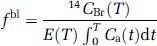Abstract
Upregulation of endothelin B (ETB) and 5-hydroxytryptamine 1B (5-HT1B) receptors via transcription has been found after experimental subarachnoid hemorrhage (SAH), and this is associated with enhanced phosphorylation of the mitogen-activated protein kinase (MAPK) extracellular signal-regulated kinase (ERK1/2). In the present study, we hypothesized that inhibition of ERK1/2 alters the ETB and 5-HT1B receptor upregulation and at the same time prevents the sustained cerebral blood flow (CBF) reduction associated with SAH. The ERK1/2 inhibitor SB386023-b was injected intracisternally in conjunction with and after the induced SAH in rats. At 2 days after the SAH, cerebral arteries were harvested for quantitative real-time polymerase chain reaction, immunohistochemistry and analysis of contractile responses to endothelin-1 (ET-1; ETA and ETB receptor agonist) and 5-carboxamidotryptamine (5-CT; 5-HT1 receptor agonist) in a sensitive myograph. To investigate if ERK1/2 inhibition had an influence on the local and global CBF after SAH, an autoradiographic technique was used. At 48 h after induced SAH, global and regional CBF were reduced by 50%. This reduction was prevented by treatment with SB386023-b. The ERK1/2 inhibition also decreased the maximum contraction elicited by application of ET-1 and 5-CT in cerebral arteries compared with SAH. In parallel, ERK1/2 inhibition downregulated ETB and 5-HT1B receptor messenger ribonucleic acid and protein levels compared with the SAH. Cerebral ischemia after SAH involves vasoconstriction and subsequent reduction in the CBF. The results suggest that ERK1/2 inhibition might be a potential treatment for the prevention of cerebral vasospasm and ischemia associated with SAH.
Keywords
Introduction
Spontaneous rupture of a cerebral aneurysm gives rise to subarachnoid hemorrhage (SAH) and carries an initial mortality of 15% to 20%. Patients who survive may, despite surgery and the current best treatment, develop significant cerebral ischemia after a period of 4 to 7 days. This condition is called cerebral vasospasm (CVS) and causes considerable morbidity and mortality (Mann, 1992). Numerous theories have been proposed to explain the pathophysiological mechanisms responsible for the late vasospasm that occurs after a SAH (Edvinsson and Krause, 2002), for example, a localized vascular mechanism, enhanced levels of ions or free radicals (Asano, 1999; Macdonald et al, 2004; Nozaki et al, 1990; Peterson et al, 1990), reduced levels of endothelial relaxant factors (Cook, 1995; Findlay et al, 1989; Kanamaru et al, 1989; Mayberg et al, 1978; Pluta, 2005), damage to perivascular nerves (Edvinsson et al, 1982, 1994; Edvinsson L, 2002) and a central nervous system dysfunction (Edvinsson L, 2002; Shiokawa and Svendgaard, 1994). In addition, there are data showing elevation of circulatory and/or local messenger molecules such as endothelin-1 (ET-1) and 5-hydroxytryptamine (5-HT) (Zervas, 1979; Zervas et al, 1975; Zimmermann and Seifert, 1998). Endothelin-1 and 5-HT are potent cerebral vasoconstrictors which mediate their effects through their respective receptors endothelin A (ETA), endothelin B (ETB) and 5-HT receptors (Masaki et al, 1994; Zimmermann and Seifert, 1998). In human cerebral arteries, it has clearly been shown that 5-hydroxytryptamine 1B (5-HT1B) is the dominant contractile 5-HT receptor (Nilsson et al, 1999) and ETA receptors for ET-1 (Nilsson et al, 1997).
We have recently revealed a novel phenomenon which might be of uttermost importance in the understanding of the successive development of vasoconstriction that occurs after SAH. In experimental SAH (Hansen-Schwartz et al, 2003a, b), there is upregulation of ETB and 5-HT1B receptors in cerebral arteries. The phenomenon is not linked to a single specific receptor subtype and may therefore explain the weak effects observed in clinical trials of specific receptor antagonists (Shaw et al, 2000; Wilkins, 1986). We have examined the intracellular pathways responsible for this receptor upregulation using a model of organ culture of cerebral artery segments. In this model, middle cerebral arteries (MCA) were incubated for 24 h, which induced upregulation of contractile ETB receptors both functionally and on the messenger ribonucleic acid (mRNA) level. This upregulation was attenuated with the extracellular signal-regulated kinase (ERK)l/2 inhibitor SB386023-b (Henriksson et al, 2004). SB386023-b inhibits the mitogen-activated protein kinase kinase kinase (MAPKKK) upstream of mitogen-activated protein kinase (MAPK)/ERK kinase (MEK) 1/2, which belongs to the raf family. It is specific for the ERKl/2 pathway, since raf binds to and activates MEK 1/2 and no other MAPKK (Henriksson et al, 2004; Schaeffer and Weber, 1999). Mitogen-activated protein kinases play a pivotal role in intracellular signalling and regulate cellular differentiation, proliferation and survival (Davis, 1993; Lewis et al, 1998). Several studies have suggested an involvement of MAPK in the pathogenesis of vasospasm after SAH (Satoh et al, 2002; Tibbs et al, 2000; Zhang et al, 2001).
The aim of the present study was to investigate the hypothesis that inhibition of the MAPK pathway ERKl/2 with SB386023-b will prevent SAH-induced decrease in cerebral blood flow (CBF) and cerebrovascular receptor upregulation. The cerebral arteries were examined using a sensitive in vitro pharmacological method, where the contractile responses to ET-1 (endothelin ETA and ETB receptor agonist), sarafotoxin 6c (S6c; selective endothelin ETB receptor agonist) and 5-carboxamidotryptamine (5-CT; specific 5-HT1 receptor agonist) were measured. To investigate if the ERKl/2 inhibition had an influence on the local CBF after SAH, an autoradiographic technique was used (Sakurada et al, 1978; Gjedde et al, 1980). In addition, the mRNA and protein levels of ET and 5-HT1B receptors were investigated by quantitative real-time polymerase chain reaction (PCR) and immunohistochemistry.
Materials and methods
All animal procedures were performed strictly within national laws and guidelines and approved by the University Animal Experimentation Inspectorate.
Rat Subarachnoid Hemorrhage Model
Subarachnoid hemorrhage was induced by a model originally devised by Svendgaard and carefully described by Prunell et al (2003).
Male Sprague–Dawley rats (350 to 400 g) were anesthetized using 5% halothane (Halocarbon Laboratories, River Edge, NJ, USA) in N2O/O2 (30:70). The rat was intubated and artificially ventilated with inhalation of 0.5% to 1.5% halothane in N2O/O2 (70:30) during the surgical procedure. The depth of anesthesia was carefully monitored and the respiration checked by regularly withdrawing arterial blood samples for blood gas analysis (Radiometer, Copenhagen, Denmark). An electric temperature probe was inserted into the rectum to record the temperature, which was maintained at 37°C. An arterial catheter to measure blood pressure was placed in the tail artery and a catheter to monitor intracranial pressure (ICP) was placed in subarachnoid space under the subocciptal membrane. At either side of the skull, 3 mm from the midline and 4 mm anteriorly from the bregma, holes were drilled through the scull bone down to dura mater (without perforation), allowing the placement of two laser Doppler flow probes to measure cortical CBF. Finally, a 27-G blunt canula with side hole was introduced stereotactically 6.5 mm anterior to the bregma in the midline at an angle of 30° to the vertical. With the aperture pointing to the right, the needle was lowered until the tip reached the scull base 2 to 3 mm anterior to the chiasma. After 30 mins of equilibration, 250 μL blood was withdrawn from the tail catheter and injected intracranially at a pressure equal to the mean arterial blood pressure (MABP) (80 to 100 mm Hg). Subsequently, the rat was kept under anesthesia for another 60 mins to allow recovery from the cerebral insult, after which catheters were removed and incisions closed. The rat was then revitalized and extubated. A subcutaneous injection of carprofen (4.0 mg/kg) (Pfizer, Denmark) was administered and the rat was hydrated subcutaneously using 40 mL isotonic sodium chloride at the end of the operation and at day one. During the period of observation the rat was monitored regularly, and if showing severe distress the animal was prematurely killed. In addition, a series of sham-operated rats were prepared. They went through exactly the same procedure as described above, with the exception that no blood was injected intracisternally. After 2 days, either harvesting of vessels or autoradiographic measurements was performed (see below for details).
Rat Subarachnoid Hemorrhage Model with Mitogen-Activated Protein Kinase Extracellular Signal-Regulated Kinase 1/2 Inhibition
This group of animals went through the same procedure as the above-mentioned SAH animals. In addition, they were treated with SB386023-b (a kind gift from Dr AA Parsons, GSK, UK) in conjunction with the operation and after the induced SAH. All animals treated with SB386023-b received 5 injections intracisternally of SB386023-b. In all, 50 μL, 10−6 mol/L of SB386023-b was injected intracisternally at 30 mins before the induced SAH and after the SAH (20 μL; 10−6 mol/L) SB386023-b was administered repeatedly after 3, 6, 24 and 32 h from the first SB386023-b injection. This dose was based on previous detailed work on isolated cerebral arteries (Henriksson et al, 2004); the dose was chosen at near max inhibition and calculation of cerebrospinal fluid volume/turnover.
Autoradiographic Measurements of Regional Cerebral Blood Flow
Local CBF was measured by a model originally described by Sakurada et al (1978) and modified by Gjedde et al (1980).
In brief, after 48 h of observation, rats in the various groups (sham, SAH and SAH treated with SB386023-b) were anesthetized using 5% halothane in N2O/O2 (30:70). The animal was intubated and artificially ventilated with inhalation of 0.5% to 1.5% halothane in N2O/O2 (70:30) during the surgical procedure. The anesthesia and the respiration were monitored by regularly withdrawing arterial blood samples for blood gas analysis. A catheter to measure MABP was placed in the right femoral artery and a catheter for blood sampling was placed in the left femoral artery. This catheter was connected to a constant velocity withdrawal pump (Harvard apparatus 22, USA) for mechanical integration of tracer concentration. In addition, a catheter was inserted in one femoral vein for injection of heparin and for infusion of the radioactive tracer. The MABP was continuously monitored and a temperature probe was inserted into the rectum to record the temperature, which was regularly maintained at 37°C. The hematocrit was measured by a hematocrit centrifuge (Beckman Microfuge 11, USA).
After 30 min of equilibration, a bolus injection of 50 μCi of 14C-iodoantipyrine 4[N-methyl-14C] (Perkin-Elmer, Boston, MA, USA) was administered intravenously. Arterial blood (122 μL) was withdrawn over 20 secs. Immediately after this the animal was decapitated, the brain removed and immersed in isopentane (JT Baker, Deventer, Netherlands) chilled to −50°C.
The arterial blood sample was transferred to liquid scintillation counting vials containing 1 mL mixture of Soluene-350 (Perkin-Elmer, Boston, MA, USA) and Isopropanol (JT Baker, Deventer, Netherlands) (1:1). After 2 h at 60°C, 0.2 mL of 30% hydrogen peroxide was added to the vials, and the samples were maintained at room temperature for 15 to 30 min. Thereafter, the samples were kept at 60°C for 30 min and 10 to 15 mL Hionic-Fluor (Perkin-Elmer, Foster, CA, USA) was added. The β-radioactivity scintillation counting was performed on the samples with a program that included quench correction (Packard 2000 CA, Hvidovre, Denmark).
The 14C activity in the tissue was determined after sectioning the brain in 20 μm sections at −20°C in a cryostat (Wild Leitz A/S, Glostrup, Denmark). The sections were exposed to X-ray films (Kodak, Denmark) together with 14C methylmethacrylate standards (Amersham Life Science, Buckinghamshire, UK), which exposed the films for 20 days. Densities of the autoradiograms were measured with a Macintosh computer equipped with an analog CF 4/1 camera (Kaiser, Germany) and a transparency flat viewer (Color-Control 5000, Weilheim, Germany). The 14C content was determined in several brain regions (see Table 1). The CBF was calculated from the brain tissue 14C activity determined by autoradiography using Gjedde et al's (1980) equation:
where fb1 is the blood flow per unit mass, CBr (T) the isotope content, E(T) the next extraction fraction of the isotope in the time from t = 0 to t = T, t = the variable time, T = the experimental time and Ca (t) is the arterial blood concentration of the isotope at time t.
Regional cerebral blood flow 48 h after subarachnoid hemorrhage
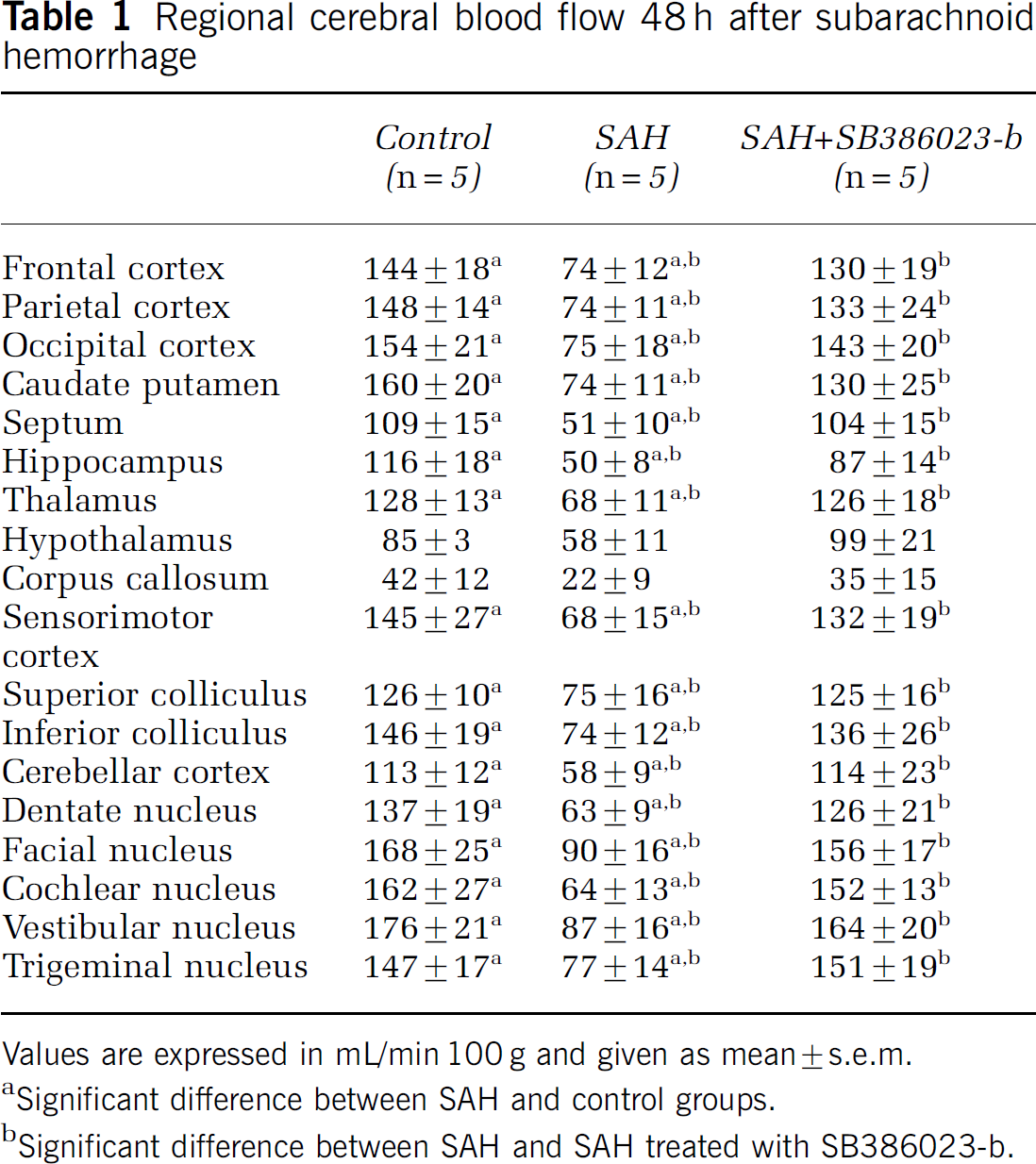
Values are expressed in mL/min 100 g and given as mean ± s.e.m.
Significant difference between SAH and control groups.
Significant difference between SAH and SAH treated with SB386023-b.
Harvest of Cerebral Arteries
After 48 h of observation, the sham, SAH and SAH treated with SB386023-b-operated rats (see above SAH model) were anesthetized with CO2 and decapitated. The brains were quickly removed and chilled in ice-cold bicarbonate buffer solution (see composition below). Under a dissection microscope, the MCAs and the basilar artery (BA) were carefully dissected free from the brain and cleared of connective tissue. The vessel segments were immediately mounted in myographs for in vitro pharmacology or snap frozen at −80°C and examined by quantitative real-time PCR or immunohistochemistry.
In Vitro Pharmacology
For contractile experiments, a myograph was used for recording the isometric tension in isolated cerebral arteries (Hogestatt et al, 1983; Mulvany and Halpern, 1977). The vessels were cut into 1-mm long cylindrical segments and mounted on two 40-μm diameter stainless steel wires in a Myograph (Danish Myo Technology A/S, Aarhus, Denmark). One wire was connected to a force displacement transducer attached to an analog-digital converter unit (ADInstruments, Oxford, UK). The other wire was connected to a micrometer screw, allowing fine adjustments of vascular tone by varying the distance between the wires. Measurements were recorded on a computer by use of a PowerLab unit (ADInstruments). The segments were immersed in a temperature-controlled buffer solution (37°C) of the following composition (mmol/L): NaCl 119, NaHCO3 15, KCl 4.6, MgCl2 1.2, NaH2PO4 1.2, CaCl2 1.5 and glucose 5.5. The buffer was continuously aerated with oxygen enriched with 5% CO2, resulting in a pH of 7.4. The vessels were stretched to an initial resting tone of 2 mN and then allowed to stabilize at this tone for 1 h. The contractile capacity was determined by exposing the vessels to an isotonic solution containing 63.5 mmol/L of K+, obtained by partial change of NaCl for KCl in the above buffer. The contraction induced by K+ was used as reference for the contractile capacity (Hogestatt et al, 1983). Only vessels responding by contraction of at least 2.0 mN to potassium for BA and 0.8 mN to potassium for MCA were included in the study. The presence of the endothelium was checked by precontracting the vessel using 5-HT (10−6,5 mol/L) (Sigma, St Louis, MO, USA) and subsequently exposing the segments to carbachol (10−5 mol/L) (Sigma, St Louis, MO, USA). A relaxant response of the precontracted tension was considered indicative of a functional endothelium (Hansen-Schwartz et al, 2003a).
Concentration-response curves were obtained by cumulative application of 5-CT (Sigma, St Louis, MO, USA) in the concentration range 10−12 to 10−5 mol/L, S6c (Sigma, St Louis, MO, USA) in the concentration range 10−12 to 10−7 mol/L and ET-1 (AnaSpec, San Jose, CA, USA) in the concentration range 10−14 to 10−7mol/L. Endothelin-1 is investigated both without and with desensitized ETB receptors achieved by exposure to S6c.
Real-Time Polymerase Chain Reaction
To quantify mRNA for the ETA, ETB and 5-HT1B receptors, RT-PCR and real-time detection monitoring the PCR products were employed. Basilar artery or MCA was homogenized in 1 mL of the RNApro™ solution (Q-BIOgene, Irvine, CA, USA) by using a FastPrep® instrument (Q-BIOgene, CA, USA). The total RNA was extracted following a protocol obtained from the FastRNA® Pro kit supplier. Reverse transcription of total RNA to cDNA was performed using the Gene Amp RT kit (PE Applied Biosystems, Foster City, USA) in a Perkin-Elmer 2400 PCR machine at 42°C for 30 min. The real-time quantitative PCR was performed with the GeneAmp SYBR Green PCR kit (PE Applied Biosystems) in a Perkin-Elmer real-time PCR machine (GeneAmp 5700 sequence detection system). The system automatically monitors the binding of a fluorescent dye to double-strand DNA by real-time detection of the fluorescence during each cycle of PCR amplification. Specific primers for the rat ETA, ETB and 5-HT1B receptor and house-keeping gene elongation factor-1 (EF-1) were designed by using the Primer Express 2.0 software (PE Applied Biosystems) and synthesized by TAG Copenhagen A/S (Copenhagen, Denmark).
Receptor primers had the following sequences:
ETA receptor
forward: 5′-GTCGAGAGGTGGCAAAGACC-3′;
reverse: 5′-ACAGGGCGAAGATGACAACC-3′;
endothelin B receptor
forward: 5′-GAT ACG ACA ACT TCC GCT CCA-3′;
reverse: 5′-GTC CAC GAT GAG GAC AAT GAG-3′;
5-hydroxytryptamine 1B receptor
forward: 5′-TCC GGG TCT CCT GTG TAC GT-3′;
reverse: 5′-GGC GTC TGA GAC TCG CAC TT-3′.
The housekeeping gene EF-1 is used as a reference, since it is continuously expressed to a constant amount in cells.
The EF-1 primers were designed as follows:
Elongation factor-1
forward: 5′-GCA AGC CCA TGT GTG TTG AA-3′;
Elongation factor-1
reverse: 5′-TGA TGA CAC CCA CAG CAA CTG-3′.
The PCR reaction was performed in 50 μL volume and started at a temperature of 50°C for 2 min, 95°C for 10 min and the following 40 PCR cycles with 95°C for 15 secs and 60°C for 1 min. To verify that each primer pair only generated one PCR product at the expected size, the real-time PCR products were separated electrophoretically on a gel. In addition, a dissociation analysis was performed after each real-time PCR run. A blank control (without template) was used in all experiments. The expected size of the amplification products were for the ETA receptor 51 base pairs, ETB receptor 86 base pairs, 5-HT1B receptor 51 base pairs and EF-1 96 base pairs. To prove that the cDNA of EF-1 and the ET and 5-HT1B receptors was amplified with a similar efficacy during real-time PCR, a standard curve were made in which the CT values were plotted against cDNA concentration on the basis of the following equation: CT = (log(1 + E))−1 log(concentration) where CT is the number of PCR cycles performed in one sample at a specific point of time, and E is the amplification efficiency with an optimal value of one. Standard curves for ETA, ETB, 5-HT1B and EF-1 were performed by dilution of cDNA sample (1:10, 1:100 and 1:1000) (data not shown).
Immunohistochemistry
The MCA and BA were dissected out and then placed onto Tissue TEK (Gibco, Invitrogen A/S, Taastrup, Denmark) and frozen on dry ice. They were then sectioned into 8-μm-thick slices. The first antibodies used were rabbit antihuman ETB (IBL, Stockholm, Sweden, 16207), diluted 1:400, goat anti-mouse 5-HT1B (Santa Cruz Biotechnologies, Santa Cruz, CA, USA, sc-1461), diluted 1:100, mouse anti-rat CD31 (Serotec, Hamar, Norway, MCA1746), diluted 1:200, and mouse anti-rat smooth muscle actin (Serotec, MCA1905T), diluted 1:100. All dilutions were performed in PBS with 10% fetal calf serum. The second antibodies used were donkey anti-mouse Cy™5 conjugated (Jackson ImmunoResearch, Cambridgeshire UK, 715-175-150) 1:100, donkey antirabbit Cy™3 conjugated (Jackson-ImmunoResearch, 711-165-152) 1:100 in PBS with 10% fetal calf serum. The antibodies were detected at the appropriate wavelength in a confocal microscopy (Zeiss, Thornwood, NY, USA). As control only secondary antibodies were used. The images were analyzed using ImageJ (http://rsb.info.nih.gov/ij/). This was performed by measuring the fluorescence in 4 to 6 different areas in each artery and the mean value for each vessel used (n = 3 in each group).
Calculations and Statistics
Data are expressed as mean ± standard error of the mean (s.e.m.), and n refers to the number of rats. Statistical analyses were performed using the nonparametric Mann-Whitney test, where P < 0.05 was considered significant.
In vitro pharmacology: Contractile responses in each segment are expressed as percentage of the 63.5 mmol/L K+-induced contraction. Emax value represents the maximum contractile response elicited by an agonist and pEC50 the negative logarithm of the drug concentration that elicited half the maximum response. For biphasic responses, Emax(1) and pEC50(1) describe the high-affinity phase, and Emax(2) and pEC50(2) describe the low-affinity phase.
Real-Time Polymerase Chain Reaction
Polymerase chain reaction experiments were performed on BA and MCA from sham, SAH and SAH treated with ERK1/2 inhibitor-operated rats. Data were analyzed with the comparative cycle threshold (CT) method (Hansen-Schwartz et al, 2003b). The CT values of EF-1 mRNA were used as a reference to quantify the relative amounts of ETA, ETB and 5-HT1B mRNA. The relative amount of mRNA was calculated with the CT values of ETA, ETB and 5-HT1B receptor mRNA in relation to the CT values of EF-1 mRNA in the sample by the formula
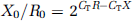
where Xo is the original amount of target mRNA.
Ro is the original amount of EF-1 mRNA, CTR is the CT value for EF-1 and CTX is the CT value for the target.
Results
Subarachnoid Hemorrhage model
The mortality rate of the animal model of SAH was 3%. In all operated rats, MABP (99 ± 6 mm Hg), partial pCO2 (39 ± 2 mm Hg), partial pO2 (102 ± 6 mmHg), hematocrit (43 ± 3 mm Hg) values and temperature were within acceptable limits during the operation. There was no difference between the three rat groups, sham, SAH and SAH, treated with SB386023-b. As a result of injecting the blood, the cortical CBF dropped over both hemispheres to 17% ± 3% of the resting flow and the ICP increased from 11 ± 1 to 123 ± 9 mm Hg. The laser Doppler blood flow and the ICP returned to the basal value within 1 h of postoperative monitoring. There was no significant difference between the two groups SAH and SAH treated with SB386023-b (Figure 1).
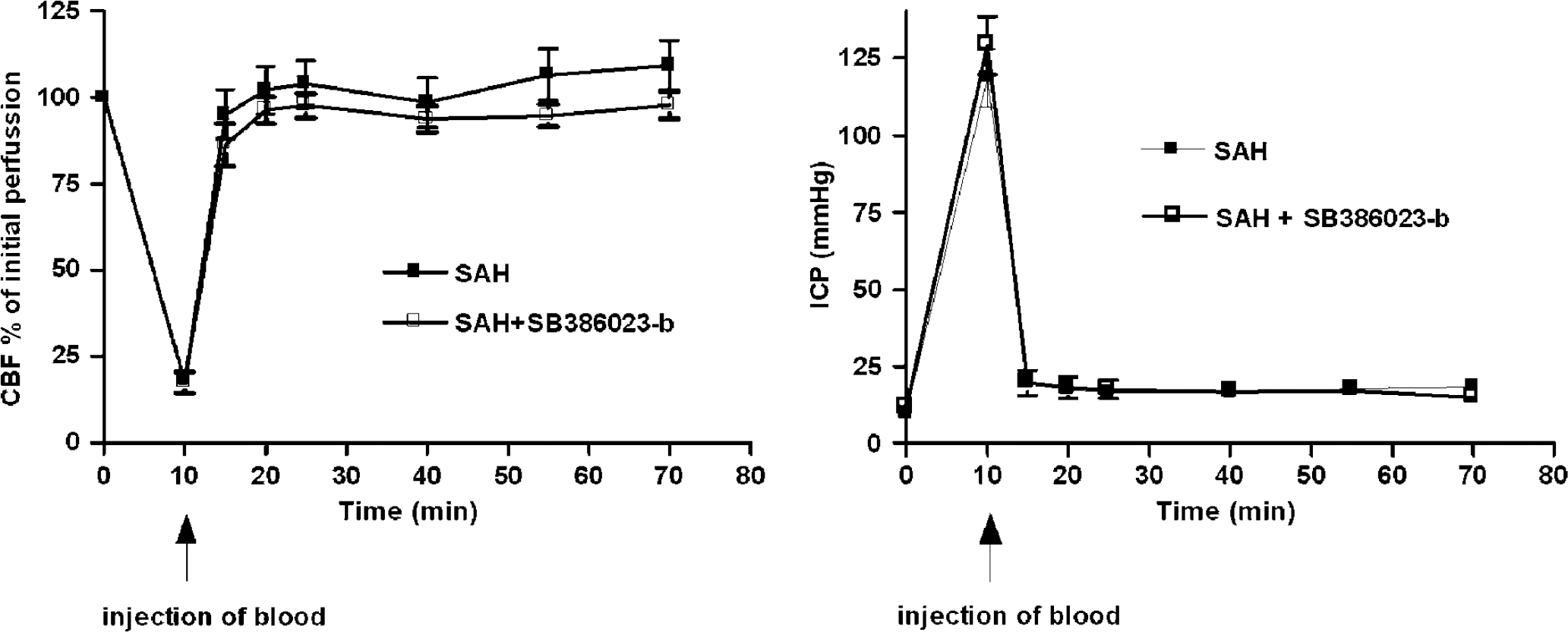
Illustration of the cortical blood flow measured with laser Dopplerflowmetry and ICP at the time of injection of blood (SAH) and 1 h after SAH.
Regional Cerebral Blood Flow (rCBF)
There was a significant global decrease in CBF in the SAH group (n = 5) compared with the control group (n = 5) from 134 ± 8 to 67 ± 4 mL/min 100 g as analyzed at 48 h. Treatment with SB386023-b in SAH (n = 5) inhibited this marked reduction in CBF compared with the SAH group (Figure 2). In 16 of the 18 brain regions examined, the SAH animals showed a marked reduction in the rCBF compared with the control-operated rats (Table 1). Treatment with SB386023-b prevented this reduction in rCBF and there was no difference as compared with the control group.
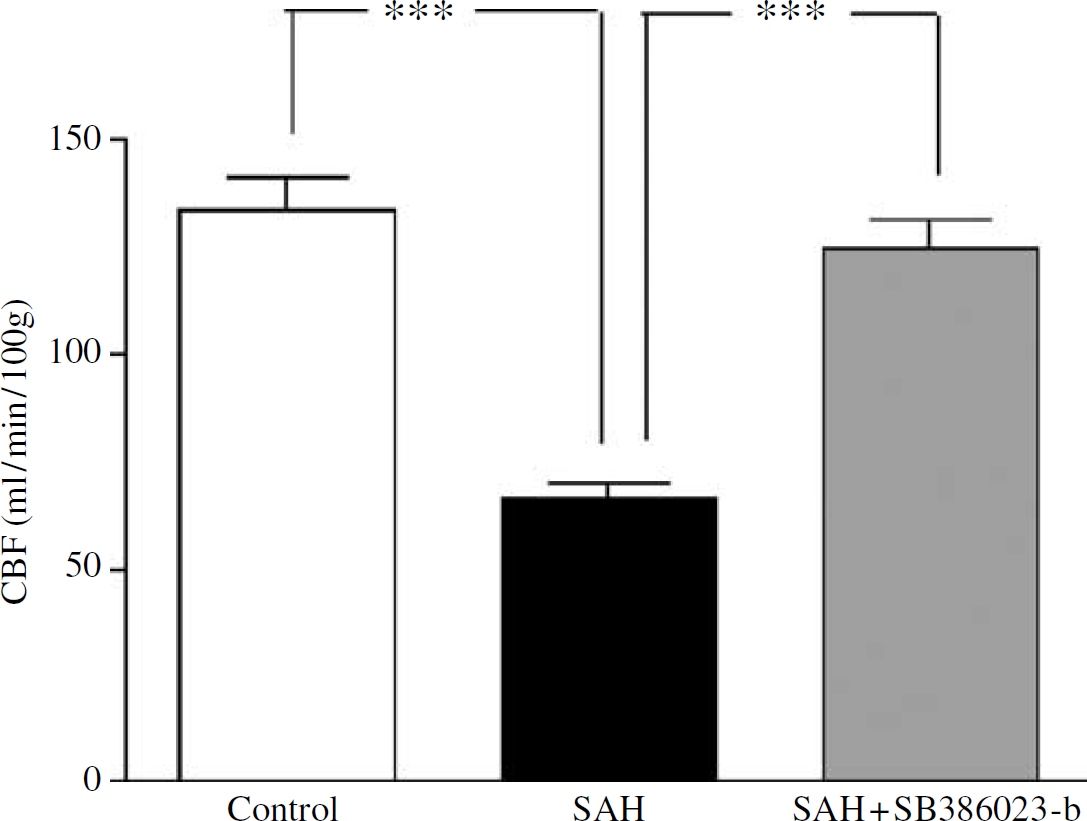
Effect of treatment with the ERK1/2 inhibitor SB386023-b on the global CBF after induced SAH (n = 5) in rats. There is a reduction in the global CBF in the SAH compared with the control rats (n = 5). Treatment with SB386023-b (n = 5) inhibited this reduction in CBF. Data were obtained by an autoradiographic method and data are expressed as mean ± s.e.m. values, ***P < 0.001.
In Vitro Pharmacology
Potassium (K+, 63.5 mmol/L)-evoked contraction of the smooth muscle cells was used as an internal control. K+-induced contractions did not differ significantly between the vessels from the three groups sham, SAH and SAH treated with SB386023-b (Table 2). Emax and pEC50 values for respective group are presented in Table 2.
Contractile effects of ET-1 and 5-CT in MCA and basilar arteries
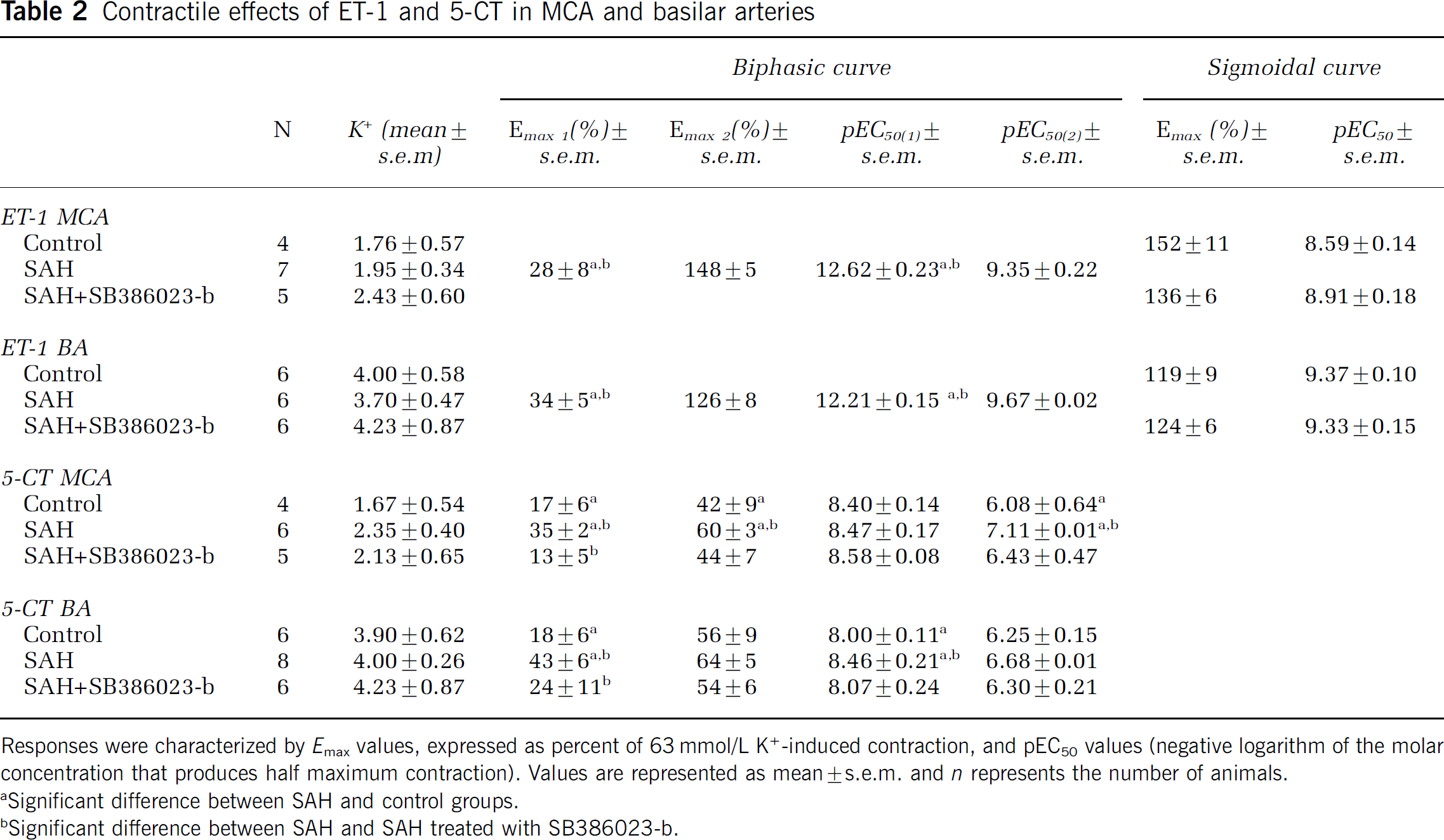
Responses were characterized by Emax values, expressed as percent of 63 mmol/L K+-induced contraction, and pEC50 values (negative logarithm of the molar concentration that produces half maximum contraction). Values are represented as mean ± s.e.m. and n represents the number of animals.
Significant difference between SAH and control groups.
Significant difference between SAH and SAH treated with SB386023-b.
Contractile response to endothelin-1: The MCA and BA from SAH (n = 9) showed a leftward shift of the curve to ET-1. This indicates an enhanced contractile response to ET-1 as compared with the sham-operated rats (n = 6), where a normal sigmoidal curve was obtained (Figures 3A and 3B). Treatment in vivo with SB386023-b of SAH rats (n = 6) showed contractile responses to ET-1 that were similar to control and significantly different compared with the SAH rats (Figures 3A and 3B).
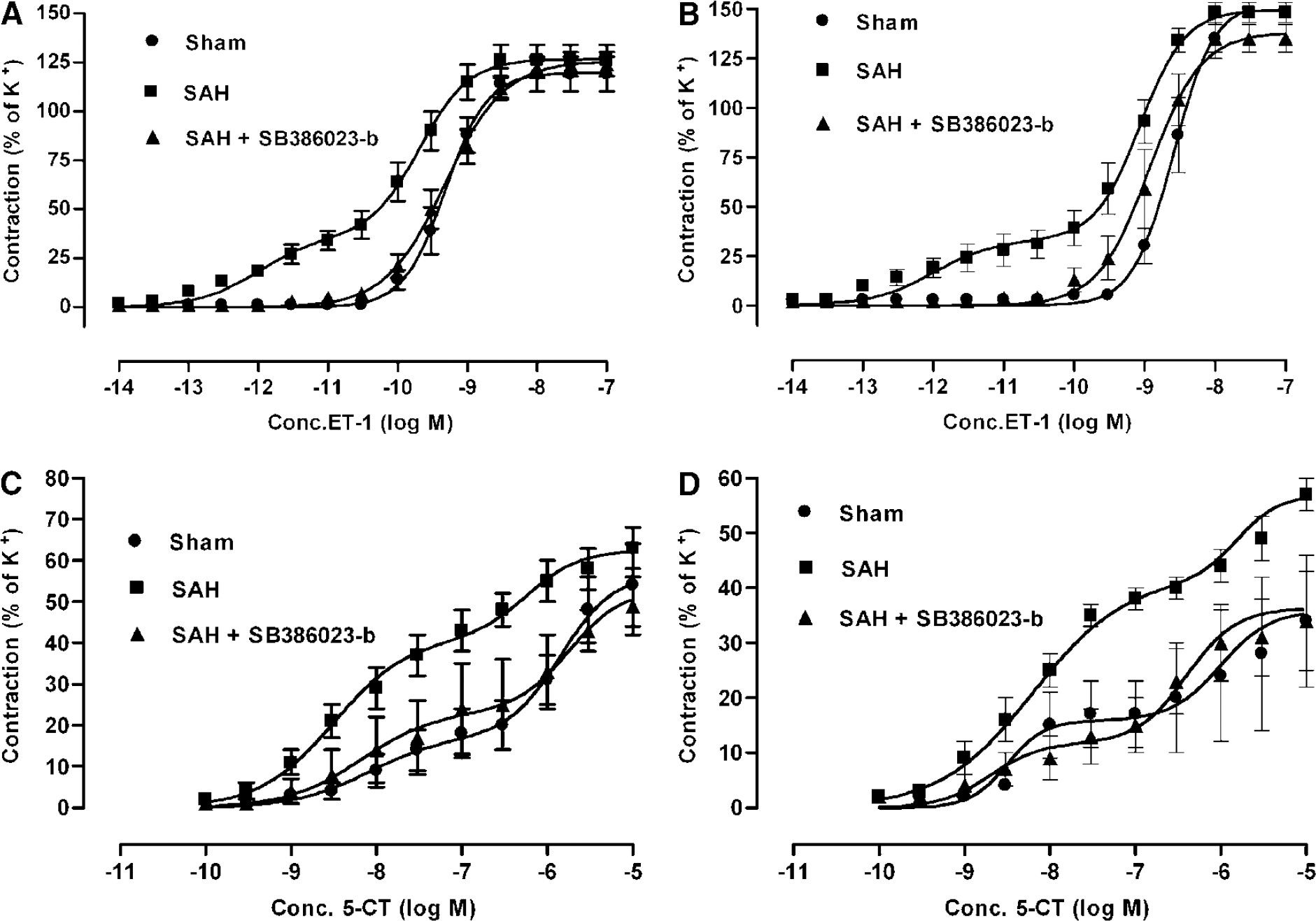
Concentration–response curves elicited by cumulative application of ET-1 and 5-CT in rat cerebral arteries. (
Previously, we have shown that this enhanced effect of ET-1 in SAH is ETB receptor dependent, since it can be blocked by the selective ETB receptor antagonist IRL1038 and is absent after S6c desensitization (Hansen-Schwartz et al, 2003b). In the present study, application of S6c alone did not give rise to any contraction either in SAH, SAH treated with SB386023-b or in sham-operated rats (data not shown). Application of S6c 60 mins before ET-1 application resulted in attenuation of the concentration-dependent contraction to ET-1. Desensitization of the ETB receptor by use of the S6c has previously been shown (Lodge et al, 1995).
Contractile response to 5-carboxamidotryptamine: 5-Carboxamidotryptamine gave rise to a biphasic concentration-dependent contraction both in MCA and BA, indicating the presence of two receptors 5-HT1B and 5-HT2A in a manner previously characterized in detail (Hoel et al, 2001). The contractile response to 5-CT was upregulated in the SAH-induced rats (n = 9) compared with the sham-operated rats (n = 6) (Figures 3C and 3D). Treatment in vivo with SB386023-b (n = 6) abolished this upregulation compared with SAH alone, making the curve similar to the one obtained in sham-operated rats (Figures 3C and 3D).
Real-Time Polymerase Chain Reaction
The standard curves for each primer pair had almost similar slopes, showing that EF-1, ETA, ETB and 5-HT1B cDNA were amplified with the same efficiency (data not shown). In each PCR experiment, a no template control was included, and there were no signs of contaminating nucleic acids in those samples. Electrophoresis verified only one product for each primer pair at the expected size (data not shown).
Since the results from the two types of brain arteries were identical, the groups of MCA and BA (n = 7 to 10) were added together for the statistical analysis. There was a significant upregulation of ETB receptor mRNA relative to the amount of EF-1 in the SAH compared with the sham-operated rats (P < 0.05). Treatment in vivo with SB386023-b prevented this upregulation (P < 0.05) and even showed a lower level of expression as compared with control (Figure 4A). The level of ETA receptor mRNA remained unchanged in SAH compared with the sham-operated rats, while treatment with SB386023-b had a tendency (P = 0.2) to decrease the level compared with SAH (data not shown). There was a significant upregulation of 5-HT1B receptor mRNA levels in SAH rats compared with sham-operated rats. Treatment with SB386023-b resulted in down-regulation of 5-HT1B receptor mRNA, as compared with SAH (Figure 4B).
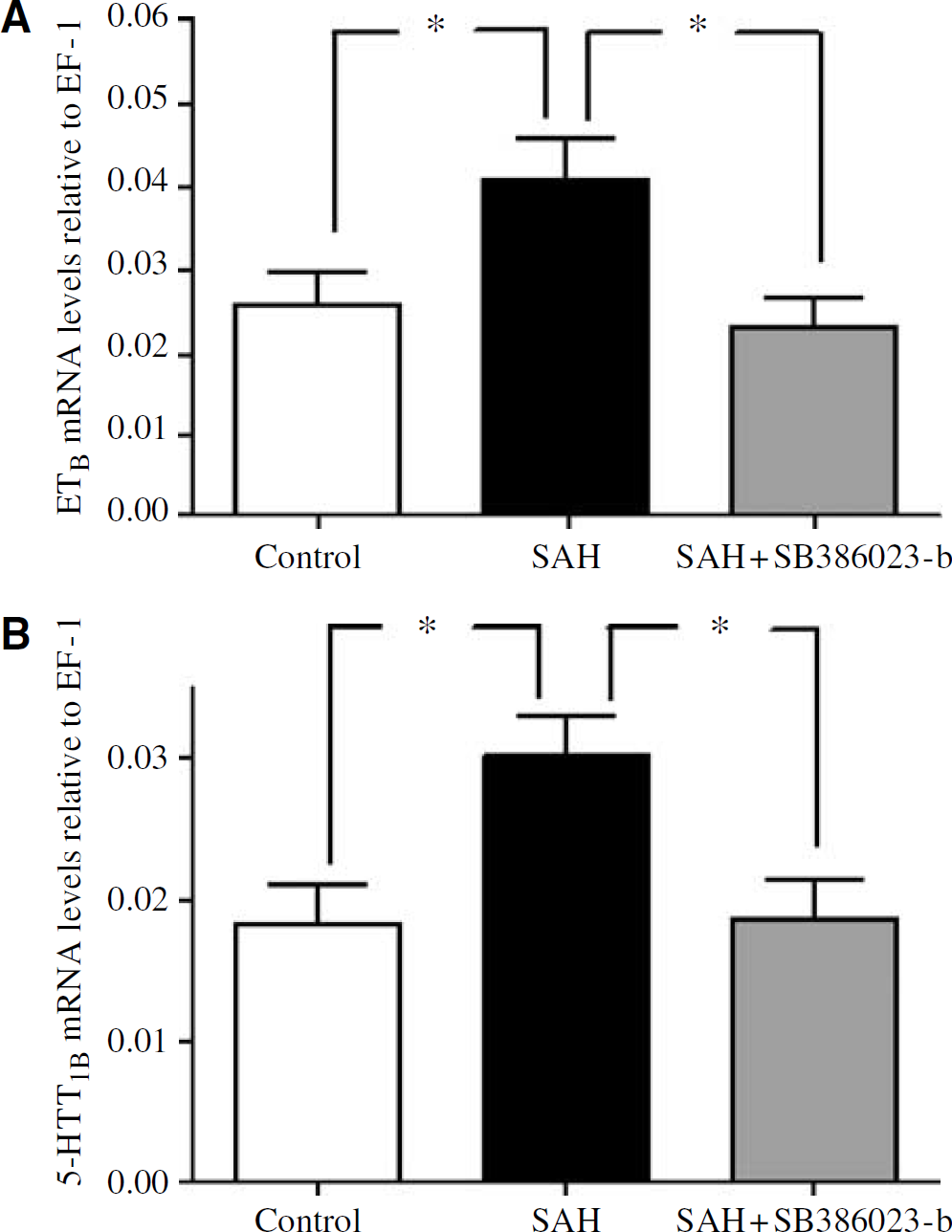
Effect of treatment with ERK1/2 inhibitor SB386023-b in cerebral arteries on the mRNA levels of 5-HT1B and ETB receptors after experimental induced SAH in rats. (
Immunohistochemistry
Selective antibodies towards the ETB and 5-HT1B receptors visualized the smooth muscle cell localization of these receptors using confocal microscopy (Figure 5). The ETB receptor protein expression was increased in SAH (166% ± 19%) as compared with sham (100% ± 10%) (P < 0.05). Similar results were seen with the 5-HT1B receptor protein level, where the expression in SAH was increased from 131% ± 18% compared with sham 100% ± 10% (P < 0.05). Treatment with the ERK1/2 inhibitor SB1386023-b prevented the upregulation of ETB (120% ± 13%) and 5-HT1B (119% ± 30%) receptor protein levels in the smooth muscle cell layer as compared with the SAH (n = 3 in each of the groups).
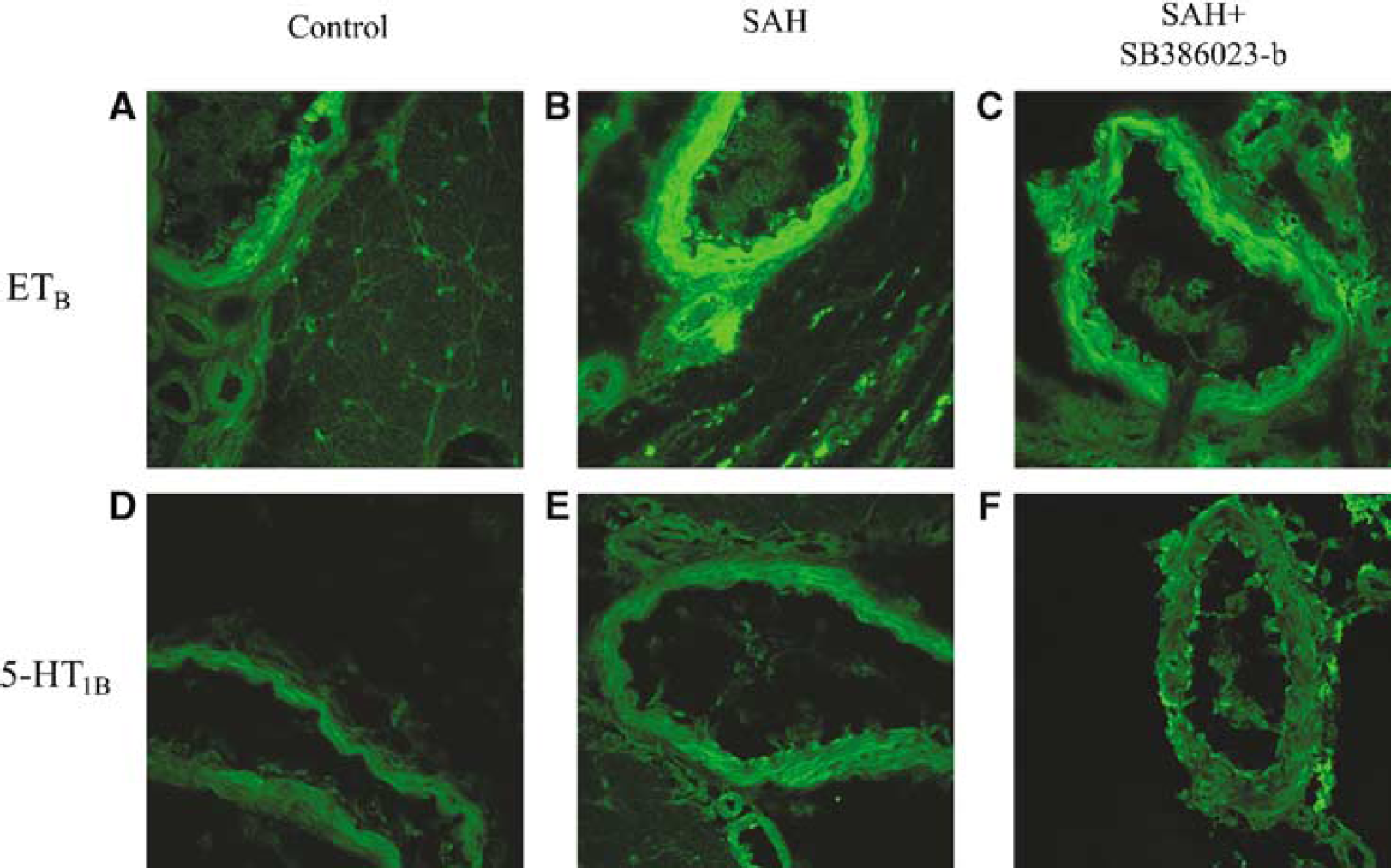
Sections from the BA showing ETB and 5-HT1B immunoreactivity in the smooth muscle cell layer. (
Discussion
This is the first study to clearly show that inhibition of ERKl/2 prevents the marked reduction in global and rCBF that is seen in conjunction with an experimental SAH. The prevention of the CBF reduction may in the clinical settings lead to a better outcome for the SAH patients, since the reduction correlates with worsening of the clinical prognosis in cerebral ischemia (Heilbrun et al, 1972; Kågström et al, 1966). The contractile responses to ET-1 and 5-CT were markedly increased in cerebral arteries after SAH. The enhanced functional response to ET-1 was ETB receptor dependent. Confocal immunohistochemistry showed that SAH induces a selective ETB receptor upregulation on the smooth muscle cells and this supports the notice of an upregulation of contractile ETB receptors. However, there was no difference between sham-operated rats and SAH rats treated in vivo with SB386023-b. This suggests that inhibition of the ERKl/2 pathway has a pivotal role in the enhanced vasoconstriction and the associated reduced CBF observed after SAH. Our main hypothesis, which is supported by the present data, suggests that SAH triggers an increase in transcription of genes coding for G-protein-coupled receptors in cerebral arteries, which translates into an increased number of contractile smooth muscle receptors. In this context, the MAPK ERKl/2 appears to have a key role. Previous reports have implicated that MAPK plays a role in CVS (Tibbs et al, 2000; Zhang et al, 2001). Cytokines and free radicals are associated with the pathogenesis of CVS after SAH (Shishido et al, 1994) and part of these effects might be related to activation of MAPKs. Protein kinase C is known to activate the MEK/ERK1/2 pathway at several levels (Schonwasser et al, 1998). This correlates with our findings, which have shown that PKC is involved in CVS and upregulation of ETB and 5-HT1B receptors (Beg et al, unpublished data).
Selective ETB receptor stimulation with S6c had no contractile effects per se. There is a point of concern to this apparent discrepancy between the observed increase in mRNA and protein levels for the ETB receptor and the apparent lack of a direct functional response to S6c alone. However, our results have clearly shown that blocking the ETB receptor through desensitization with S6c attenuates the first upregulated phase, while it does not affect the second phase, indicating the presence of a functional ETB receptor. This is in agreement with a previous SAH study, where the ETB receptor is selectively blocked by an ETB receptor antagonist IRL1038, and results similar to ours were obtained (Hansen-Schwartz et al, 2003b). We do not possess exact data to suggest the nature of the relationship between the two receptors. A possible mechanism can be a dimerization mechanism between ETA and ETB receptors that operates to form a receptor heterodimer in the recognition of ET-1, a typical bivalent ligand (Harada et al, 2002).
The present results are the first in vivo data obtained using quantitative real-time PCR analysis, and they are in agreement with the findings of downregulated ETB and 5-HT1B receptor function after treatment with the ERKl/2 inhibitor SB386023-b. This points to the involvement of ERK1/2 upstream from the transcriptional level in the signal transduction pathway leading to the appearance of contractile ETB and 5-HT1B receptors. In organ culture, we have previously examined the involvement of ERK1/2 and the stress-activated protein kinase pathways in the upregulation of ETB receptors. Extracellular signal-regulated kinase 1/2 was the only one that was activated in cerebral (Henriksson et al, 2004) and mesenteric arteries after organ culture (Uddman et al, 2003) and shown to be involved in the ETB receptor upregulation. Inhibition of the ERK1/2 with the inhibitor SB386023-b in cerebral arteries decreased the functional response of ETB receptor upregulation, ETB mRNA and ETB protein levels (Henriksson et al, 2004). In the present study, we have shown with confocal immunohistochemistry that ETB receptor antibodies bind specifically on the smooth muscle cells of cerebral arteries and that SAH upregulates this signal. This increase in expression of ETB receptors is normalized by treatment with SB386023-b. We have studied the early phase of ETB receptor upregulation in mesenteric arteries after organ culture in detail (Moller et al, 2002). There was a significant increase in receptor mRNA already at 3 h, which reached a plateau at 24 h. The functional responses appeared later and continued beyond 48 h. Transcriptional or translational inhibitors abolished both responses, indicating an upregulation on a transcriptional level. The same phenomenon occurs in rat cerebral arteries (Henriksson et al, 2003). Consequently, such an increase in cerebrovascular receptor expression may lead to an increase in vascular tone after an SAH, which may in turn cause late cerebral vasoconstriction and reduced CBF, with subsequent development of cerebral ischemia and neuronal death.
Another point of interest is that SB386023-b had no effect at all on the contractility induced by ET-1 or 5-CT when it was administered before the induction of SAH (data not shown). It was also observed that there was no difference in the cortical CBF in the acute phase at induction of SAH between the two groups, SAH and SAH treated with SB386023-b. These results indicate that the positive effect that we have observed after treatment with SB386023-b arises when it is administered after induction of SAH, which is clinically relevant. However, further studies need to be performed to investigate the therapeutic window of this approach.
In conclusion, this study has shown that ETB and 5-HT1B receptors are upregulated in SAH and that this upregulation is prevented by administration of the ERK1/2 inhibitor SB386023-b. SB386023-b also prevents the reduction in regional and global CBF seen after SAH. These findings suggest that the MAPK ERK1/2 pathway is a novel therapeutic target in treatment of CVS and ischemia after SAH.
Footnotes
Acknowledgements
The authors are grateful for the scientific help from Professor NA Svendgaard, Professor N Diemer and the technical assistance of M Nielsen.