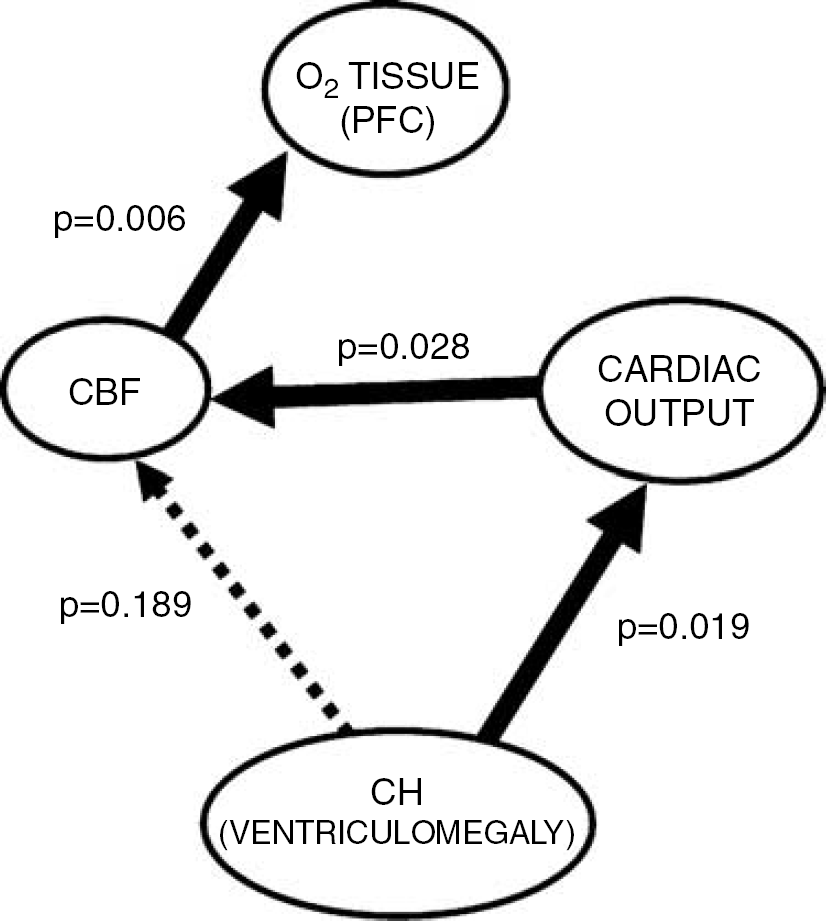
Summary diagram illustrating the relationship between ventriculomegaly, cardiac function, CBF, and oxygen delivery in our experimental (canine) model of chronic obstructive hydrocephalus. The degree of ventriculomegaly in CH was significantly correlated with CO and not directly related to CBF. In turn, CBF was directly correlated with oxygen delivery in the PFC.
Discussion
This study shows parallel changes in cardiac function, systemic blood flow, and CBF after surgical induction of hydrocephalus. The degree and pattern of CBF reduction and recovery were directly related to changes in cardiac function over time and not to changes in either CSF volume or pressure. While similar reductions in cardiac function and CBF in hydrocephalic and control animals during the early period may reflect surgical effects, the further decline of CO and CBF in only hydrocephalic animals at 16 weeks suggests that the effect may be directly related to the hydrocephalus. Finally, regional CBF reduction was significantly correlated with increased ICC and decreased tissue oxygen levels, suggesting that the observed changes in CBF were physiologically significant.
Using our experimental animal model of CH, we confirmed and expanded earlier findings for CSF volume, pressure, and compliance from our laboratory (Johnson et al, 1999; Fukuhara et al, 2001; Luciano et al, 2001) and baseline regional CBF measures that were consistent with earlier findings using the identical microsphere technique (Reinhardt et al, 2001). Further, the induced hydrocephalus used in this study resulted in a significant global reduction in CBF, which is also consistent with global decreases reported in earlier experimental studies in cats (Higashi et al, 1986; da Silva et al, 1995) and rats (Jones et al, 1993; Klinge et al, 2003; Kawamata et al, 2003) using different animal models of hydrocephalus and different methods of blood flow measurement. A similar blood flow reduction has been observed in patients with adult CH (or normal pressure hydrocephalus (NPH)) using a variety of techniques such as Xe clearance with contrast CT, Kety–Schmidt dilution, and SPECT, and PET imaging (for a review, see Owler and Pickard, 2001)). Similar to these earlier studies, we found the greatest reduction in CBF in frontal cortical areas and caudate nucleus.
Cerebral blood flow reduction in CH is often considered to be primarily due to changes in ICP and compression and stretch of vessels distorted by ventriculomegaly. Previous anatomical studies have characterized progressive ventriculomegaly as the distortional expansion of the anterior and inferior horns of the lateral cerebral ventricles, suggesting that hydrocephalus may result from differential ventricular expansion relating to force vectors acting on adjacent cortical structures (Greitz et al, 1992). However, while earlier reports found a relationship between CBF and CSF volume (Wozniak et al, 1975; Mabe et al, 1990; Nakada et al, 1992; Goh and Minns, 1995) and pressure (Zierski, 1987), the present study found no correlation between the severity (either CSF volume or pressure) of hydrocephalus and the average CBF of eight brain areas investigated. The exception to this was that the individual CBF measure for the caudate nucleus was correlated with VV. The particular sensitivity of the caudate to increased lateral VV may result from generally high regional flow rate and proximal location to the ventricle, and may explain motor and gait deficits found prominently in clinical CH.
Although this study cannot differentiate between autoregulatory system damage or the breaching of its adaptive limits, the degrees of CO and CBF reduction (∼30% and 50%, respectively) shown in this study match or exceed the autoregulation limits suggested in other studies (Davis and Sundt Jr., 1980; Tranmer et al, 1992) (Larsen et al, 1994; Kadas et al, 1997; Ursino and Giulioni, 2003; Van Lieshout et al, 2003; Lakin et al, 2003). The concept of an ‘overwhelmed’, but not damaged, autoregulatory system may be further suggested by recovery of response with treatment of hydrocephalus. In an early study we found that the response to hyperventilation (CO2 or chemical autoregulation) as manifested by oxygen saturation change was absent in hydrocephalic animals, where baseline oxygen level was already low. With CSF shunting, the response to CO2 changes was restored along with baseline oxygen levels. These experimental findings are consistent with clinical evidence that cerebral autoregulation may be simply overwhelmed, and not destroyed, in CH (Czosnyka et al, 2002, 2003; Minhas et al, 2004).
The cause of the observed changes in cardiac function was likely different in the early versus the late time periods of this study. Cranial and cardiac procedures performed in this investigation, which were identical in both experimental and control groups, likely resulted in surgical trauma responsible for decreased cardiac function and blood flow in the early (2-week) time point. This finding was consistent with clinical and experimental studies that have shown changes in CBF relating to either cranial or cardiac injury (Gruhn et al, 2001; Williams et al, 2001; Nortje and Menon, 2004). By comparison, differences in cardiac function and blood flow between groups in the late phase were more likely associated with hydrocephalus-induced brain distortion and injury. At this last time point studied (16 weeks), the hydrocephalic group showed a significant divergence from the SC group in blood flow, cardiac function and CVP measures. Taken together, this suggests a decrease in CBF associated with decreased cardiac function specific to the hydrocephalus. This could not be attributed to changes in weight or behavior since both groups were similar (and normal) across all time points. Unfortunately, evolution of this hydrocephalus-specific effect could not be followed past 16 weeks since this was the latest time point investigated. Finally, this study found a significant correlation between ventricular size and CO in the hydrocephalus group, indicating that ventricular enlargement might be affecting cardiac function perhaps through alteration of autonomic function.
Previous studies have shown that neurogenic injury may be directly responsible for cardiac dysfunction. Neurologic studies have shown ECG abnormalities to be associated with cerebral infarction, glioma, head trauma, intraparenchmyal hemorrhage, meningitis, seizures, and headaches (Cropp and Manning, 1960; Hersch, 1964; Talman, 1985; Marion et al, 1986; Elrifai et al, 1996). Furthermore, brain injury relating to subarachnoid hemorrhage has shown pathologic changes in cardiac tissue, including contraction band necrosis, myocardial enzyme release, and elevated sympathetic activity (Greenhoot and Reichenbach, 1969; Ibayashi et al, 1986; Lieb et al, 1996; Mayer et al, 1999; Masuda et al, 2002). Though not the main focus of the current study, we did not find ECG or any microscopic pathologic changes in cardiac tissue in any animal with CH. While severe increases in ICP have been shown to be associated with abnormal ECG (Dicker et al, 1983; Rudehill et al, 1987a, b ; Biswas et al, 2000; van Aken et al, 2003), we report a gradual, nonsignificant increase in ICP in our animal model, which would most likely have insignificant clinical implications. Evidence for a heart–brain relationship relating to CH comes mostly from studies investigating cardiac abnormalities and disease that report secondarily changes in CSF space (Graff-Radford and Godersky, 1987; Casmiro et al, 1989; for a review, see Krauss et al, 1996). Our data support the possibility of cardiac function changes in hydrocephalus and further suggest that CNS-induced cardiac changes in turn affect CBF.
Hydrocephalus decreased oxygen saturation levels in the DLPFC, and this decrease in tissue oxygen delivery was significantly correlated with decreased CBF in this same region. This finding validates and gives functional significance to our observed decrease in CBF. Our finding for the level of CBF after 16 weeks after CH induction (19 to 28 μL/g min, mean 24 μL/g min) is considerably less than the minimum CBF of 50 to 60 μL/g min required for adequate oxygen delivery to the brain (Van Lieshout et al, 2003). Cortical areas with blood flow deficits of this magnitude have been shown to be associated with cellular dysfunction, including anaerobic glycolysis and decreased glucose metabolism, and have often been referred to clinically as an ischemic penumbra (Marshall, 2004).
Finally, we found that ICC was inversely related to CO, CVP, and blood flow in the early phase. Intracranial compliance (ΔV/ΔP), described as the brain's ability to accommodate changes in volume with respect to pressure, is influenced by arterial and venous pressures, blood volume, cellular and extracellular matrices, and surrounding meninges. This study suggests that a decreased CBF may result in decreased brain turgor and a more compliant brain. A more compliant brain may in turn make it more susceptible to further compression by the ventricular system.
This increased compliance is an apparent discrepancy with clinical evidence, which suggests a decreased brain compliance in the hydrocephalus. Decreased compliance has also been suggested in other neurologic disorders such as Alzheimer's disease and normal pressure hydrocephalus (Bateman, 2004, 2005), and migraine headaches (Rupprecht et al, 2001; de Hoon et al, 2003; Silvestrini et al, 2004), where CBF may be affected. Indeed, in our own earlier study (Fukuhara et al, 2001), we show decreased cranial compliance in CH in agreement with that found clinically. This apparent discrepancy is based on the measurement of the volume/pressure relationship under different conditions. We found that with fully expanded ventricles the compliance is decreased, but with CSF drainage and lower pressures the compliance is increased relative to control animals (Fukuhara et al, 2001). These findings are consistent with the hydrocephalus altering the nonlinear pressure-volume curve both by increasing compliance in the earlier ‘adaptive’ phase of the curve, and by decreasing compliance in the steep ‘non-adaptive’ phase. Taken together, these observations are all consistent with a softer brain in hydrocephalus, which appears less compliant when studied in the compressed state. Finally, these findings are consistent with the common concept of a role for CBF and CVP in ICC and an abnormality in hydrocephalus.
Summary/Conclusion
Using an experimental model of chronic obstructive hydrocephalus, this investigation found quantitative changes in cardiac function, cerebral and systemic blood flow, oxygen delivery, and ICC, as each relate to the progression and severity of the hydrocephalus. Overall, we report a close association between blood flow and cardiac function. Initially, we found significant decreases in cardiac function and blood flow for both experimental and SC animals, which later recovers in control animals and further decline in hydrocephalus animals. The results from this study suggest the following: (1) even in developing hydrocephalus, characterized by increased CSF VV and pressure, cardiac function plays a dominant role in CBF; (2) cerebral autoregulation may be overwhelmed, but not likely damaged, in CH; (3) CH is associated with cardiac dysfunction and decreased CBF; (4) decreased CBF may be responsible for impaired oxygen delivery and increased ICC.
While cerebral pressure and morphology changes in hydrocephalus undoubtedly play an important role in CBF and diminished brain function, the concurrent measurement of systemic hemodynamics performed in this study shows that the heart continues to have a strong influence over the brain during times of vulnerability. The brain also influences the heart, and alterations in cardiac function associated with the hydrocephalus may further impair CBF. The heart–brain interaction in the hydrocephalus will need to be further studied to develop effective treatment strategies.
