Abstract
For brain tissue regeneration, any scaffold for migrated or transplanted stem cells with supportive angiogenesis is important once necrotic brain tissue has formed a cavity after injury such as cerebral ischemia. In this study, a new porous gelatin–siloxane hybrid derived from the integration of gelatin and 3-(glycidoxypropyl) trimethoxysilane was implanted as a three-dimensional scaffold into a defect of the cerebral cortex. The porous hybrid implanted into the lesion remained at the same site for 60 days, kept integrity of the brain shape, and attached well to the surrounding brain tissues. Marginal cavities of the scaffolds were occupied by newly formed tissue in the brain, where newly produced vascular endothelial, astroglial, and microglial cells were found with bromodeoxyuridine double positivity, and the numbers of those cells were dose-dependently increased with the addition of basic fibroblast growth factor (bFGF) and epidermal growth factor (EGF). Extension of dendrites was also found from the surrounding cerebral cortex to the newly formed tissue, especially with the addition of bFGF and EGF. The present study showed that a new porous gelatin–siloxane hybrid had biocompatibility after implantation into a lesion of the central nervous system, and thus provided a potential scaffold for cell migration, angiogenesis and dendrite elongation with dose-dependent effects of additive bFGF and EGF.
Introduction
Although the regenerative potential of mammalian central nervous system (CNS) has long been believed to be extremely limited (Ramon y Cajal, 1928), the recent discovery of neural stem cells (NSC) has given rise to a new approach for restorative medicine even in the CNS (Snyder et al, 1997). In addition, stem cells from other sources such as adult bone marrow stromal cells (BMC; Chen et al, 2001), umbilical cord cells (UCC; Chen et al, 2003), and adipotissue cells (Kang et al, 2003) have been shown to have the ability to protect brain tissue from injury, to promote exogenic regeneration, and to reconstitute the neural network by their transplantation. Furthermore, it has been shown that neurotrophic or growth-promoting factors or gene transfer by a plasmid and virus vector promoted endogenous neurogenesis (Nakatomi et al, 2002), and results of combination studies by transplantation of stem cells in which the gene for a neurotrophic factor was transferred were recently reported (Kurozumi et al, 2004). These strategies for regenerative therapies have mainly focused on the acute or subacute phase of brain injury, and the necrotic core was therefore not saved or repaired because of migrated or transplanted cell loss (Snyder and Park, 2002). Any scaffold for migrated or transplanted cells with supportive angiogenesis could greatly reduce such problems.
Biodegradable scaffolds are important as biocompatible conduits for tissue repair and regeneration. For many organs other than the CNS, the usefulness of natural biodegradable polymers, such as collagen, gelatin, fibrin and alginates, or synthetic biodegradable polymers, has been investigated (Griffith, 2002). In the clinical setting, a three-dimensional interconnected porous scaffold has been used for reconstructive surgery of bone defects because of tumor resection, trauma, or congenital abnormality (Kaito et al, 2005). Even in the field of CNS, amputated axons of the spinal cord could be elongated through an alginate sponge implanted in the gap to re-enter the other stump of the transected cord (Kataoka et al, 2001). Hyaluronic acid porous hydrogel has been implanted into defects of the cerebral cortex (Hou et al, 2005; Tian et al, 2005). As a result, regeneration from large loss in the brain tissue could be enhanced not by simple implantation of a scaffold, but rather by additive growth factors such as basic fibroblast growth factor (bFGF) and epidermal growth factor (EGF) to further potentiate cell proliferation.
In this study, therefore, a new porous gelatin–siloxane hybrid derived from the integration of gelatin and 3-(glycidoxypropyl) trimethoxysilane (GPSM) was chosen as a biodegradable scaffold (Ren et al, 2001a). This is a three-dimensional scaffold with both strength and slow biodegradation easily controlled by the amount of the bridging bonds or cross-link density between gelatin chains and GPSM molecules without any inhibitory effect on cell proliferation in vitro (Ren et al, 2002). Thus, the present study was designed to determine whether the implantation of porous gelatin–siloxane hybrids could establish a meaningful scaffold for regeneration of a cerebral cortical defect, and whether addition of bFGF and EGF enhances cell genesis after the cerebral defect (Nakatomi et al, 2002; Jin et al, 2003).
Materials and methods
Preparation and Filling of a Scaffold with Basic Fibroblast Growth Factor and Epidermal Growth Factor
A hybrid porous scaffold was prepared using gelatin and silicate through a procedure of sol–gel processing, post-gelation, and freeze-drying as described previously (Ren et al, 2002). This material was degraded about 25% by 35 days of Tris buffer incubation (Ren et al, 2001b). Pore size of the resulting hybrid scaffold was 190 ± 15 mm. Before implantation, the scaffold was cut into cubic blocks of 2 × 2 × 5 mm (20 mm3) in size.
Solutions of bFGF and EGF (Roche Diagnostics, Manheim, Germany) were prepared with concentrations of 0, 100, and 500 mg/mL. The aqueous solutions of bFGF (10 mL) and EGF (10 mL) were mixed and dropped onto 20 mm3 of porous gelatin–siloxane hybrids, and then the hybrids were left at 25°C for 1 h for impregnation of bFGF and EGF into the porous hybrids. The bFGF and EGF solutions were almost totally absorbed into the porous hybrids through this impregnation process.
Animals and Surgical Procedure
All experimental procedures were approved by the Animal Committee of the Graduate School of Medicine, Dentistry, and Pharmacy, Okayama University. Male Wistar rats at the age of 12 weeks (body weight: 250 to 280 g) were anesthetized with an intraperitoneal injection of pentobarbital (10 mg/250 g rat) and placed in a stereotaxic apparatus. A piece of bone over the right frontal cortex was excised with a dental drill, and an approximately 2 × 5 mm defect in the superficial brain was made by cutting and aspiration under a surgical microscope. The defect was positioned at 3.0mm anterior to 2.0 mm posterior of the bregma (5 mm in length) and 2.0 to 4.0 mm (2 mm in width) lateral to the midline, and the depth of the lesion was approximately 2.0 mm below the skull surface. All meninges overlying the cavity were carefully removed. The block of porous gelatin–siloxane hybrids with or without bFGF and EGF was placed into the lesion immediately or at 30 days after making the defect. The defect was subsequently covered by skull bone removed at the beginning of the surgery, and the wound was closed by Aron Alpha, followed by sawing the scalp. All of the above surgical procedures were performed in a sterile manner.
To identify the newly produced cells, bromodeoxyuridine (BrdU; Sigma, St Louis, MO, USA) dissolved in saline was intraperitoneally injected (50 mg/kg) at 1, 2, and 3 days after implantation of the scaffold twice per day (total dose of 300 mg/kg rat). For examination of timing at scaffold implantation between acute and chronic phase after brain injury and of the efficacy of bFGF and EGF, the experimental schedule, which is making a defect in the cerebral cortex, implantation timing of scaffold, BrdU injection, and killing, was programmed as summarized in Figure 1.
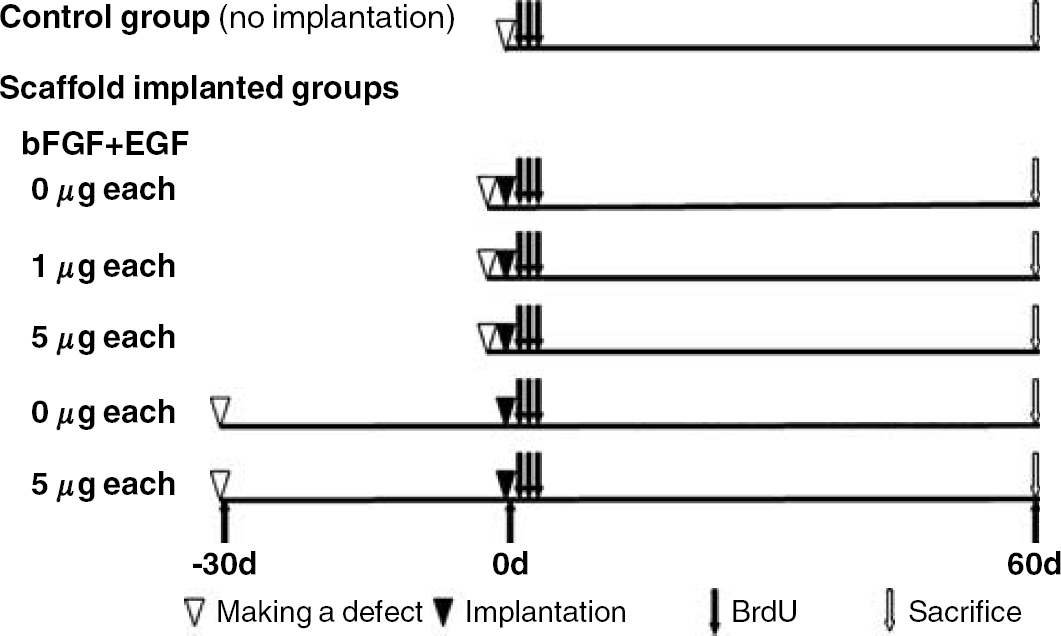
Experimental schedule for making a defect in the cerebral cortex, implantation of a scaffold, bromodeoxyuridine (BrdU) injection, and killing. After making a cerebral cortical defect, the rats received only BrdU (control group) or additional scaffold implantation (scaffold implanted groups) immediately or 30 days after making the defect.
Preparation and Evaluation of Brain Tissues
Under deep anesthesia with pentobarbital (20 mg/250 g rat), the rats (n = 5 in each group) were transcardially perfused with 5 U/mL heparinized saline followed by 4% paraformaldehyde in phosphate buffer (PB). The whole brain was subsequently removed and immersed in the same fixation for 12 h at 4°C. After washing out paraformaldehyde by sucrose solution in PB, the brains were rapidly frozen by dry ice. Coronal brain sections of 20 μm in thickness at the caudate level were prepared by using a cryostat and mounted on a silane-coated slide glass.
For quantitative analysis of the newly formed tissue, the sections were stained with hematoxylin and eosin (HE) and observed with a light microscope (Olympus BX-51; Olympus Optical). The area of the new tissue formed in the space of the implanted porous scaffold was measured in four sections by pixel counting using a computer program of Photoshop 6.0, and the volume was calculated. The volumes of the newly formed tissue in the experimental groups were statistically analyzed using a one-way analysis of variance (ANOVA) with post hoc test.
Single Immunohistochemical Analysis
To identify the types of cells in newly formed tissue, single immunohistochemical analysis was performed for N-acetylglucosamine oligomers (NAGO), glial fibrillary acidic protein (GFAP), ionized calcium-binding adapter molecule-1 (Iba1), neuron-specific nuclear protein (NeuN) and BrdU. Lycopersicon esculentum lectin (LEL) is a glycoprotein with affinity for NAGO, which mature vascular endothelial cells express (Porter et al, 1990; Augustin et al, 1995), and GFAP, Iba1, NeuN are markers of differentiating astroglial, microglial (Imai et al, 1996) and neuronal cells, respectively. Bromodeoxyuridine was used for the detection of newly synthesized DNA. The slides were first incubated in 1% hydrogen peroxide to quench endogenous peroxidase activity. For detection of BrdU, the slides were incubated in 2 mol/L HCl at room temperature for 30 mins to denature DNA. After blocking nonspecific reaction by bovine serum albumin (BSA), the slides were incubated with LEL or the first antibody at room temperature for 2 h. Lycopersicon esculentum lectin and the first antibodies were chosen with each dilution as follows: biotinylated LEL (Vector Laboratories, Burlingame, CA, USA) at 1:200, mouse monoclonal anti-GFAP antibody (Chemicon, Temecula, CA, USA) at 1:200, rabbit polyclonal anti-Iba1 antibody (a gift from Dr Imai Y at Ehime University, Touon, Japan) at 1:200, mouse monoclonal anti-NeuN antibody (Chemicon) at 1:200, and mouse monoclonal anti-BrdU antibody (Oncogene Research Products, Boston, MA, USA) at 1:200. The slides were washed with phosphate-buffered saline (PBS) and then incubated with biotinylated anti-mouse IgG antibody (Vector Laboratories) or biotinylated anti-rabbit IgG antibody (Vector Laboratories) at 1:200 for 1 h at room temperature. They were subsequently incubated with avidin–biotin–peroxidase complex (Vector Laboratories) for 30 mins and then developed using diaminobenzidine as a peroxidase substrate. In each study, a set of sections was stained in a similar way without the primary antibody as a negative control.
Double Fluorescent Immunohistochemistry
To determine whether BrdU-labeled cells differentiate into vascular endothelial, astroglial, and microglial cells, double immunofluorescent studies were performed for BrdU plus LEL or GFAP or Iba1. To examine axonal growth into the regenerative tissue, a double immunofluorescent study for BrdU plus microtubule-associated protein 2 (MAP2), a marker of the neural dendritic process, was also performed.
To make BrdU easily detected, the slides were first treated with HCl and BSA as described previously and then incubated with sheep polyclonal anti-BrdU antibody (Biodesign, Saco, MA, USA) at 1:200 and LEL or other antibodies for GFAP, Iba1, and MAP2. After washing in PBS, the slides were incubated with FITC-labeled anti-sheep IgG antibody (Sigma) at 1:200 simultaneously with Texas red avidin D (Vector Laboratories) at 1:50 or Texas red-labeled anti-mouse IgG antibody (Vector Laboratories) at 1:200 or tetramethylrhodamine isothiocyanate-labeled anti-rabbit IgG antibody (Sigma) at 1:200. The slides were then covered with VECTASHIELD Mounting Medium with 4′, 6′-diamidino-2-phenylindole (Vector Laboratories). To confirm the specificity of the primary antibody, a set of sections was stained in a similar way without primary antibodies. The treated sections were scanned with a confocal microscope equipped with an argon and HeNe1 laser (LSM-510; Zeiss, Jena, Germany). Sets of fluorescent images were acquired sequentially for the red and green channels to prevent crossover of signals from green to red or from red to green channels.
Cell Counting
To evaluate the results of single immunohistochemical analysis quantitatively, the positively stained cells for NAGO, GFAP, Iba1, and BrdU in the newly formed tissue within the scaffold were counted in four coronal sections per rat brain. In the double fluorescent studies, the double-positive cells were counted in the same manner. Results are expressed as means ± standard deviation. The numbers of cells in the experimental groups were compared using one-way ANOVA with post hoc test.
Results
Shape of the Brain with or without Scaffold Implantation
The fresh brain in the control group without implantation showed distortion of the cerebral cortex and enlargement of the ipsilateral lateral ventricle (LV), resulting from the defect of the cerebral cortex for 60 days (Figures 2A and 2C). However, scaffolds made of porous gelatin–siloxane hybrid that had been implanted into the lesion area of the defected cerebral cortex remained in the same site for 60 days after implantation (Figure 2B). The structure of the porous scaffold, which was observed before implantation by a scanning electron microscope (JEOL JSM-6300CX), as shown in the inset of Figure 2B, remained almost intact in the brains of all rats (Figure 2D).
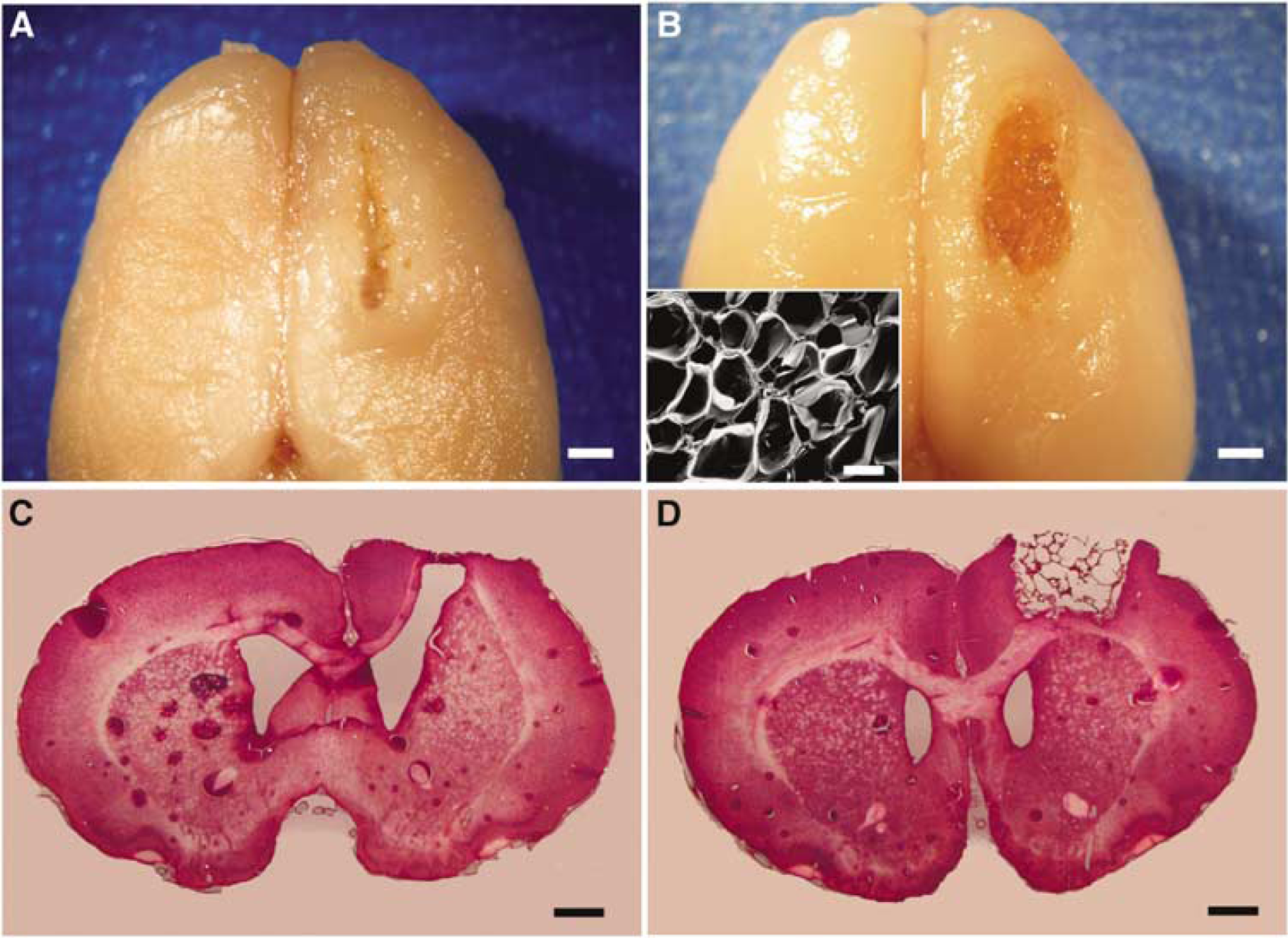
Whole brain (
Newly Formed Tissue After Scaffold Implantation
Newly formed tissue was not found in the brain without implantation of a scaffold after making a defect (Figure 3A). In contrast, the implanted scaffolds were well attached to the surrounding brain tissues, and the marginal cavities of the scaffolds were occupied by newly formed tissue in the brain (Figures 3B to 3F, insets). The new tissues were mainly formed at the site of the corpus callosum (CC; Figures 3B to 3F) and partly at the flanking cerebral cortices (Figures 3B to 3D). The volume of new tissue was dose-dependently increased with treatment of bFGF and EGF (Figures 3B to 3D and 3G). Implantation of the scaffold after 30 days of the primary defect resulted in only a small increase of new tissue (Figure 3E), but still amplified by additive bFGF and EGF (Figure 3F). Quantitative analysis (Figure 3G) showed that the volume of new tissue in the scaffolds was 1.46±0.37 mm3 when scaffolds were implanted immediately after making the defect (Figure 3G(b)), and the volumes increased to 2.10±0.23 (P = NS) and 2.77±0.79 mm3 (*P < 0.05) with 1 or 5 μg each of bFGF and EGF, respectively (Figures 3G(c) and 3(d)). However, the volume of new tissue was 0.70±0.16 mm3 when scaffolds were implanted 30 days after making the primary defect (Figure 3G(e)), and the volume slightly increased to 1.10±0.32 mm3 with 5 μg each of bFGF and EGF, significantly smaller than that in the group in which scaffolds were implanted immediately after making the defect and treated with 5 μg each of bFGF and EGF (†P < 0.05) (Figure 3G(f)).
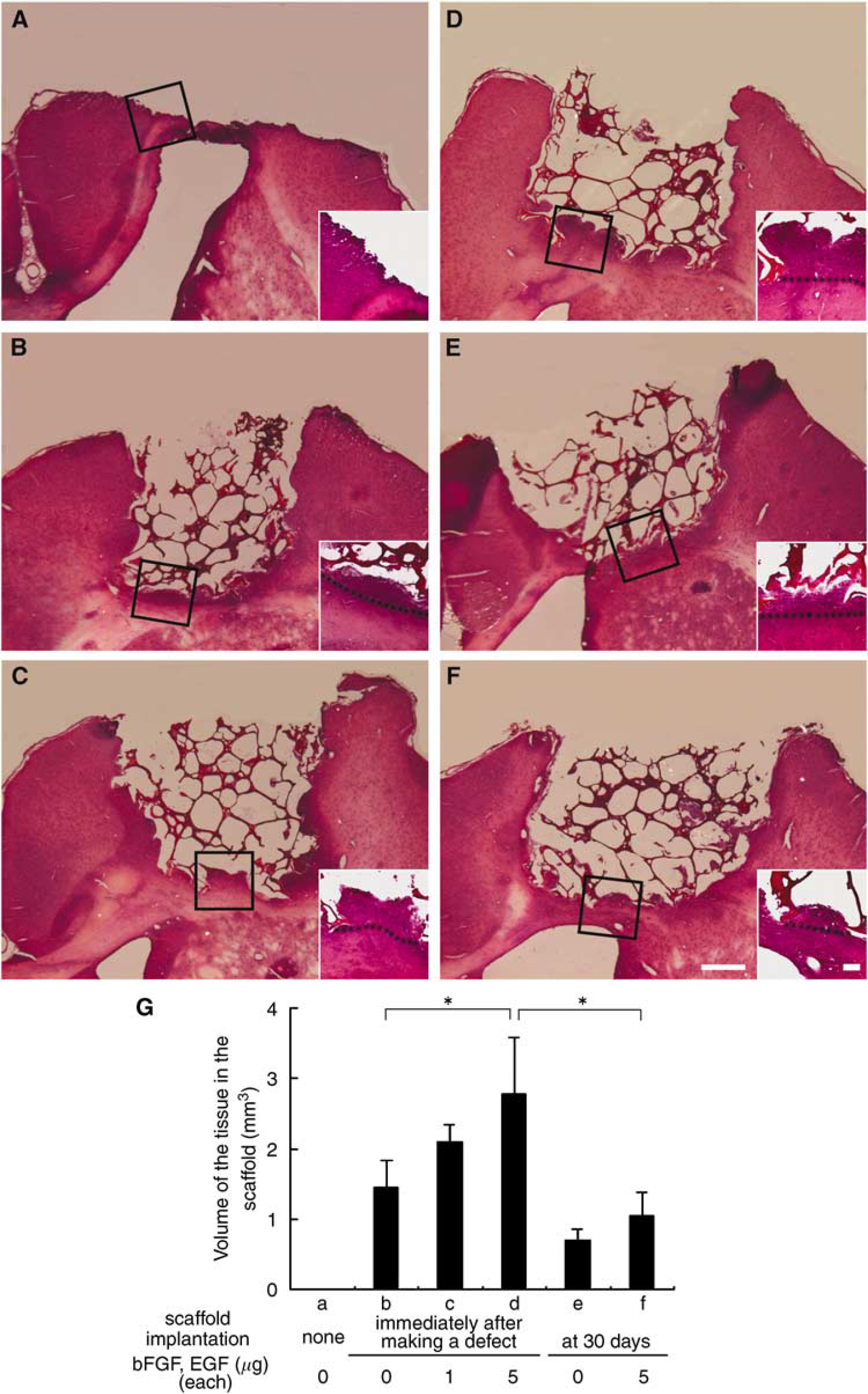
Representative hematoxylin and eosin (HE)-stained coronal sections without (
Components of the Newly Formed Tissue
Single immunohistochemistry revealed that NAGO-positive cells representing vascular endothelial cells showing round or tramlines were present in the newly formed tissue (Figure 4, left column) and that GFAP-positive cells dominated the newly formed tissue in the scaffold cavity (Figure 4, central column), where a small number of Iba1-positive cells were also found (Figure 4, right column). The three types of cells existed with a uniform distribution in the marginal cavities of the scaffolds in each experimental group (Figure 4). Conversely, NeuN-positive cells were not detected in the newly formed tissue (data not shown). The numbers of these three types of cells in the new tissue were dose-dependently increased with treatment of bFGF and EGF.
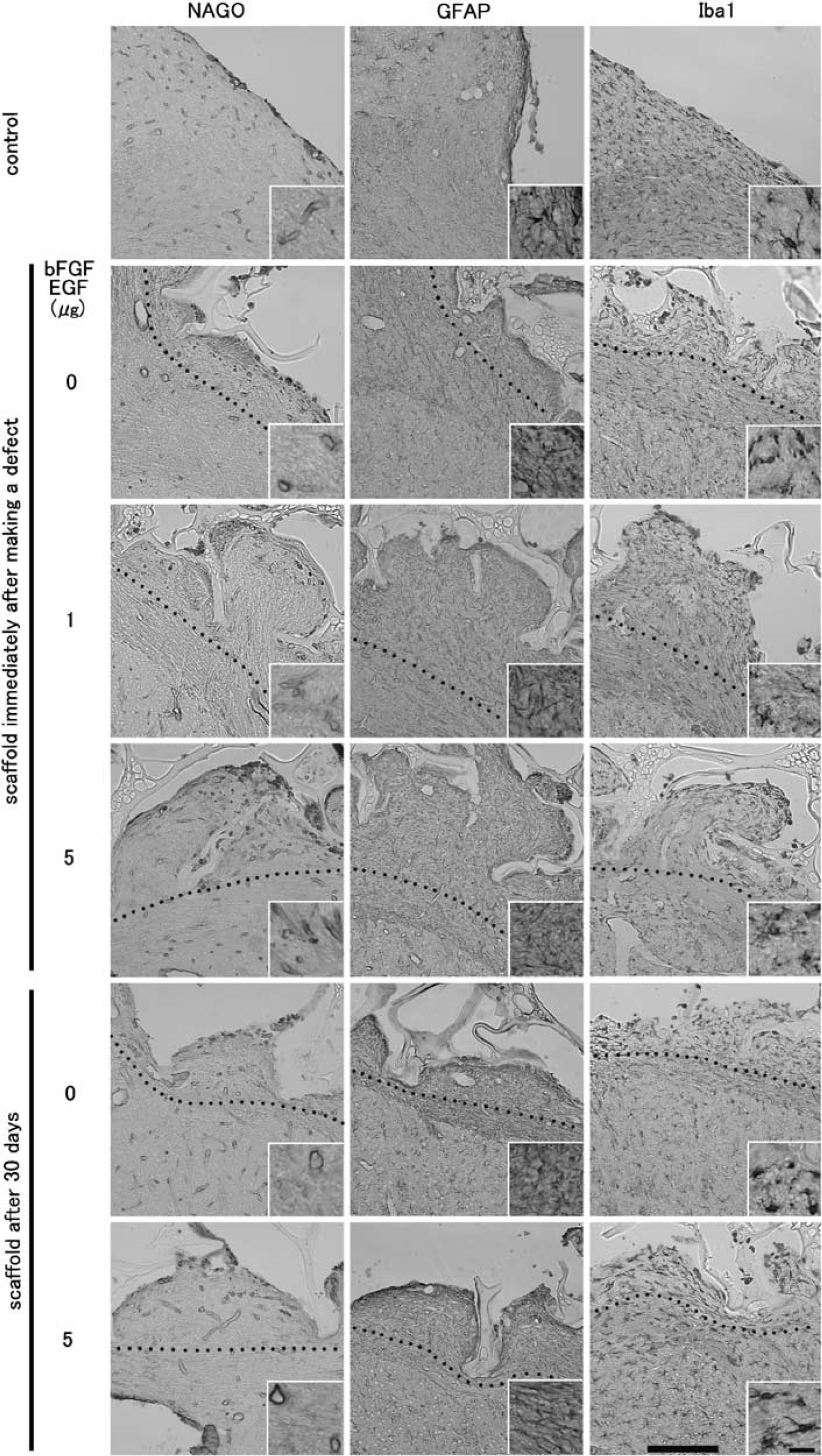
Representative photomicrographs of single immunohistochemistry for N-acetylglucosamine oligomers (NAGO), glial fibrillary acidic protein (GFAP), and ionized calcium-binding adapter molecule-1 (Iba1) of newly formed tissue at the site of the corpus callosum. Black dotted lines show the margin of the scaffold and original brain. Note uniform distribution of these three types of stainings in the marginal cavity of the scaffold in each experimental group. Insets show magnifications. Scale bars: panel, 200 μm; inset, 20 μm.
Quantitative analysis (Figure 5) showed that the numbers of NAGO-, GFAP-, and Iba1-positive cells in new tissue within the scaffolds were 30.8±8.6, 337.4±30.4, and 66.2±7.9, respectively, when the scaffolds were implanted immediately after making the defect (Figures 5A to 5C(b)), and that the numbers increased to 38.8±8.4, 420.6±38.9, and 124.3±6.6 (P = NS) with 1 μg each of bFGF and EGF and to 73.6±4.9, 620.8±63.3, and 153.8±12.0 (*P < 0.05, when implanted immediately with no bFGF and EGF or with 1 μg each of bFGF and EGF) with 5 μg each of bFGF and EGF (Figures 5A to 5C(c and d)). However, the numbers were 15.3±7.6, 169.3±35.4, and 41.5±5.8, respectively, when scaffolds were implanted 30 days after making the primary defect (Figures 5A to 5C(e)) and slightly increased to 23.3±16.6, 230.0±55.2, and 68.8±16.2 with 5 μg each of bFGF and EGF, although these numbers were significantly smaller than the numbers in the group in which scaffolds were implanted immediately after making the defect with 5 μg each of bFGF and EGF (*P < 0.05) (Figures 5A to 5C(f)).
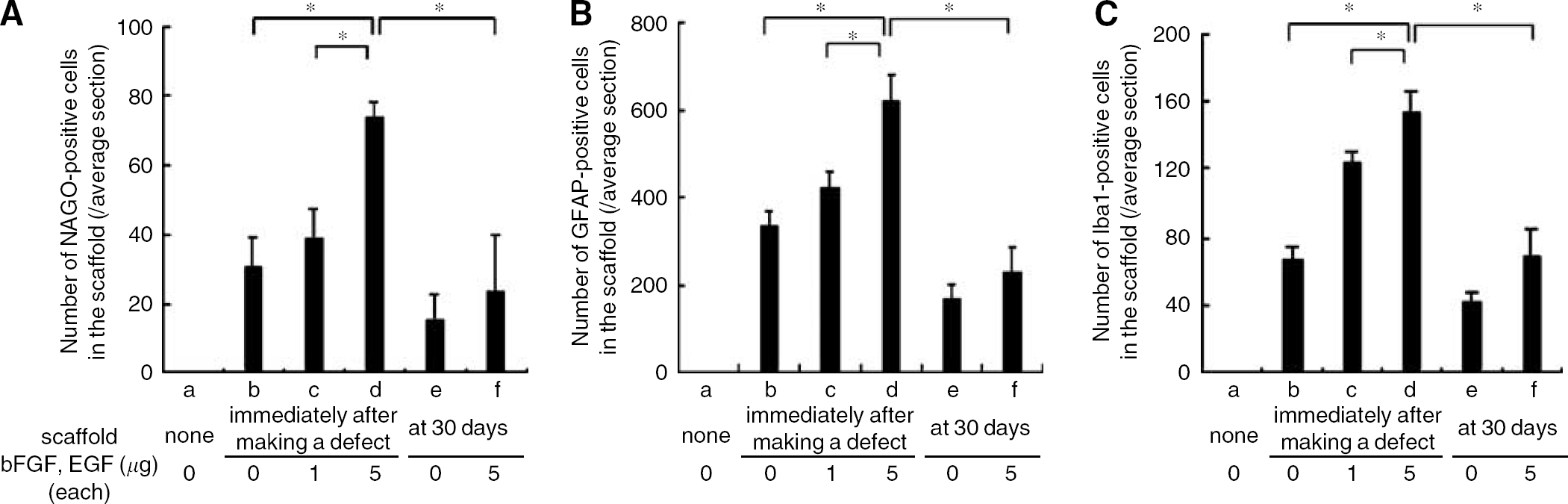
Numbers of N-acetylglucosamine oligomers (NAGO)-(
Distribution of Bromodeoxyuridine-Labeled Cells
Figure 6 shows the distribution of BrdU-labeled cells around the area with scaffold implantation. In each experimental group, many BrdU-labeled cells were accumulated in and around the newly formed tissue mainly at the site of the CC (Figure 6B, arrowheads), while a few cells were also found at the flanking cerebral cortex surrounding the scaffold (Figures 6A and 6C, arrowheads). Small numbers of BrdU-labeled cells were also found in the subventricular zone (SVZ) of the LV (Figure 6D, arrowheads) and also within and around the CC close to (Figure 6E, arrowheads) and distal from (Figure 6F, arrowheads) the LV. The number of BrdU-labeled cells in the newly formed tissue was dose-dependently increased with treatment of bFGF and EGF. Quantitative analysis (Figure 6H) showed that the number of BrdU-labeled cells in new tissue within the scaffolds was 41.4±17.2 when scaffolds were implanted immediately after making the defect (Figure 6H(b)), and that the number increased to 53.6±28.4 (P = NS) and to 89.5±23.2 (*P < 0.05) with 1 or 5 μg each of bFGF and EGF (Figure 6H(c and d)). However, the number was 18.3±8.5 when scaffolds were implanted 30 days after making the primary defect (Figure 6H(e)) and slightly increased to 28.9±12.8 with 5 μg each of bFGF and EGF, although the number was significantly smaller than the number in the group in which scaffolds were implanted immediately after making the defect with 5 μg each of bFGF and EGF (*P < 0.05; Figure 6H(f)).
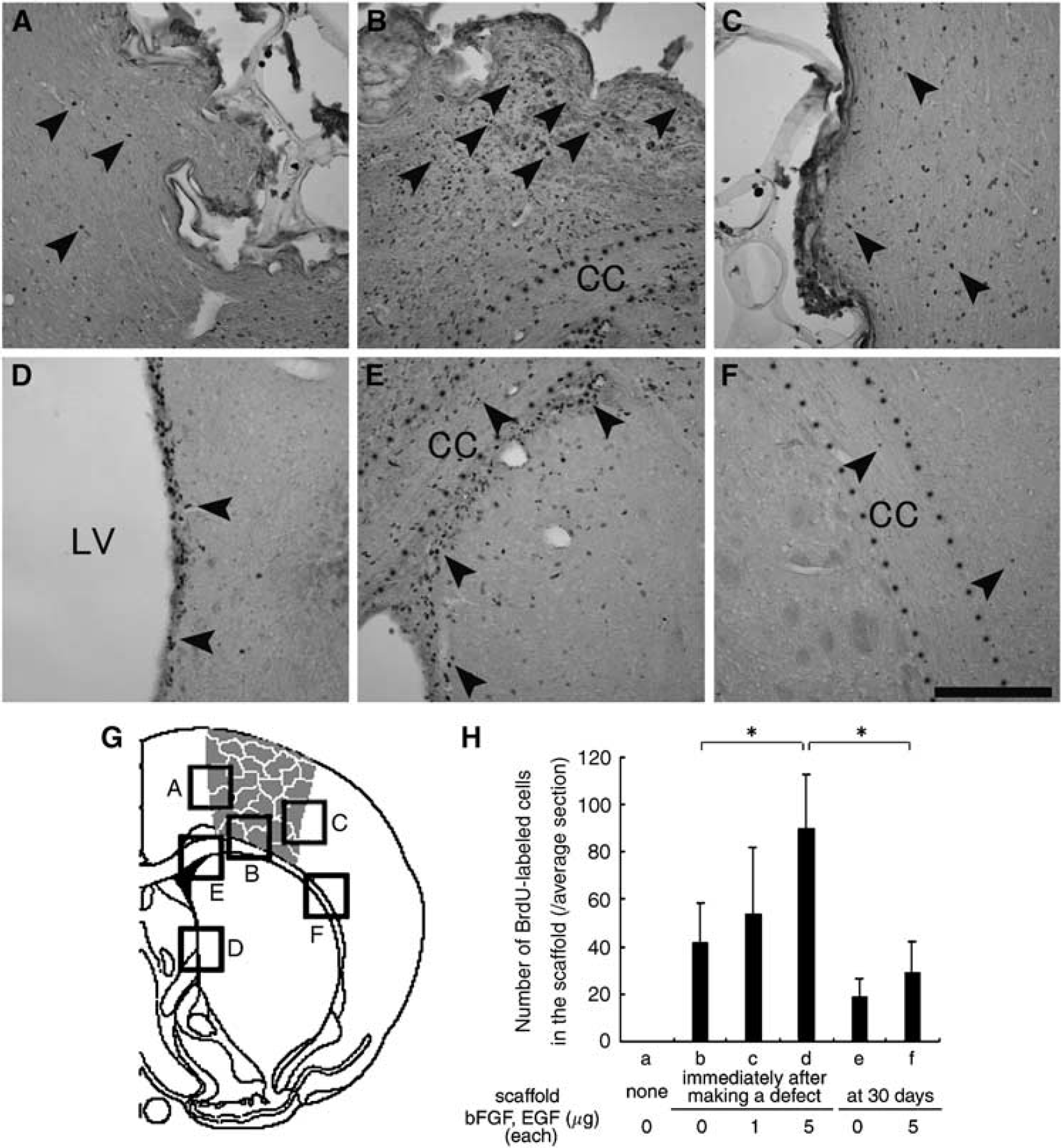
Distribution of bromodeoxyuridine (BrdU)-labeled cells in the group in which a scaffold was implanted immediately after making a defect with 5 μg each of basic fibroblast growth factor (bFGF) and epidermal growth factor (EGF) (
Phenotypes of Bromodeoxyuridine-Labeled Cells
The double fluorescent study showed that BrdU-labeled cells (Figure 7, green color) were double positive with a few NAGO (Figures 7A and 7B, red), many GFAP (Figures 7C and 7D, red), and a few Iba1 (Figures 7E and 7F, red). In new tissue formed in the scaffold implanted immediately after making a defect, the number of double-positive cells for NAGO were 1.2±0.8 and 3.6±0.9 without (Figure 7A) and with (Figure 7B) 5 μg each of bFGF and EGF, 14.5±2.9 and 42.1±3.2 for GFAP (Figures 7C and 7D), and 2.0±0.7 and 3.2±0.8 for Iba1 (Figures 7E and 7F), respectively.
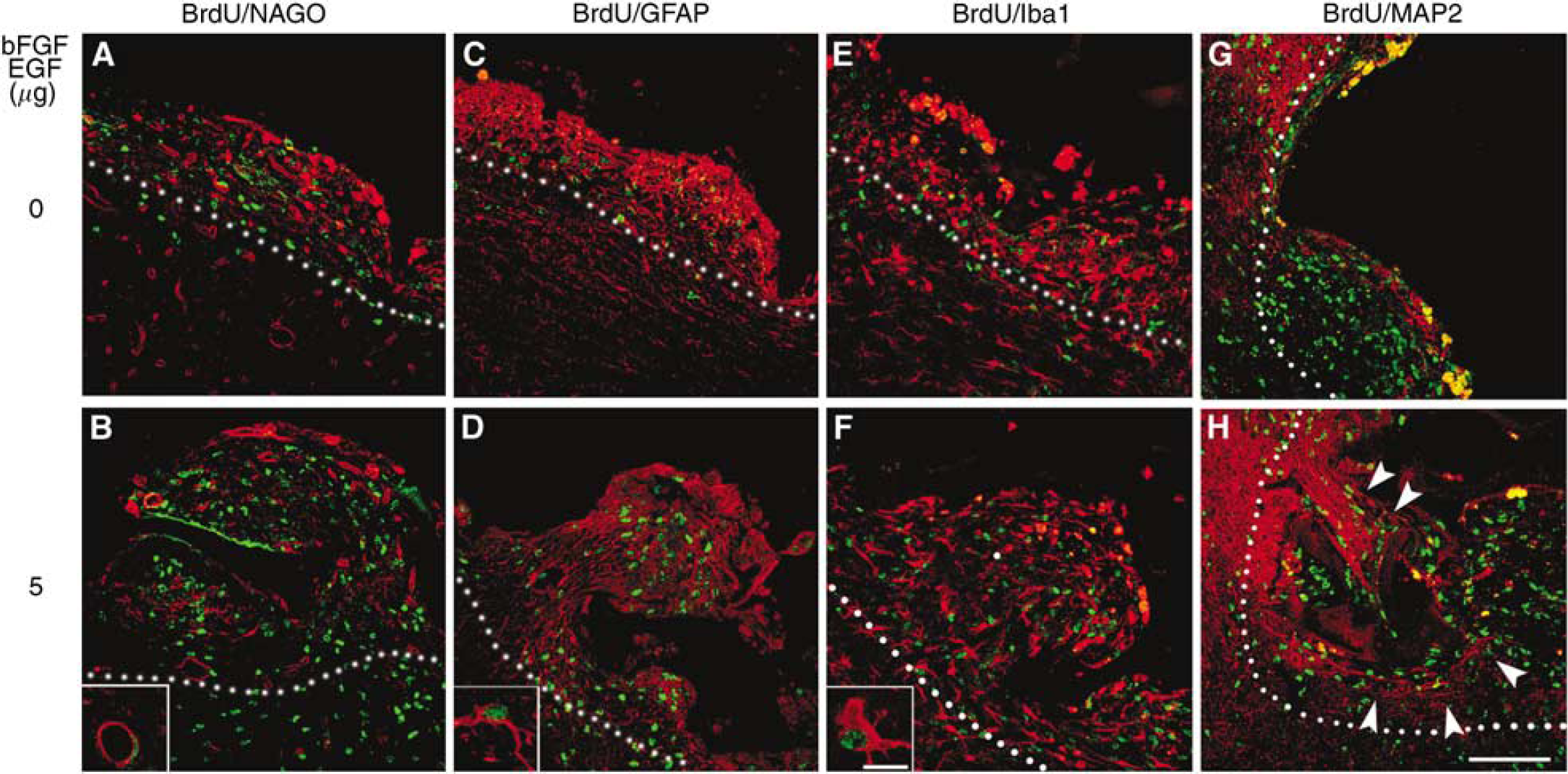
Double fluorescent study for bromodeoxyuridine (BrdU) (green) and N-acetylglucosamine oligomers (NAGO) (
Extension of dendrites that were MAP2 positive (Figures 7G and 7H, red) was observed from the surrounding cerebral cortex to the newly formed tissue, notably in sections in which the scaffold was implanted immediately after making a defect with 5 mg each of bFGF and EGF (Figure 7H, white arrowheads).
Discussion
In severe CNS injury such as cerebral infarction, the damaged area eventually changes from necrotic tissue to a cystic cavity with extensive tissue loss at the chronic stage. Although exogenously transplanted cells such as NSCs, ES cells, BMCs, and UCCs may be very powerful tools for tissue reconstruction, there are various problems, including tumorigenic transformation, immunological rejection, and ethical concerns for the source (Vats et al, 2005). Of major problem is that most transplanted cells cannot survive in the environment of the brain lesion (Snyder and Park, 2002). For this reason, any scaffold for migrated or transplanted cells with supportive angiogenesis should be important in the space of lost tissue. A scaffold with the function of extracellular matrix (ECM) with additive trophic factors could provide a new environment to the brain tissue for regeneration.
In the present study, therefore, a new porous gelatin–siloxane hybrid was used as a three-dimensional scaffold to support cell regeneration and axonal regrowth. The present scaffold material was designed to be porous and permeable so as to allow the ingress of nutrients and removal of waste products and to allow the ingrowth of vascular tissue to ensure the survival of the newly generated cells. In addition, the present scaffold material is biodegradable, its biodegradability easily controlled by being changing the proportions of gelatin and siloxane, allowing for the defect tissues to regrow and be occupied by new cells (Ren et al, 2002). Furthermore, gelatin is capable of holding various trophic factors by forming of polyion complex and to release gradually (Ikada and Tabata, 1998), an advantage that was used in the present study. The porous gelatin–siloxane hybrid was not found to evoke an inflammatory reaction, which inhibits tissue regeneration. In fact, the number of Iba1-positive cells representing macrophages/microglias in new tissue formed within the scaffold was similar to that in the normal cerebral cortex in each experimental condition (Figure 4, right column), suggesting no additive inflammatory and immune reactions (Rosengren et al, 1997).
It became evident in the present study that implantation of the scaffold itself is very important for keeping integrity of brain shape (Figures 3A versus 3B to 3F). Of great interest is that new tissue formation in the scaffold was greater at the CC site than at the flanking site (Figures 3B to 3F), suggesting that new cells mainly came from the LV through the CC. Immunohistochemical analysis revealed that the distribution of each cell type in the scaffold is obviously different from the distributions of cells around the intrinsic brain tissue (Figure 4), with a large number of BrdU-labeled cells accumulated in and around the tissue in the scaffold (Figure 6). Although it is not clear at present whether these cells are derived from the SVZ, bone marrow (Hess et al, 2002; Li et al, 2001), or de novo mitosis (Lin et al, 1998; da Cunha et al, 1993), the findings imply that the tissue was newly constituted by proliferation of cells after the injury, probably to regenerate the injured tissue.
In terms of timing to implant the scaffold (Figures 1, 3 to 6), the volume of newly formed tissue was obviously increased in cases of immediate implantation after making the defect compared with that in cases of implantation at 30 days. In the chronic stage of brain injury, glial scar formation may greatly prevent brain tissue from regenerating by inhibiting angiogenesis, cell migration, and axonal growth. The development of a method for preventing such scar formation at acute to chronic stages and for absorbing such a scar at the chronic stage might enable effective scaffold implantation in the future.
With addition of bFGF and EGF, tissue regeneration was promoted (Figures 3B to 3D, 3G(b to d)) with a dose-dependent increase in the number of BrdU-labeled cells (Figure 6). The present study showed that the number of NAGO-positive cells representing vascular endothelial cells in the scaffold was dose-dependently increased (Figures 4 (left column) and 5A(b to d)), and that a few cells were double labeled with BrdU (Figures 7A and 7B). Both growth factors have an effect on vascular endothelial proliferation, the effect of bFGF being much stronger (Gospodarowicz et al, 1978; Schweigerer et al, 1987). The addition of bFGF and EGF also dose-dependently increased the numbers of GFAP- and Iba1-positive cells (Figures 4, 5B(b to d) and 5C(b to d)), suggesting astroglial and microglial cell genesis in the newly formed tissue in the scaffold. Since BrdU-labeled cells in the scaffold were mainly found at the site of the CC (Figure 6), which were also promoted by the addition of bFGF and EGF, it is possible that generation of these cells was enhanced by diffusion of bFGF and EGF from the scaffold to around the SVZ of the LV (Jin et al, and thus they migrated into the scaffold through the CC. However, the absence of NeuNpositive cells in the newly formed tissue indicates a preferential differentiation in this scaffold system to glial cells. A different induction factor to change differentiation from glial cells to neuronal cells could resolve this issue in the future. Transplantation of the scaffold with exogenous NSCs is also feasible (Park et al, 2002; Tate et al, 2002).
The double immunofluorescent study revealed the phenotypic order of newly generated cells in the scaffold to be astrocytes > microglia < vascular endothelia, which is the same even with the addition of bFGF and EGF (Figures 7A to 7F). However, addition of bFGF and EGF increased the number of double-positive cells by 1.6- to 3.0-fold (Figures 7A, 7C, 7E versus 7B, 7D, 7F). Astrocytes can secrete molecules such as neurocan (Deguchi et al, 2005), phosphacan (Asher et al, 2000), and myelin-associated proteins such as Nogo (Mingorance et al, 2004) which inhibit axonal growth. Although astrocytes were the main component of the newly formed tissue in the scaffold (Figure 7D), the extension of dendrites was observed from the surrounding cerebral cortex to the newly formed tissue in the case of addition of bFGF and EGF (Figure 7H). This suggests amelioration of such an inhibitory action of astrocytes for tissue regeneration by bFGF and EGF. Future trials with more potent inducers of axonal and dendrite elongation could greatly improve synaptic rearrangement in the scaffold tissue by using a recently developed scaffold material such as hyaluronic acid porous hydrogel (Hou et al, 2005; Tian et al, 2005), which is a major constituent of the ECM of the brain.
Stroke affects more than 500,000 people each year in the United States (Smith et al, 2005). They suffer from severe disabilities, but the therapeutic approach remains only palliative. If the brain tissue could be restored, the disabilities should be ameliorated in such patients. Although we cannot clinically use this material in its present study, further studies may make this material a novel tool for tissue regeneration.
In summary, the present study showed that a new porous gelatin–siloxane hybrid had biocompatibility after implantation into a lesion of the CNS, and that this material provided a potential scaffold for cell migration, angiogenesis, and dendrite elongation with a dose-dependent effect of additive bFGF and EGF. Further investigation is needed to determine whether new tissue regeneration in the cerebral defect could be further enhanced by means of a scaffold with a multiple delivery system by controlled release of neurotrophic or growth-promoting agents and whether scaffold implantation can also work well for tissue regeneration by controlling scar problems.
Footnotes
Acknowledgements
The authors thank Dr Imai Y (Ehime University, Japan) for his generous gift of the antibody against Iba1.
