Abstract
Occlusive cerebrovascular disease leads to brain ischemia that causes neurological deficits. Here we introduce a new strategy combining mesenchymal stromal cells (MSCs) and ex vivo hepatocyte growth factor (HGF) gene transferring with a multimutated herpes simplex virus type-1 vector in a rat transient middle cerebral artery occlusion (MCAO) model. Gene-transferred MSCs were intracerebrally transplanted into the rats' ischemic brains at 2h (superacute) or 24 h (acute) after MCAO. Behavioral tests showed significant improvement of neurological deficits in the HGF-transferred MSCs (MSC-HGF)-treated group compared with the phosphate-buffered saline (PBS)-treated and MSCs-only-treated group. The significant difference of infarction areas on day 3 was detected only between the MSC-HGF group and the PBS group with the superacute treatment, but was detected among each group on day 14 with both transplantations. After the superacute transplantation, we detected abundant expression of HGF protein in the ischemic brain of the MSC-HGF group compared with others on day 1 after treatment, and it was maintained for at least 2 weeks. Furthermore, we determined that the increased expression of HGF was derived from the transferred HGF gene in gene-modified MSCs. The percentage of apoptosis-positive cells in the ischemic boundary zone (IBZ) was significantly decreased, while that of remaining neurons in the cortex of the IBZ was significantly increased in the MSC-HGF group compared with others. The present study shows that combined therapy is more therapeutically efficient than MSC cell therapy alone, and it may extend the therapeutic time window from superacute to acute phase.
Keywords
Introduction
Occlusive cerebrovascular disease often causes global ischemia of the brain and results in neuropathological changes. Several methods have been proposed to augment brain reorganization, including the stimulation of endogenous processes through pharmacologic or molecular manipulation, gene therapy, behavioral and rehabilitation strategies, and the provision of new substrates for recovery through cell therapy.
Bone marrow contains the precursors of nonhematopoietic tissues that are referred to as mesenchymal stem cells or marrow stromal cells (MSCs) (Friedenstein et al, 1978). Marrow stromal cells are characterized by the ability to self-renew in a number of nonhematopoietic tissues, and by their multipotentiality for differentiation into various tissues, such as fibroblasts, bone, muscle, and cartilage (Caplan and Bruder, 2001; Phinney, 2002). Additionally, they share some characteristics of neurons and astrocytes when cultured in vitro (Kim et al, 2002) or after being implanted into the central nervous system in vivo (Chopp et al, 2000; Nakano et al, 2001; Li et al, 2001, 2002; Chen et al, 2002a, b). Marrow stromal cells can also secrete growth factors and cytokines into the soluble stromal and neurochemicals into the brain (Li et al, 2002; Chen et al, 2002a, b), cross the blood–brain barrier (BBB) and migrate throughout the brain preferentially to areas that have suffered damage (Chen et al, 2000; Li et al, 2000, 2001; Damme et al, 2002). Many previous researchers have reported on mesenchymal stromal cell (MSC) transplantation as a source for autoplastic therapies and improvement in functional recovery after stroke (Chen et al, 2000; Li et al, 2000, 2001, 2002; Rempe and Kent, 2002; Kurozumi et al, 2004).
Hepatocyte growth factor (HGF) is a disulfide-linked heterodimeric protein that was initially purified and cloned as a potent mitogen for hepatocytes and a natural ligand for the c-met proto-oncogene product (Nakamura et al, 1984; Matsumoto and Nakamura, 1996). Subsequently, several functions have been ascribed to HGF, including antiapoptosis, angiogenesis, motogenesis, morphogenesis, hematopoiesis, tissue regeneration in a variety of organs, and the enhancement of neurite outgrowth (Matsumoto and Nakamura, 1997; Hayashi et al, 2001; Sun et al, 2002a, b; Jin et al, 2003). It has also been reported that HGF administration could inhibit the BBB destruction, decrease brain edema, and provide a neuroprotective effect after brain ischemia (Miyazawa et al, 1998; Hayashi et al, 2001; Shimamura et al, 2004).
Recent experimental studies suggest the possibility that gene transduction into MSCs could enhance their existing therapeutic potential (Chen et al, 2000; Kurozumi et al, 2004). Here, we evaluate the efficiency and effects of gene transduction into MSCs using a replication-incompetent herpes simplex virus type-1 (HSV1764/4-/pR19) vector disabled by the deletion of three critical genes for viral replication encoding infected cell polypeptide (ICP)4, ICP34.5, and virion protein (VP16) (vmw65). This vector contains HSV latency-associated transcript (LAT) promoter and two kinds of enhancer elements: cytomegalovirus (CMV) enhancer and Woodchuck posttranscriptional regulatory elements (WPRE). The availability of this vector has already been examined in the nervous system (Coffin et al, 1998; Palmer et al, 2000; Lilley et al, 2001).
In the present study, we intracerebrally transplanted MSCs in which a gene of interest was transferred with this HSV-1 vector ex vivo into a rat transient middle cerebral artery occlusion (MCAO) model under superacute and acute therapeutic time phase, and investigated whether such combined therapy could improve the effects of ischemia.
Materials and methods
Donor Cell Preparation
Marrow stromal cells of adult Wistar rats were prepared following the method described by Azizi et al (1998). In brief, the marrow of rat tibias and femurs was extruded with 10mL of alpha-MEM (Sigma Chemical Co., St Louis, MO, USA) and cultured in the same medium supplemented with 10% fetal bovine serum (FBS), 2 mmol/L L-glutamine, and antibiotic–antimycotic 1mL/100mL (GIBCO Invitrogen, Carlsbad, CA, USA) at 37°C, 98% humidity and 5% CO2. After 48 h, the nonadherent cells were removed by replacing the medium, and the adherent cells were continuously subcultured as MSCs. The fifth to seventh passages were used for the following experiments.
HSV1764/-4/pR19-Hepatocyte Growth Factor Virus and Propagation
One of the authors of the current study (Coffin) constructed the prototype HSV1764/4/pR19GFP virus and has previously described this vector's characteristics (Palmer et al, 2000; Lilley et al, 2001), which are also described briefly in the Introduction. In the present study, the green fluorescent protein (GFP) gene was replaced with a full-length rat HGF complementary DNA (cDNA) tagged with the KT3 (SV (simian virus)40 large, T antigen) epitope (ratHGFKT3) (Sun et al, 2002b), and the authenticity of this vector (pR19ratHGFKT3WPRE) was confirmed by sequence analysis. Homologous recombination was performed in M49 cells by cotransfection of plasmid pR19ratHGFKT3WPRE DNA and HSV1764/-4/pR19GFP viral DNA. White plaques were selected and purified three times, and replication-incompetent viruses were propagated as described previously (Palmer et al, 2000). We ultimately obtained the HSV1764/-4/pR19-HGF virus (HSV-HGF) with a titer of 2 × 108 pfu/mL for use in the present experiments.
Ex Vivo Gene Delivery to MSCs
The cultured MSCs from the fifth to seventh passages were infected with the virus suspension by incubation for 1 h. After infection, the virus suspension was changed to normal culture medium for MSCs and continuously cultured for the subsequent 24 h before transplantation.
Our previous experiments show that the transduction efficiency of the GFP gene into the MSCs with our HSV-1 vector is more than 50% even with a multiplicity of infection (MOI) of 5. Here we set the MOI at 5 for the desired gene transfer to MSCs ex vivo.
Hepatocyte Growth Factor Detection with Enzyme-Linked Immunosorbent Assay (ELISA) In Vitro
We prepared 1.6 × 105 MSCs in each well of a six-well dish. The MSCs were transferred with HGF gene by infection with HSV-HGF at MOIs of 0, 0.1, 1, 5, and 10. At 1 h after infection, the infected MSCs were successively incubated with normal culture medium for another 24 h. The culture supernatant and cells were then individually collected through centrifugation. The HGF protein concentrations in MSC culture supernatant and in MSC extracts prepared using 50 mmol/L Tris-HCl (pH 7.4), 150 mmol/L NaCl, 1% Triton X-100, 1 mmol/L phenylmethylsulfonylfluoride (PMSF) (Wako, Osaka, Japan), 2 μg/ml antipain (Peptide Institute Inc., Osaka, Japan), 2 μg/ml leupeptin (Peptide Institute), and 2 μg/ml pepstatin (Peptide Institute) were determined by ELISA using an anti-rat HGF polyclonal antibody (Tokushu Meneki, Tokyo, Japan) as described (Sun et al, 2002b).
Transient Middle Cerebral Artery Occlusion Animal Model
Experiments were performed on 8-week-old male Wistar rats weighing 250 to 280 g. We induced transient MCAO using the previously described method of intraluminal vascular occlusion (Longa et al, 1988). In brief, a length (18.5 to 19.0 mm, determined according to the animal's weight) of 4-0 surgical nylon suture was gently advanced from the external carotid artery into the lumen of the internal carotid artery until it reached the proximal segment of the anterior cerebral artery. After 2 h of MCAO the animals were reanesthetized, and reperfusion was achieved by withdrawing the nylon suture.
The rats were subjected to transient MCAO for 2 h to produce a consistent and reproducible ischemic lesion in the unilateral striatum and cortex.
Intracerebral Transplantation of MSCs
At 2 or 24 h after the onset of MCAO (i.e., on reperfusion), the animals were placed in a stereotactic head holder (model 900, David Kopf Instruments, Tujunga, CA, USA) under inhalation anesthesia. MSCs were intracerebrally transplanted by inserting a 26-gauge needle with a Hamilton syringe into the right striatum (anteroposterior (AP) = 0mm; lateral to midline (ML) = 2.0 mm; vertical to dura (DV) = 4.5 mm) from bregma, based on the atlas given by Paxinos et al (1985). There were 1 × 106 cells in total 10-μl fluid volumes that transplanted into each animal over a 10-min period. No immunosuppressive drugs were used in any animal.
Experimental Groups
In this study, there were seven experimental groups: groups 1 and 5 were treated with phosphate-buffered saline (PBS); groups 2 and 6 were treated with untreated MSCs only; group 3 was treated with the GFP-transferred MSCs (MSC-GFP); and groups 4 and 7 were treated with HGF gene-transferred MSCs (MSC-HGF).
Groups 1 to 4 were treated 2 h after MCAO (superacute phase) and groups 5 to 7 were treated 24 h after MCAO (acute phase).
Behavioral Testing
The rats of groups 1 to 4 (n = 6) were subjected to a modified neurological severity score (mNSS) test (Schallert et al, 1997) to evaluate neurological function before MCAO, at 2h after MCAO, and at 1, 4, 7, 14, 21, 28, and 35 days after MCAO. The rats of groups 5 to 7 (n = 6) were subjected to mNSS before MCAO and at 0, 1, 4, 7, and 14 days after MCAO. These tests are battery of motor, sensory, reflex, and balance tests, which are similar to the contralateral neglect tests in humans. The higher the score, the more severe the neurological deficit (Chen et al, 2001).
Infarction Volume
We stained the brains of groups 1, 2, and 4 (n = 6) and groups 5 to 7 (n = 5) with 2,3,5-triphenyltetrazolium chloride (TTC) (Wako Pure Chemical Industries, Osaka, Japan) to detect the infarction volume of each group at 3 and 14 days after treatment. Briefly, the rats' brains were removed and cut into seven equally spaced (2 mm) coronal sections. These sections were immersed in a 2% solution of TTC at 37°C for 20 mins to reveal the infarcted areas. This procedure is known to reliably mark ischemic damage even at 14 days after MCAO (Bederson et al, 1986; Kurozumi et al, 2005).
The disposition of the ischemic area was evaluated by calculating the hemispheric lesion area using imaging software (Scion Image, version Beta 4.0.2; Scion Corp., Frederick, MD, USA). To avoid overestimation of the infarct volume, the corrected infarct volume (CIV) was calculated as CIV =[LT-(RT-RI)] ×d, where LT is the area of the left hemisphere, RT is the area of the right hemisphere, RI is the infarcted area, and d is the slice thickness (2 mm) (Raymond et al, 1990). Relative infarct volumes are expressed as a percentage of contralateral hemispheric volume.
Terminal Deoxynucleotidyltransferase (dUTP) Nick End-Labeling (TUNEL) Staining and Immunohistochemical Assessment
Sample Preparation: At different time points, rats of groups 1, 2, and 4 were reanesthetized and transcardially perfused with saline, followed by 4% paraformaldehyde in PBS. The brain tissues were cut into seven equally spaced coronal blocks. The tissues were processed and 10-μm cryosections were cut.
Immunohistochemical Staining: We can detect three kinds of HGF in this study: the endogenous HGF secreted by the rat ischemic brain tissue after stoke (en-HGF), the exogenous HGF secreted by the transplanted MSCs (ex-HGF-1), and the exogenous HGF delivered from the HSV-HGF (ex-HGF-2). For the immunohistochemical staining of HGF, the whole rats' brain sections of groups 1, 2, and 4 were prepared on days 2 and 14 after treatment. Rabbit anti-rat HGF primary antibody (prepared by some of the authors of this article, and belonging to the Division of Molecular Regenerative Medicine, Osaka University Graduate School of Medicine, Japan) was used to detect the three kinds of HGF (mixed); a KT3 primary monoclonal antibody (1:1000) (Covance Research Products, Berkeley, CA, USA) was used to detect the ex-HGF-2; a biotinylated universal secondary antibody (VECSTAIN Elite ABC Kit, PK-6200, Vector Laboratories, Burlingame, CA, USA) and a goat anti-rabbit IgG affinity-purified rhodamine-conjugated secondary antibody (1:200) (Chemicon International, Temecula, CA, USA) were also used here. Reaction products were visualized with the VECSTAIN Elite ABC Kit (PK-6200) and a DAB Substrate Kit (Vector Laboratories, Burlingame, CA, USA). To detect the donor MSCs, bisbenzimide (Hoechst 33258; Polysciences, Eppelheim, Germany) was used to fluorescently label cell nuclei in vitro. Some sections were counterstained with hematoxylin and observed under a normal light microscope (VB-S20 Multiviewer System, Keyence, Osaka, Japan and Microphot-FXA, Nikon Corp., Tokyo, Japan), and some were directly observed by a fluorescence microscope (BX-50-34-FLAD1, Olympus). The donor MSCs could be detected under ultraviolet (UV) light with blue fluorescence as marked by Hoechst 33258.
To visualize the remaining neurons in the cortex of the ischemic boundary zone (IBZ) ofgroups 1, 2, and 4 (n = 3), 7 days after treatment, microtubule-associated protein 2 (MAP-2) was used as the first antibody (1:500) (Chemicon International Inc., CA, USA). Negative control slides for each animal received identical preparation for immunohistochemical staining, except that primary antibodies were omitted.
Terminal Deoxynucleotidyltransferase Nick End-Labeling Staining: At 7 days after treatment, coronal cryosections (10-μm thick) of each rat of groups 1, 2, and 4 (n = 3) were stained by the TUNEL method for in situ apoptosis detection (ApopTag kit, Chemicon International, USA). Specifically, after postfix slides were incubated in a mixture containing terminal deoxynucleotidyl transferase and anti-digoxigenin-rhodamine (Red). Then, they were counterstained with bisbenzimide (Hoechst 33258), which stains blue for each nucleus. The total numbers of TUNEL-positive cells and Hoechst counter-staining positive cells were individually counted in 2 slides from each brain, with each slide containing five random fields from the IBZ, under an × 20 objective of the fluorescence microscope system (BX-50-34-FLAD1, Olympus), using a 3-CCD color video camera (Keyence VB-7010, Keyence, Osaka, Japan).
Statistical Analysis
Data are presented as means±standard deviations (s.d.). Data from the behavior test (mNSS) were evaluated with repeated-measures analysis of variance (ANOVA), with subsequent Fisher's protected least significant difference (PLSD) test. StatView 5.0 software (SAS Institute, Cary, NC, USA) performing the Student's t-test was used to test the CIV data and the difference in means of percentage of the apoptosis-positive cells and the remaining neurons. A difference with a probability value of P≤0.05 was considered to be statistically significant.
Results
Quantification of Hepatocyte Growth Factor Analysis with Enzyme-Linked Immunosorbent Assay In Vitro
As a result, the HGF concentration was approximately 15 times higher in the culture supernatant than in the cell extract at the same MOI, and its increase was correlated with an increase in MOI. Although normal MSCs can produce HGF protein at 0.4 ng/1.6 × 105 cells/24 h, after the MSCs were infected with HSV-HGF at an MOI of 5, they were found to produce HGF protein at 2.4 ng/1.6 × 105 cells/24 h (Figure 1).
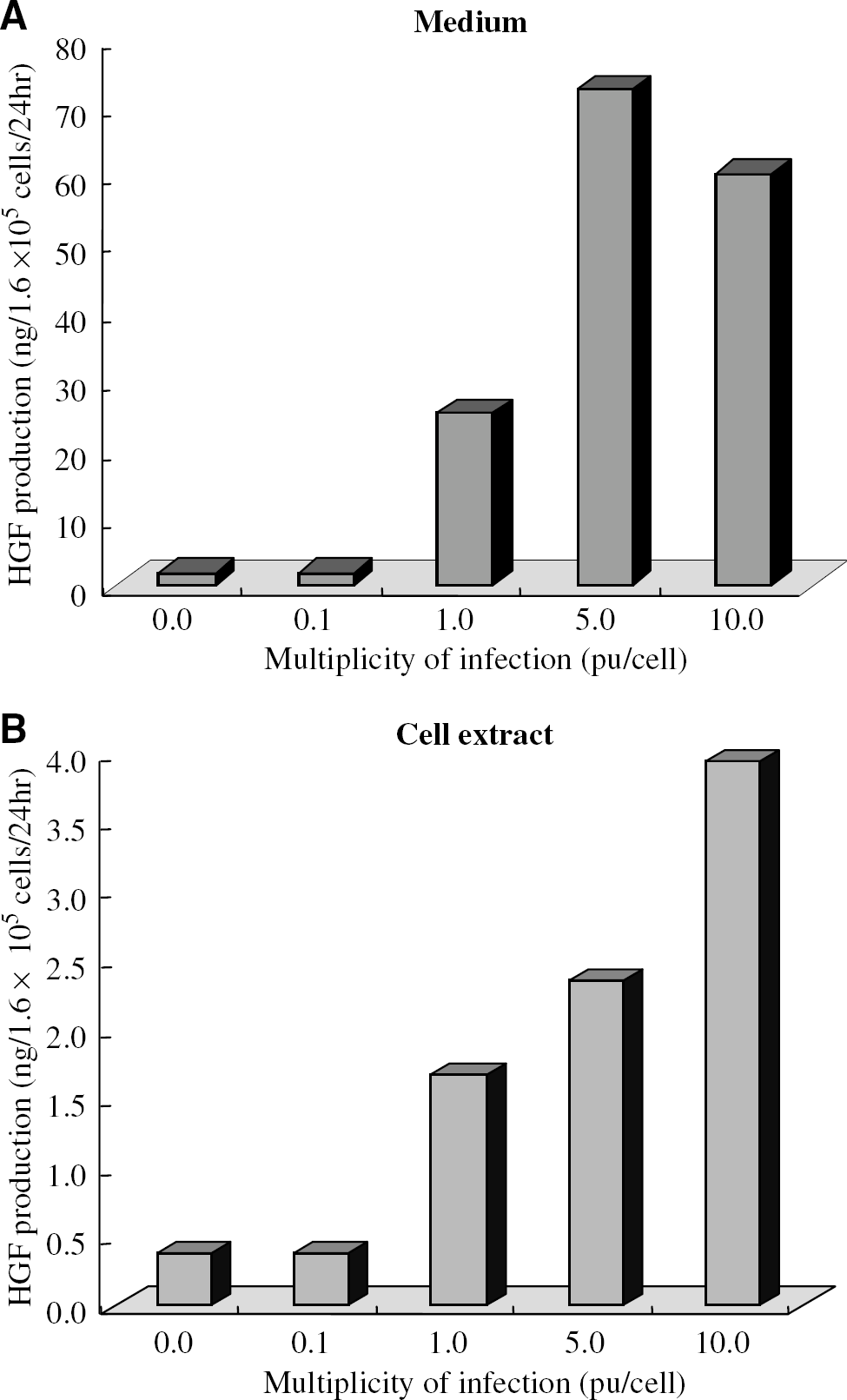
Enzyme-linked immunosorbent assay to determine HGF concentration in vitro. Hepatocyte growth factor concentrations were detected in MSC culture supernatant (
Neurological Outcome
No significant difference in neurological function was detected among all the groups just before cell transplantation. Significant differences of functional recovery were found in group 1 individually compared with group 2 (days 14 to 35, P < 0.05), with group 3 (days 21 to 35, P < 0.05), and with group 4 (days 4 and 7, P 0.05; days 14 to 35, P 0.01) during the observation periods after the superacute transplantation (Figure 2A), and in group 5 individually compared with group 6 (day 14, P < 0.05), with group 7 (day 7, P < 0.01 and day 14, P < 0.01) after the acute transplantation (Figure 2B). Interestingly, we observed significant differences of functional recovery on day 14 among all the superacute treated groups including the MSC-GFP group, which served as a control for ex vivo nontherapeutic gene transduction (P < 0.05). Exceptionally, there was no significant difference only between the MSC-only and the MSC-GFP groups at that time point (Figure 2A). We also found significant neurological recovery on day 14 in the combined therapy group treated even in the acute phase, compared with the MSC-only group treated in the superacute phase (Figure 2C). Also, significant difference of functional recovery on day 14 was found among the groups treated in the acute phase (Figure 2B).
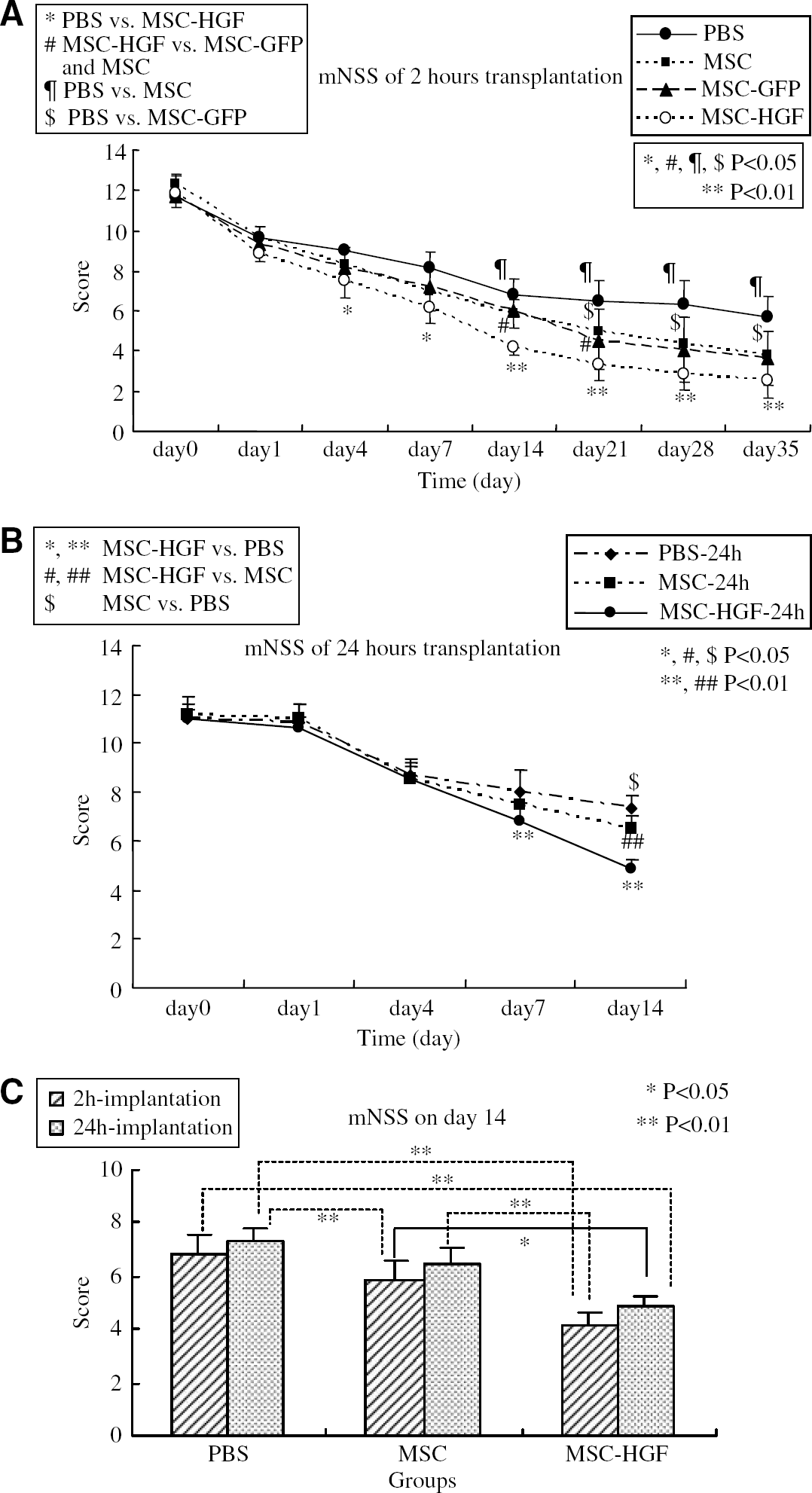
Behavioral functional test (mNSS) before and after MCAO. Groups 1 and 5: treated with PBS; groups 2 and 6: treated with MSC-only; group 3: treated with MSC-GFP; groups 4 and 7: treated with MSC-HGF (n = 6 per group). The rats of groups 2 to 7 received 1.0 × 106 cells via intracerebral transplantion in 10 μl PBS. (
Quantitative Analysis of Infarct Volume
We compared the infarction areas in coronal sections of groups 1, 2, and 4 on day 3 (Figure 3A) and day 14 (Figure 3B), and compared those of groups 5 to 7 on the same time points by TTC staining, and expressed lesion volume as a percentage of contralateral hemispheric volume. At 3 days after treatment, significant difference of %CIV was only detected in the MSC-HGF group compared with the PBS group (34.52% ± 3.44% versus 41.83% ± 6.25%, P < 0.05), both of which were treated in the superacute phase (Figure 3C). However, on day 3 there was no significant difference of %CIV among any group that was treated in the acute phase (Figure 3C), while on day 14 there were significant reductions of %CIV in the rats of the MSC-HGF group compared with not only the PBS group but also the MSC-only group treated in the both therapeutic phases (Figure 3D). Also on day 14, the rats treated with MSC-only showed significant reduction in %CIV compared with the PBS group that was treated in the superacute phase (Figure 3D).
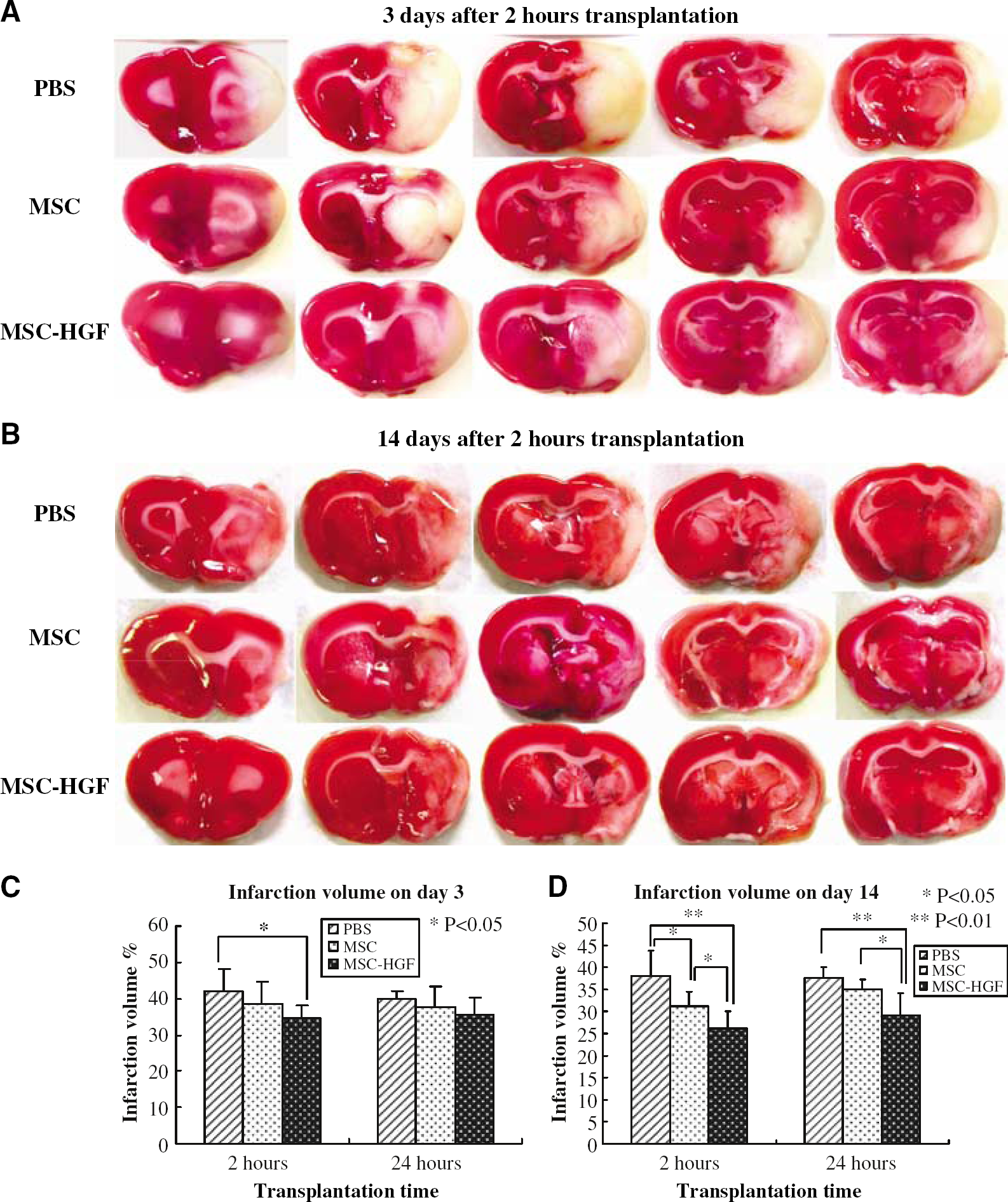
Infarction volume detected by TTC staining. (
Hepatocyte Growth Factor and herpes simplex virus type Gene-Transferred Hepatocyte Growth Factor Detection In Vivo
The macrographs presented in Figure 4 showed that mixed HGF protein was diffusely overexpressed in almost the whole ipsilateral brain in the MSC-HGF group compared with other groups, not only on day 2 (column A) but throughout at least the first 2 weeks (column C) after treatment. The microphotographs presented in column B of Figure 4 showed that high HGF expression in the MSC-HGF group could be detected in both the ipsilateral cortex and the ipsilateral basal ganglia at 2 days after treatment. Nevertheless, almost no HGF expression could be detected on the contralateral hemisphere in any treatment group (Figure 4).
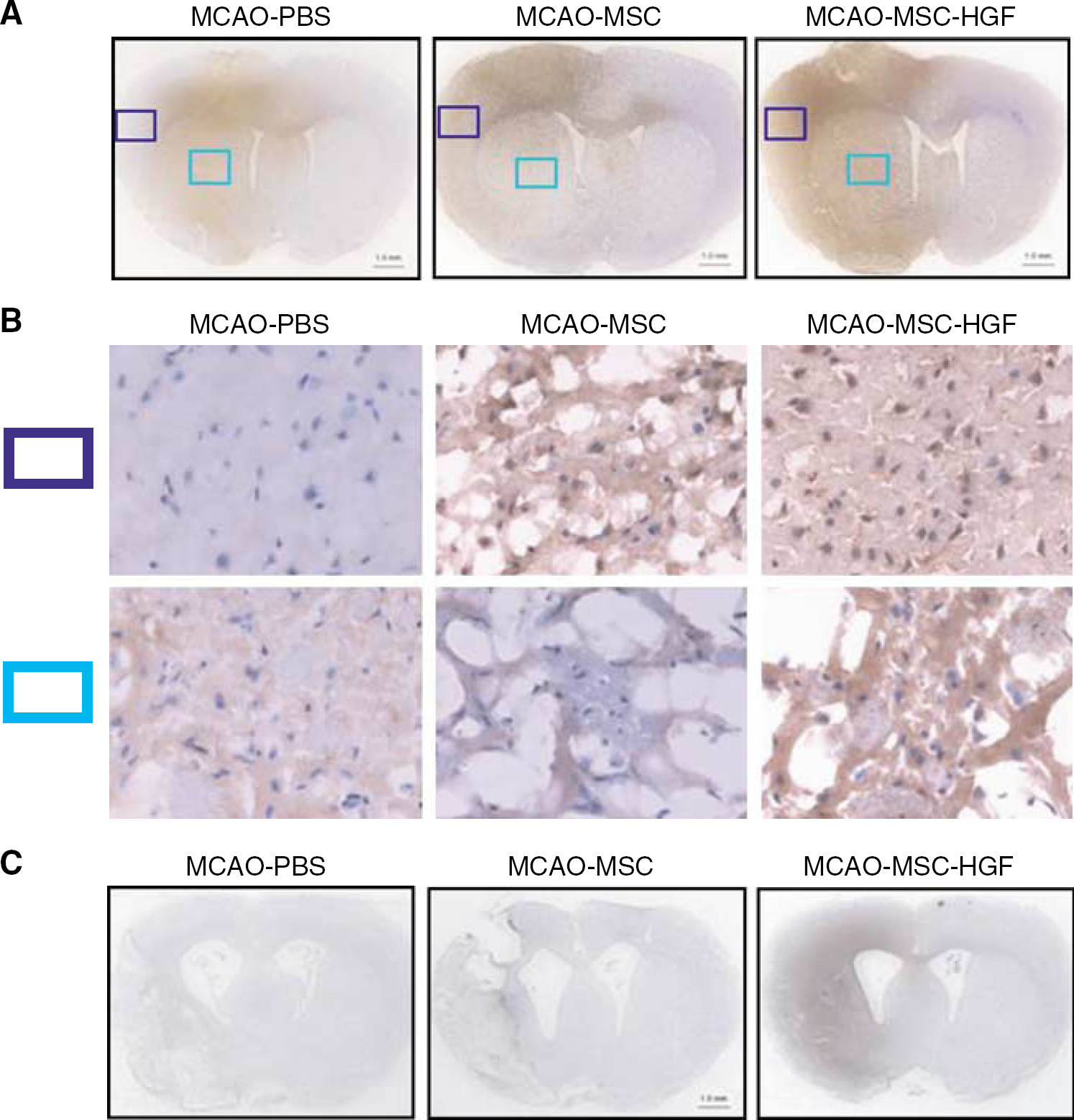
Immunohistochemistry for HGF expression in vivo. Mixed HGF expression (brown color) in the ischemic brains of groups 1, 2, and 4 detected by immunohistochemistry on days 2 (row A) and 14 (row C). Scale bar: 1.0mm. The upper and lower rows identified in B (original magnification, × 400) show the cortex and basal ganglia, respectively, of the images shown in row A.
Fluorescent staining of groups 1, 2, and 4 on day 14 (Figure 5, column C) also showed higher mixed HGF expression in the MSC-HGF group than that of the other groups. Also, we could detect donor MSCs with blue fluorescence expression by direct observation under UV light (Figure 5, column B). We could identify the HGF expression with red fluorescence in both the transplanted cells and the intercellular space in the transplantation area.
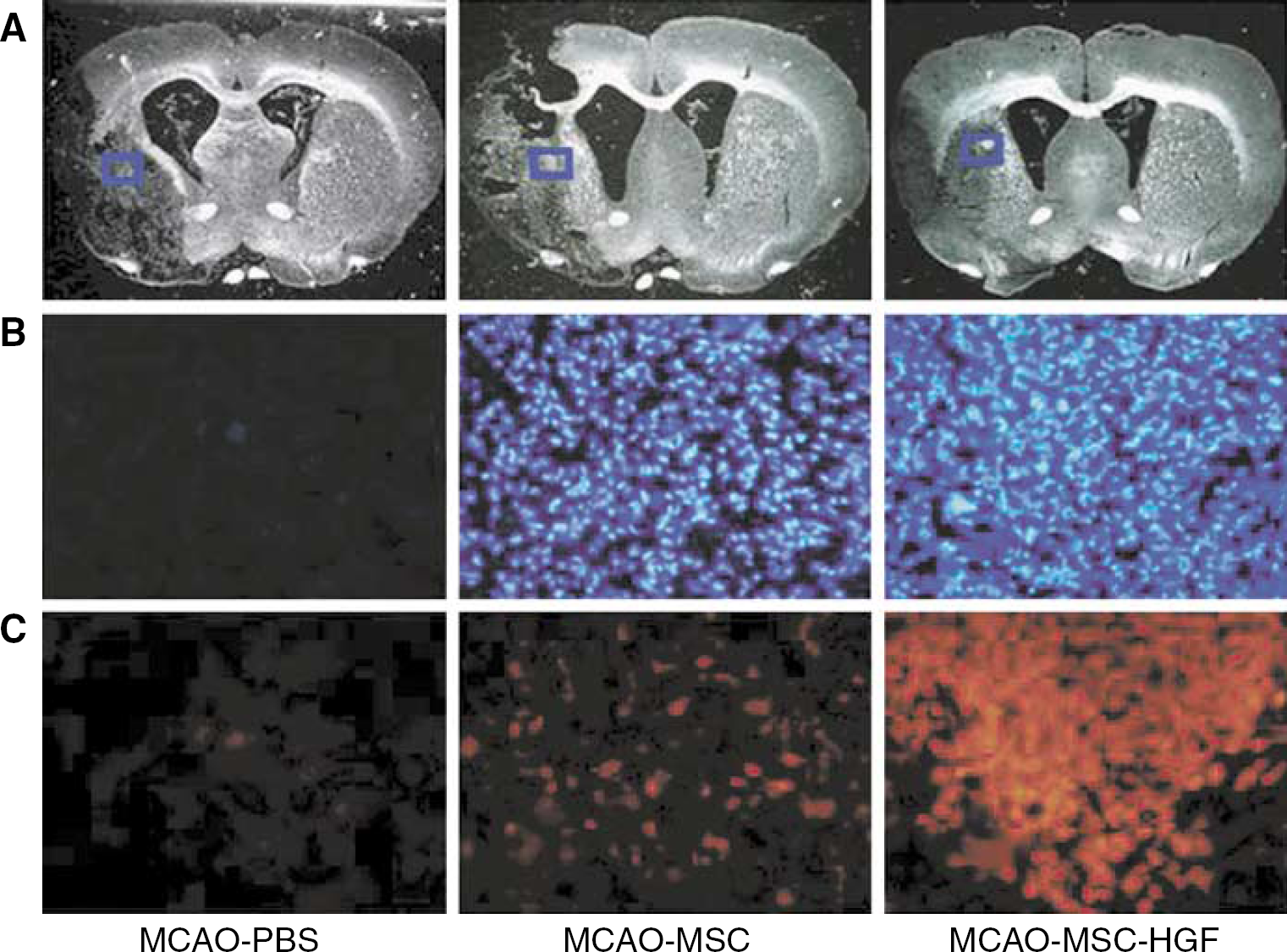
Expression of HGF and identification of transplanted MSCs. Photographs in row C show mixed HGF expression in the ipsilateral brain of groups 1, 2, and 4 with red fluorescence, and photographs in row B show transplanted donor MSCs of groups 2 and 4 with blue fluorescence, at 2 weeks after treatment. The microphotographs shown in rows B and C have the same size and higher power magnification than the blue squares in row A. Original magnification, × 200.
Furthermore, we detected ex-HGF-2 expression, which was transferred from HSV-HGF by anti-KT3 staining (Figures 6D to 6F) of the implantation area. As a result, we had identified ex-HGF-2 expression both in the HGF gene-transferred MSCs (arrows in Figure 6G) and in the intercellular space of the transplantation area (arrowheads in Figure 6G) only in the MSC-HGF group (Figure 6F) even 14 days after transplantation. Additionally, we confirmed that MSC itself can also secrete HGF in vivo (Figure 6B).
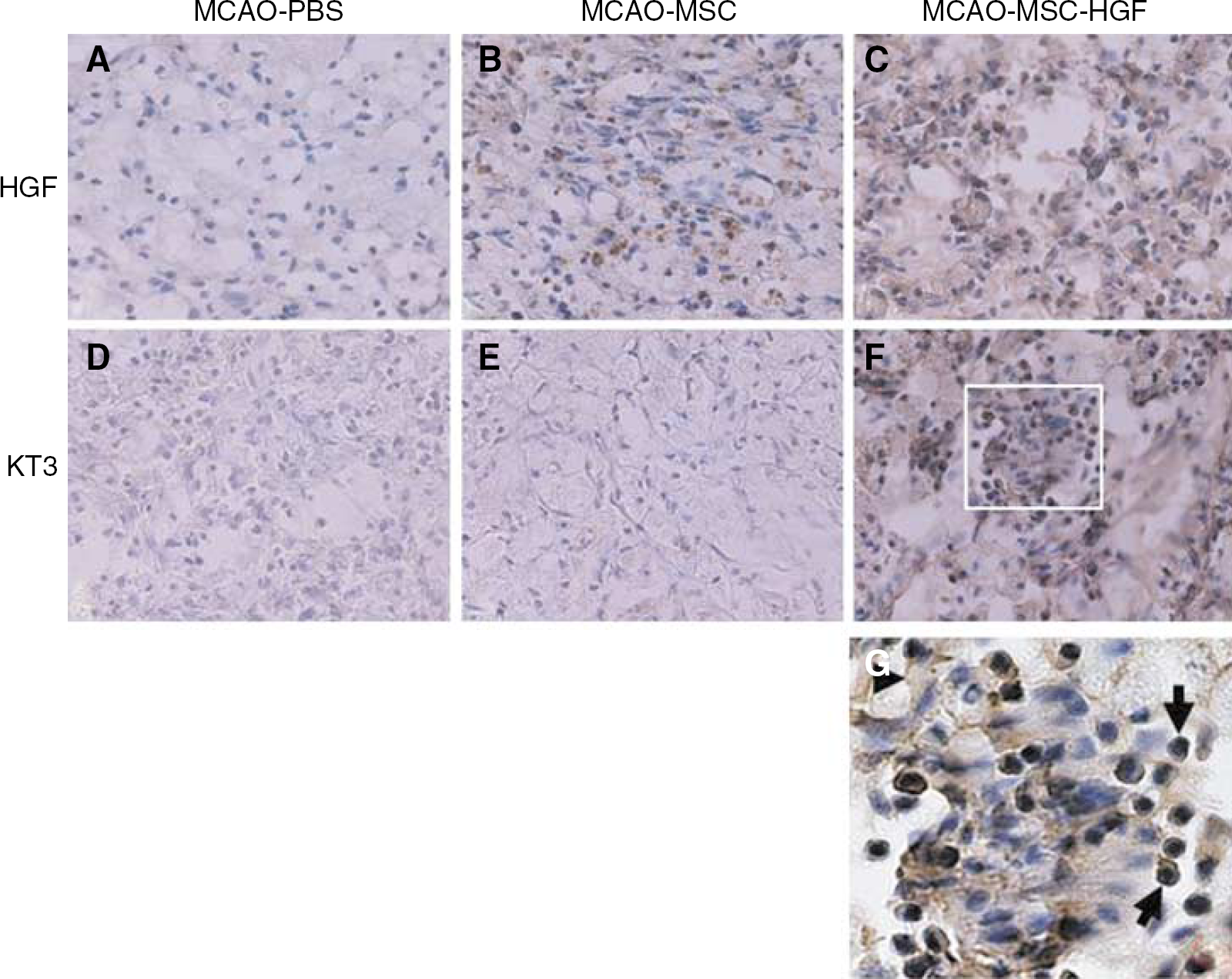
Immunohistochemistry for HSV-1 vector-transferred exogenous HGF and mixed HGF expression. The upper column (A–C) shows mixed HGF expression in groups 1, 2, and 4 with anti-rat HGF immunostaining, and the lower column (D–F) shows ex-HGF-2 expression with anti-ratHGFKT3 immunostaining at 2 weeks after treatment. Original magnification, × 200. (G) is the enlarged white square in (F), arrows mark HGF expression in the transplanted MSCs and an arrowhead marks HGF expression in the intracellular space.
Antiapoptosis
Using TUNEL staining (Figure 7, columns B and C), apoptotic cells with red fluorescence were counted in the IBZ 7 days after treatment, while cells were counted in the same area with blue fluorescence by Hoechst 33258 nuclei marking. In this area we could not detect transferred MSCs; therefore, counterstained cells seemed to be host-derived. The percentage of apoptotic host cells was significantly decreased in the MSC-HGF group (4.92% ± 2.15%) compared with the PBS group (22.12% ± 4.28%, P < 0.01) and MSC-only group (10.73% ± 5.64%, P < 0.01). However, there was also significant decrease of apoptotic cells between the MSC-only group (10.73% ± 5.64%) and the PBS group (22.12% ± 4.28%, P < 0.01) (Figure 7C).
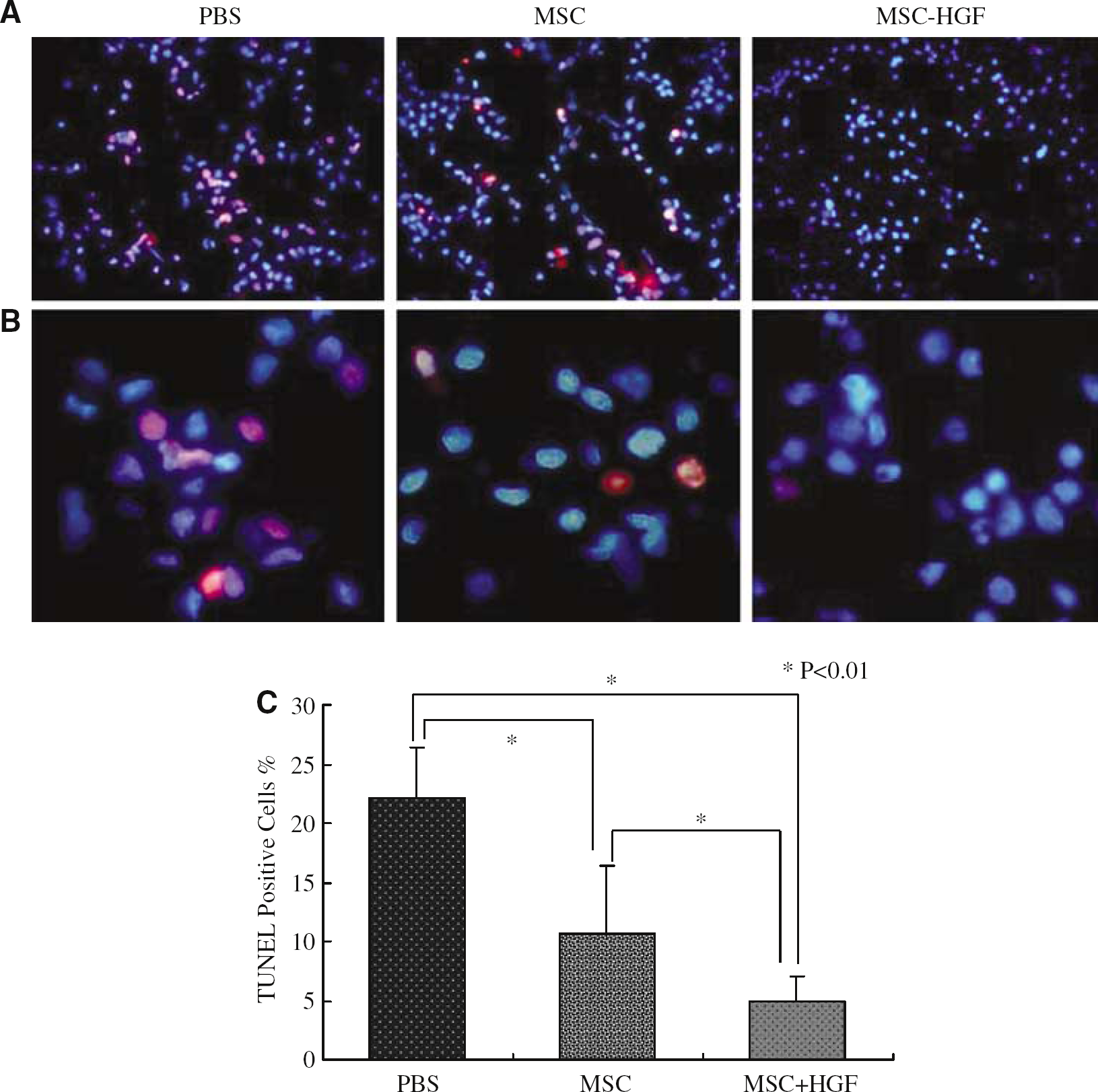
Apoptotic cells in the IBZ with TUNEL staining. (
Neuroprotection
Immunohistochemical staining revealed the remaining neurons of the host with MAP-2 neuronal marker 7 days after treatment (Figure 8A). The percentage of remaining neurons in the cortex of IBZ significantly increased in the MSC-HGF group (20.73% ± 2.38%) compared with the PBS group (7.75% ± 1.58%, P < 0.01) and the MSC-only group (12.13% ± 3.05%, P < 0.01). Also, the significant increase of remaining neurons was found in the MSC-only group (12.13% ± 3.05%), in comparison with the PBS group (7.75% ± 1.58%, P < 0.01) (Figure 8B).
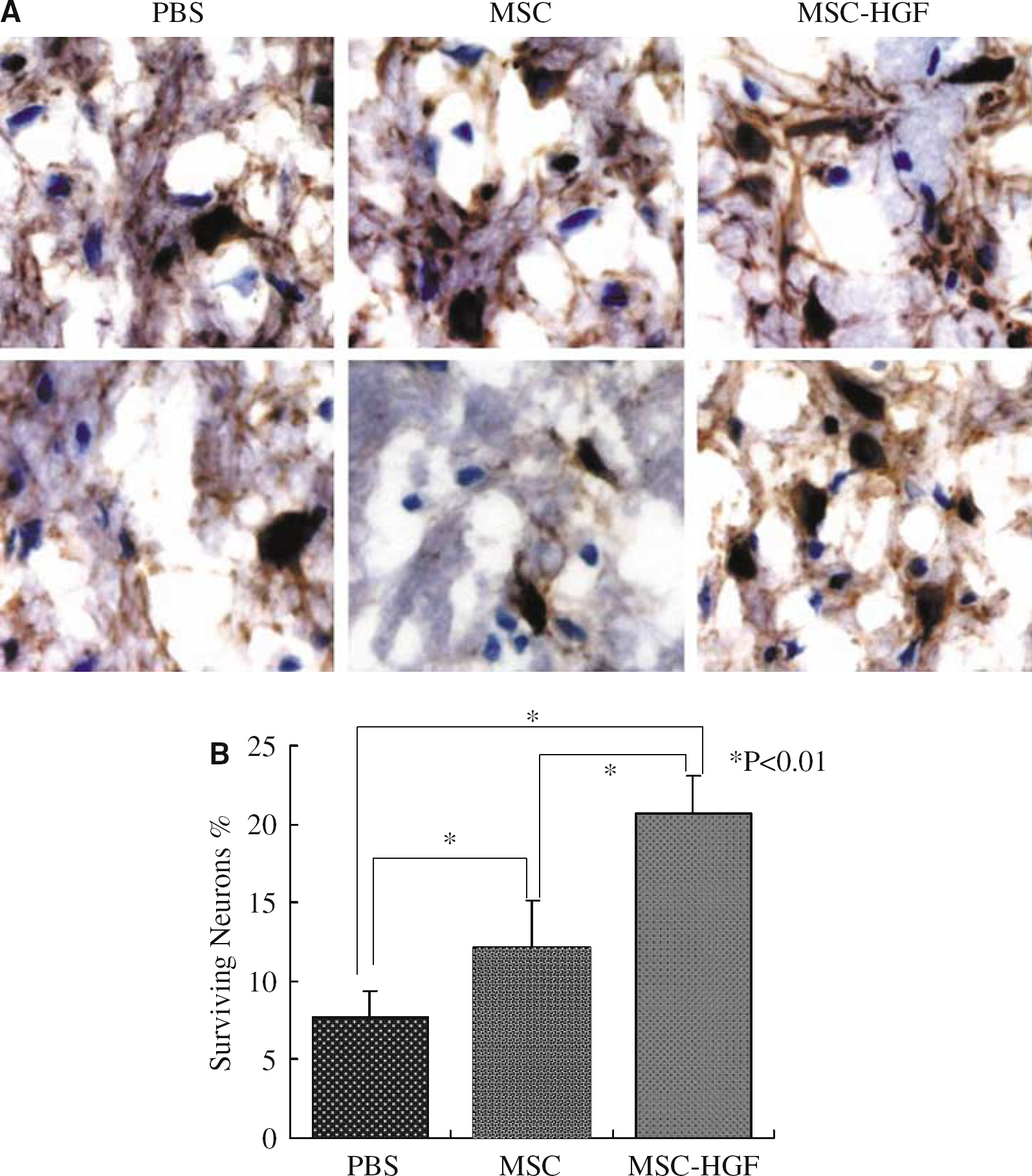
Remaining neurons in the cortex of IBZ with MAP-2 immunostaining. (
Discussion
Brain ischemia initiates a cascade of events that produces neuronal death and leads to neurological deficits. To prevent brain injury after ischemia, some studies have focused on cell therapies by using embryonic stem cell. But ethical and logistical problems make it unlikely that such therapy could serve as a source of material for therapeutic transplants. Recently, MSC transplantation was reported as a source of autoplastic therapies which not only improve functional recovery after stroke but also have a low risk of tumorigenesis and do not provoke immune reactions (McIntosh and Bartholomew, 2000; Li et al, 2002). In the present study, rats of the MSC-only and MSC-HGF groups also showed more significant neurological functional recovery than those of the PBS group.
It is well known that the efficiency of gene transduction to such MSC populations is low, even with virus vectors such as an adenovirus (Ad) (Conget and Minguell, 2000). To date, Kurozumi et al (2004) have reported the relatively high efficiency of gene transduction using fiber mutant Ad vector, but the peak level of expression was transient because the Ad vector would not integrate the gene of interest into the genome of the host cells. Lentivirus could express a high efficiency of gene transduction into MSC, but its biosafety remains uncertain because of its origin, the human immunodeficiency virus (Trono, 2000). Retroviruses, which have the ability to integrate the gene of interest into the chromosomes of the host cells, also show a relatively high efficiency of gene transduction to MSC. However, a note of warning was stressed against the potential rise of a neoplasm with a retrovirus-based vector (Pages and Bru, 2004).
In the present study, by the in vitro HGF ELISA data and histological detection, we showed that our HSV-1 vector had successfully transferred the gene of interest to the MSC population with high efficiency in vitro, and gene-transferred MSCs had successfully functioned in vivo to express and maintain a high level of the gene of interest. We confirmed that the increased HGF expression on day 14 was primarily due to the ex-HGF-2 expression that was proven by anti-KT3 staining, as the HSV-1 vector-transferred HGF cDNA was tagged with KT3 epitope. Also, such ex-HGF-2 protein was produced within the HGF gene-transferred MSCs and secreted in the intercellular space diffusely in the combined therapy group.
Furthermore, there were no significant differences in functional recovery between the MSC-only group and the MSC-GFP group during the whole detection time course. Also, no obvious difference of apoptosis and the dividing ability was observed between naive MSCs and the HGF gene-transferred MSCs in the current study in the first 2 weeks after transplantation (data not shown). It may indicate that gene transfer with HSV-1 vector ex vivo would not influence the survival and dividing abilities and the therapeutic efficiency of MSCs after transplantation.
So far, to reduce the disability resulting from stroke, some studies have focused on the development of neuroprotective agents such as brain-derived neurotrophic factor, the fibroblast growth factor that effectively prevents delayed neuronal death after transient brain ischemia (Kurozumi et al, 2004; Watanabe et al, 2004). Recently, overexpression of HGF that can improve the neurological sequalae by neuroprotection, reduce the infarct volume, and the likelihood of brain edema after stroke was reported (Miyazawa et al, 1998; Tsuzuki et al, 2000; Hayashi et al, 2001; Shimamura et al, 2004). It suggested that HGF should be one of the most potent growth factors for treating brain ischemia.
To detect the therapeutic efficiency of combined therapy, we tried to treat brain ischemia in the superacute and acute therapeutic phases. Both of them showed significant improvement of neurological deficits compared with MSC-only cell therapy. We got the same result as that Shimamura et al (2004) had reported, that HGF had the therapeutic efficiency of reducing the infarction volume after transient MCAO. We also found on day 14 that the MSC-only treated group could significantly reduce the infarction volume under the superacute treatment compared with the PBS-treated group, but not under the acute treatment. This was the same as the Chopp's group had reported, that transplanted MSCs to the transient MCAO model 24 h after ischemia occurred had improved neurological function recovery, but not significantly decreased infarction area (Chen et al, 2000, 2001; Li et al, 2000, 2001), while our data of the superacute treatment showed the contrast result. We thought that it might have been caused by the different therapeutic time window. Furthermore, we found significant neurological recovery of the rats treated with combined therapy on day 14, 24 h after MCAO occurred (acuter phase), than the rats of the MSC-only group treated even 2 h after MCAO occurred (superacute phase). It indicates that our combined therapeutic method may extend the therapeutic time window for treating brain ischemia at least until 24 h after the onset of MCAO, while compared with the MSC-only cell therapy. To treat transient ischemia, both the combined therapeutic method and superacute therapeutic time window might be important.
Mesenchymal stromal cell-only therapy also showed significant improvement of functional outcome and decrease of infarction volume when cells were administered 2h after stroke. A more likely mediator of short-term benefit may reflect increased production of growth factors, including neurotrophins adjusted to the needs of the compromised tissue with an array of reducing host cells' apoptosis in the IBZ, including neurons, and promoting functional recovery of the remaining neurons (David and Thomas, 2002; Chopp and Li, 2002). After stroke, cerebral tissue reverts to an earlier stage of development and thus becomes highly responsive to stimulation by cytokines, trophins, and growth factors from the invading MSCs (Chopp and Li, 2002). The MSCs may simply provide the resources required by the ontogenous cerebral tissue to stimulate cerebral remodeling.
In the present study, the combined therapy group showed more therapeutic benefit than the MSC-only cell therapy. Hepatocyte growth factor gene-modified MSCs may also behave as small molecular factories, secrete an array of cytokines and trophic factors over an extended period and not in a single bolus dose, directly involved in promoting plasticity of the ischemic damaged neurons or in stimulating glial cells to secrete neurophins. Marrow stromal cells secrete many cytokines known to play a role in hematopoiesis (Dormady et al, 2001), and also supply autocrine, paracrine, and juxtacrine factors that influence the cells of the marrow microenvironment themselves (Haynesworth et al, 1996). The interaction of MSCs with the host brain may lead MSCs and parenchymal cells also to produce abundant trophic factors, which may contribute to recovery of function lost as a result of a lesion too (Williams et al, 1986). We speculate that HGF gene-modified MSCs also had carried out such ways not only to produce extended and abundant exogenous HGF, but also a variety of other cytokines and trophic factors, and interact with each other in an anatomically distributed, tissue-sensitive, and temporally ongoing way.
Other functions of HGF include reducing the BBB destruction without exacerbating cerebral edema, decreasing intracranial pressure, inducing angiogenesis, and interacting with other kinds of neurotrophic factors; cytokines that are secreted by MSCs themselves may also take part in improving the neurological recovery after stroke. We also speculate that the various cytokines secreted from MSCs or MSC-HGF activate the proliferation and differentiation of endogenous neural stem and progenitor cells in the subventricular zone, such as Chopp and Li (2002) had reported. Also, transplanted MSCs themselves might differentiate into some kinds of central never system cells (Woodbury et al, 2000). Actually, we also found some MSCs expressing glial phenotype 4 weeks after transplantation only in the combined therapy group (data not shown), which might suggest that HGF gene transduction could influence transplanted MSC differentiation. But tissue regeneration might be another part of the mechanisms that induced recovery after stroke mainly occurs in the chronic therapeutic time course.
Anyhow, our MSC-HGF combined therapy enhanced the therapeutic efficiency than the MSC-only cell therapy for stroke in rats treated in both the superacute and acute phases. The target gene was successfully transferred to MSCs with the HSV-1 virus vector in vitro, and later the gene-modified MSCs served as both a therapeutic material and a vector platform that continuously carried the target gene into the brain and functioned in vivo. This method might be safer than direct gene transfer with viral vectors for in vivo treatments, more therapeutically efficient than MSC-only cell therapy, extend the therapeutic time window from superacute to at least 24 h after ischemia happened, and also could be used as a post-treatment method for stroke. Although the best therapeutic time schedule, the administration route of MSCs and best cytokine gene (or cocktail of the genes) should be explored for better clinical application. It may require a broad array of treatments to prevent neurological disorders in brain ischemia, which may offer a promise for human clinical treatment in future.
