Abstract
Oxygen therapy for ischemic stroke remains controversial. Too much oxygen may lead to oxidative stress and free radical damage while too little oxygen will have minimal therapeutic effect. In vivo electron paramagnetic resonance (EPR) oximetry, which can measure localized interstitial partial oxygen (pO2), can monitor penumbral changes of pO2. Therefore, we used EPR to study the effects of oxygen therapy in a rat model of 90-mins middle cerebral artery occlusion (MCAO). We found that 95% normobaric O2 given during ischemia was able to maintain penumbral interstitial pO2 levels close to the preischemic value while it may cause a two-fold increase in penumbral pO2 level if given during reperfusion. Elevation of the penumbra pO2 to preischemic physiologic level during MCAO significantly reduced infarction volume, improved neurologic function, decreased the generation of reactive oxygen species (ROS), and reduced matrix metalloproteinase (MMP)-9 expression and caspase-8 cleavage in the penumbra tissue of rats brain treated with oxygen. These results suggest that maintaining penumbral oxygenation by normobaric oxygen treatment during ischemia lead to neuroprotection, which is further reflected by the decreased production of ROS, MMP-9, and caspase-8.
Introduction
Cerebrovascular disease is the third leading cause of death and an important cause of hospital admission and long-term disability in most industrialized populations (Bonita et al, 1990). It is well established that acute ischemic stroke produces an irreversibly damaged ischemic core and a surrounding potentially salvageable penumbra (Astrup et al, 1981), which is the target of ischemic stroke therapy (Fisher, 1997). Oxygenation status in the penumbra is critical to neuronal survival. We have shown in our previous work (Liu et al, 2004) that in vivo and real-time monitoring of penumbral interstitial partial pressure of oxygen (pO2) by electron paramagnetic resonance (EPR) is achievable and that penumbral pO2 can be raised by normobaric hyperoxia treatment. Increasing oxygen levels in the penumbra can improve the metabolic status and help penumbral tissue to survive, but it may also bring forth oxygen-related damage. Therefore, ‘guided’ (with monitoring of tissue oxygen) oxygen therapy, which maintains penumbral interstitial pO2 at the preischemic level, may be a solution to the long debated issue of oxygen therapy in stroke (Roos et al, 1998; Rusyniak et al, 2003).
Over-oxygenation during hyperbaric oxygen (HBO) therapy may result in oxidative stress and free radical damage. It has been suggested that the detrimental effects of exposure to high concentrations of oxygen are due to the abundance of reactive oxygen species (ROS) (Jamieson et al, 1986). An increase in free radicals in the blood from persons undergoing HBO exposure was directly demonstrated by electron paramagnetic resonance spectroscopy (Narkowicz et al, 1993). Oxidative stress may further activate MMPs (Asahi et al, 2000; Gasche et al, 2001; Sugawara et al, 2001) and caspases (Morita-Fujimura et al, 2001; Anantharam et al, 2002), which cause blood–brain barrier break-down and neuronal death, respectively.
Recent experiments have shown that normobaric hyperoxia is beneficial when administered in transient cerebral ischemia in animals (Singhal et al, 2002a, b ) although this does not appear to be the case for permanent cerebral ischemia (Li et al, 1994). Therefore, the effects of normobaric oxygen therapy need to be actively investigated. Here we evaluated the therapeutic effects of normobaric hyperoxia treatment using EPR-guided oxygen therapy. Specifically, we investigated: (i) whether penumbral tissue pO2 could be modulated by varying the oxygen content in the inspired gas, (ii) whether raising the penumbral pO2 during ischemia to the physiologic level would afford any neuroprotection, and (iii) whether raising penumbral pO2 would negatively or positively affect the factors that are associated with the cerebral injury due to ischemia. We found that maintaining the oxygenation status of the penumbra at preischemic levels during ischemia results in a protective effect and decreased ROS production, attenuated MMP-9 expression, and decreased caspase-8 cleavage in the penumbra.
Materials and methods
Animal Handling
The Laboratory Animal Care and Use Committee of the UNM HSC approved all experimental protocols. A total of 138 male Sprague–Dawley rats (Charles River Laboratory, Wilmington, MA, USA) weighing 310 to 330 g were used for this study. After four animals excluded due to anesthetic accidents or other unexpected accidents, 134 animals were used for final analysis. The animals were maintained in a climate-controlled vivarium with a 12-h light-dark cycle with free access to food and water.
For all surgical procedures, 4.0% isoflurane in N2:O2 (70%:30%) was used for anesthesia induction, and 1.75% for anesthesia maintenance. Rats were anesthetized throughout preischemia, ischemia and reperfusion. Physiologic monitoring during the procedure comprised measurement and maintenance of core (rectal) temperature at 37.5°C±0.5°C using a heating pad. Mean blood pressure was also monitored using Advanced Blood Pressure Monitor System (Harvard Apparatus, Holliston, MA, USA).
Implantation of Electron Paramagnetic Resonance Oximetry Probe Lithium Phthalocyanine
Before implantation of the microscopic EPR oximetry probe lithium phthalocyanine (LiPc), the ischemic penumbra was predetermined, as described previously (Liu et al, 2004). Lithium phthalocyanine crystal (approximate diameter 0.2 mm) was placed using the same coordinators at 0.5 mm posterior, 2 mm lateral to bregma, and 5.5 mm down from skull for monitoring of penumbral pO2 (Liu et al, 2004) since the animal model, surgical conditions, animal species, and body weight were the same as in our previous studies. Rats were allowed to recover from implantation 48 to 72 h before induction of cerebral ischemia. The correct assignment of the implantation site was further confirmed post mortem by TTC staining. The LiPc material was a gift from Dr Harold Swartz (NIH In Vivo EPR Center, Dartmouth College, NH, USA).
Animal Model of Focal Cerebral Ischemia and Reperfusion
Middle cerebral artery occlusion (MCAO) followed by reperfusion was conducted using the intraluminal model (Koizumi et al, 1986). Briefly, the external carotid artery (ECA), internal carotid artery (ICA) and pterygopalatine artery (PPA) of the ICA were exposed. A silicone rubber coated monofilament nylon suture with a diameter of 0.37 mm (Doccol Co., Albuquerque, NM, USA) was inserted into the ICA via a cut on the ECA. The suture was advanced along the ICA to the extent of 19 to 20 mm from the bifurcation. The actual length of the inserted suture was adjusted individually according to a sensation of resistance. Reperfusion was produced by gently withdrawing the suture out of ECA. For all animals used in this study, successful MCAO was confirmed post mortem by 2% 2,3,5-triphenyltetrazolium chloride (TTC) stain as previously described (Liu et al, 2002), or confirmed in vivo by laser Doppler flowmetry (LDF) as described previously (Liu et al, 2004). The MCAO animal model success rate is 100% without any accident of intracranial bleeding.
Three groups of animals (non-MCAO, hyperoxia during MCAO and hyperoxia during reperfusion) were used additionally to study the effect of hyperoxia treatment on cerebral blood flow in penumbra, core, contralateral side and the nonischemic rats. There are 4 animals in each group. The coordinators for penumbra are 0.5 mm posterior and 3 mm lateral to bregma, for core are 0.5 mm posterior and 5.5 mm lateral to bregma. Continuous monitoring of blood flow for 20-mins was performed both before and during hyperoxia treatment. The average blood flow value in the last 5-mins was used for analysis.
Normobaric Hyperoxia Therapy
To determine the optimal inspired oxygen concentration for the oxygen therapy, a preliminary experiment was performed. A total of 20 animals were preimplanted with LiPc particles to penumbra position 3 days before the experiment. Under anesthesia, femoral arteries were cannulated with heperinized PE-50 tubing for taking blood samples. Middle cerebral artery occlusion model was induced using the same procedure as described above. During 90-mins ischemia, animals in each group were allowed to breathe the mixed gas with a different oxygen concentration while blood gas and penumbral tissue oxygen levels were measured at the same time. Five different mixed gases were studied, including 30%O2 + 70% N2, 70% O2 + 30% N2, 95%O2 + 5°% N2, 100% O2, and 95% O2 + 5%CO2, with four rats in each group.
To assess the short-term therapeutic effects of hyperoxia treatment animals were randomized into three groups: nontreatment control, hyperoxia intervention during 90-mins ischemia, and hyperoxia intervention during 90-mins reperfusion, with 11 animals in each group. Normobaric hyperoxia treatment was initiated by switching inspired gas to carbogen (95%:5%, O2:CO2) and terminated by switching back to regular anesthetic gas N2O:O2 (70%:30%). Hyperoxic gas was delivered during 90 mins ischemia or 90 mins reperfusion period. Real-time EPR monitoring of penumbral interstitial pO2 was conducted during preischemia, 90-mins ischemia and 90-mins reperfusion (see following). After completion of 90-mins reperfusion, animals were evaluated by Rogers' eight-point neurologic scale, which has been systematically studied and shows excellent correlation with both cortex and striatum histopathologic outcome (Rogers et al, 1997). Infarction volume was evaluated 24-h after ischemia (see below).
Two additional groups, nontreatment control and hyperoxia during 90-mins ischemia, were used to assess the longer term effect of hyperoxia treatment, with 4 animals in each group. Rogers' eight-point neurologic score and infarction volume were evaluated at 72 h after ischemia.
Real-time Electron Paramagnetic Resonance Oximetry
Real-time monitoring of penumbral pO2 was conducted using a Bruker EleXsys E540 EPR spectrometer equipped with an L-band bridge (Bruker Instruments, Billerica, MA, USA), as described previously (Liu et al, 2004). Typical EPR acquisition parameters include microwave power 10 mW, microwave frequency 1.07 GHz, magnetic field center 380 G, scan range 2.0 G, and modulation amplitude at less than one-third of the EPR line-width. Interstitial pO2 was measured continuously throughout the experiment, and reported at the time points shown.
Tissue Processing and Infarction Volume Measurement
Rats were decapitated at 24-h, or 72-h after MCAO according to experimental design. Brains were removed and chilled with ice water for 5 mins and sectioned into 1 mm thick slices using brain matrix and stained by immersion into 2% 2,3,5-triphenyltetrazolium chloride (TTC) stain as previously described (Liu et al, 2002). Digital pictures were taken and the infarction volumes were calculated using Image Pro Plus (version 4.1) software. Infarction volume was expressed as the percent of the infarcted tissue as compared with the entire brain.
Dihydroethidium Detection of Superoxide
To evaluate superoxide production during oxygen treatment, we used Dihydroethidium (HEt) as an indicator for superoxide production (Chan et al, 1998; Murakami et al, 1998). In total, 25 animals were used for this purpose, with five animals in each group. There were two nonischemic control groups consisting of groups of normoxia and hyperoxia. Dihydroethidium was also injected in three of the ischemic groups consisting of groups of normoxia, hyperoxia during ischemia and hyperoxia during reperfusion. Rats were administered 0.5 mg HEt (Molecular Probes, Eugene, OR, USA) at a concentration of 1 mg/mL by i.v. injection 5-mins before induction of cerebral ischemia. All HEt treated animals were killed immediately after 90-mins reperfusion by intraaortic perfusion fixation while anesthetized. Brains were removed and immersed in 10% formalin for a minimum of 24 h of postfixation. After postfixation the brains were sectioned into 50-micron sections with a vibratome. Brain sections were treated with 0.2% (W/V) sodium borohydride (Sigma) in PBS for 20 mins and then transferred into PBS for rinsing (Clancy and Cauller, 1998). Sections were mounted on glass slides with coverslips using Gel/Mount mounting media (Fisher Scientific). Tissue sections were viewed using a microscope (Olympus BH2-RFCA) equipped with epifluorescence optics using excitation = 510 to 550 nm and emission > 580 nm for ethidium (ET) detection. The definition for penumbra and core was the same as previously reported in our paper (Liu et al, 2002) according to the cell morphology changes in the early stage. Digital images were acquired with a digital camera (Olympus MLH 020550) by the Olympus MagnaFire Camera Imaging and Control software and analyzed using Image Pro Plus software.
High-pressure Liquid Chromatography with Electrochemical Detection of 8-hydroxy-2′-deoxyguanine Formation
Oxidative damage caused by hydroxyl radicals was evaluated using 8-hydroxy-2′-deoxyguanine (8-OHdG) as a biomarker. In total, 18 rats were used for the 8-OHdG study. Animals were allotted to three groups, namely, normoxia, hyperoxia during ischemia, and hyperoxia during reperfusion, with six rats in each group. Animals were killed via decapitation under deep anesthesia by isoflurane at 24 h after ischemia. The brain was rapidly removed, and penumbral tissues were sampled as previously described (Liu et al, 2004). The sample was freshly prepared or frozen at −80°C. Extraction of DNA and subsequent HPLC-ECD measurement were performed according to the techniques described previously (Hel-bock et al, 1999). In brief, the sample was first homogenized in 1 ml of homogenizing buffer (0.32 mmol/L sucrose, 5 mmol/L MgCl2, 10 mmol/L Tris-HCl, pH 7.5, and 0.1 mmol/L Desferal) and then DNA was precipitated and purified. After digestion by Nuclease P1 (3.3 mg/mL) and alkaline phosphatase (1U/μL), the supernatant was transferred to a 30,000-Da microfiltration tube, and centrifuged for 30 mins at 10,000 g and 4°C. For quantitative measurement of 8-OHdG, a 50 pL sample was injected into the HPLC apparatus (ESA, Chelmsford, MA, USA). Isocratic analysis was performed on a CoulArray system (model 5600) equipped with a dual piston pump (model 582) and PEEK pulse damper. The sample was separated on a 15 cm × 4.6 mm, 3 μm LC-18-DB column (Supelco, Bellefonte, PA, USA). The mobile phase consisted of 50 mmol/L KH2PO4 (pH 5.5 with NaOH) containing methanol (95:5, vol/vol). The detector potentials were set at 100/400 mV (versus potential difference) and flow-rate at 1.0ml/min. The 2′-deoxyguanine (dG) was detected with a UV detector (model 520, ESA, Chelmsford, MA, USA) at 260 nm. The level of 8-OHdG in the sample DNA was expressed as the number of 8-OHdG per 106 dG.
Matrix Metalloproteinase and Caspase Assessment
To explore the possible mechanism of the protective effect of normobaric hyperoxia, expression of MMPs and caspases were assessed. Three groups of rats were used for this purpose, normoxia, normobaric hyperoxia during ischemia, and normobaric hyperoxia during reperfusion, with six animals in each group. At 24 h after ischemia rats were decapitated and the brains were removed quickly. The penumbra and contralateral tissue were sampled as previously described (Liu et al, 2004). Expression of MMP-2 and MMP-9 was assessed by gelatin zymography (Planas et al, 2001). Caspase-8 cleavage was assessed by Western blot.
The frozen tissues were homogenized with a lysing buffer (Tris-Cl: 50 mmol/L pH 7.6, NaCl: 150 mmol/L, CaCl2: 5 mmol/L, Brij-35: 0.05%, NaN3: 0.02%, Triton X-100: 1%). The protein concentration in the homogenate was determined using Bradford reagent (Bio-Rad Laboratories, Hercules, CA, USA). Matrix metalloproteinane-2 and MMP-9 in homogenate were concentrated with gelatin-sephrose beads (Gelatin Sephrose 4B, Amersham Biosciences, Piscataway, NJ, USA) and analyzed by gelatin zymography as previously reported (Planas et al, 2001). Western blotting was performed using samples subjected to gelatinolytic extraction. Proteins (300 μg) from extracted samples were electrophoresed in 15 SDS-PAGE acrylamide gels, transferred onto nitrocellulose membranes (BioRad Laboratories, Hercules, CA, USA), and incubated for 1 h in TBS containing 5% nonfat milk and 0.1% Tween-20 at room temperature. Membranes were then incubated overnight with polyclonal primary antibody against cleaved caspase-8 (1:1,000, Chemicon, Temecula, CA, USA), then washed in TBS with 0.1% Tween-20, incubated for 1 h at room temperature with HRP-conjugated antirabbit antibody (1:1,000, Cell Signaling Technology, Beverly, MA, USA), and revealed using the SuperSignal West Pico chemiluminescent kit (Pierce Biotechnology, Rockford, IL, USA) following the manufacturer's instructions. To control sample loading and protein transfer, the membranes were stripped and rehybridized to assess β-actin (1:4,000, Sigma, St Louis, MO, USA). The amount of cleaved caspase-8 was standardized in proportion to β-actin.
Statistics
ANOVA for repeated measures and Sheffe multiple comparison were used for the analyses of pO2 changes 127 during ischemia and reperfusion, and hyperoxia intervention. One-way ANOVA and Sheffe multiple comparisons were used for analyses of infarction volume. Kruskal—Wallis analysis of ranks and Mann—Whitney U-test were used for neurologic scores. Two-tailed Student's t-test was used to compare the changes in 8-OHdG, MMPs, and caspases in the penumbra between the hyperoxia treated group and the control group. Significance was considered with P < 0.05.
Results
Inspired Oxygen Concentration and Penumbral Tissue Partial Pressure of Oxygen
To investigate the effect of inspired oxygen concentration on the penumbral tissue pO2, and to determine the inspired oxygen concentration that can increase the penumbral tissue pO2 to the preischemic level, we treated the animals with gases containing various percentages of O2. Rats breathing 30%, 70%, 95%, 100% O2 balanced with nitrogen, and 95% O2 balanced with CO2 inspired oxygen were found to have a penumbral pO2 of 12.3, 17.4, 34.1, 38.2, and 31.8 mm Hg, respectively (Table 1). Blood gas pO2 and blood pH were also monitored for these rats during the oxygen treatment, as shown in Table 1. The results show that the animals treated with 95% or 100% O2 were able to maintain the penumbral pO2 close to the preischemic level (about 34 mm Hg). Furthermore, rats breathing 95% O2 balanced with 5% CO2 had a relatively normal blood pH and breathing rhythm as compared with those rats breathing 95% O2 mixed with 5% N2 or pure oxygen. Therefore, 95% O2 mixed with 5% CO2 (carbogen) was chosen as a means for normobaric hyperoxia treatment in this study.
Blood gas and penumbral pO2 measurement
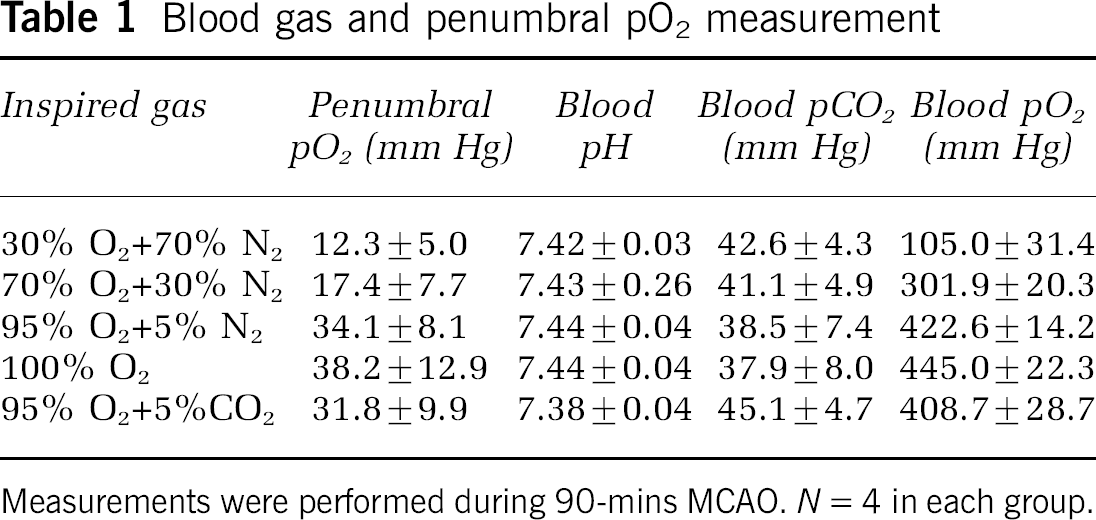
Measurements were performed during 90-mins MCAO. N = 4 in each group.
Cerebral Blood Flow and Blood Pressure Monitoring of Animals
During ischemia, LDF regional cerebral blood flow dropped to 17.9% ± 2.1% of the preischemic level, and after reperfusion the blood flow was restored to 90.7% ± 5.4% of the preischemic level, which is consistent with the reported values in rats under similar conditions (Sugo et al, 2002; Westermaier et al, 2003). Mean blood pressure remained constant throughout the procedure within the range of 82±14 mm Hg.
Changes of Penumbral Interstitial Partial Pressure of Oxygen during Hyperoxia Treatment
As shown in Figure 1, preischemic interstitial pO2 levels in the penumbral region were stable at three measuring points and there was no significant difference of preischemic pO2 levels among the three groups. Preischemic penumbral pO2 levels were 31±6 to 36±10 mm Hg when animals were breathing N2O:O2 (70%:30%). After the occlusion of middle cerebral artery, the penumbral pO2 level decreased rapidly within the first 5 mins to a level of 12 to 15 mm Hg if the animal was left untreated, as in the normoxia control group, and the hyperoxia group in which oxygen was delivered during reperfusion. Conversely, in the group in which 95% O2 was given during ischemia, penumbral pO2 was maintained close to preischemic levels. ANOVA showed a significant difference between the treated and untreated groups (P < 0.05). After reperfusion, the penumbral interstitial pO2 level in the untreated control group returned slowly to a level that remained lower than the preischemia value (approximately 76% of preischemic level), as shown in our earlier report (Liu et al, 2004). However, when the animal breathed 95% O2 on reperfusion, the penumbral interstitial pO2increased rapidly to a level that was approximately twice the preischemic level. ANOVA showed a significant difference (P < 0.05) between the hyperoxia intervention group and the untreated control group at and after 10-mins of reperfusion. In the group of hyperoxia delivered during ischemia, although penumbralp O2levels were maintained close to preischemic level during ischemia, the penumbralp O2decreased slowly when inspired gas was switched back to 30% O2 on reperfusion and approached the level of the untreated control group after 30 mins.
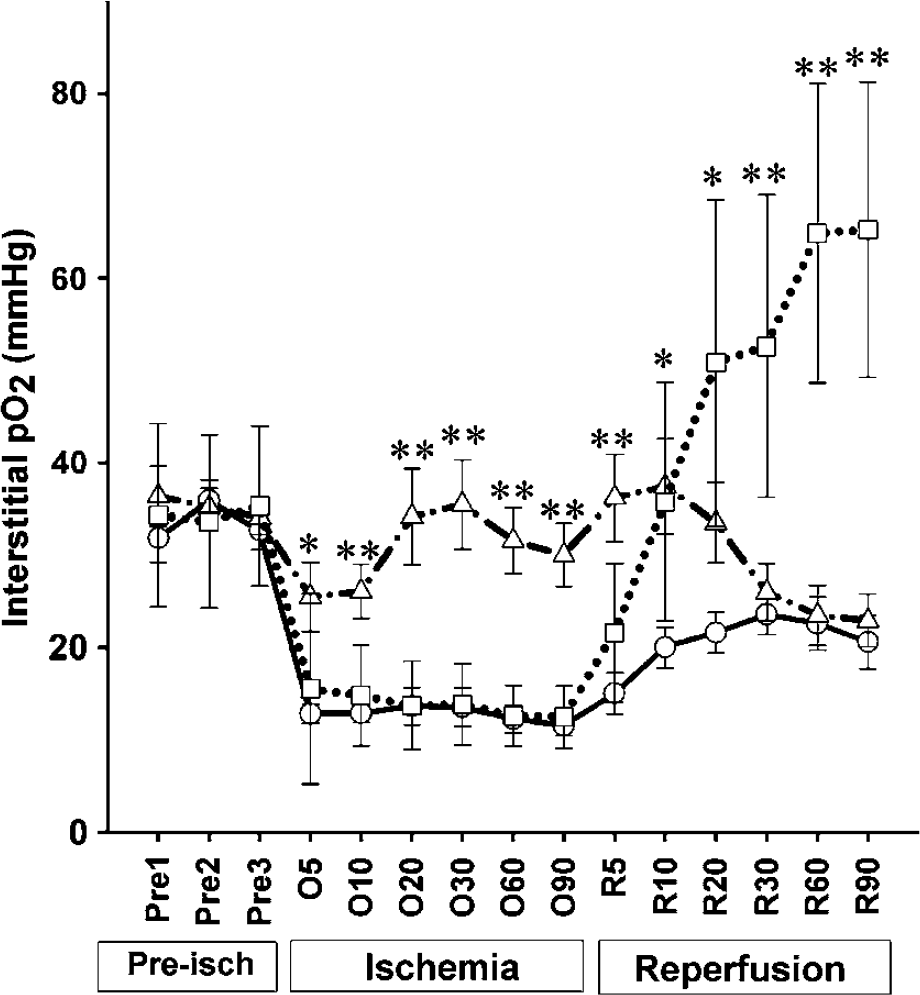
Penumbral interstitial O2 level in normoxia and hyperoxia rats during cerebral ischemia and reperfusion. Circle (○), group of normoxia. Triangle (▵), group of normobaric hyperoxia treatment during 90-mins ischemia. Square (□), group of normobaric hyperoxia treatment during 90-mins reperfusion. Pre1–Pre3, three preischemic measurement points. 05-090, 5 to 90 mins after occlusion of middle cerebral artery. R5–R90, 5 to 90 mins after reperfusion. Single asterisk indicates significant difference (P < 0.05) when compared with normoxia group. Double asterisks indicate significant difference (P < 0.05) when compared with the other two groups. Data are expressed as Mean ± s.d., n = 11 in each
Effects of Normobaric Hyperoxia on Infarction Volume and Neurologic Scores
Infarction volume in the untreated group was 19% of the entire brain volume when measured at 24 h postischemia. As shown in Figure 2A, hyperoxia intervention during 90-mins ischemia reduced infarction volume by 40%. ANOVA showed a significant difference between the control group and group of hyperoxia delivered during ischemia (P < 0.0.5). Interestingly, hyperoxia delivered during 90-min reperfusion also reduced the infarction volume by 15%; however, this difference was not shown to be statistically significant. The reduction of infarction volume was maintained when the rats were evaluated at 72 h after ischemia (Figure 2C). The infarction volume evaluated at 72 h after ischemia is relatively smaller than that observed at 24 h, probably due to the attenuated edema and the enhanced microglia infiltration (Lehrmann et al, 1997).
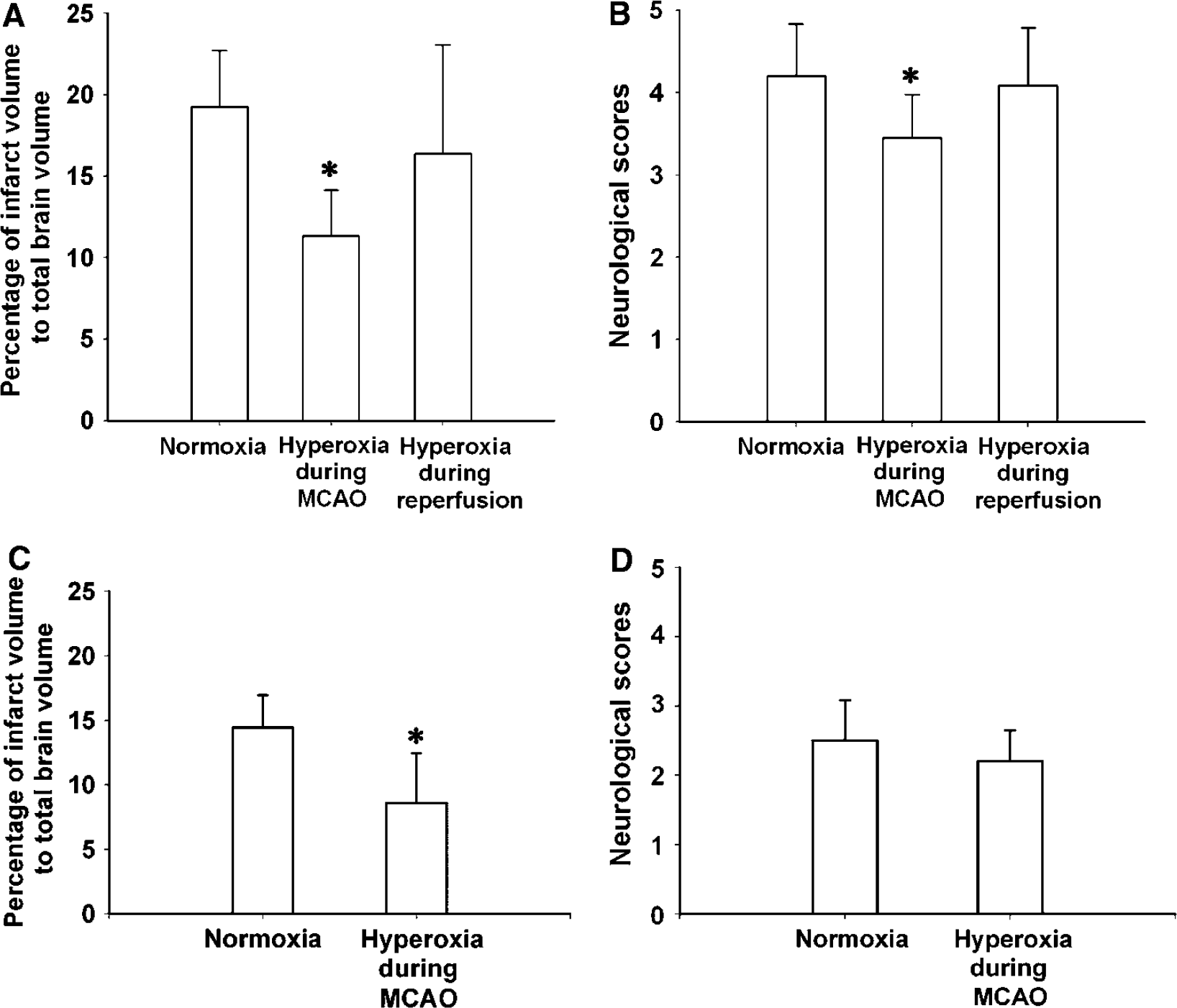
Effect of normobaric hyperoxia on infarction volumes and neurologic scores in normoxia and hyperoxia treated rats. (
The results of the neurologic function assessment using Rogers' 8-point neurologic scale demonstrated (Figure 2B) that the normoxia animals had an average score of around 4.2, when measured after 90 mins reperfusion. Significant improvements (P < 0.05) in neurologic function were found in the group of hyperoxia delivered during ischemia, but not in the group of hyperoxia delivered during reperfusion (P > 0.05). The scores were reduced by approximately 0.8 points for the group that received hyperoxia treatment during ischemia. When evaluated at 72 h after ischemia, the neurologic scores for both groups improved considerably, and the hyperoxia treated group remained to be better than the control group, although the difference in scores was not statistically significant (Figure 2D).
Superoxide Production after Normobaric Hyperoxia
The fluorescent dye, HEt, reacts with superoxide to produce ethidium (Et), which has been widely used as a biomarker of superoxide production. Almost no Et-like signal was observed in the nonischemic control group (normoxia) as well as in the nonischemic hyperoxia group (Figures 3A and 3B). Little signal was observed in the contralateral side of animals subject to cerebral ischemia and reperfusion (Figure 3C). The Etlike signal was markedly increased and lit up the shape of neuronal bodies in the ischemic penumbra in animals subject to 90-mins ischemia and 90-mins reperfusion (Figure 3D), suggesting the production of superoxide during cerebral ischemia and reperfusion. Hyperoxia treatment made no difference in the intensity of the Etlike signal regardless of whether it was delivered during ischemia or during reperfusion, as shown in Figures 3E and 3F.
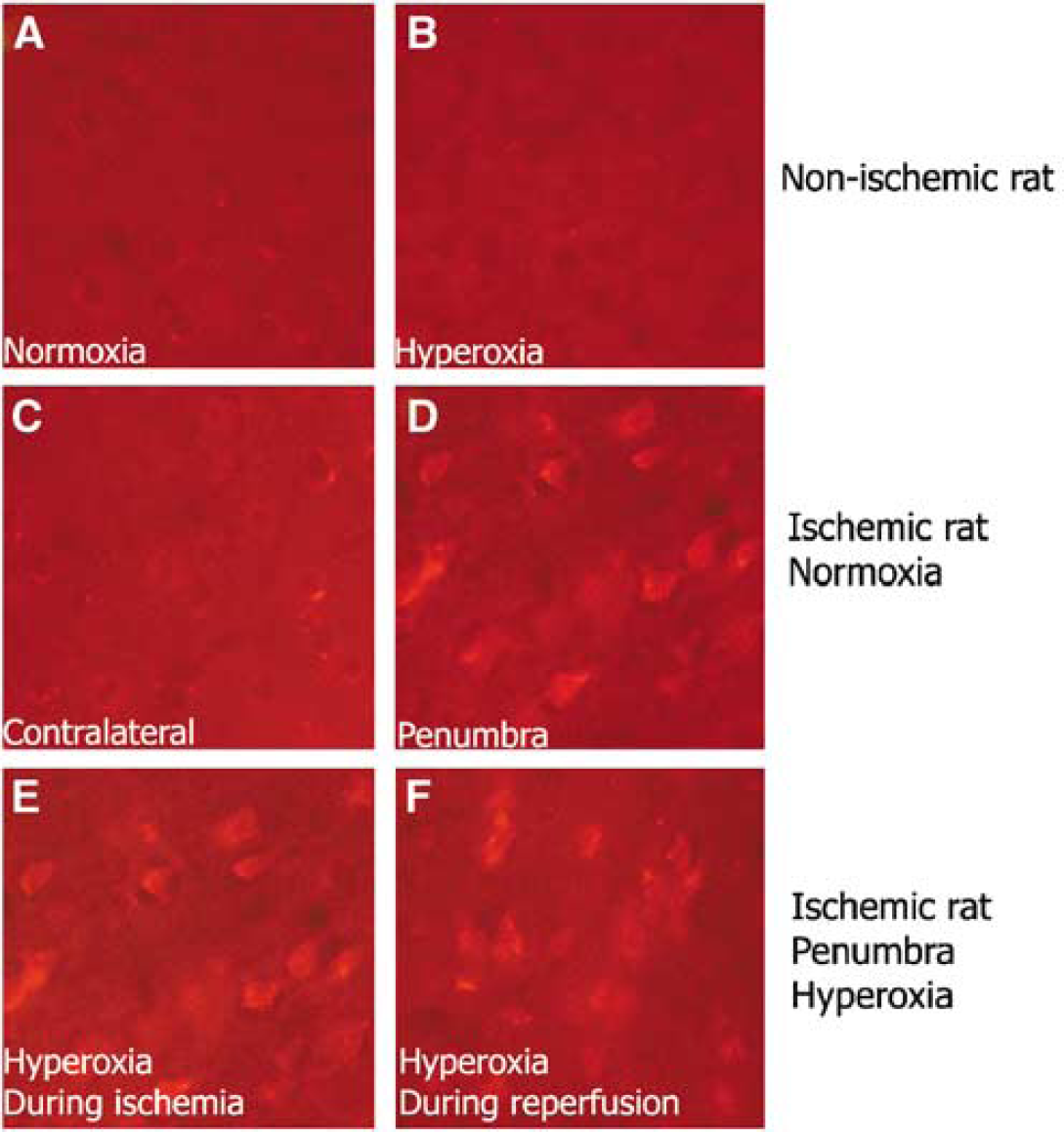
Dihydroethidium detection of superoxide production in ischemic rats with normoxic or hyperoxic treatment. Bright red Et-like signal indicates superoxide production. Normoxia, rats breathing 30% oxygen. (
8-hydroxy-2′-deoxyguanine Measurement by High-pressure Liquid Chromatography with Electrochemical Detection
Although the use of the fluorescence method with HEt demonstrated the generation of superoxide after cerebral ischemia and reperfusion, as a qualitative technique, it was unable to differentiate any potential change in the free radical production due to hyperoxia treatment. We next utilized the production of 8-OHdG as a highly quantitative method for the generation of hydroxyl radicals, which are derived from superoxide. As shown in Figure 4, cerebral ischemia and reperfusion markedly increased the production of 8-OHdG in the ischemic penumbra, as compared with the contralateral side (which has values similar to those obtained in nonischemic rats). Hyperoxia treatment with 95% O2 during ischemia significantly (P < 0.05) decreased penumbral 8-OHdG production in comparison to the normoxia group. It is also interesting to note that hyperoxia treatment given on reperfusion was not found to increase 8-OHdG production.
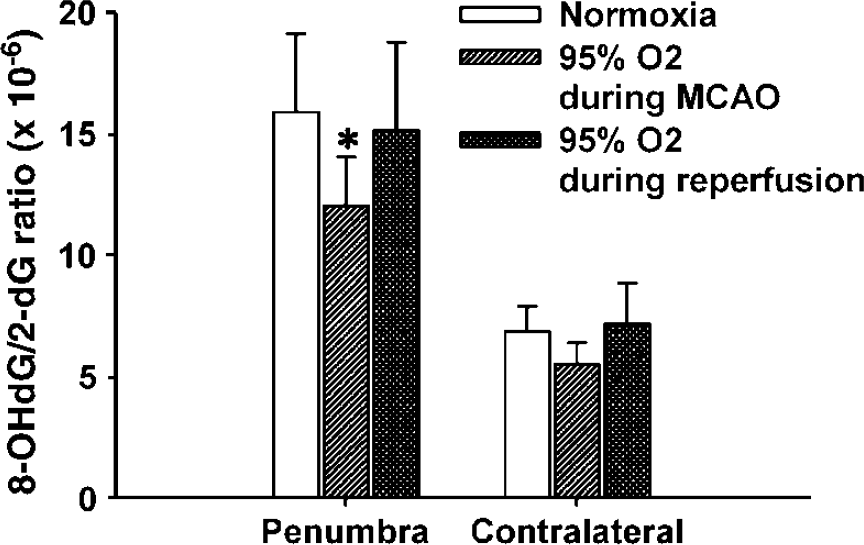
Effect of hyperoxia therapy on 8-OHdG production in penumbra and contralateral side of normoxia and hyperoxia groups. Normobaric hyperoxia was delivered during 90-mins ischemia or during 90-mins reperfusion. The production of 8-OHdG was significantly reduced in the group of hyperoxia delivered during ischemia (P < 0.05). The 8-OHdG level is presented as a ratio to 2-dG × 10−6. Data are expressed as Mean±s.d., n = 6 in each group. Ischemia and reperfusion induced a significant increase of 8-OHdG production in penumbra in normoxia and hyperoxia groups. Asterisk, P < 0.05 when compared with the same region of normoxia group.
Matrix Metalloproteinase Expression and Caspase Activation
Since the enhanced function of MMPs and cleavage of caspases are two well-characterized mechanisms leading to cerebral injury that are regulated through free radical generation, we hypothesized that decreased free radicals generation due to hyperoxia treatment may attenuate the expression and activation of MMPs and caspase in the ischemic penumbra. These processes may in part be involved in the mechanism of the protective effect of normobaric hyperoxia. Figure 5 reveals that cerebral ischemia under normoxia conditions considerably increased the levels of both MMP-2 and MMP-9 in the ischemic penumbra, as compared with the contralateral side of the ischemic brain. In the normoxia group, MMP-9 levels in the penumbra were approximately four times of those in the contralateral side. Hyperoxia treatment significantly (P < 0.05) decreased the penumbral MMP-9 levels by 41% and 59% during MCAO and reperfusion, respectively (Figure 5A). In contrast, the MMP-2 levels were not affected by the hyperoxia treatment during MCAO, but decreased significantly in response to hyperoxia treatment during reperfusion (Figure 5B).
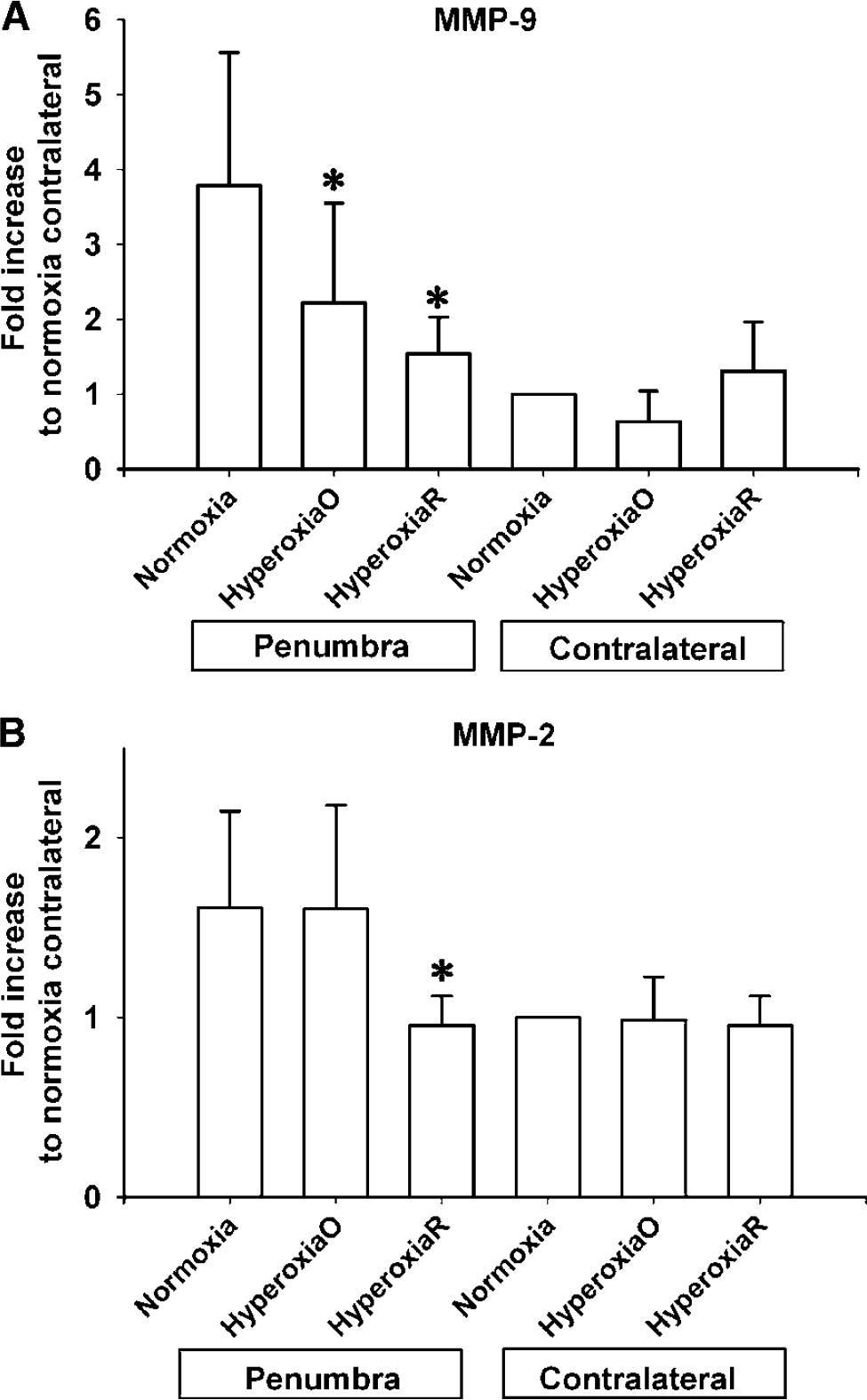
Effect of hyperoxia therapy on the expression of MMP-9(
Using caspase cleavage in the contralateral side of the normoxia group to normalize caspase levels in the penumbra of the normoxia group and in both sides of the hyperoxia group, we found that acute focal cerebral ischemia induced a substantial increase in the levels of 22 kDa caspase-8 in the penumbra (Figure 6). Hyperoxia treatment significantly (P < 0.05) decreased caspase-8 cleavage in the penumbra by 31% and 25% during MCAO and reperfusion, respectively. Interestingly, increased caspase cleavage was observed in the contralateral side on hyperoxia treatment.
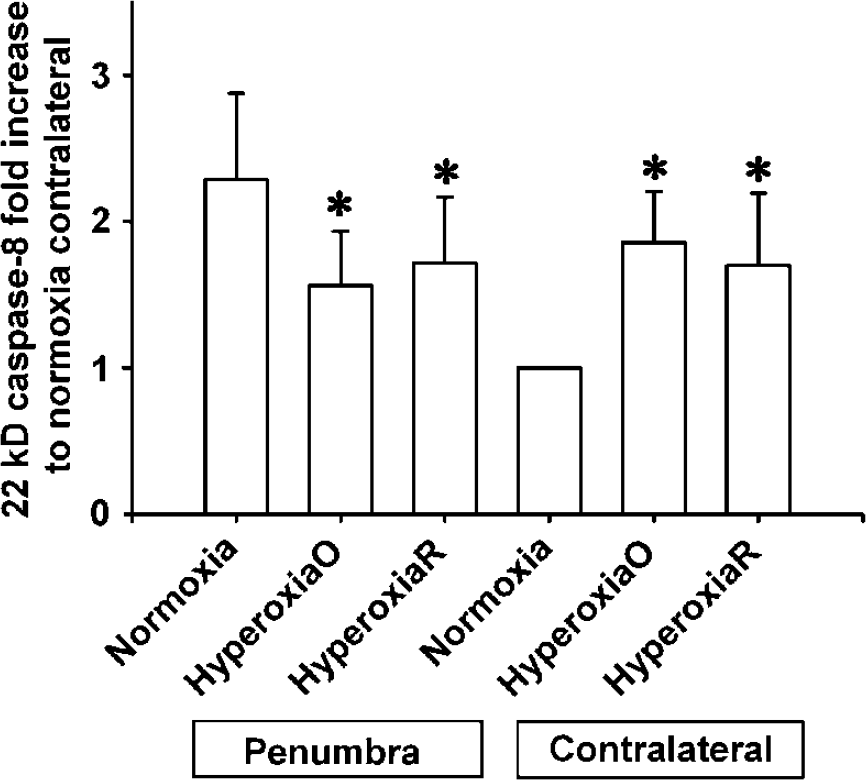
Effect of hyperoxia therapy on penumbral 22-kDa caspase-8 activation. Normobaric hyperoxia was delivered during 90-mins ischemia (HyperoxiaO) and 90-mins reperfusion (HyperoxiaR); it significantly reduced caspase-8 activation in penumbra, but increased caspase-8 activation in the contralateral side (P < 0.05). Caspase-8 activation is expressed as fold increase in comparison to contralateral side of normoxia group. Data are expressed as Mean±s.d., n = 6 in each group. Asterisk, P < 0.05 compared with the same region of normoxia group.
The Effect of Normobaric Hyperoxia on Cerebral Blood Flow
Our results showed (Figure 7) that compared with the pretreatment values, normobaric hyperoxia decreased cerebral blood flow in non-MCAO rats by nearly 40%, and also the blood flow in the contralateral side of ischemic rats by 20%. Administration of 95% oxygen during ischemia increased penumbral blood flow by 30% of the value before treatment, and the same treatment decreased penumbral blood flow by 10% when hyperoxia was delivered during reperfusion. Normobaric hyperoxia has no significant effect on the blood flow in core region.
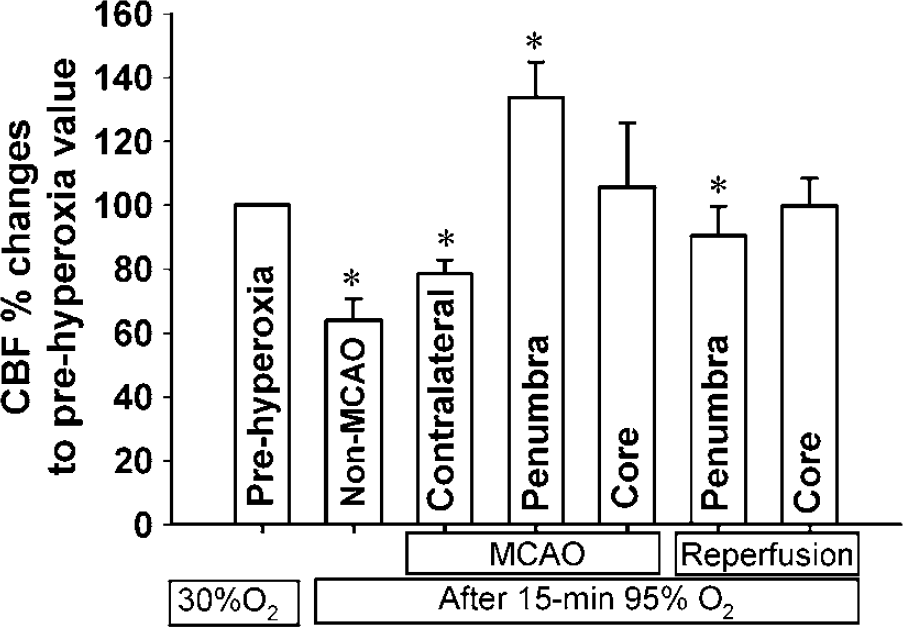
Effect of normobaric hyperoxia treatment on cerebral blood flow measured by laser Doppler. Hyperoxia significantly reduced cerebral blood flow in non-MCAO animals and in the contralateral side of MCAO animals. Hyperoxia delivered during MCAO significantly increased penumbral blood flow while it significantly decreased the blood flow when delivered during reperfusion. The blood flow in the core region was not affected by hyperoxia during both MCAO and reperfusion. Blood flow was normalized as percent changes to the prehyperoxia values in each group, and expressed as Mean ± s.d., n = 4 in each group. Asterisk, P < 0.05 compared with prehyperoxia value.
Discussion
The results presented in this study demonstrate that normobaric hyperoxia (95% O2 with 5% CO2) administered immediately after the occlusion of MCA for 90 mins maintained penumbral oxygenation status close to preischemic levels, which significantly reduced infarction volume and improved neurologic function. Further experiments showed that the improved penumbral oxygenation levels resulted in reduced ROS production, decreased MMP and caspase cleavage in the penumbral tissue.
The efficiency of hyperoxia treatment in improving penumbral oxygenation is critical to the outcome of hyperoxia therapy. To attain the benefits of normobaric hyperoxia treatment, the interstitial pO2 level must be raised to, and probably not higher than, a proper range. Most studies concerning oxygen therapy were conducted without monitoring interstitial pO 2 level, and as such, they are ‘blinded’ oxygen therapies. Blinded oxygen therapy may result in a great range of therapeutic effects, due to either over-oxygenation or under-oxygenation. With EPR oximetry-guided oxygen therapy, we found that 95% normobaric oxygen is indeed able to maintain the penumbral interstitial pO2 at close to preischemic levels. This maintenance of penumbral pO2 in the physiologic range during ischemia is likely responsible for the observed neuroprotective effects of the hyperoxia treatment. The results reported here underscore the importance of monitoring the actual interstitial pO2 during oxygen therapy. Magnetic resonance imaging (MRI) (McCoy et al, 1996) may potentially be helpful for ‘guided’ clinical oxygen therapy although obvious technical hurdles for measuring brain interstitial oxygen levels accurately in humans remain.
Reactive oxygen species may be generated when tissue oxygen level is too high or too low. Our results (Figure 1) indicate that acute cerebral ischemia decreased penumbral tissue pO2 level to approximately 25% of the preischemic level and that hyperoxia treatment can maintain penumbral pO2 at the levels close to the preischemic value. In this study we have shown that, contrary to common expectation, the normobaric hyperoxia treatment actually decreases ROS production in the penumbra when penumbralp O2 is maintained close to the preischemic level (Figure 4). In cerebral ischemia, ROS are involved in the activation of MMPs (Asahi et al, 2000; Gasche et al, 2001; Sugawara et al, 2001) and caspases (Morita-Fujimura et al, 2001; Anantharam et al, 2002). The decreased ROS production in penumbra may contribute to the attenuated expression of MMP-9 and the decreased cleavage of caspase-8 that were observed in this study. This result is consistent with the most recent study by Kim et al (Kim et al, 2005), in which they found that pro-MMP-9 level in the ischemic brain at 6 h after ischemia is significantly less in animals receiving normobaric hyperoxia comparing with control rats. However, it is still unclear whether the changes in MMP-9 and caspase-8 expression contribute to the neuroprotection seen in hyperoxia therapy, or conversely, that the neuroprotection, and/or the reduced ROS production lead to the changes in MMP-9 and caspase-8 in the penumbral tissue. It is likely that the changes of MMP-9 and caspase-8 are due to the direct effect of hyperoxia treatment or indirect effect of reduced ROS production because when the treatment was delivered during reperfusion it resulted in no significant reduction in infarction volume, but still altered the levels of MMP-9, MMP-2 (Figures 5A and 5B) and caspase-8 (Figure 6). Interestingly, the caspase-8 cleavage in the contralateral hemisphere of hyperoxia treated animals was also increased. Since the tissue pO2 in the contralateral side is highly elevated due to the hyperoxia treatment, the observation may suggest that hyperoxia could induce the apoptotic process in the nonischemic region. It is not clear whether this is a transient phenomenon, or a detrimental event that would lead to permanent injury. Further study is required to elucidate the mechanism of hyperoxia induced caspase-8 cleavage and related apoptosis, as well as its implication.
It is worth pointing out that hyperoxia seems more tolerable to the brain than hypoxia. This is demonstrated by the findings that during hyperoxia treatment ROS production in the contralateral side was not increased (Figures 3 and 4) although a pO2 level much higher than the physiologic values has been achieved there. Furthermore, when 95% oxygen was delivered during reperfusion, although it resulted in a pO2 level in penumbra that is twice of the preischemic level we failed to find a concomitant increase in ROS generation (Figures 3 and 4). These results are further supported by a recent report that normobaric hyperoxia treatment during focal cerebral ischemia–reperfusion does not increase oxidative stress, as measured by heme oxygenase-1 induction and protein carbonyl formation (Singhal et al, 2002b). Together, these findings suggest that under appropriate conditions normobaric hyperoxia treatment would not cause observable increase in the production of ROS, while it may actually decrease ROS generation in the penumbra during ischemia. Since ROS generation is a major contributor to the ischemia-induced brain injury, it is reasonable to speculate that combined therapy of the administration of antioxidants in conjunction with hyperoxia treatment could potentially bring about even greater neuroprotection than individual treatment alone.
In our study, significant reduction of infarction volume was found only when the treatment was administered immediately after ischemia and the reduced infarction volume was maintained for up to 72 h after ischemia. This result is similar to the report on human study (Singhal et al, 2005). The failed neuroprotection for 90-mins hyperoxia delivered during reperfusion is possibly due to the length of ischemia and the late starting time of treatment. Considering currently no reported study indicating injury from early initiation of normobaric hyperoxia, it is potentially conceivable that early initiation of normobaric oxygen therapy may produce greater benefits. The optimized oxygen dose, the appropriate starting time, and the duration of normobaric oxygen treatment for different conditions of cerebral ischemia will likely vary significantly, and should be studied specifically in the future. Since oxygen can be delivered in an ambulance or even at the patient's home, it could be used in the acute stage to expand the therapeutic window for the patient for further aggressive treatment in the hospital.
The mechanism for normobaric hyperoxia in the production of the observed neuroprotective effect remains unknown. It is believed that hyperoxia produces benefits through the favorable alteration of metabolic environments and perfusion conditions in the ischemic region (Singhal et al, 2002b). Our results showed that hyperoxia treatment when delivered during ischemia actually increased penumbral blood flow while it decreased penumbral blood flow if delivered during reperfusion. The result is consistent with Singhal's most recent report on human (Singhal et al, 2005). The improved blood flow may contribute to the neuroprotection by hyperoxia. The attenuated of MMP-9 expression and caspase-8 cleavage observed in this study indicate that MMP and caspase-8 may play a role in the hyperoxia-induced neuroprotection.
In conclusion, the present study demonstrates that normobaric hyperoxia therapy with 95% O2 given during ischemia maintains the penumbral pO2 at the physiologic levels, reduces infarction volume, and improves neurologic function in acute focal cerebral ischemia. The improved penumbral oxygenation status leads to reduced ROS production, decreased expression of MMP-9, and decreased cleavage of caspase-8 in the penumbra.
Footnotes
Acknowledgements
The authors thank Professor Harold Swartz from Dartmouth Medical School for providing LiPc.
