Abstract
Bone marrow-derived cells (BMDCs) contribute to revascularization after ischemia. However, the mechanisms by which BMDCs support vessel remodeling after cerebral ischemia are not clear. Using mouse chimeras that express enhanced green fluorescent protein in reconstituted bone marrow, we investigated the role of BMDCs in revascularization and brain repair after middle cerebral artery occlusion of murine brain. After ischemia, two populations of BMDCs were observed, one in the brain parenchyma and another associated with the vasculature. The number of BMDCs that infiltrated the brain parenchyma peaked at 7 days and persisted through 14 days, the last time point observed. The majority of BMDCs were characterized as microglia, based on cell-type-specific marker expression. We observed a robust angiogenic response after cerebral ischemia. Bone marrow-derived cells associated with remodeling blood vessels were negative for endothelial markers, but were surrounded by basal lamina and expressed desmin and vimentin, identifying these cells as pericytes. Quantification of BMDCs that expressed desmin revealed increasing desmin expression with time. Perivascular associated BMDCs that expressed desmin were immunoreactive for the angiogenic factors vascular endothelial growth factor and transforming growth factor-β. These findings suggest that pericytes are recruited from the periphery and are involved in blood vessel stabilization during ischemiainduced angiogenesis.
Introduction
Bone marrow-derived cells (BMDCs) contribute to revascularization and tissue regeneration in a wide range of ischemic pathologies (Rafii et al, 2003), including myocardial infarction (Hamano et al, 2001; Kocher et al, 2001; Orlic et al, 2001), limb ischemia (Iba et al, 2002; Ikenaga et al, 2001; Taguchi et al, 2003), retinal degeneration (Grant et al, 2002; Otani et al, 2002) and stroke (Beck et al, 2003; Chen et al, 2003; Hess et al, 2002; Taguchi et al, 2004; Zhang et al, 2002). Bone marrow is a rich source of stem and progenitor cells that can mobilize to ischemic sites (Rafii et al, 2003). After cerebral ischemia, BMDCs rapidly infiltrate the brain, where they have been reported to give rise primarily to microglia (Beck et al, 2003; Hess et al, 2004; Priller et al, 2001), or endothelial cells (Hess et al, 2002; Zhang et al, 2002) and a limited number of cells that express astrocytic (Eglitis et al, 1999) and neuronal markers (Cogle et al, 2004; Hess et al, 2002, 2004). The recruitment of BMDCs after stroke may represent an attempt at endogenous self-repair. Bolstering the selective recruitment of bone marrow-derived stem and progenitor populations may lead to therapeutic opportunities to stimulate revascularization and repair, or provide avenues for therapeutic gene delivery.
The formation of new blood vessels and vascular remodeling occurs after cerebral ischemia, and is likely to be important for minimizing cell death and restoring blood flow to damaged areas (Hayashi et al, 2003; Krupinski et al, 1994; Wei et al, 2001). Recent evidence indicates that ischemiainduced angiogenesis in the adult brain involves the proliferation of preexisting endothelial cells, with a small contribution by circulating marrow-derived endothelial progenitor cells (Beck et al, 2003; Zhang et al, 2002). Intravenous administration of enriched populations of endothelial progenitors (Taguchi et al, 2004) or bone marrow stromal cells (Chen et al, 2003) enhances both angiogenesis and functional recovery after stroke. The angiogenic effect of these cells appears to be mediated primarily via paracrine cytokine and growth factor signaling, rather than via widespread incorporation of donor progenitor cells into the growing vasculature. The response to intravenous administration of CD34+ cells stimulates both intrinsic angiogenic and neurogenic responses, which appear to be tightly linked (Peterson, 2004; Taguchi et al, 2003).
Angiogenesis involves complex signaling mechanisms, with a high degree of spatial and temporal coordination among multiple cell types (Jain and Duda, 2003). Successful revascularization of ischemic brain must not only involve the formation of new vessels but also their stabilization and maturation through recruitment of support cells and reconstitution of blood–brain barrier properties. Pericytes are support cells of the microvasculature critically involved in initiation (Balabanov et al, 1996; Diaz-Flores et al, 1994) and sprouting (Nehls et al, 1992; Ozerdem and Stallcup, 2003). In addition, pericytes are important in the stabilization of nascent capillaries and prevention of vascular regression (Allt and Lawrenson, 2000; Gerhardt and Betsholtz, 2003). During angiogenesis, pericytes are recruited to growing microvessels, where they directly inhibit endothelial cell proliferation and migration through contact and secretion of growth-inhibitory factors (Antonelli-Orlidge et al, 1989; Diaz-Flores et al, 1994; Hirschi and D'Amore, 1997; Orlidge and D'Amore, 1987). Pericyte loss results in endothelial cell apoptosis and destabilization of the microvasculature. Global loss of pericyte function results in perinatal lethality due to severe microvascular leakage and hemorrhage (Hellstrom et al, 1999; Lindahl et al, 1997). In brain capillary endothelial cells, pericyte-derived angiopoietin-1 induces the expression of tight junction proteins required for the formation and maintenance of the blood–brain barrier (Hori et al, 2004). Thus, pericyte recruitment is likely to play an important role in vascular remodeling after stroke, although the mechanisms underlying pericyte recruitment and function in the ischemic brain have not been extensively studied.
In the present study, we utilized green fluorescent protein (GFP) bone marrow chimeric mice, coupled with immunocytochemistry and high-resolution confocal microscopy, to investigate the migration and phenotypic differentiation of BMDCs after mild focal ischemia. Similar to previous reports, we observed rapid and massive influx of BMDCs into the brain parenchyma, where they gave rise primarily to resident microglial cells. However, we also observed recruitment of BMDCs in association with the neovasculature, where they gave rise to pericytes expressing the angiogenic factors, vascular endothelial growth factor (VEGF) and transforming growth factor (TGF)-β. These observations add to the understanding of how BMDCs contribute to angiogenic mechanisms in the ischemic brain, and might have important implications for therapeutic targeting of angiogenesis after stroke.
Materials and methods
Bone Marrow Transplantation
Donor bone marrow was harvested from 6- to 10-week-old C57BL/6-TgN (ACTβEGFP) mice that express GFP under a β-actin transcriptional promoter in all tissues except red blood cells and hair (Okabe et al, 1997). Mice were overdosed with halothane and killed by decapitation to remove tibias and femurs. Bone marrow was flushed out of the tibias and femurs by using 5 mL serum-free Dulbecco's modified Eagle medium (DMEM) and a 26-gauge needle attached to syringe. The cells were dispersed by gentle aspiration, rinsed by centrifugation at 2000 rev/min and resuspended in DMEM at a concentration of 3 × 107 cells/mL.
Recipient C57BL/6J mice (The Jackson Laboratory, Bar Harbor, ME, USA) between 6 and 8 weeks of age received 9.0 Gy irradiation from a dual 127Cs source within a gamma irradiator, and immediately received 6 × 106 bone marrow cells via tail vein injection. All recipient mice received 10 mg/L polymyxin and 100 mg/L neomycin via the drinking water for 14 days after transplantation to prevent infection.
Middle Cerebral Artery Occlusion (MCAO)
At 6 weeks after bone marrow reconstitution, recipient mice were subjected to mild transient focal ischemia by right MCAO using the intraluminal thread method (n = 35). Anesthesia was induced with 1.5% isoflurane and maintained with 1.0% isoflurane in 1-L O2, using a vaporizer (Summit Medical Equipment, Bend, OR, USA). The right common carotid artery (CCA) was exposed by a midline incision in the neck and the internal and external CAs were isolated. The external carotid artery was ligated with 6-0 silk suture. A 6-0 rounded tip nylon suture was introduced into the CCA 10 to 11 mm proximal to the bifurcation of the CCA. The suture was advanced through the internal carotid artery to occlude the middle cerebral artery (MCA). Successful MCAO resulted in an 80% decrease of blood flow to the right cerebral hemisphere, as assessed by laser Doppler analysis. After 30 mins, the suture was withdrawn and the CCA ligated with a silk suture above the point of suture insertion. Mice were allowed to undergo reperfusion via the Circle of Willis for 1, 2, 3, 7 or 14 days (n = 7/time point) before being killed for analysis. Sham animals (n = 3) were anesthetized and a suture introduced to the CCA, but the suture was not advanced. A second control group comprised GFP bone marrow chimeras that did not receive any subsequent treatment (no surgery controls; n = 3). Mice were subcutaneously injected with 0.05 mg/kg Bupronex for analgesia before surgery and 1 h after surgery.
Histology
Mice were overdosed with sodium pentobarbital administered intraperitoneally (150 mg/kg) and transcardially perfused with approximately 10 mL of phosphate-buffered saline (PBS) containing 0.1% procaine and 2 U/mL heparin, followed by approximately 50 mL of 2% paraformaldehyde containing 0.075 mol/L lysine and 0.01 mol/L sodium periodate. Brains were post-fixed overnight, cryoprotected in 30% sucrose and sectioned at 16 μm thickness in the coronal plane using a cryostat. For histology, every 12th section was obtained and 20 sections at 160 μm consecutive intervals were collected for each animal through striatum.
Fluorojade and TUNEL Staining
Fluorojade staining was used to detect degenerating neurons (Schmued et al, 1997). Histological sections were immersed in 100% ethanol for 3 mins, followed by 70% ethanol and distilled water for 1 min. The slides were transferred to a 0.06% solution of potassium permanganate for 15 mins, followed by 30 mins in a solution containing 0.001% fluorojade (Histo-chem Inc., Jefferson, AR, USA) in 0.1% acetic acid. Slides were rinsed in distilled water, air dried and coverslipped using Permount (Fischer Scientific, Fairlawn, NJ, USA) mounting medium.
Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) staining was performed on histological sections as per the manufacturer's protocol (NeuroTACs II, Trevigen, Inc., Gaithersburg, MD, USA), using a fluorescein isothiocyanate (FITC)-conjugated streptavidin as a fluorophore. Sections in which the deoxynucleotidyl-transferase was omitted served as negative controls. To distinguish the phenotype of TUNEL-positive cells, some sections were further processed for NeuN or GFAP immunohistochemistry as described below.
Immunohistochemistry
Nonspecific staining was blocked by preincubation of histological sections in PBS containing 1.0% Tween-20 and 10% normal goat or donkey serum and 1% bovine serum albumin. Primary antibodies and dilutions were as follows: mouse anti-NeuN (1:100; Chemicon International, Temucula, CA, USA) and mouse anti-HuC/HuD (1:10; Molecular Probes, Eugene, OR, USA) for neurons, rabbit anti-GFAP (1:500; Accurate Chemical, Westbury, NY, USA) for astrocytes, rat anti-CD11b (1:50; Accurate Chemical) and Iba-1 (1:500; Wako Chemicals, Richmond VA, USA) for microglia/macrophages, rabbit anti-laminin (1:500; Sigma, St Louis, MO, USA) for basement membrane, rabbit anti-von Willebrand Factor (1:500; Dako Cytomation, Carpenteria, CA, USA) and mouse anti-CD31 (1:200; Chemicon) for endothelial cells, rabbit anti-desmin (1:500; Chemicon), mouse anti-vimentin (1:400; Dako Cytomation) for pericytes, rabbit anti-Ki67 (1:500; Dako) for proliferating cells, rabbit anti-VEGF (1:200; Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA), mouse anti-TGF-β (1:50; R&D Systems, Minneapolis, MN, USA). Appropriate secondary antibodies conjugated to Cy3 (1:250; Jackson ImmunoResearch, West Grove, PA, USA) were used for detection. To Pro-3, iodide (1:500 Molecular Probes) was used to label the nuclei. Omission of primary antibody or incubation with pre-immune IgG served as negative controls.
Image Analysis and Cell Counts
The numbers of both GFP+ and TUNEL+ cells were determined by digitally capturing 50 random fields from 5 sections within the ischemic and contralateral striatum using an Olympus fluorescent microscope with an ×40 objective, attached to a SPOT digital camera. BIOQUANT Image Analysis Software (R&M Biometrics, Inc.) was used to quantitate the number of positive cells and area per field. The number of cells/mm2 was calculated by dividing the number of cells counted within the field by the area of the field. Similarly, the number of laminin-reactive blood vessels was estimated by capturing 5 fields using an ×40 objective throughout the ischemic and contralateral striatum. For colocalization of GFP with various histological markers, 100 GFP+ cells per animal were sampled for each cell type-specific marker, using a Zeiss LSM510 confocal microscope with an ×100 objective. Cells that appeared to express both GFP and a cell type-specific marker were subjected to optical sectioning through the z-plane at 1-μm intervals. Because neural differentiation of BMDCs is thought to be rare, to look for colocalization with neural markers, all GFAP-, NeuN- and Hu-positive cells analyzed in the ischemic striatum were evaluated for GFP expression using ×20 magnification before optical sectioning through the z-plane of cells that appeared colocalized. The number of blood vessels that were associated with bone marrow-derived pericytes over time was estimated by visualizing 50 random blood vessels stained immunohistochemically against laminin within the ischemic and contralateral striatum and scoring each blood vessel positive if a GFP+ cell was contained within the basal lamina of that blood vessel.
Statistical Evaluations
Statistical evaluation of cell count comparisons was performed using Prism 3.0 (Graphpad Software Inc., San Diego, CA, USA). For two-group comparisons, Student's t-tests were performed. For three-group comparisons, one-way ANOVAs were applied to detect differences among the groups, followed by a Tukey multiple pairwise comparison test. P-values of <0.05 were considered significant. All data are presented as means ± s.e.m.
Results
Time course of Bone Marrow-Derived Cells Infiltration After Focal Ischemia
Green fluorescent protein bone marrow chimeras received transient occlusion of the right MCA for 30 mins, followed by reperfusion for 1, 2, 3, 7 or 14 days (n = 7 mice per group). As previously described (Endres et al, 1998), mild focal ischemia for 30 mins resulted in delayed neuronal cell death, which appeared to peak by 72 h of reperfusion, as assessed histologically utilizing fluorojade staining for degenerating neurons and TUNEL staining (Figure 1). TUNEL-positive cells were positive for the neuronal marker NeuN (Figure 1B) and very rarely were TUNEL-positive cells (1 to 2 cells/section) colocalized with the astrocyte marker GFAP (data not shown). Terminal deoxynucleotidyl transferase dUTP nick end labelling-positive cells were not observed after 7 or 14 days of reperfusion, although light fluorojade staining persisted (data not shown). Approximately 75% to 80% of mice exhibited an ischemic lesion after surgery. Mice that exhibit no degeneration appear similar to sham surgery, no surgery mice and contralateral striatum with very few peripheral cells within the brain parenchyma. Only mice that showed positive indices for neuronal death via fluorojade or TUNEL were used for further analysis (n = 4 to 7/time point).
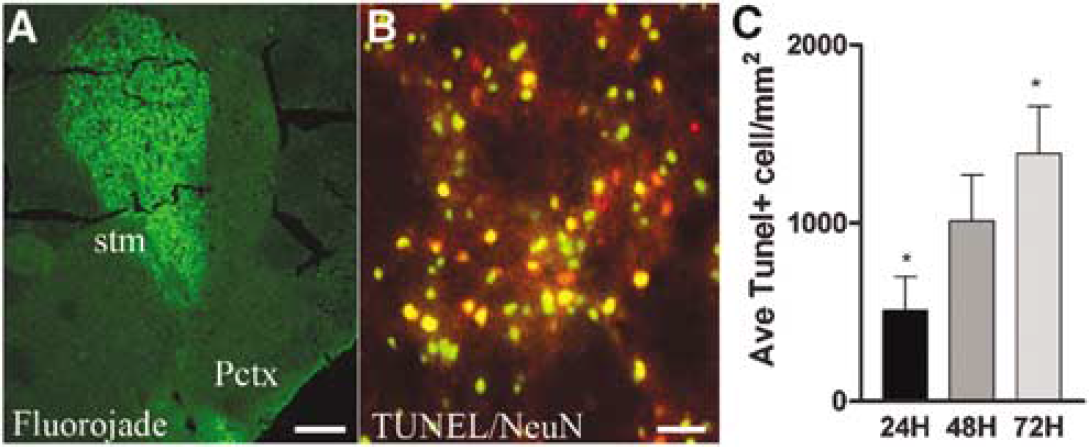
Cell death after ischemia. (
In sham-operated mice, occasional BMDCs were distributed throughout the brain parenchyma or near blood vessels. Transient focal ischemia evoked robust infiltration of GFP+ BMDC into the striatal parenchymal and perivascular regions (Figure 2). The density of GFP+ cells was significantly increased in the ischemic striatum by 2 days after occlusion (129.8 ± 41.79 cells/mm2) compared with nonischemic striatum (10.13 ± 3.693 cells/mm2). After 7 days of reperfusion, the density of GFP+ cells was approximately 85-fold greater in ischemic versus nonischemic striatum (700.25 ± 146.10 versus 8.925 ± 2.225 cells/mm2, respectively), and remained elevated out to 14 days, which was the longest time point studied. Green fluorescent protein-positive BMDCs that were associated with parenchyma versus perivascular compartments displayed distinct characteristics and were therefore analyzed separately for phenotype and proliferation, as described below.
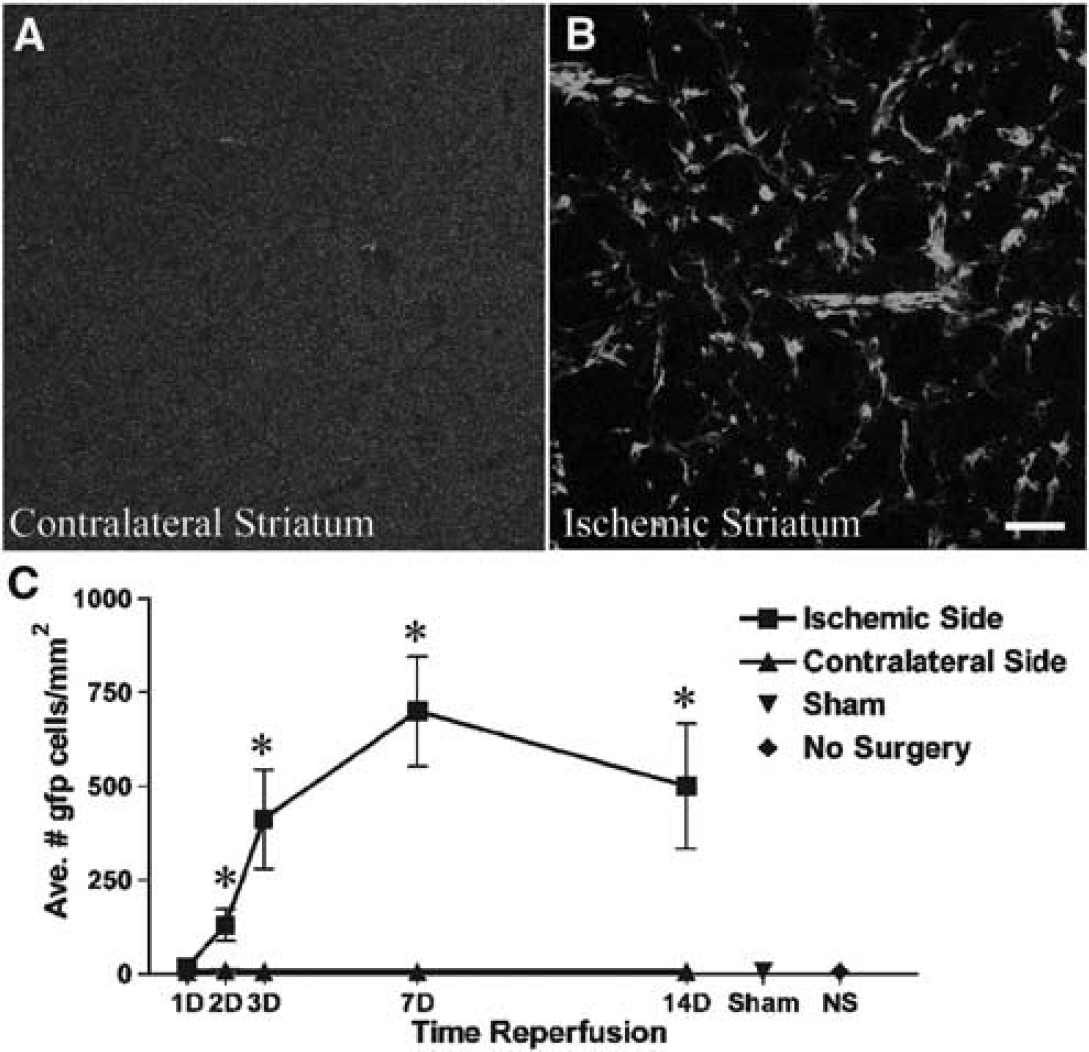
Bone marrow-derived cells infiltrate areas of ischemic injury. (
Parenchymal Bone Marrow-Derived Cells Express Microglial Markers and are not Proliferative
The majority of parenchymal BMDCs expressed the pan-hematopoietic marker, CD45, and the monocyte/microglia markers, CD11b (not shown) and Iba-1 (Figure 3). The percentage of GFP+ cells that coexpressed Iba-1 increased with time after ischemia from approximately 50% at day 1 of reperfusion to approximately 75% at 14 days reperfusion. Similarly, the percentage of GFP+ cells that expressed CD45 increased from 75% to almost 100% at day 14, suggesting that BMDCs retain their hematopoietic phenotype in brain parenchyma, as has been reported previously in noninjured and aspiration-induced lesion (Vallieres and Sawchenko, 2003). In addition to Iba-1-positive microglia, CD45-positive cells include all mature hematopoietic cells, including neutrophils, dendritic cells and lymphocytes. At no time point did we observe GFP+ cells colabeled with the neuronal markers, NeuN or Hu, or GFP+ cells colabeled with the astrocyte marker, GFAP.
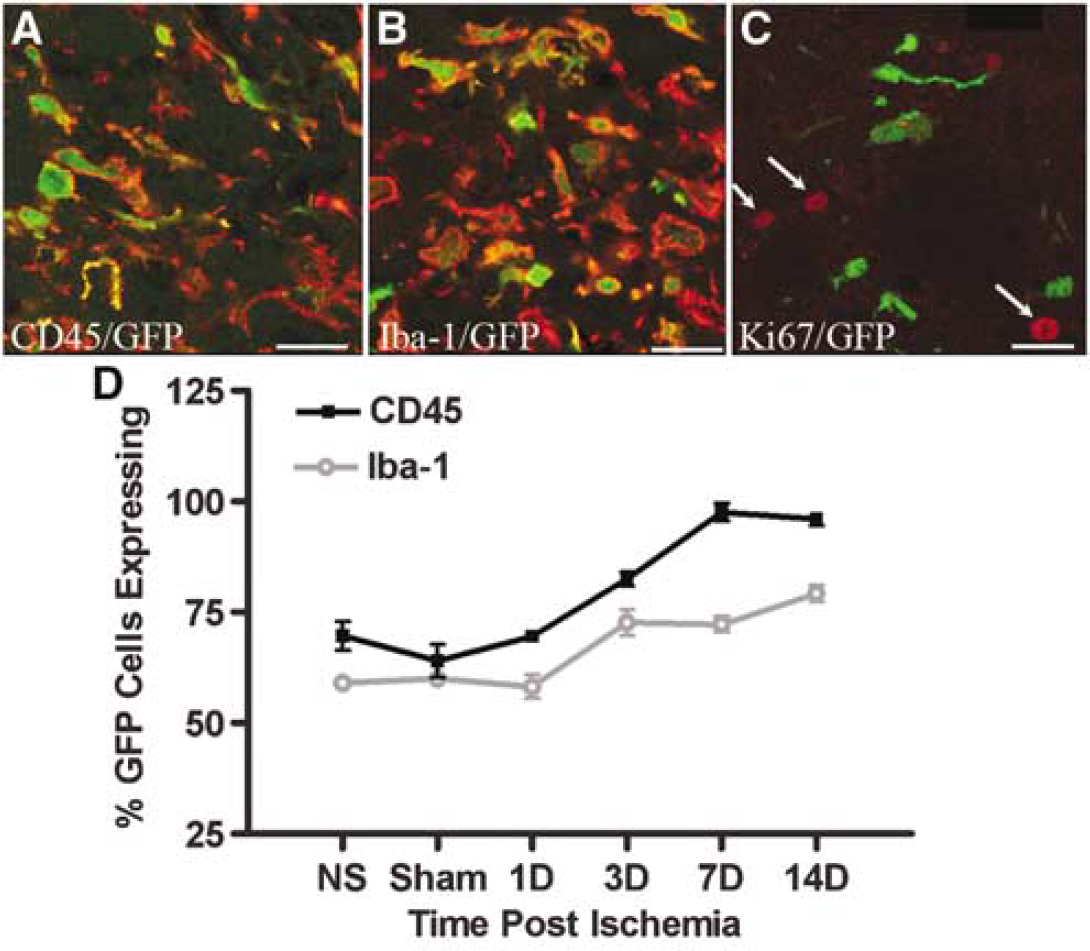
Phenotypic characterization of parenchymal BMDCs. Confocal micrographs of CD45 (
To determine the contribution of proliferation to the increased numbers of GFP+ BMDCs within the ischemic brain, we immunostained histological sections using an antibody directed against a proliferation marker, Ki67 (Figure 3C). Ki67 is a nuclear protein and is expressed by cells in all phases of the cell cycle except G0 (Brown and Gatter, 2002). Only occasional GFP+ cells within the parenchyma were immunopositive for Ki67 at any time point analyzed, indicating that these cells undergo a negligible amount of proliferation once they have entered the brain parenchyma. However, we did observe several GFP– parenchymal cells that were Ki67 positive on day 3 of reperfusion, which may represent endogenous glia proliferation after ischemia.
Perivascular Bone Marrow-Derived Cells Undergo Proliferation and Give Rise to Pericytes
To determine whether perivascular GFP+ cells were associated with ischemiainduced angiogenesis, we assessed the morphology and number of laminin + vessels at each time point after transient focal ischemia (Hayashi et al, 2003; Rosenstein et al, 1998). Increased numbers of laminin immunoreactive vessels within the ischemic striatum became significant by day 3 of reperfusion compared with contralateral striatum (92.73 ± 11.46 compared with 40.09 ± 4.284; P < 0.001). Numbers of laminin + vessels continued to increase within the ischemic striatum compared with the contralateral striatum to day 14 (149.40 ± 7.072 compared with 39.11 ± 3.472 (Figures 4A, 4B and 4D)). These vessels were thin walled and dilated, indicative of new vessel growth (Zhang et al, 2000). The growing vasculature was also marked by a proliferative burst at 3 days after transient MCAO, as assessed by Ki67 immunolabeling. Both GFP+ cells and cells that were negative for GFP and associated with the growing vasculature displayed Ki67 immunoreactivity at day 3 (Figure 4C). Few Ki67 immunopositive cells were observed surrounding the vasculature at later time points.
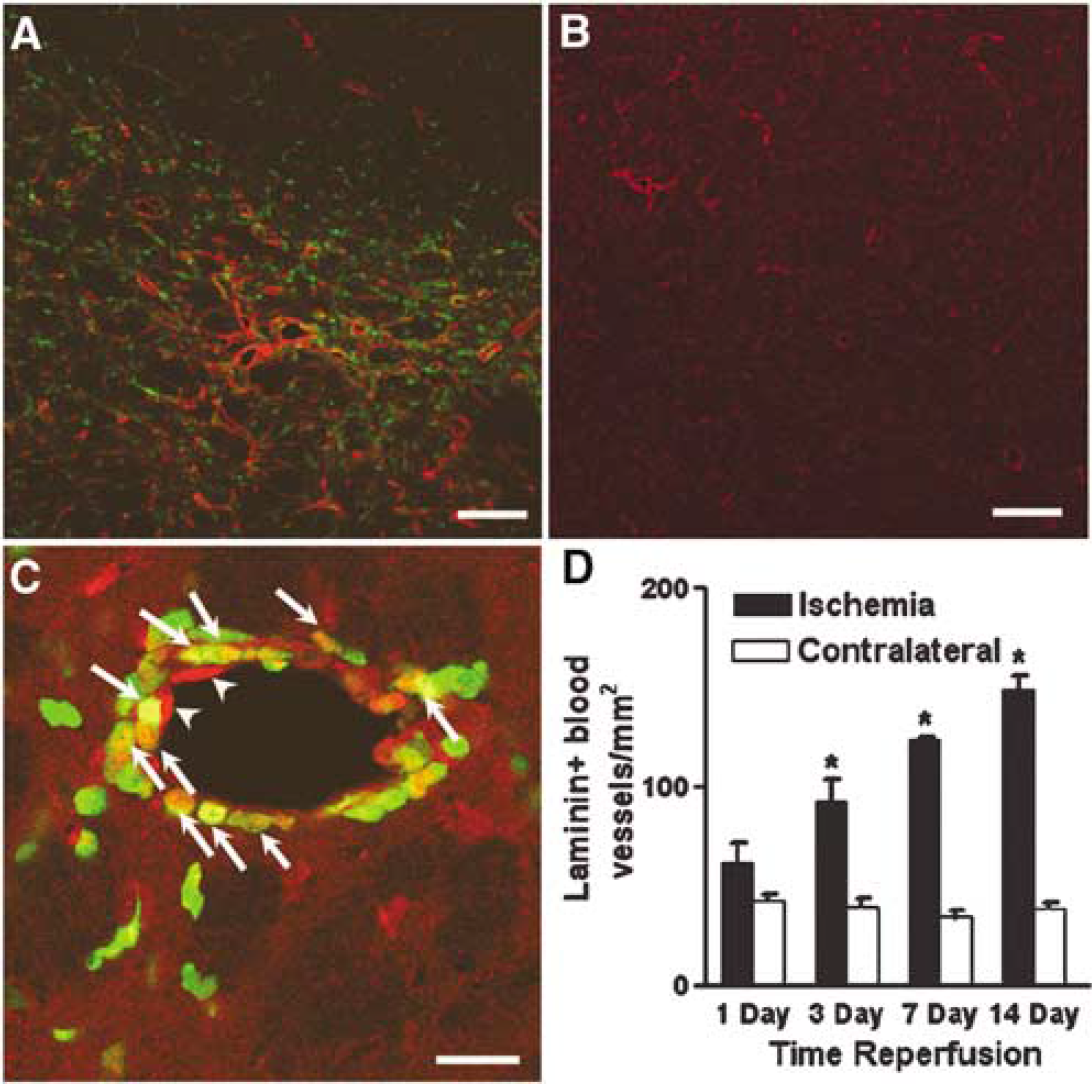
ischemiainduced angiogenesis after MCAO. Laminin reactivity (red) is upregulated on the ischemic striatum (
In addition to many perivascular microglia, a subset of GFP+ cells associated with the neovasculature displayed morphological and phenotypic characteristics of pericytes. Green fluorescent protein-positive pericytes were located within the endothelial basal lamina and formed tubes (Figure 5A). These cells were negative for the endothelial cell markers vonWillebrand factor (Figure 5B) and CD31 (not shown), but were positive for the pericyte markers desmin (Figures 6A–6D) and vimentin (not shown). The percentage of GFP+ cells associated with the neovasculature that coexpressed the pericyte marker, desmin, reached significance by day 3 and displayed a striking increase with time after ischemia, reaching approximately 39% by 14 days (Figure 6E). Furthermore, the percent of blood vessels that had at least one associated GFP+ cell surrounded by basal lamina in a pericyte-like location also reached significance on day 3 in comparison to contralateral striatum (11.17% ± 1.465% compared with 1.320% ± 0.660%; P < 0.001). The percentage of vessels with a GFP+ cell in a pericyte-like position increased to 19.83% ± 1.465% by day 14 after ischemia in comparison to contralateral striatum, which remained relatively stable at 1.320% ± 0.661% vessels with GFP+ pericytes (Figure 6F). These data suggest a continued recruitment and maturation of bone marrow-derived pericytes after ischemia.
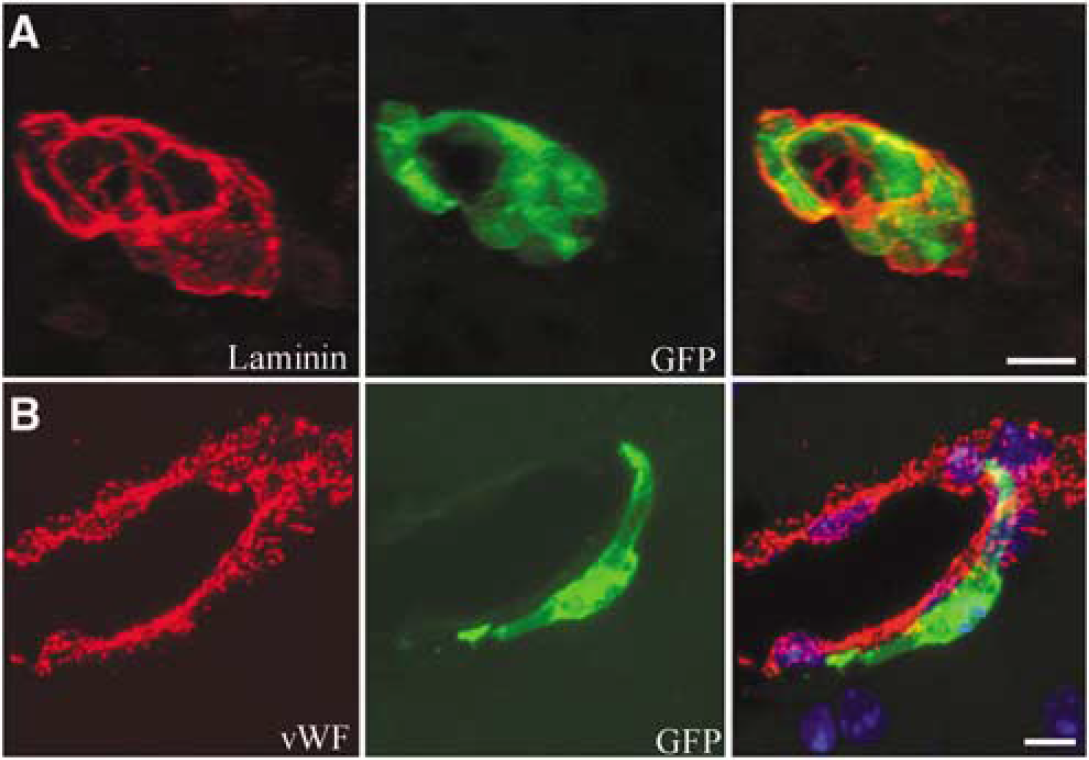
Bone marrow-derived cells associated with the vasculature are not endothelial cells. (
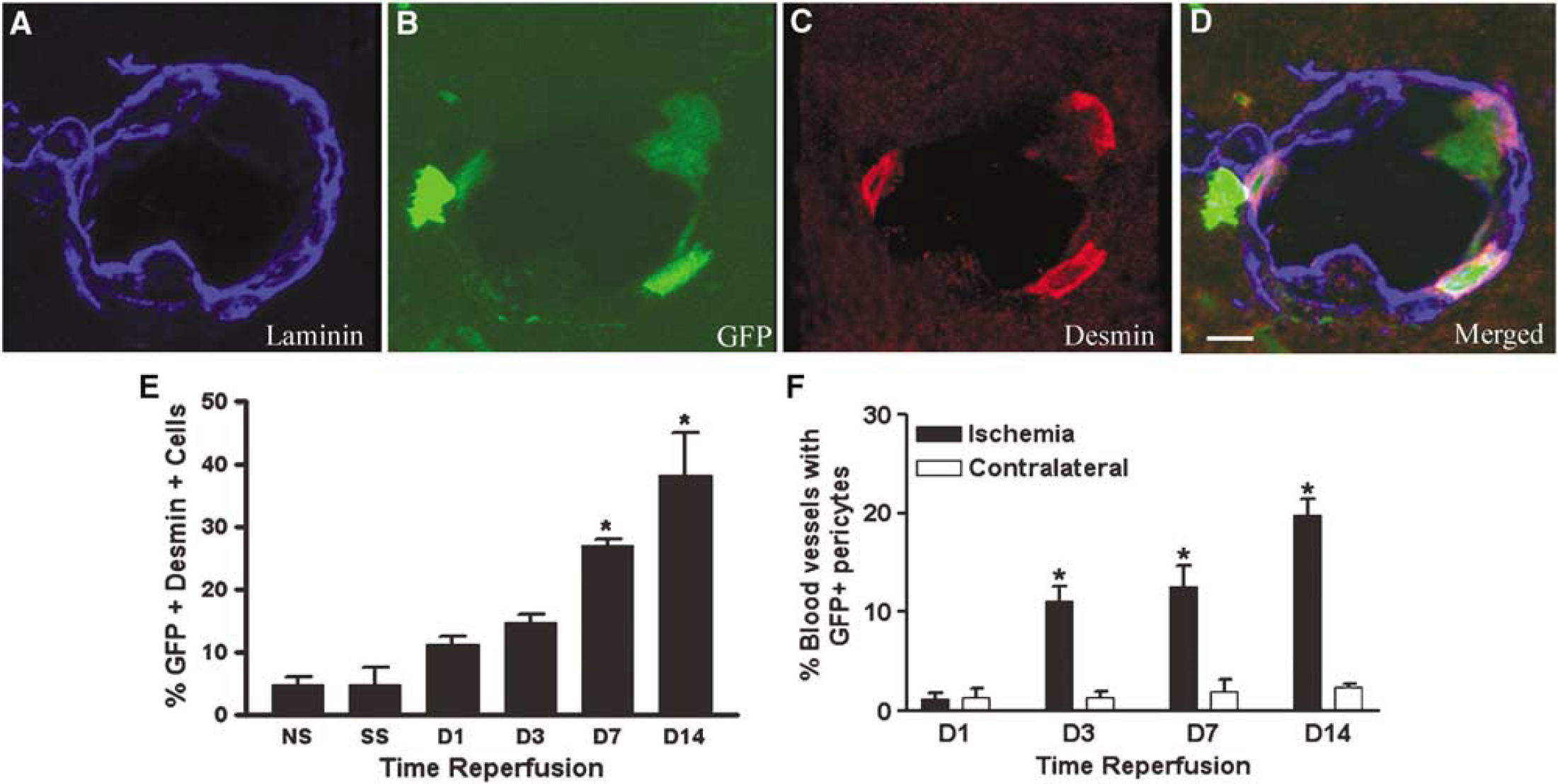
Bone marrow-derived cells express the pericyte marker desmin. Confocal projection image illustrating (
The maturation of nascent vasculature requires intricate signaling between endothelial cells and pericytes that involve both TGF-β and VEGF expression by pericytes, which inhibit migration and proliferation of endothelial cells and support their survival, respectively. Immunostaining revealed that GFP+ cells that colabeled with desmin also expressed TGF-β or VEGF (Figure 7). By day 14 after ischemia, 58.50% ± 6.198% and 40.12% ± 3.987% of GFP+ cells associated with the vascular expressed TGF-β and VEGF, respectively. These findings suggest that although BMDCs do not widely incorporate as endothelial cells into the growing vasculature, pericytes involved in vessel maturation and stabilization appear to be recruited from circulating BMDCs.
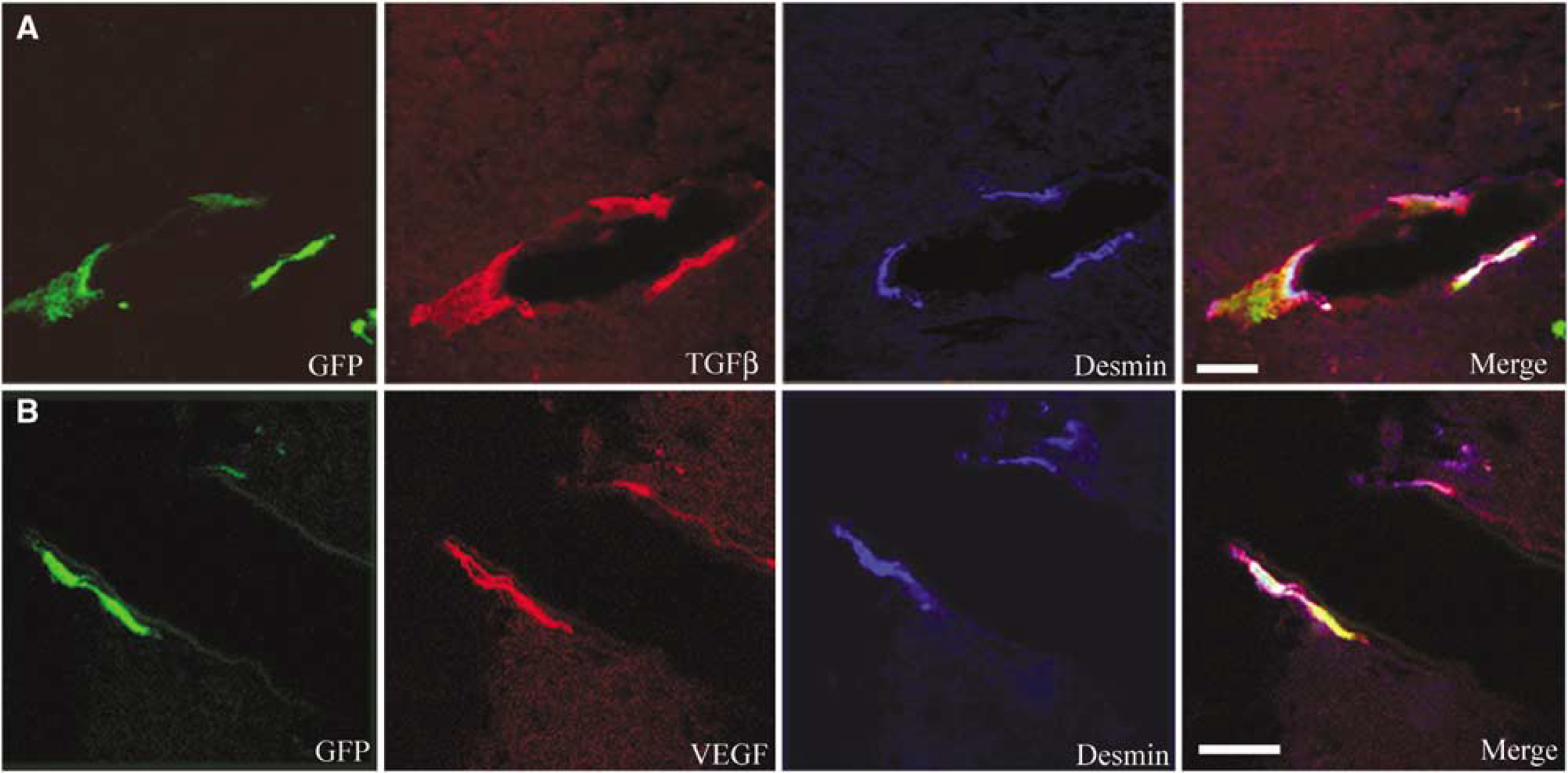
Vascular associated BMDCs express angiogenic factors. (
Discussion
Our studies show that mild focal ischemia stimulates massive influx of BMDCs into both parenchymal and perivascular compartments within the area of ischemic damage. Within the injured parenchyma, BMDCs gave rise to microglia, which are mostly nonproliferative. Perivascular BMDCs associated with growing vasculature display a proliferative burst concomitant with that of existing endothelial cells. Many of the BMDCs associated with the neovasculature are specifically identified as pericytes, and express the angiogenic factors TGF-β and VEGF. These results suggest that pericytes are recruited from the circulation during ischemiainduced angiogenesis, and are likely to be involved in stabilizing the architecture of newly formed vessels. These observations add to the mounting evidence that BMDCs are intricately involved in revascularization and repair processes in the ischemic brain.
Mild cerebral ischemia induced a rapid and massive influx of BMDCs into the CNS parenchyma, which peaked at 7 days and remained elevated at 14 days. The majority of these cells comprise parenchymal microglia, based on morphological and immunophenotypic criteria. In GFP chimeric mice, BMDCs have been estimated to give rise to 40% of all microglia in uninjured brain by 1 year after bone marrow reconstitution (Hess et al, 2004). Our studies further show that the increase in marrow-derived microglia in response to ischemia is due to rapid influx of cells and not due to proliferation of existing marrow-derived microglia, because few GFP+ microglia were immunopositive for the proliferation marker, Ki67. Although our studies did not extend beyond 14 days after ischemia, previous studies suggest that these cells may persist at increased levels for up to 6 months (Beck et al, 2003).
Beyond their role in phagocytosis and clearing of cellular debris, microglia have been purported to exert both beneficial and deleterious effects after brain injury, through production of both neurotrophic and neurotoxic factors, respectively. In a mouse MPTP-lesion model of Parkinson's disease, marrow-derived microglia almost uniformly express the inducible form of nitric oxide synthase, iNOS (Kokovay and Cunningham, 2005), which participates in neuronal degeneration in this injury model (Dehmer et al, 2000). Further studies seem warranted to determine whether marrow-derived microglia are neurotoxic, or possess properties distinct from those of endogenous microglia in repair processes after stroke.
It has been well established that, under baseline conditions, BMDCs migrate into the brain and take up residence within the perivascular compartment (Bechmann et al, 2001a, b ; Hickey, 1991; Hickey and Kimura, 1988). We observed an increased number of BMDC associated with the perivascular compartment after cerebral ischemia. Endothelial cells and GFP+ perivascular cells underwent a burst of proliferation at 3 days after cerebral ischemia, with very little subsequent proliferation, as assessed by Ki67 immunostaining. During this period, the ischemic striatum was also marked by the appearance of thin-walled, dilated capillaries, which are indicative of new vessel formation (Zhang et al, 2000). The accumulation of GFP+ cells around the neovasculature suggests that these cells might be involved in vascular remodeling and angiogenesis. In peripheral tissues, accumulation of monocytes/macrophages around collateral vessels is thought to be involved in collateral artery growth through secretion of growth factors, cytokines and proteases (Arras et al, 1998; Heil et al, 2002; Ziegelhoeffer et al, 2004).
Previous studies have shown that new vessel formation after cerebral ischemia occurs primarily via angiogenesis, through the proliferation and migration of endothelial cells from existing blood vessels, with minimal contribution from bone marrow-derived endothelial progenitor cells (Beck et al, 2003; Hess et al, 2004; Zhang et al, 2002). Similarly, we did not observe GFP+ cells expressing endothelial cell markers at any time point analyzed. Although one report suggested that marrow-derived cells give rise to approximately 42% of all endothelial cells in GFP+ vessels within the periinfarct region (Hess et al, 2002), subsequent studies have only identified occasional, rare marrow-derived endothelial cells (<1% of BMDCs) in bone marrow chimeras subjected to stroke (Beck et al, 2003; Hess et al, 2004; Zhang et al, 2002), or in normal mice subjected to stroke, with subsequent intravenous infusion of marrow stromal cells or endothelial progenitors enriched from peripheral blood (Taguchi et al, 2004). Therefore, it is possible that our analysis was not exhaustive enough to identify these rare marrow-derived endothelial cells. Similarly, we did not observe GFP+ cells colabeled with astrocytic or neuronal markers, which has also been reported to rarely occur (Corti et al, 2002; Hess et al, 2002, 2004).
High-resolution confocal microscopy revealed that many of the GFP+ BMDCs associated with the vasculature were pericytes, based on multiple criteria used for identification of these cells (Allt and Lawrenson, 2001; Gerhardt and Betsholtz, 2003), including morphology, position on the abluminal surface of capillaries, encasement by the endothelial basal lamina and expression of the pericyte markers, vimentin and desmin. The number of desmin+ marrow-derived pericytes increased with time after ischemia to reach approximately 38% by day 14. These cells are unlikely to arise from cell fusion processes, because cell fusion is extremely rare (1 in 105 cells) (Terada et al, 2002). In addition, because desmin is not expressed by all brain pericytes, these numbers might be an underestimation of BMDC contribution. Green fluorescent protein marker infidelity could also result in an underestimation of donor cells in brain (Anderson et al, 2005). Pericytes function to stabilize newly formed capillaries, by preventing endothelial cell apoptosis and vascular regression (Carmeliet, 2003; Distler et al, 2003), and influencing the formation of the blood–brain barrier (Berezowski et al, 2004; Hori et al, 2004). After brain injury, pericytes have been observed to migrate away from the capillary wall into the brain parenchyma, a process which may accelerate vascular damage and leakage (Dore-Duffy et al, 2000; Gonul et al, 2002).
Although the origin of pericytes has not been well studied, it is known that during development pericytes are recruited to growing vessels by endothelial platelet-derived growth factor (PDGF-B), through engagement of the pericyte platelet-derived growth factor receptor beta (PDGFR-β). Genetic deletion of PDGF-B or PDGFR-β in mice results in a severe deficit in pericytes and microvascular leakage and hemorrhage (Leveen et al, 1994; Lindahl et al, 1997; Soriano, 1994). After cerebral ischemia, PDGFR-β is specifically upregulated in pericytes, indicating that PDGF-B is also involved in pericyte recruitment in brain ischemia (Renner et al, 2003). Once recruited, pericytes directly inhibit endothelial cell proliferation and migration through activation of TGF-β expressed on the pericyte cell surface. This process involves proteolytic conversion of latent TGF-β through pericyte–endothelial cell contacts, which occur via holes in the basement membrane and formation of peg-and-socket contact between endothelial cells and pericytes (Orlidge and D'Amore, 1987; Sato and Rifkin, 1989). Endothelial cell maturation and vessel stability also occur through production of angiopoietin-1 by pericytes, which engages endothelial tie-2 receptor tyrosine kinase (Ramsauer and D'Amore, 2002; Ramsauer et al, 2002; Uemura et al, 2002). In brain endothelial cells, pericyte-derived angiopoietin-1 induces the expression of endothelial tight junction proteins (Hori et al, 2004). During retinal vascularization, astrocyte-derived VEGF directs sprouting of vessels. Recent studies have shown that pericytes are also induced to express VEGF concomitant with their maturation, which may contribute to vessel stabilization by replacing VEGF supplied by the surrounding tissue (Ramsauer and D'Amore, 2002), and thereby preventing endothelial cell apoptosis. Our observation that marrow-derived pericytes express both TGF-β and VEGF strongly suggests that these cells are actively engaged in vascular remodeling within the ischemic region.
The relationship of pericytes to perivascular and parenchymal microglia is unclear. There are reports that pericytes can leave the endothelial basement membrane and migrate into the perivascular space (Dore-Duffy et al, 2000; Thomas, 1999), or into the brain parenchyma to give rise to resident microglia (Bechmann et al, 2001b). That pericytes, perivascular cells and microglia express an overlapping set of immunophenotypic markers has led many investigators to conclude that all three cell types are derived from a common monocyte progenitor (Guillemin and Brew, 2004; Thomas, 1999). Others contend that these cell types remain distinct from one another in both placement and function within the brain, and that pericytes are derived from vascular smooth muscle cells (Gerhardt and Betsholtz, 2003; Kida et al, 1993). Our observation that BMDCs give rise to perivascular cells, pericytes and parenchymal microglia after cerebral ischemia suggests that all three cell types can be derived from bone marrow in the adult brain, but whether of a common cell origin is not clear. These observations are likely to be important for therapeutic targeting of angiogenesis in stroke, because promotion of pericyte recruitment from the peripheral circulation might be essential for stabilization and maturation of the neovasculature in the ischemic brain.
Footnotes
Acknowledgements
The authors gratefully acknowledge NIH support from NINDS 1 R21 NS43646 and R01 NS047373. We thank Ashleigh Martin and Mark Garcia for their assistance with histology. Images in this paper were generated in the UNM Cancer Center Fluorescence Microscopy Facility, which received support from NCRR 1 S10 RR14668, NSF MCB9982161, NCRR P20 RR11830, NCI R24 CA88339, NCRR S10 RR19287, NCRR S10 RR016918, the University of New Mexico Health Sciences Center and the University of New Mexico Cancer Center.
