Abstract
Brain arteriovenous malformations (BAVMs) are a potentially life-threatening disorder. Matrix metalloproteinase (MMP)-9 activity was greatly increased in BAVM tissue specimens. Doxycycline was shown to decrease cerebral MMP-9 activities and angiogenesis induced by vascular endothelial growth factor (VEGF). In the present study, we determined the dose-response effects of doxycycline and minocycline on cerebral MMP-9 using our mouse model with VEGF focal hyperstimulation delivered with adenoviral vector (AdVEGF) in the brain. Mice were treated with doxycycline or minocycline, respectively, at 1, 5, 10, 30, 50, or 100 mg/kg/day through drinking water for 1 week. Our results have shown that MMP-9 messenger ribonucleic acid (mRNA) expression was inhibited by doxycycline starting at 10 mg/kg/day (P < 0.02). Minocycline showed more potent inhibition on MMP-9 mRNA expression, starting at 1 (P < 0.005) and further at more than 30 (P < 0.001) mg/kg/day. At the enzymatic activity level, doxycycline started to suppress MMP-9 activity at 5 mg/kg/day (P < 0.001), while minocycline had an effect at a lower dose, 1 mg/kg/day (P < 0.02). The inhibition of cerebral MMP-9 mRNA and activity were highly correlated with drug levels in the brain tissue. We also assessed the potential relevant signaling pathway in vitro to elucidate the mechanisms underlying the MMP-9 inhibition by tetracyclines. In vitro, minocycline, but not doxycycline, inhibits MMP-9, at least in part, via the extracellular signaling-related kinase 1/2 (ERK1/2)-mediated pathway. This study provided the evidence that the tetracyclines inhibit stimulated cerebral MMP-9 at multiple levels and are effective at very low doses, offering great potential for therapeutic use.
Introduction
Recent data suggest that brain arteriovenous malformation (BAVMs) are associated with excessive vascular remodeling due to pathologically increased angiogenesis (Gaetani et al, 1999; Hashimoto et al, 2001; Todor et al, 1998). Our earlier observation showed increased levels of matrix metalloproteinase (MMP)-9 relative to tissue inhibitors of metalloproteinases in surgical BAVM specimens (Hashimoto et al, 2003). By affecting vascular stability, MMP-9 may play a key role in vascular malformation growth and spontaneous hemorrhage.
Development of strategies to stabilize the vascular wall can be applied to lessen the incidence and morbidity of cerebral hemorrhage (or re-hemorrhage) in BAVM patients. Matrix metalloproteinase inhibitors have shown promise in treating neoplastic diseases (Sternlicht and Bergers, 2000) and manipulating vascular phenomena. Targeted deletion of the MMP-9 gene in murine models of abdominal aortic aneurysms (AAA) specifically suppresses experimental aneurysm formation (Pyo et al, 2000). In clinical trials, oral tetracycline-class drugs decreased MMP in AAAs and carotid plaques (Axisa et al, 2002; Baxter et al, 2002; Curci et al, 2000). While doxycycline has been evaluated most extensively for vascular diseases, minocycline is also emerging as clinically applicable nonspecific MMP inhibitors that possess neuroprotective properties (Yong et al, 2004).
As a first step in pursuing this therapeutic strategy for brain vascular malformations, we proposed to test the hypothesis that doxycycline and minocycline suppress the stimulated cerebral MMP-9 expression in a dose-dependent manner in a murine cerebral angiogenic model (Yang et al, 2003). Smooth muscle cells are one of the important components of vascular structure and play a crucial role in vascular remodeling. The migration of smooth muscle cells is thought to be one of the initial events in angiogenesis promoted by MMPs. We used human aorta smooth muscle cell to determine whether there is a differential underlying mechanism of MMP-9 inhibition by the tetracyclines. Such work will provide, at least in part, theoretical underpinning for MMP inhibitors to decrease vascular remodeling and rupture in BAVM patients.
Materials and methods
Animals and Treatment
Animal use was approved by the university committee of animal research. Adult male, CD-1 mice, weight 30 to 35 g, were purchased from Charles River Laboratory (Wilmington, MA). The control and AdVEGF groups received AdlacZ, the viral vector control, and AdVEGF, recombinant human VEGF165 complementary deoxyribonucleic acid (cDNA) delivered with adenoviral vector, respectively. Mice (n = 4 to 6 per group) with VEGF gene transduction in the brain were treated with doxycycline or minocycline, respectively. The method for adenoviral-mediated VEGF gene transfer in the brain was described previously (Yang et al, 2003). Briefly, after induction of anesthesia with ketamine and xylazine (intraperitoneal), mice were placed in a stereotactic frame (David Kopf Instruments, Tujunga, CA). Hamilton syringe was inserted through a burr hole 1 mm lateral to the sagittal suture, 1 mm posterior to bregma, and 3 mm under the cortex. In all, 2 μl of adenoviral suspension with 2.88 × 106 particles of either AdlacZ or AdVEGF was injected stereotactically into the right caudate putamen. In the treatment group, doxycycline or minocycline (Sigma Co., St Louise, MO) was administered starting on the day of AdVEGF injection. The doses were administered at 1, 5, 10, 30, 50, and 100 mg/kg/day through drinking water for 1 week. Our previous data have shown that VEGF expression increased at day 5 in the mouse brain after AdVEGF transduction, with the MMP expression peaking at 1 week (Lee et al, 2004a). The mice were allowed free access to food and water, with a 12-h alternating light-dark cycle (0800 to 2000h).
Tissue Collection
Coronal sections of brain tissues, including 1 mm anterior and posterior to the injection site, were quickly frozen in liquid nitrogen and stored at —80°C. For MMP activity assessment, tissue homogenates were centrifuged at 10,000g for 20 min at 4°C, and the supernatants of the homogenates were used for zymography. For mRNA assessment, total RNA was extracted using Trizol reagent (GIBCO Life Technologies, Grand Island, NY, USA).
Measurement of Plasma and Tissue Tetracycline Levels
Levels of doxycycline and minocycline in the brain tissue were analyzed with a LC/MS/M system (Quattro Ultima, Micromass., Beverly, MA, USA) using the electrospray/positive ionization mode. Multiple reaction monitor (MRM) was set at 458.2 > 441.0m/z for minocycline, 444.9 > 427.8m/z for doxycycline, and 320.2 > 247.2m/z for chloroquine (as the internal standard for minocycline assay). The column was a Hypersil C8 column (4.6 × 50 mm, 3 mm particle size). For analysis of doxycycline, the mobile phase was acetonitrile/water/TFA/ 10% (NH4)2HPO4, w/v (35:65:0.1:0.01, v/v) with isocratic elution. For analysis of minocycline, a gradient elution was used with mobile phase A composed of acetonitrile/water/TFA/10% (NH4)2HPO4, w/v (15:75:0.1:0.01, v/v) and mobile phase B composed of acetonitrile/water/TFA/10%(NH4)2HPO4, w/v (35:65:0.1:0.01, v/v). Brain samples were homogenized with water and then treated with acetonitrile for protein precipitation before injecting to LC/MS/MS for drug analysis.
Human Aortic Smooth Muscle Cell Culture (HASMCs)
Human aortic smooth muscle cell cultures were purchased from Cell Systems (St Katharinen, Germany). Their homogenous nature was confirmed through immunostaining with anti-smooth muscle α-actin (Chemicon, Temecule, CA, USA). Human aortic smooth muscle cell cultures were incubated in DME medium with 10% fetal bovine serum (GIBCO, Grand Island, NY, USA) and maintained at 37°Cin5% CO2/95% ambient mixed air. Human Aortic Smooth Muscle Cell Culture experiments were performed on five to eight passages.
During the VEGF, doxycycline, and minocycline experiments, the cultured HASMCs grew to 80% confluence. These cells were incubated in serum-free medium for 24 h after removing 10% FBS medium. Human aortic smooth muscle cell cultures were then treated with VEGF (20 ng/ml) and with doxycycline at 10 and 20 μmol, respectively, at the same time for 24 h. Aliquots of conditional medium were collected for zymographic analysis.
For the phospho-extracellular signaling-related kinase (ERK)1/2 expression study, HASMCs grew to approximately 80% confluence, and were made quiescent with serum-free medium for 24 h. After being pretreated with doxycycline or minocycline, VEGF was added. Cells were incubated for 15 mins for assessment of ERK1/2 expression according to our previous study (Yao et al, 2004). PD98059, a known inhibitor of ERK1/2 pathways, served as the positive control of the study. Cells were washed twice with PBS and scraped into a lysis buffer.
The cytotoxicities of doxycycline and minocycline on HASMCs were determined using a methylthiazoltetrazolium (MTT) cleavage assay (Hanelt et al, 1994). Human aortic smooth muscle cell cultures were treated with doxycycline and minocycline at increasing concentration in 0.2% bovine serum albumin (BSA)/Dulbecco's modified Eagle's medium (DMEM). After incubation for 36 h with 20 ng/mL VEGF, 2 mg/mL MTT solution was added. Finally, MTT-containing medium was aspirated off and 150 μL of dimethyl sulfoxide (DMSO) solution was added. As a result, living cells formed crystals due to the presence of MTT. Crystals were solvated by DMSO and the absorbance was measured at 570 nM using a microplate reader.
Gelatin Zymography
Equal amounts of sample proteins were separated by electrophoresis on 10% zymogram gels (Invitrogen, Carlsbad, CA, USA). The gels were subsequently stained with colloidal blue stain (Invitrogen, Carlsbad, CA, USA). Proteolytic bands in zymography were quantified by scanning densitometry using KODAK image analysis software (Eastman Kodak Co., Rochester, NY, USA).
Real-Time Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR)
Complementary deoxyribonucleic acid was synthesized using TaqMan Reverse Transcription Reagents (Applied Biosystems, Foster City, CA, USA). Mouse MMP-9 and galactosidase (GUS) Taqman oligonucleotide primers were purchased from Applied Biosystems (Foster City, CA, USA). Real-time PCR reactions were performed by use of an ABI-Prism 7000 sequence detector. Results were analyzed using Sequence Detection Software (Applied Biosystems, Foster City, CA, USA). Levels of MMP-9 expression of mRNA were normalized to GUS as an internal control.
Western Blot Analysis
Equal amounts of protein (20 μg/lane) for each sample were electrophoresed through a 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gel, and blotted onto a nitrocellulose membrane (Amersham, Piscataway, NJ, USA). The membrane was blocked by 5% non-fat milk solution in Tris-buffered saline (TBS)/0.1% Tween-20 for 1 h at room temperature. p-Extracellular signaling-related kinase 1/2 signals were detected by incubating membranes with rabbit polyclonal p-ERK1/2 antibodies overnight (Cell Signaling, Beverly, MA, USA). Horseradish peroxidase (HRP)-labeled anti-rabbit IgG secondary antibody (Amersham, Piscataway, NJ, USA) was added for 1 h. The membrane was exposed to Kodak film (Eastman Kodak Co., Rochester, NY, USA). Bands were scanned and semiquantified by densitometry.
Statistical Analysis
Data are expressed as mean ± s.d. MMM-9 activity and mRNA expression, and the amount of p-ERK1/2 between different groups, were analyzed using ANOVA followed by Fisher's protected least significant difference (PLSD) test. Correlations of MMP-9 inhibition and brain tissue levels of tetracyclines were tested using z-test. A P-value < 0.05 is considered as statistically significant difference.
Results
Doxycycline and Minocycline Inhibition of Cerebral Matrix Metalloproteinase-9 Messenger Ribonucleic Acid
Both doxycycline and minocycline showed dose-dependent inhibition of MMP-9 mRNA expression in the mouse brain with focal hyperstimulation of AdVEGF, while minocycline appeared to have more potent effects (Figure 1). Cerebral MMP-9 mRNA expression induced by AdVEGF hyperstimulation was inhibited by doxycycline starting at 10 mg/kg/ day, corresponding to 37% inhibition of the MMP-9 mRNA in the AdVEGF-only group (P < 0.02). At higher doses of doxycycline at 50 and 100 mg/kg/ day, MMP-9 mRNA were further decreased (P < 0.002). Compared with doxycycline, minocycline inhibited MMP-9 mRNA expression at lower dosages, which started at 1 mg/kg/day (P < 0.005) and further at higher doses from 30 mg/kg/day (P < 0.001), corresponding to 38% and 63% inhibitions of MMP-9 in the AdVEGF group, respectively. There were significant differences of MMP-9 mRNA in the groups treated with minocycline at 1, 5, and 10 mg/kg/day compared with the groups treated with minocycline at 30, 50, and 100 mg/kg/day, respectively (P < 0.05).
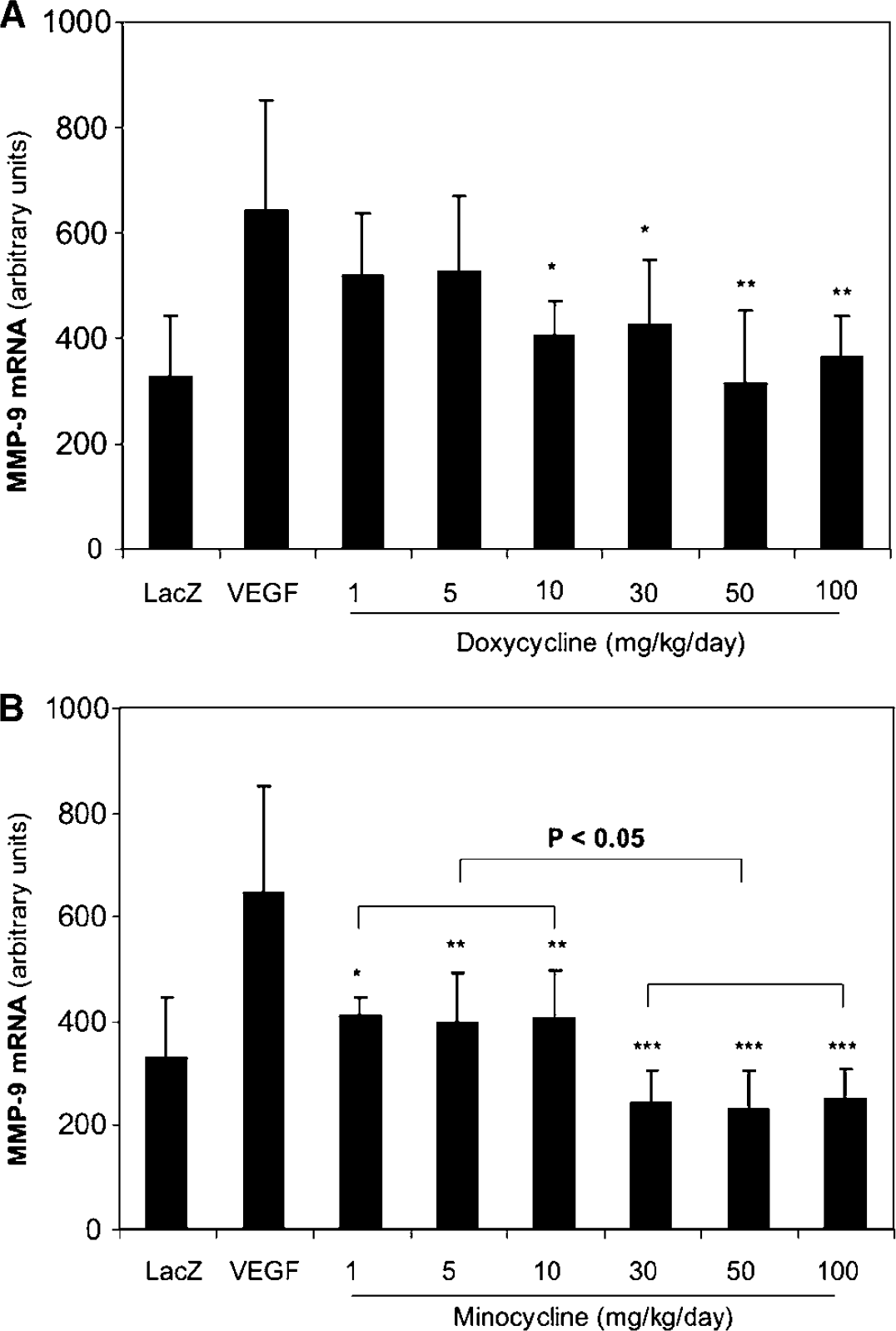
Inhibition of VEGF induced cerebral MMP-9 mRNA in a dose-dependent manner.
Doxycycline and Minocycline Inhibition of Cerebral Matrix Metalloproteinase-9 Activity
The tetracyclines suppressed AdVEGF-induced cerebral MMP-9 activities in a similar dose-dependent pattern, but at lower drug dosages than the effective dose on MMP-9 mRNA expression (Figure 2). Doxycycline suppressed MMP-9 activity starting at 5 mg/kg/day, corresponding to 60% inhibition of MMP-9 activity in the AdVEGF-only group (P < 0.05). In a similar pattern of the effects at the mRNA level, minocycline showed the effect starting at lower dosage than doxycycline at 1 mg/kg/day, corresponding to 44% inhibition of MMP-9 activity in the AdVEGF-only group (P < 0.02), and further inhibition at higher dosages.
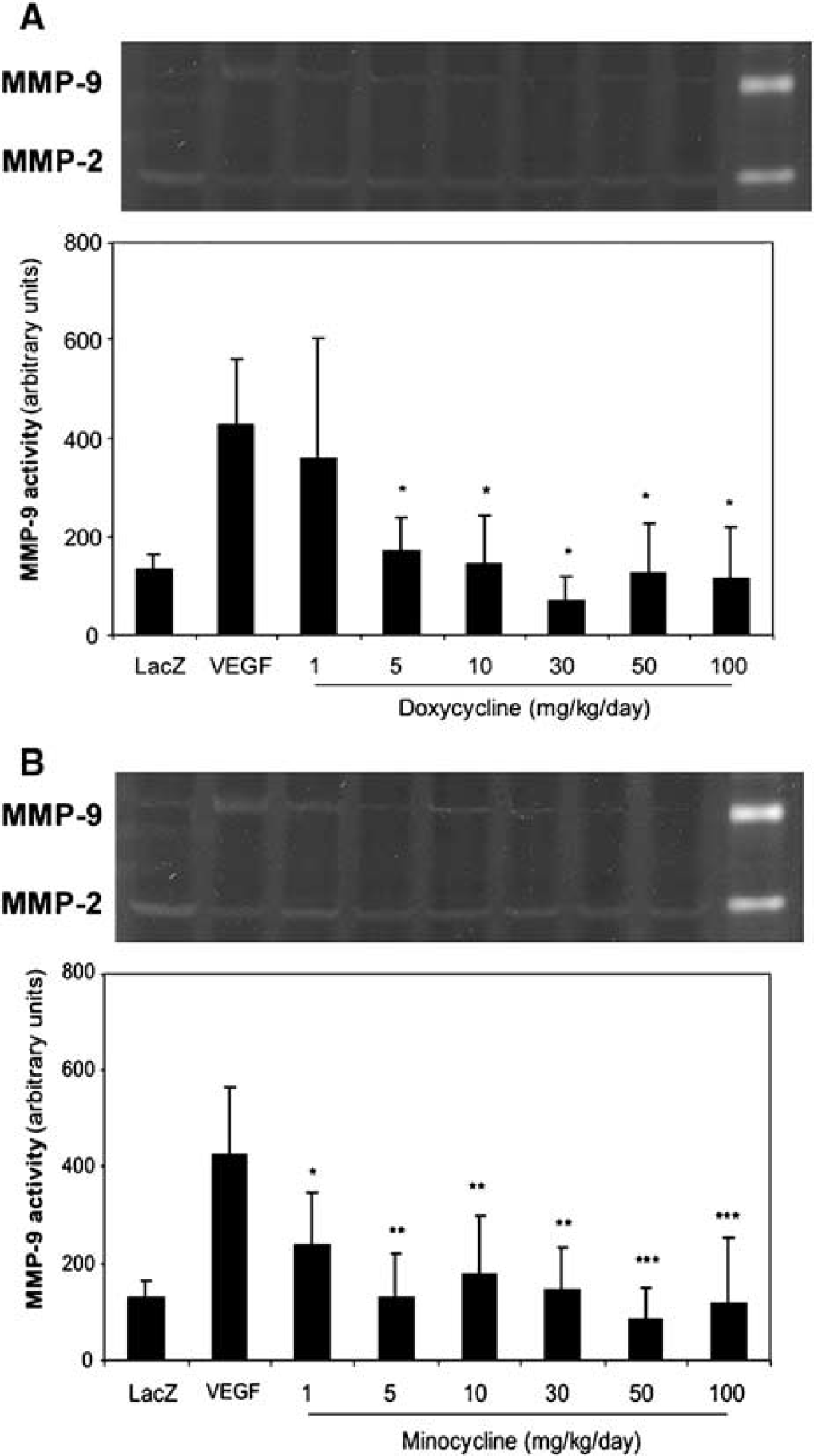
Inhibition of VEGF induced cerebral MMP-9 activity in a dose-dependent manner.
Correlation of Matrix Metalloproteinase-9 Inhibition and Levels of Tetracyclines in the Brain
To verify the drug uptake by the animals, we measured brain levels of tetracycline in several groups of mice. There were considerable variations in the drug levels among animals in the same groups. In general, minocycline appeared to have higher concentrations in the brain tissue than doxycycline at the doses that we examined. The plateau of brain levels of tetracyclines in the 100 mg/ kg/day groups was noted, while at other doses administered the brain levels of tetracyclines increased with increased oral doses correspondingly. Retrospectively, we found that the specimens with minocycline and doxycycline at 100 mg/kg/day were harvested in the afternoon, whereas the other groups were harvested in the morning (data not shown).
Despite the wide variation of tetracycline drug levels in the brain, the inhibition of cerebral MMP-9 mRNA and activity were highly correlated with the tissue levels of doxycycline and minocycline with P < 0.01 (Figure 3) in individual animals. In particular, the correlation between MMP-9 mRNA inhibition and minocycline was extremely significant with r2 = 0.789, P < 0.0001 (Figure 3B).
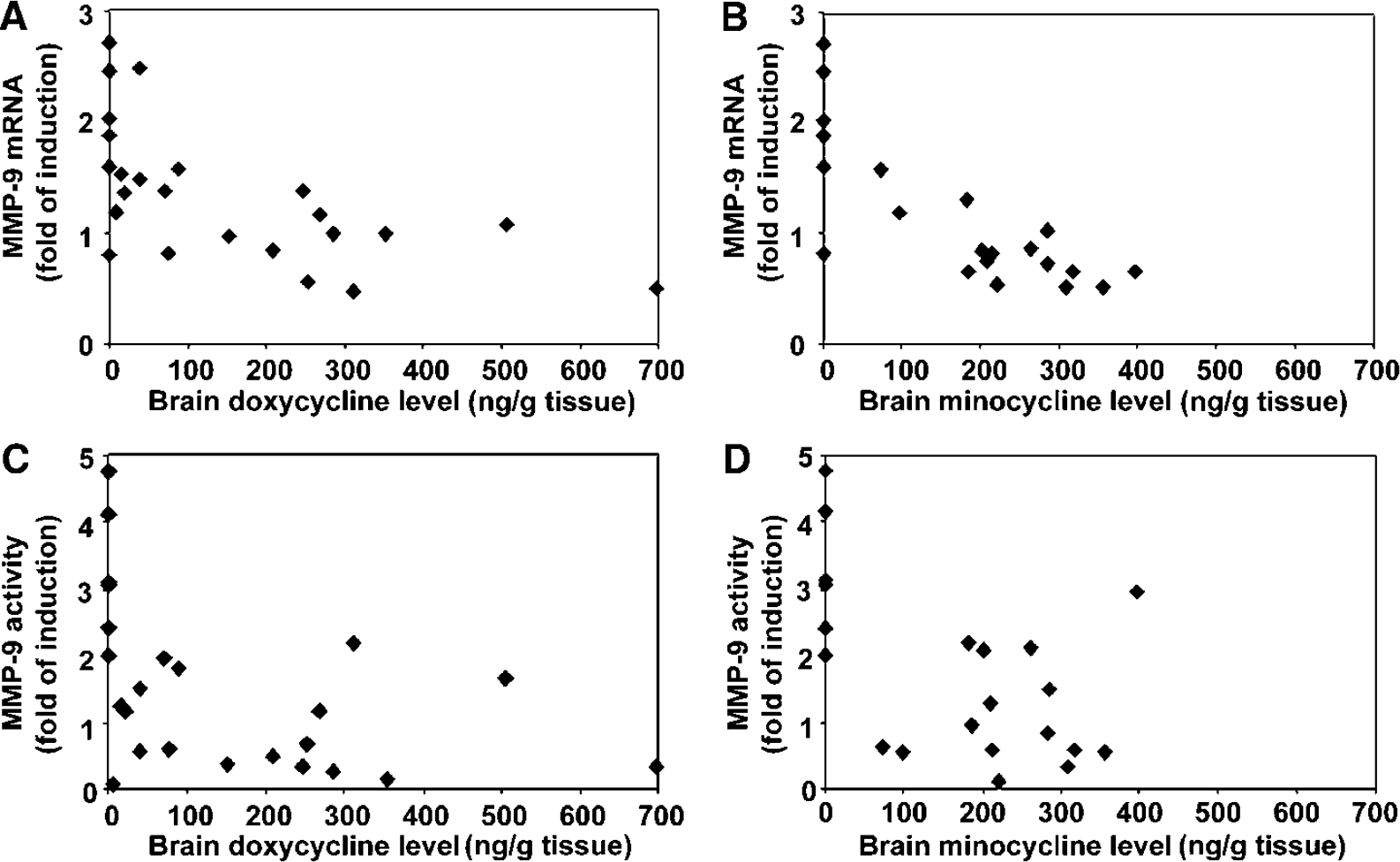
Correlation of MMP-9 inhibition and tetracycline brain tissue levels.
Inhibition of Matrix Metalloproteinase-9 Activity in Vascular Endothelial Growth Factor-Treated Human Aortic Smooth Muscle Cell Cultures
To determine whether doxycycline blocks MMP-9 activities in VEGF-treated HASMCs, we tested two different doses of doxycycline in VEGF-treated HASMCs. The results showed that doxycycline could inhibit MMP-9 in a dose-dependent manner (P < 0.05, Figure 4).
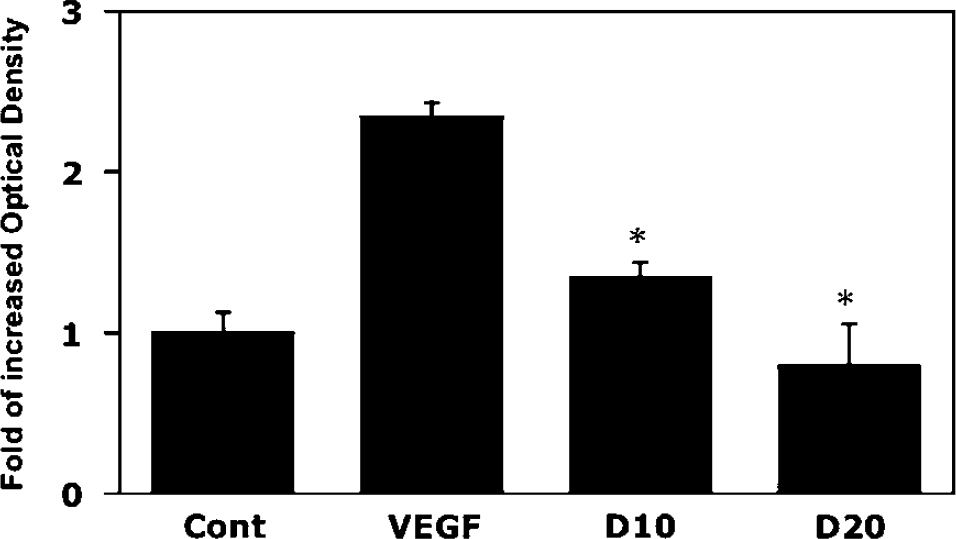
Doxycycline inhibits VEGF-induced MMP-9 activities in HASMCs. Effects of doxycycline on VEGF-induced MMP activities in HASMCs were determined by MMP zymographic assay. After pretreatment of serum-free HASMCs for 24 h, HASMCs were treated with VEGF (20ng/ml) and doxycycline for 24 h. Bar graphs represent the quantitative zymograms. D10 = doxycycline 10mmol, D20 = doxycycline 20mmol. Data show five independent experiments with duplicates, and are expressed as mean ± s.d. *P < 0.05, doxycycline treatment groups versus VEGF-alone group.
To rule out HASMC death as the cause for inhibition of MMP activity, we evaluated cell viability using MTT test. Our results show that doxycycline or minocycline at the concentrations of 60 μmol/L was not cytotoxic to confluent HASMCs. Increased concentration of doxycycline at 100 μmol/ L resulted in a 40% reduction of viability (data not shown), suggesting that the dose range in our experiments is safe, and the inhibition of doxycycline or minocycline on HASMC MMP activity was not due to the cytotoxicity caused by the agents.
Inhibition of Extracellular Signaling-Related Kinase 1/2 in Vascular Endothelial Growth Factor-Treated Human Aortic Smooth Muscle Cell Cultures
We next explored the mechanism that doxycycline and minocycline regulate VEGF-induced MMP activation in HASMCs. Western blot analysis of signaling pathway of ERK1/2 showed that VEGF could induce ERK1/2 phosphorylation. Minocycline in the doses of 15 to 30 μm significantly downregulated ERK1/2 phosphorylation in VEGF-treated HASMCs compared with the VEGF-only group (P < 0.05, Figure 5). However, doxycycline could not inhibit ERK1/2 phosphorylation. These results suggest that minocycline may inhibit VEGF-induced HASMC MMP activity at least partially through the ERK1/2 pathway, but not doxycycline.
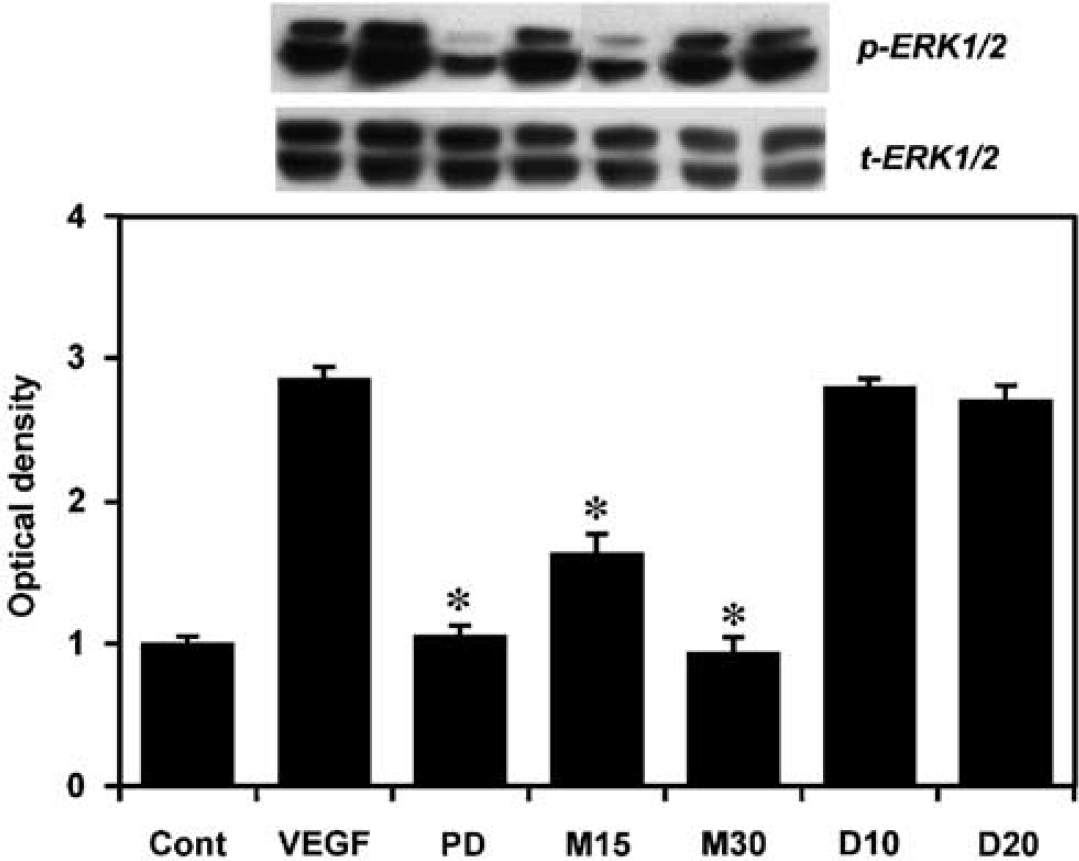
Minocycline inhibits ERK1/2 pathway in VEGF-stimulated HASMC. Effects of doxycycline and minocycline on VEGF-induced ERK1/2 expression in HASMCs were determined by Western blot analysis. After pretreatment of serum-free HASMCs for 24 h, HASMCs were treated with doxycycline and minocycline for 30 min, and then treated with VEGF (20 ng/ml) for 24 h. Bar graphs show Western blot analysis results. Minocycline, but not doxycycline, inhibited VEGF-induced ERK1/2 phosphorylation in the HASMCs. Cont= control group, PD = PD98059, M15 = minocycline 15μmol, and M30= minocycline 30μmol, D10 = doxycycline 10μmol, D20 = doxycycline 20μmol. Data are relative values compared with the control group, shown as mean ± s.d.; n = 5. *P < 0.05, treatment groups versus VEGF-alone group.
Discussion
The present study has shown that (1) doxycycline and minocycline inhibit cerebral MMP-9 in a dose-dependent manner; (2) the inhibition of MMP-9 takes place at both mRNA and proteolytic activity levels, and highly correlates with brain levels of tetracyclines; (3) the inhibition of MMP-9 by minocycline and doxycycline might be mediated through different signaling pathways.
While the therapeutic potential of doxycyline on MMP activity associated with pathogenesis of vascular diseases has been studied, little to no comparable data exist for minocycline, which is increasingly implicated in the treatment of central nervous system diseases. Doxycycline treatment was associated with reduction of MMP-9 levels in the aortic wall in patients with abdominal aortic aneurysms and showed dose-dependent reduction of MMP-9 in cultured human mononuclear phagocytes, one of the major sources of MMP-9 in human and experimental aneurismal tissues (Curci et al, 2000). In a mouse abdominal aortic aneurysm model, doxycycline at 10, 50, and 100 mg/kg/day doses through drinking water produced 33%, 44%, and 66% reductions in aneurysm growth, respectively (Prall et al, 2002). More importantly, the serum levels present in the mice allowed for a 33% to 66% reduction in aortic growth and were comparable to the plasma levels achieved in human subjects with 100 mg of doxycycline twice daily. These data implied that MMP inhibition by tetracyclines in a range of drug dosage similar to antimicrobial dose in human may have functional effect on the vascular wall remodeling. In our earlier study, doxycycline treatment at 30 mg/kg/day through drinking water inhibited the induced MMP-9 activity in association with cerebral angiogenesis stimulated by VEGF in the mouse brain (Lee et al, 2004a). In the present study, we have chosen a wide range of tetracycline oral doses similar to the study of mouse abdominal aortic aneurysms, but further extended the dose to as low as 1 mg/kg/day.
Studies of patients with acute coronary syndromes and chronic periodontitis also reported metalloproteinase inhibition with subantimicrobial daily doses of doxycycline at 20 mg (Brown et al, 2004; Lee et al, 2004b). We have shown that both tetracycline derivatives can inhibit the VEGF-induced cerebral MMP-9 activity in a dose-dependent manner and that the inhibition started at even lower doses, that is, doxycycline and minocycline at 5 and 1mg/kg/ day, respectively. The fact that our results are in agreement with findings in other organ systems suggested that cerebral MMP-9 can be inhibited by the tetracyclines at the doses comparable to, or even much lower than, the 100mg twice daily conventional dosage as antibiotics.
Our measurements of tetracycline levels in the brain tissue after oral administration through drinking water have shown wide distributions, presumably reflecting the variability of intake and absorption between individual animals, as well as the relatively short half-life of tetracyclines in rodents. The optimal doses of tetracyclines and the delivery of the drugs to the brain remain a concern, although the tetracyclines, especially minocycline, have been studied in experimental models for a wide range of neurological diseases. Fagan et al (2004) reported highly variable minocycline levels in the plasma and cerebrospinal fluids after intraperitoneal injection, and concluded that intravenous administration was safer and more reliable for acute neuroprotection. Another recent study showed considerable intermouse variability after oral administration of minocycline and doxycycline at 0.5 to 2.0mg/mL (Smith et al, 2003). It was noticed that the half-life of minocycline is 2 to 3 h in the rodents (Andes and Craig, 2002; Colovic and Caccia, 2003), which is much shorter than that in human at 15 to 20 h (Saivin and Houin, 1988). After intraperitoneal injection, minocycline levels in the brain peaked at 2 h and continued to decline thereafter (Smith et al, 2003). If we assume that mice primarily take the drugs through the drinking water while they are active during the dark cycle (2000 to 0800 h), the continuous decrease of their levels in the brain after the peak may help to explain the variation in our data, especially the relatively lower than expected drug levels in our 100mg/kg/ day groups harvested later in the day. Nevertheless, the inhibition of cerebral MMP-9 correlated well with the measured drug levels in the brain in spite of the variability in the drug disposition for tetracyclines.
The precise mechanism of MMP inhibition by tetracyclines remains unclear. Studies showed that tetracycline antibiotics are pluripotent drugs that inhibit the activity of MMP-9 and affect many cellular functions, including proliferation, migration, and vascular remodeling. Generally, tetracyclines are thought to act via both direct and indirect mechanisms. Several mechanisms have been proposed by previous studies, suggesting that the inhibition may be due to modulations including via gene transcription, scavenging reactive oxygen species, or Zn2+ binding (Ryan et al, 2001). Our previous study showed that minocycline inhibiting MMP-9 activity is also through inhibiting ERK and PI3K pathways, suggesting that the effects of minocycline could be through multiple pathways (Yao et al, 2004). Here, we showed that both doxycycline and minocycline inhibited cerebral MMP-9 mRNA expression and that gelatinase activities were further inhibited with lower treatment dose in doxycycline and higher degree in minocycline treatment, suggesting that the effects are at gene transcription level and at protein translation and/or post-translational levels. Nevertheless, this result of complementary combination of inhibition at multiple levels confirms that tetra-cyclines are particularly effective in influencing MMP-9 levels in the brain.
In our study, minocycline appeared to be a more potent inhibitor of cerebral MMP-9 than doxycycline at both mRNA and activity levels. The difference in potency was especially notable at the mRNA levels and lower drug dosages. Minocycline inhibited cerebral MMP-9 mRNA expression to 62% of VEGF stimulation group starting at 1 mg/kg/day, and further to 37% at 30 mg/kg/day. In contrast, doxycycline started the inhibition at a higher dose at 10 mg/kg/day. In a similar trend, at the activity level, minocycline started to inhibit cerebral MMP-9 at 1 mg/kg/day, compared with doxycycline at a higher dose of 5 mg/kg/day. Our results are consistent with other studies showing that minocycline is more effective in treating diseases in the central nervous system than doxycycline, which has been attributed to minocycline's higher lipid solubility, and thus better penetration of the blood-brain barrier (BBB) (Barza et al, 1975). Both doxycycline and minocycline have been detected in the CSF and brain tissue after oral administration under normal physiological conditions, but not their less lipid-soluble pro-type, tetracycline. It is well known that VEGF causes BBB leakage. Although there is a good chance for a better drug penetration into brain tissue under VEGF hyper-stimulation, as in our model, currently there is insufficient data to characterize it. Nevertheless, it would be a favorable event for drug treatment if it does exist.
In addition to the difference in pharmacokinetics, the inhibition of MMP-9 by the tetracyclines might be mediated through different biochemical pathways. Studies of inflammatory signaling suggested that minocycline and doxycycline might have differential effects in different species and cell types (Amin et al, 1996). Compared to doxycline, minocycline has more potent neuroprotection, which was also attributed to the difference in its affinity with enzymes or cellular structures associated with ischemic damage, and better brain penetration (Yrjanheikki et al, 1998). Earlier studies reported that ERK pathways, an important mechanism in growth-factor-stimulated angiogenesis, contributed to the transcriptional regulation of MMP-9 (Cho et al, 2000). A recent study from our group has shown that minocycline exerted multiple inhibitory effects on vascular smooth muscle cell migration, including inhibition of MMP expression through the ERK1/2 pathway (Yao et al, 2004). In the present study, doxycycline did not affect the ERK pathway, in contrast to downregulation of ERK1/2 phosphorylation in VEGF-treated HASMCs by minocycline. This finding may help explain, at least partially, the fact that minocycline is more potent in the inhibition of MMP-9 mRNA induced by VEGF stimulation, although this is merely an example of differences in the biological mechanism. There are other possible mechanisms of MMP-9 inhibition, and doxycycline may inhibit MMP-9 through alternative pathways. A recent study in human corneal epithelial cells has shown that doxycycline markedly inhibited the tumor growth factor (TGF)-beta1-induced production of MMP-9 and activation of the Smad, c-Jun N-terminal kinase, ERK, and p38 mitogen-activated protein kinase signaling pathways (Kim et al, 2005). More studies still need to be performed to clarify relevant mechanisms in the cerebral vasculature.
In summary, we have clarified the dose-response relationship for inhibiting stimulated cerebral MMP activity with doxycycline and minocycline, supported by significant correlation of the degree of MMP inhibition and the levels of the tetracycline derivatives in the brain. The present study has provided promising evidence for future studies to evaluate the use of tetracyclines to decrease the rate of spontaneous brain hemorrhage in patients harboring BAVMs, in which MMPs could be a key contributing factor.
Footnotes
Acknowledgements
The authors wish to thank Voltaire Gungab and members of the UCSF BAVM study project for their assistance in data collection and manuscript preparation.
