Abstract
Infection, inflammation, and hyperthermia associated with cerebral ischaemia are known to contribute to enhanced neuronal cell loss and more severe behavioural deficits. Because neonatal exposure to an immune challenge has been shown to alter the severity of inflammatory and febrile responses to a further immune challenge experienced in adulthood, we hypothesised that this could also alter temperature responses and neuronal survival after ischaemia. Thus, male Sprague–Dawley rats were treated at postnatal day 14 with a single injection of the bacterial endotoxin lipopolysaccharide (LPS) and were examined as adults for temperature changes, behavioural deficits, and neuronal cell loss associated with global cerebral ischaemia after a two-vessel occlusion (2 VO). Neonatally LPS-treated rats showed behavioural differences in a novel object exploration paradigm, as well as altered temperature responses to the 2 VO compared with neonatally salinetreated controls. Interestingly, these neonatally LPS-treated rats also showed increased cell loss in the central nucleus of the amygdala, a region that is important in the processing of emotional responses, but that is not usually examined in animal models of cerebral ischaemia. No differences were seen in the CA1, CA3, or dentate gyrus regions of the hippocampus. This work shows the importance of examining brain regions other than the hippocampus in association with global ischaemia. We also highlight the importance of the early period of development in programming an animal's ability to deal with injury such as cerebral ischaemia in adulthood.
Introduction
It is currently well accepted that inflammation and alterations to the immune system can profoundly affect health and can increase susceptibility to heart disease and stroke. Infection, for example, is a predisposing risk factor in humans for the development of a brain infarct when it is experienced within the week preceding the ischaemia (Macko et al, 1996). In particular, respiratory tract infections may trigger or augment large-vessel atherothromboembolic or cardioembolic stroke (Paganini-Hill et al, 2003). Body temperature, too, is a major factor in neuronal survival after cerebral ischaemia (Kuroiwa et al, 1990; Coimbra et al, 1996a, b; Corbett and Thornhill, 2000). The hyperthermia typically associated with ischaemia has been shown to contribute substantially to long-term neuronal cell loss in animal models (Kuroiwa et al, 1990; Baena et al, 1997; Ginsberg and Busto, 1998) and in humans (Ginsberg and Busto, 1998). Thus, hyperthermia is associated clinically with more severe symptoms of neural injury, while maintained hypothermia during and after cerebral ischaemia is neuroprotective (Colbourne et al, 1997; Ginsberg and Busto, 1998; Thornhill and Corbett, 2001; Felberg et al, 2001; Al Senani and Grotta, 2002).
Important recent investigations from this laboratory have determined that an immune challenge to neonatal rats, in the form of the endotoxin lipopolysaccharide (LPS), leads to an attenuation of the hyperthermic response to a similar immune challenge in adulthood, as well as an attenuation of other elements of the inflammatory response such as activation of cyclooxygenase (COX)-2 in the brain (Boisse et al, 2004). These findings may have implications for recovery after stroke, because both neutrophilic and mononuclear inflammation alter the outcome of cerebral ischaemia (Hallenbeck et al, 1986; Giulian and Robertson, 1990; Kochanek and Hallenbeck, 1992), while chemical mediators of inflammation such as COX-2 and interleukin-1 may also play an important role in responses to ischaemia (Yamasaki et al, 1995; Planas et al, 1999; del Zoppo et al, 2000; Buchan et al, 2000).
We therefore hypothesised that the attenuated hyperthermic response to inflammation seen in adult rats exposed to LPS as neonates could also be reflected in altered temperature responses to ischaemia. These effects may therefore result in altered neuronal survival and behavioural correlates of ischaemic damage after cerebral ischaemia in these neonatally immune challenged rats.
On postnatal day 14 (P14), rats underwent an intraperitoneal injection of either (a) pyrogen-free saline (controls) or (b) LPS. Once these rats reached adulthood, a well-established model of brief forebrain ischaemia, the bilateral carotid artery occlusion (two-vessel occlusion—2VO) with hypotension (Smith et al, 1984b; McBean et al, 1995; McBean and Kelly, 1998), was then used to induce global cerebral ischaemia. These rats were then assessed for temperature changes and behavioural deficits associated with global cerebral ischaemia, as well as for cell loss in relevant brain regions.
Materials and methods
Animals
Pregnant Sprague–Dawley rats (Charles River) were maintained at 22°C on a 12 h light/dark cycle (7:00 am to 7:00 pm) with pelleted rat chow and water available ad libitum. Ten days after birth, that is, P 10, litters were culled to 12 pups. All litters were weaned at P 21 and male rats were kept and housed 3 to 4 animals per cage until they reached approximately 8 to 10 weeks of age. All procedures were thus conducted on male rats and were in accordance with the Canadian Council on Animal Care regulations and approved by the local University of Calgary Animal Care Committee.
Early Life Manipulations
At P14, animals were removed from their mother for approximately 5 mins and subjected to intraperitoneal injections of either LPS (Escherichia coli; serotype 026:B6, L-3,755; Sigma, St Louis, MO, USA; 100 µg/kg) in 1 mL/kg pyrogen-free saline, or to an equivalent volume of pyrogen-free saline. Ears were clipped for identification. Pups were then returned to their home cages and were left undisturbed except for weaning and the usual cleaning and feeding procedures, until approximately 8 to 10 weeks of age. Approximately equal numbers of animals from each litter received LPS or saline, and during the interval between injection and experimentation, care was taken to ensure that each cage contained both LPS and saline pretreated pups. Experiments were conducted on animals from five litters over a period of 6 months.
Surgery
Approximately 6 to 8 weeks after injection, 27 animals were brought to a separate temperature-controlled testing room within our laboratory, which had the same environmental parameters as the general housing facility, and they were allowed to acclimatise overnight. On the morning of the following day, all animals were briefly anaesthetised with halothane (induced at 4% in a Plexiglass chamber and maintained at 2% by means of a face mask) and silicone-coated temperature data loggers (SubCue Dataloggers; Calgary, Alberta, Canada) were implanted into the abdominal cavity. After a day of recovery, animals were subjected to either sham surgery or to a brief forebrain ischaemia (Smith et al, 1984b; McBean et al, 1995; McBean and Kelly, 1998). The rats were deprived of food for approximately 18 h overnight before the 2VO or sham surgery to normalise blood glucose, which can affect neurologic outcome after ischaemia (Voll and Auer, 1988; Marie and Bralet, 1991; Garnier et al, 1999). On the day of the 2VO or sham surgery, animals were anaesthetised using halothane, and the tail artery cannulated for blood pressure monitoring. During surgery, brain temperature was estimated using a thermocouple thermometer in the ear and was maintained at 37.0°C ± 0.3°C by means of an overhead heating lamp. To induce ischaemia, a ventral midline neck incision was made allowing the isolation of the common carotid arteries. Both common carotid arteries were occluded simultaneously for 20 mins with nontraumatic microarterial clips, and the blood pressure reduced to and maintained between 50 and 55 mm Hg for the duration of the surgery by means of fine adjustments of the halothane anaesthesia (McBean et al, 1995; McBean and Kelly, 1998). A 20 mins duration of carotid artery occlusion was identified in preliminary trials as necessary and sufficient to produce consistent neuronal cell damage in the CA1 region of the hippocampus, one region particularly vulnerable in global ischaemia (Pulsinelli et al, 1982; Smith et al, 1984a; Colbourne et al, 1999). After the clips were removed, blood pressure was allowed to recover to presurgical pressures and the arteries were visually inspected to ensure reperfusion. Sham-treated animals were subjected to the same surgery, but no occlusion of the arteries was made. Immediately after surgery the animals were returned to their home cages and allowed to recover undisturbed.
Neurologic Deficit Testing
One and seven days after surgery, animals were assessed for sensorimotor deficits as modified from that described previously (Sarti et al, 2002). Animals were assessed during the light phase of the circadian cycle between 10:00 am and 1:00 pm by an examiner who was masked to the type of neonatal treatment and surgery the rats had received. Rats were assessed for deficits in (i) spontaneous activity in the home cage, (ii) symmetry in the movement of four limbs, (iii) forepaw stretching, (iv) climbing, (v) body proprioception, (vi) response to vibrissae touch. Each animal was given a score out of six, where for each test, zero was equal to no neurologic deficit and one was equal to some deficit.
Open Field Testing
Just before implantation of the abdominal temperature data loggers, as well as 1 and 7 days after the 2VO to induce ischaemia, that is, in three separate trials, all animals were assessed for locomotor activity and anxiety using an open field paradigm (Igarashi and Takeshita, 1995). The circular open field was of 90 cm diameter and had white inside walls, 45 cm high. The arena was placed in a brightly lit room on an off-white coloured floor with a black grid that divided the area into 12 sections. Each animal was placed in the centre of the arena and observed for locomotion (number of grid-lines crossed), number of rearings, number of instances of grooming, and number of faecal pellets deposited in a period of 5 mins. Animals were assessed during the light phase of the circadian cycle between 10:00 am and 1:00 pm by an examiner who was masked to the type of treatment the rats had received. For each test, the examiner stood in the same position relative to the arena.
Object Exploration Testing
Immediately on the conclusion of the open field tests, rats underwent a novel object exploration test for evaluation of nonspatial working memory (Ennaceur and Delacour, 1988; Ennaceur and Meliani, 1992; Roy and Chapillon, 2004). On the first exploration trial, two identical objects (500 mL glass beakers) were introduced into the middle of the open field arena. The rats were allowed to explore the objects for 5 mins. They were scored during this time for each of the two objects separately in seconds spent with the nose directed towards and less than 2 cm away from the object (Pawlak and Schwarting, 2002; Schroder et al, 2003). During the second trial, that is, 1 day after the 2VO or sham surgery and immediately after the second open field test, the left-hand side (relative to the examiner) object was replaced with a novel object (a square blue plastic container of approximately the same size as the 500 mL beaker) and the rats were scored for 5 mins. For the third trial, that is, 7 days after the 2VO and immediately after the third open field test, the left-hand side object was replaced with a third object (a round brown cane basket of approximately the same size as the 500 mL beaker) and the rats were again scored for 5 mins. Objects and the open field arena were cleaned thoroughly with virkon between each animal.
Perfusions
Seven days after the 2VO or sham surgery, animals were deeply anaesthetised with Na pentobarbital (80 to 100 mg/kg intraperitoneally) and perfused with 4°C phosphate-buffered saline via the left cardiac ventricle, followed by 10% phosphate-buffered formalin. A brief (7 days) survival time was used as this has been shown to reveal close to maximum cell damage (Colbourne et al, 1999).
Histology
After perfusion, brains were blocked and immersed in 10% buffered formalin. Blocks were then embedded in paraffin and 10 µm coronal sections were cut and mounted onto polylysine-coated slides for subsequent staining. The extent of the ischaemic injury was quantified by histologic analysis of remaining hippocampal and central amygdala (CeA) cells in toluidine blue-stained coronal sections. The sections were first deparaffinised, then rehydrated and stained with toluidine blue. Neuronal damage was evaluated by an experimenter who was masked to the type of treatment the animals had received. Surviving neurons were counted in the CA1, the CA3, and dentate gyrus subfields of the hippocampus in four 10 µm sections 120 µm apart, from and caudal to −4.2 mm relative to bregma. Surviving neurons were also counted in the CeA in four 10 µm sections, 120 µm apart, from and caudal to −2.3 mm relative to bregma. The four values obtained for each subfield of the hippocampus and for the CeA were summed to obtain a total for each region, and the data for each group of animals were expressed as the mean ± standard error of the mean.
Data Analysis
Scores for neurologic deficits, open field, and object exploration for the groups treated with saline or LPS as neonates were compared using a two-way repeated measures (RM) analysis of variance (ANOVA) for each of the sham and 2VO groups, with neonatal treatment as the between factor and trial as the repeated measure. Where a significant interaction or effect of trial was found, this was followed by a one-way RM ANOVA and then, where significant, by a Dunnett's post hoc comparison comparing subsequent trials to the first. Where a significant effect of treatment was found, this was followed by Student's unpaired t-tests as appropriate. For the object exploration test, the total change in exploration time between trials one and two after the 2VO was compared between neonatally saline- and LPS-treated groups using a Student's unpaired t-test. Fever data were calculated as a fever index (°C × h) between 1 and 13 h after the 2VO. Thus, the temperature at each 15 mins interval was used to calculate a baseline temperature for the hour before surgery. The mean change from this baseline was then calculated per hour for each animal and these data summed for the 12 h. Fever indices for neonatally saline-and LPS-treated animals after the 2VO were then compared using a Student's unpaired t-test. Numbers of surviving neurons from each brain region were compared between neonatally saline- and LPS-treated animals using Student's unpaired t-tests. In each case, statistical significance was assumed when P < 0.05. Data are presented as mean ± standard error of the mean.
Results
Mortality
Neonatal exposure to LPS had no significant effect on animal mortality during or after surgery. Two of the 14 animals (14%) in the group exposed to saline as neonates died prematurely, one during the surgery and one during the recovery period. Both animals were of those subjected to the 2VO. Only one of the 13 animals (8%) in the neonatal LPS group died prematurely, and this occurred during the recovery period after the 2VO. Animals that died prematurely were omitted from subsequent analyses.
Neurologic Score
No neurologic deficits were found for any of the animals tested using the neurologic score as described in the Materials and methods section. Data are therefore not shown.
Open Field
Sham-treated animals: Of the sham-treated animals, both the neonatally saline (n = 6)- and LPS (n = 6)-treated rats showed an habituation in their rearing (F(2,10) = 32.42, P < 0.0001; Figure 1A) and locomotor (F(2,10) = 19.69, P < 0.0001; Figure 1B) responses to the open field test. No differences were seen, either in rearing (Figure 1A), locomotor activity (Figure 1B), grooming, or number of faecal pellets deposited (data not shown), between the neonatally saline-and LPS-treated rats either before or after sham surgery.
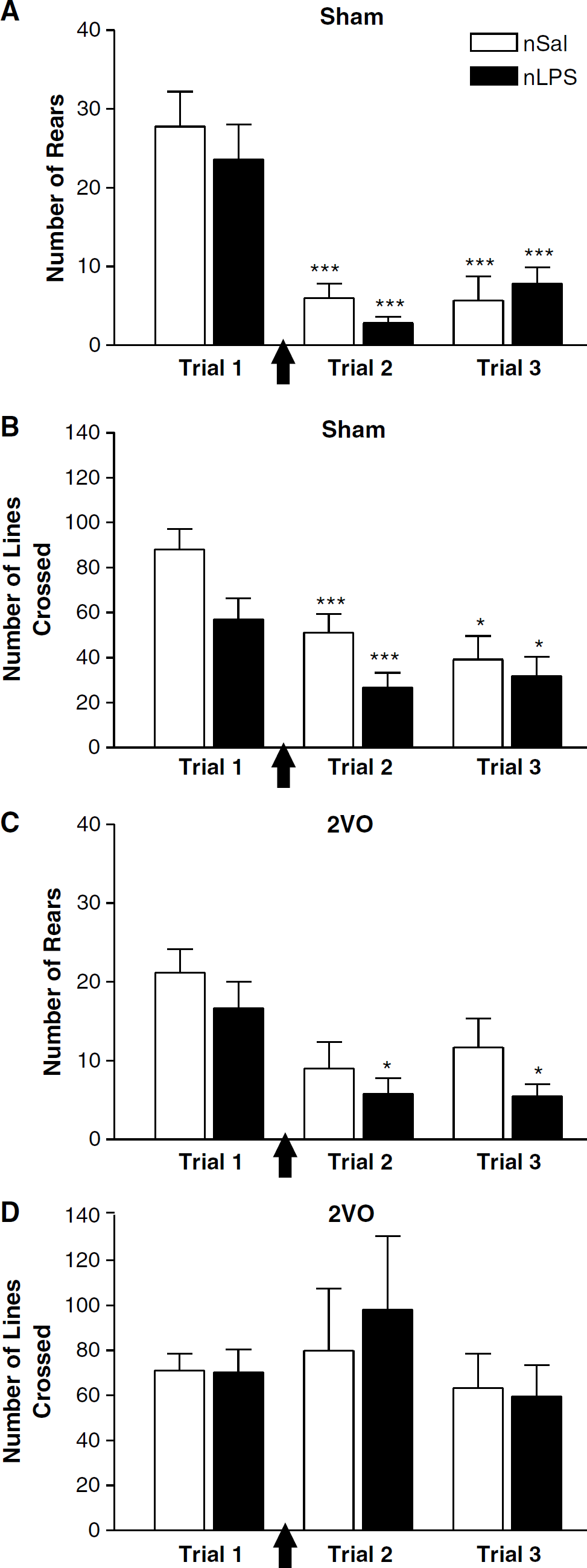
Open field behaviour in animals treated as neonates with saline (nSal—white bars) or lipopolysaccharide (nLPS—black bars) before and after sham surgery or two-vessel occlusion (2V0). (
Two-vessel occlusion animals: Importantly, the first trial scores for rearing, locomotor activity, grooming, and number of faecal pellets in the animals assigned to the 2VO group were very similar to those seen with the sham-treated animals.
As was seen with the sham-treated groups, there were no differences between the neonatally saline (n = 6)- and LPS (n = 6)-treated rats exposed to the 2VO in rearing (Figure 1C), locomotor activity (Figure 1D), grooming, or number of faecal pellets (data not shown). A habituation of rearing (F(2,10) = 9.456, P < 0.01) did occur in the neonatally LPS-treated animals (F(2) = 7.376, P < 0.05) in the trial 1 day after (P < 0.05) as well as 7 days after surgery (P < 0.05) with respect to the presurgery trial, but this was not statistically significant in the rats treated as neonates with saline (Figure 1C). Interestingly, the predilection for decreased locomotor activity after the first trial that was observed with the sham-treated animals was abolished by the ischaemia. That is, no habituation with trial was seen in either the neonatally saline- or LPS-treated groups that underwent the 2VO (Figure 1D).
Object Exploration
Sham-treated animals: The time spent exploring the introduced objects in the first object exploration test differed significantly depending on the neonatal treatment the animals had received (F(1,10) = 5.218, P < 0.05; Figure 2A, Table 1). Neonatally LPS-treated animals spent significantly less time exploring the two objects introduced to the open field arena than their salinetreated counterparts (t = 2.871; df = 10; P < 0.05). Neither group of rats showed a right or left side bias in the first novel object trial, both exploring the two objects equally (Table 1). There was also a tendency, albeit nonsignificant, for the neonatally LPS-treated rats to explore the rest of the open field area, as well as the introduced objects, less than the salinetreated rats did (Table 1). No differences were seen in rearing (Table 1), grooming, or in the number of faecal pellets produced (data not shown) between the two groups in this first object exploration trial.
Object exploration and exploration-associated activity in animals treated as neonates with saline (nSal) or LPS (nLPS) before and after sham surgery or two-vessel occlusion (2VO)
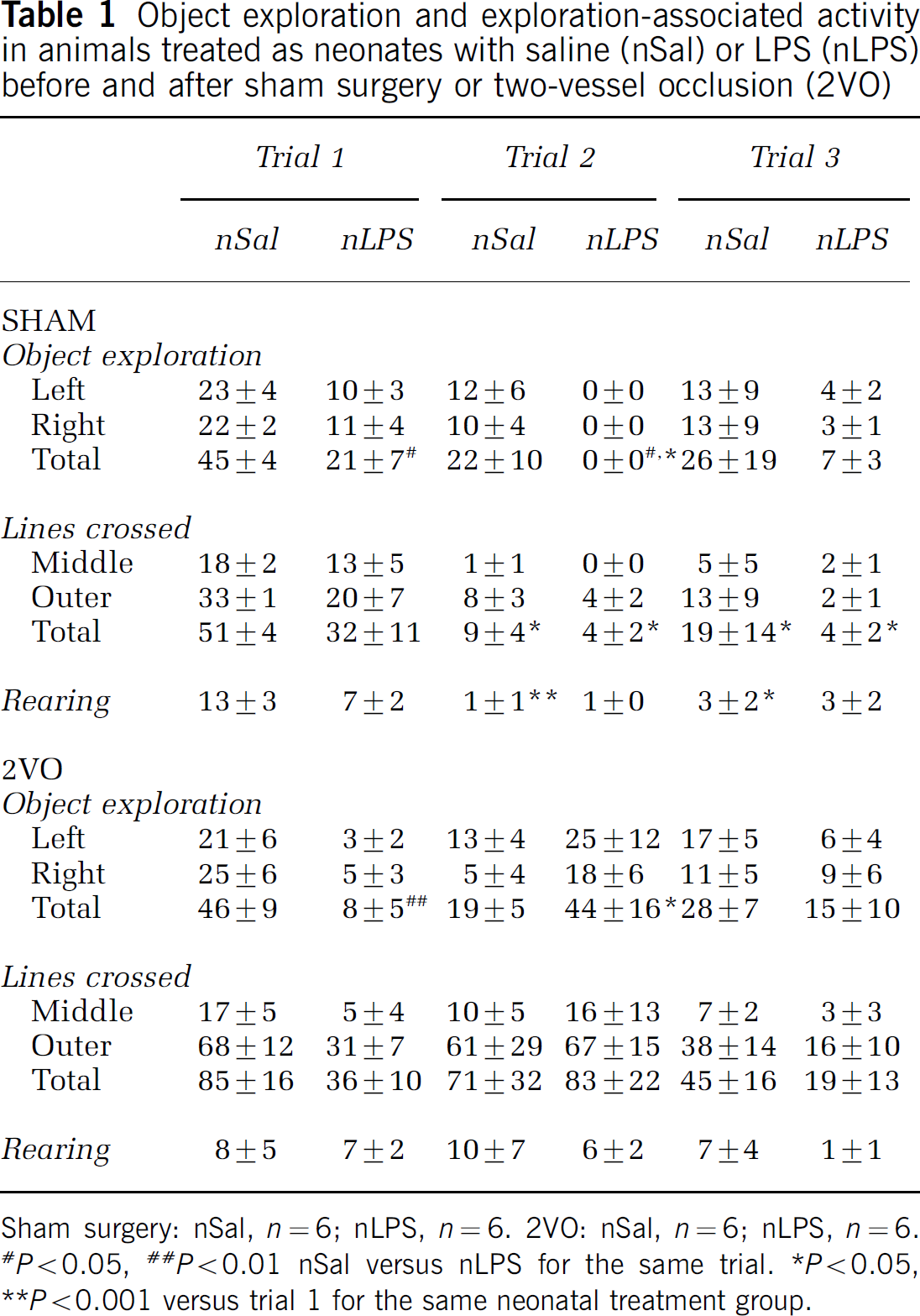
Sham surgery: nSal, n = 6; nLPS, n = 6. 2VO: nSal, n = 6; nLPS, n = 6. *P 0.05, <**P 0.01 nSal versus nLPS for the same trial. *P < 0.05, <**p 0.001 versus trial 1 for the same neonatal treatment group.
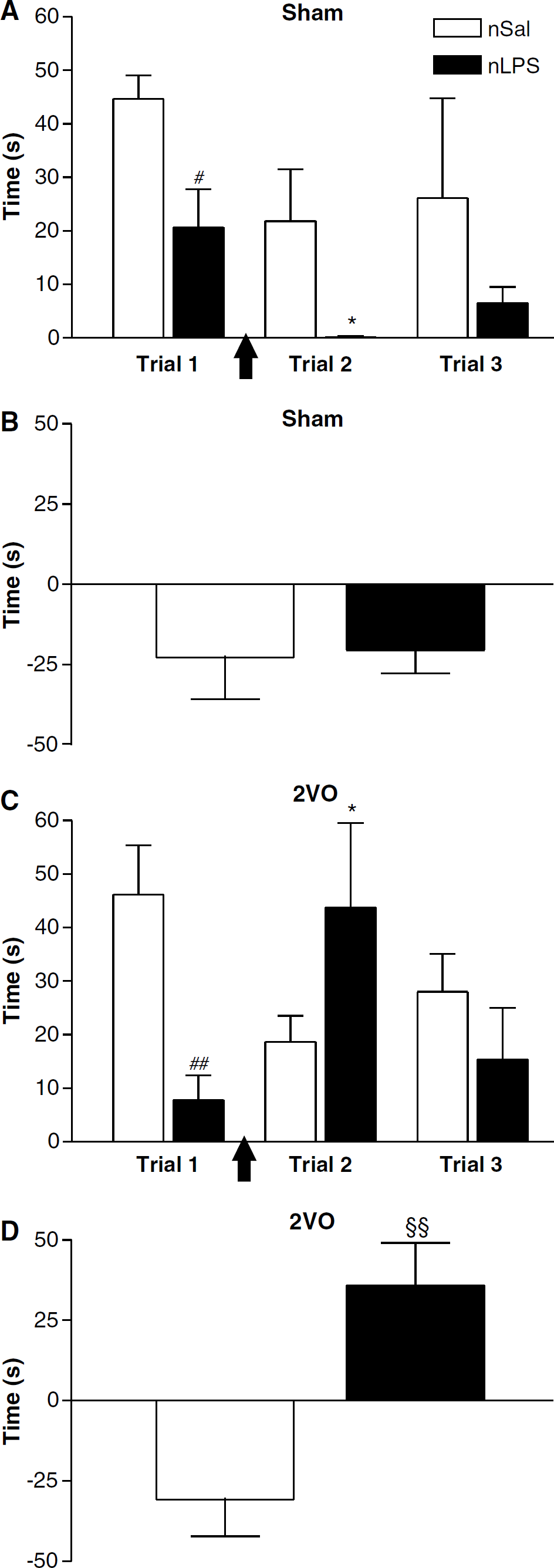
Object exploration in animals treated as neonates with saline (nSal—white bars) or lipopolysaccharide (nLPS—black bars) before and after sham surgery or two-vessel occlusion (2VO). (
There was a substantial reduction in the time spent exploring both the introduced objects in the second and third trials in sham-treated animals (F(2,10) = 3.865, P < 0.05; Figures 2A and 2B). In the second trial, the pattern observed previously was maintained, with neonatally salinetreated animals exploring both objects more than the LPS treated. Indeed, none of the neonatally LPS-treated animals showed any exploration of either the familiar or the novel object in the second trial. However, by the third trial, both groups exercised some exploration of the introduced objects. In the neonatally salinetreated group, where some exploration of the introduced objects was seen in the postsurgical trials, there was no bias towards exploring the novel object more than the familiar one. Each of the left-hand side and right-hand side objects was explored equally (Table 1).
Associated locomotor activity of the open field arena in the presence of the novel objects was also significantly reduced over the three trials in both groups of sham-treated animals compared with the first trial (F(2,6) = 16.65, P < 0.001), as was rearing (F(2,6) = 14.08, P < 0.001; Table 1).
Two-vessel occlusion animals: The propensity for neonatally LPS-treated rats to explore the introduced objects less than the salinetreated group was also seen in the animals that were to undergo the 2VO (t = 3.729; d = 10; P < 0.01, following P < 0.01 with the two-way RM ANOVA; F(2,10) = 7.504; Figure 2C). Interestingly, the second object exploration trial, 1 day after the 2VO, revealed that any propensity for decreased exploratory activity with two trials in neonatally LPS-treated rats was reversed with the ischaemia (Figures 2C and 2D). The neonatal LPS animals explored both objects for more time than they had previously, indeed the same amount of time as had the neonatally salinetreated animals before the ischaemia. In contrast, the neonatally salinetreated rats explored both objects significantly less than they had before ischaemic injury. This change in time spent exploring the objects was thus significantly different between the neonatal saline and neonatal LPS groups (P < 0.01; Figure 2D).
Despite previous exposure in trial 1 to the left-hand side object, both groups of rats explored each of the left and right objects for the same amount of time in the second trial (Table 1). The propensity for associated locomotor activity and rearing to be reduced over the three trials was abolished by ischaemia in both groups of animals compared with the first trial (Table 1).
In the third object exploration trial, 7 days after surgery, the rats again explored both objects equally and there was no difference between neonatally salinetreated rats and the neonatally LPS-treated ones (Figure 2C, Table 1).
Body Temperature
Before the 2VO or sham surgery, temperatures and circadian rhythms of the rats treated as neonates with LPS were identical to those treated as neonates with saline (Figure 3A). During surgery, our efforts to maintain ear temperatures of 37°C ± 0.3°C resulted in consistent body temperatures of between 36.1°C and 36.5°C (Figure 3B).
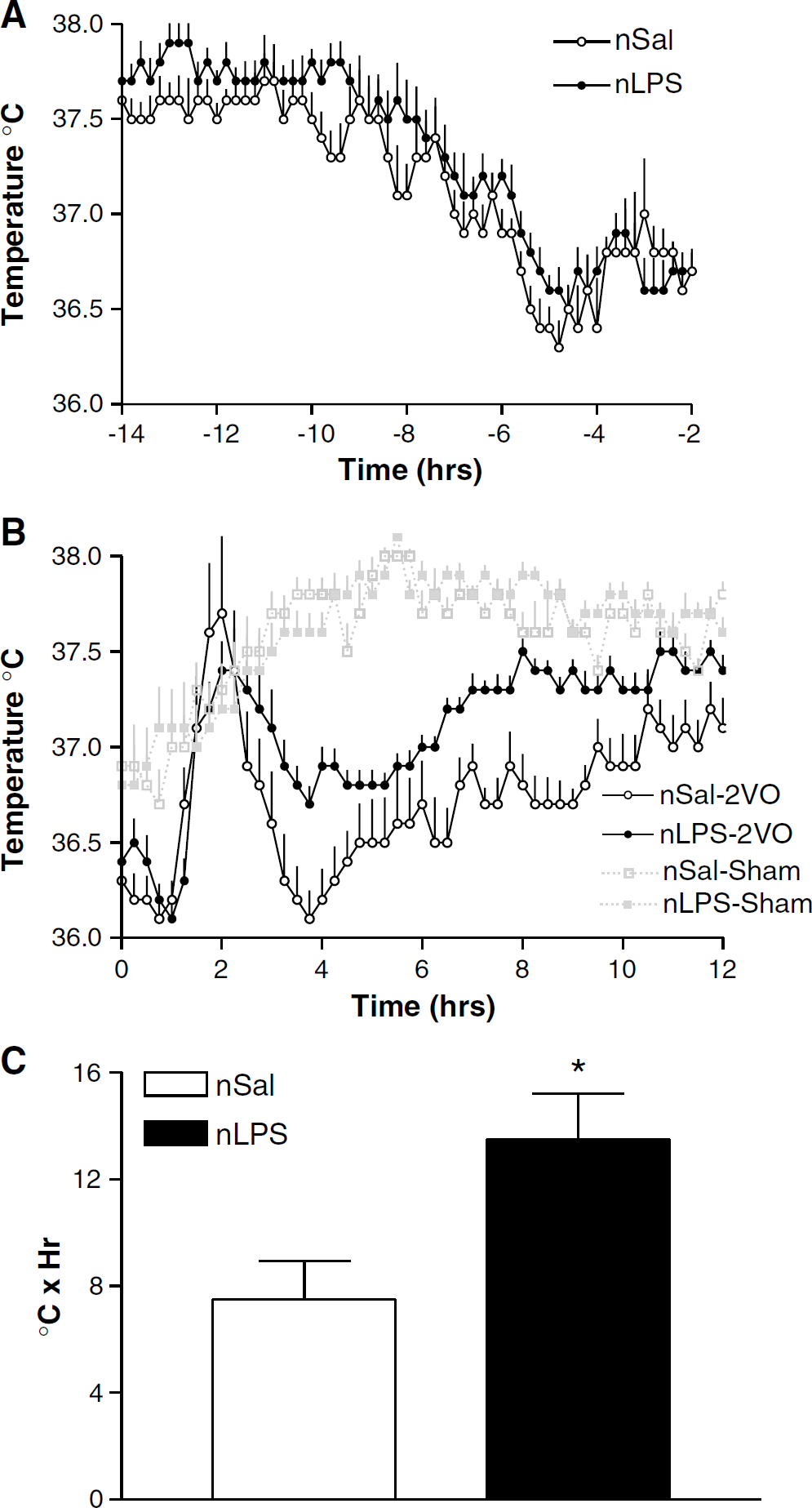
Body temperatures in animals treated as neonates with saline (nSal:
Sham surgery had little effect on body temperature in either group of rats. The febrile responses to the ischaemic injury, however, did differ depending on the neonatal treatment to which the animals were exposed. Both the neonatally salinetreated and the neonatally LPS-treated animals showed a sharp 1.3°C to 1.5°C increase in temperature immediately after surgery, peaking at 1 h after the end of surgery and returning towards baseline by 4 h after surgery (Figure 3B). A comparison of the fever indices revealed that the body temperatures of the neonatally salinetreated rats were maintained significantly lower than the neonatally LPS-treated animals for up to 12 h after the ischaemic injury (P < 0.05, Student's unpaired t-test; Figure 3C), and both groups sustained their body temperature lower than the sham-treated animals for the same period (Figure 3B).
At 13 h after the 2VO, the body temperatures of the 2VO rats were again not different and were no longer different from those of the sham-treated animals. Temperatures and their usual circadian rhythms were maintained until the end of the experiment (data not shown).
Cell Counts
Hippocampus: In sham-treated animals, similar numbers of pyramidal cells were found in all regions of the hippocampus assessed, irrespective of the neonatal treatment they had received (Figures 4A, 5A, and 5B). The 2VO was associated with reduced numbers of intact pyramidal cells in the CA1 region of the hippocampus in both the neonatally salinetreated and neonatally LPS-treated rats. In both cases, numbers of healthy cells were reduced by approximately 50% in ischaemic animals compared with their sham-treated counterparts (Figure 4). A slight reduction in numbers of intact cells was also seen in the CA3 and dentate gyrus subfields of the hippocampus in ischaemic animals. This cell loss was less substantiative than that seen in the CA1 region but was also independent of the neonatal treatment received (Figure 5).
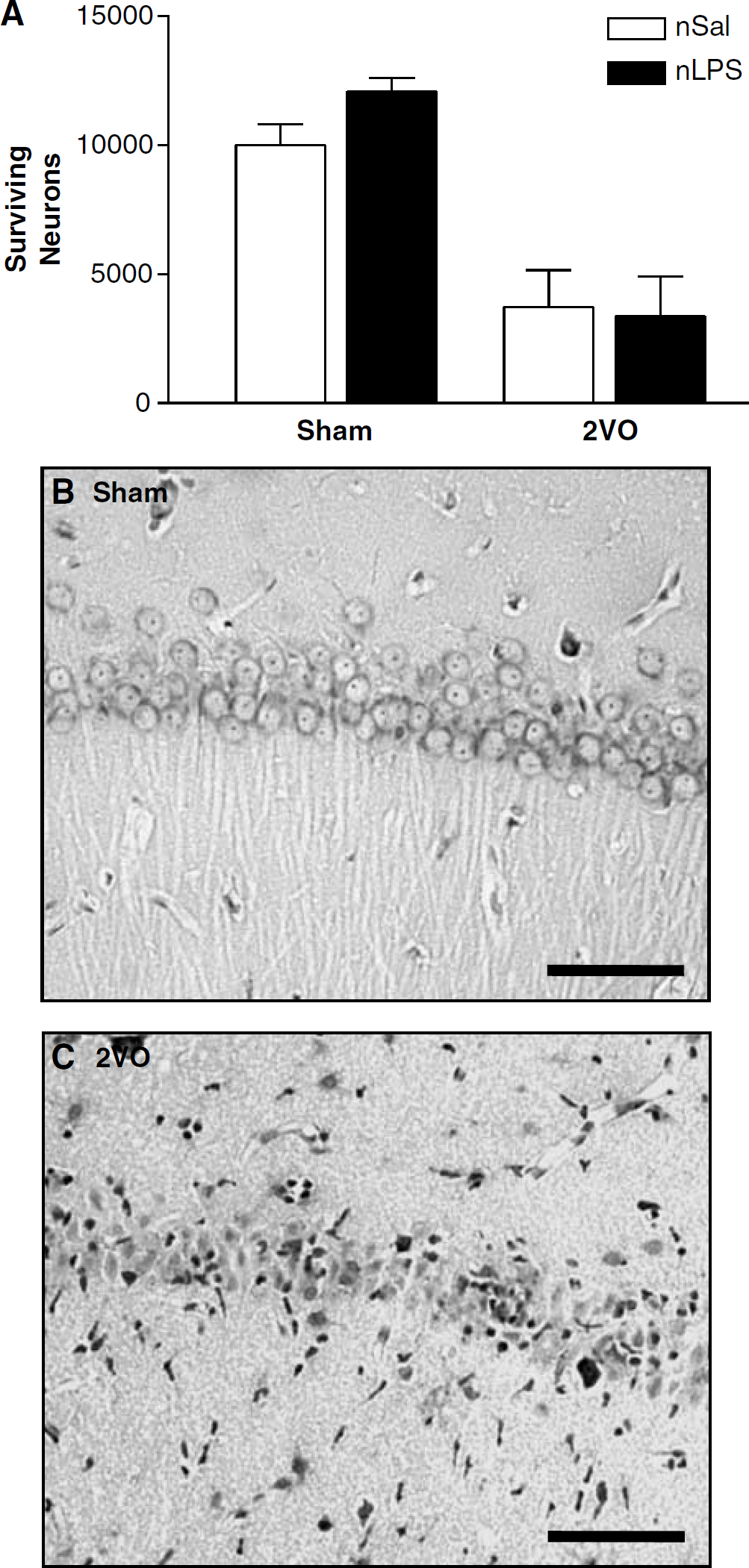
Surviving CA-1 hippocampal pyramidal cells 7 days after the two-vessel occlusion (2VO). (
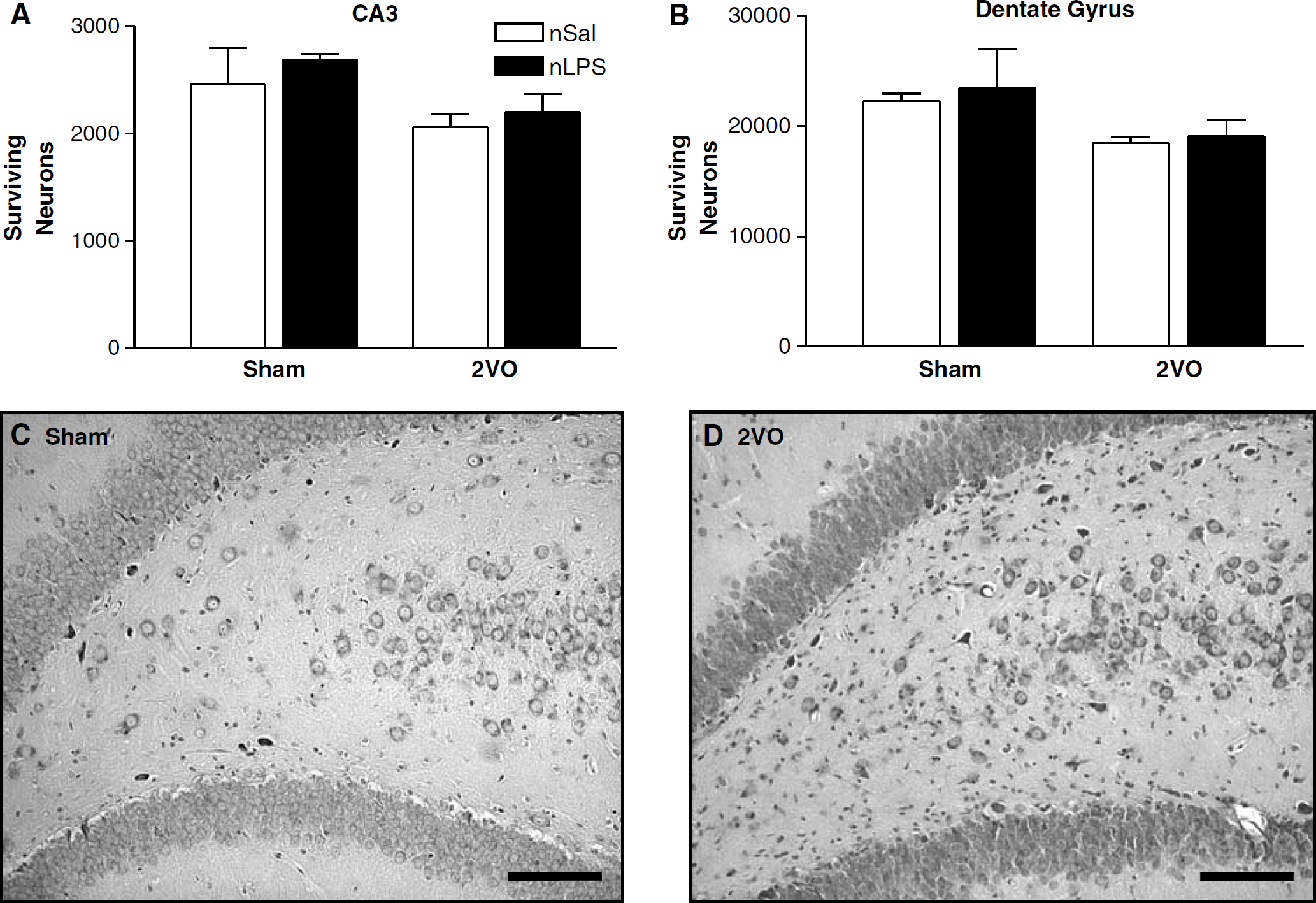
Surviving CA-3 and dentate gyrus hippocampal pyramidal cells 7 days after the two-vessel occlusion (2VO). (
Central amygdala: The finding of altered object exploration as a factor of neonatal treatment in the behavioural tests led to an evaluation of the damage in the CeA. As with the hippocampus, similar numbers of cells were found in this region in the sham-treated animals irrespective of the neonatal treatment they had received (Figure 6). Interestingly, the amount of ischaemic damage seen after the 2VO in the CeA was dependent on the neonatal treatment received, as was shown by a significant effect with a Student's unpaired t-test. Thus, neuronal cell loss was more pronounced in the animals that had received LPS as neonates compared with their neonatally salinetreated counterparts (t= 2.916, df = 8, P < 0.05; Figure 6).
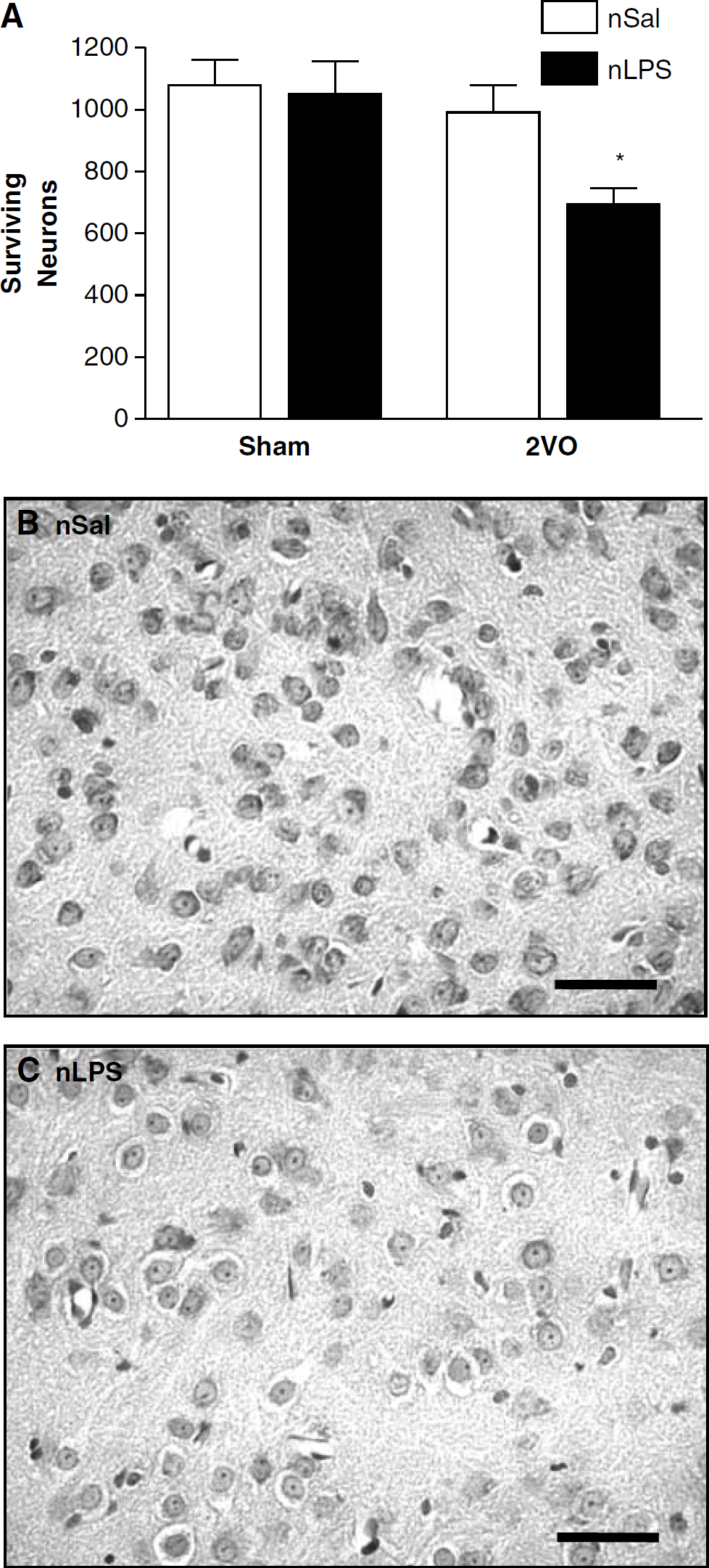
Surviving CeA neurons 7 days after the two-vessel occlusion (2VO). (
Discussion
The results of the present study highlight the importance of the early period of development and the impact it can have on an animal's ability to deal with injury in later life. We have shown that rats treated as neonates with a model of bacterial infection, in what is, to our knowledge, the longest preconditioning paradigm heretofore reported in association with stroke, show altered temperature and behavioural responses to brief forebrain ischaemia in adulthood, as well as altered neuronal cell death compared with neonatally salinetreated control rats.
Body temperatures of rats treated as neonates with saline were approximately 1°C lower than those of neonatally LPS-treated rats for at least 12 h after the 2VO to induce ischaemia. In addition, the severely reduced object exploration displayed by the neonatally LPS-treated animals before ischaemia was spectacularly reversed 1 day after the induction of cerebral ischaemia. When numbers of surviving neurons were examined in the CA1, CA3, or dentate gyrus regions of the hippocampus, no differences were seen between the two neonatal treatments. It therefore seemed that an immune challenge experienced as a neonate did not alter neuronal survival after cerebral ischaemia. However, when we also examined the CeA, a region intimately involved in fear and anxiety responses to novel situations (Sah et al, 2003), we found that neonatally LPS-treated animals had significantly more damage in this region than the neonatally salinetreated controls.
Effects of Neonatal Immune Challenge on Behaviour after Ischaemia in Adulthood
The outcome of the object exploration test, together with the rats' performances in the open field paradigm allows us to make inferences about some aspects of the rats' behaviour. The first-trial reluctance of the neonatally immune challenged animals to approach the introduced objects compared with neonatally salinetreated rats is a robust and very interesting finding. Intriguingly, it implies that an occurrence as simple and common as an infection experienced during a critical period of development has lasting effects on an animal's psychological responses to novel situations. While the novel object exploration test is generally used as a test for memory, it should be emphasised, as noted by Roy and Chapillon (2004), that lack of exploration of a novel object in a task such as this may not necessarily reflect impairment of memory processes, but may also have a strong psychological reactivity component. These results may therefore indicate that these neonatally LPS-treated animals respond to novel situations in a more passive and fearful manner.
Importantly, the cerebral ischaemia induced by the 2VO had a pronounced effect on the rats' behavioural responses to the novel object exploration task. Whereas in neonatally salinetreated animals, the 2VO had no effect on object exploration behaviour, the ischaemia did have a marked effect on the neonatally LPS-treated animals. Previously minimally exploring animals explored, after ischaemia, at least as much as their salinetreated counterparts had in the first trial. Thus, it is evident that the psychological mechanisms that were altered in these animals by a neonatal infection are profoundly sensitive to an ischaemic insult.
The present results could be interpreted as the neonatal immune challenge having some effect on the animals' ability to recover acutely from the cerebral ischaemia. The hypothermia that was observed in the neonatally LPS-treated rats in the 12 h after the cerebral ischaemia was not as pronounced in the neonatally salinetreated rats, perhaps indicating a better ability in these animals to regulate temperatures at normal levels after the surgery. However, as both groups of animals displayed similar levels of activity in the open field tests, other factors are likely to be involved in these behavioural differences.
Effects of Neonatal Immune Challenge on Temperature After Ischaemia in Adulthood
One of the most interesting findings of the present investigation is the elevated body temperatures of the neonatally LPS-treated animals relative to the neonatally salinetreated ones after the 2VO. It is possible that this comparatively smaller hypothermia may have contributed to, or caused, the reduced CeA cell survival and the behavioural differences seen in the neonatally LPS-treated animals. It has previously been established that hypothermia maintained for 24 h or longer after cerebral ischaemia can be neuroprotective (Corbett et al, 2000; Colbourne et al, 2003). The shorter duration of the hypothermic response to the 2VO in the present study may account for the lack of differential neuroprotection between LPS and saline pretreated animals in the CA1 and CA3 regions of the hippocampus. However, few investigations examined brain regions other than these divisions of the hippocampus and perhaps there are other areas that are more sensitive to changes in temperature, even over the short term. As discussed in the Introduction, hyperthermia has been shown to contribute substantially to long-term neuronal cell loss after ischaemia (Kuroiwa et al, 1990; Baena et al, 1997; Ginsberg and Busto, 1998). We have previously shown that animals treated as neonates with LPS have elevated basal levels of COX-2, the rate-limiting enzyme in the production of the pyrogenic prostaglandin E2, at least in the hypothalamus (Boisse et al, 2004), and it is possible that an elevated basal COX-2 may have, in the present investigation, led to a smaller reduction in postischaemic temperatures, and to enhanced neuronal cell loss. Indeed, elevated COX-2 has been associated, in transgenic mice constitutively expressing human COX-2 in the striatum, cerebral cortex, and hippocampus, with increases in infarct size after focal ischaemia (Dore et al, 2003), while COX-2 inhibitors can protect against the effects of ischaemia (Candelario-Jalil et al, 2005).
Effects of Neonatal Immune Challenge on Neuronal Survival After Ischaemia in Adulthood
One of the brain regions most sensitive to cerebral ischaemia in rodent models and humans is the hippocampus, particularly the CA1 subdivision (Pulsinelli et al, 1982; Colbourne et al, 1999). The present investigation highlights that the usual focus on CA1 pyramidal neurons may fail to disclose important information on alterations in neuronal cell death elsewhere in the brain. In the present investigation, although substantial CA1 cell death was seen, verifying the efficacy of the manipulation, no differences were seen between the neonatally saline- and LPS-treated groups. We therefore thought it pertinent to examine cell survival in brain regions more closely involved in the kinds of behavioural changes seen in the present study.
The amygdala, particularly the CeA, is known for its role in governing reactions to psychological situations and plays a critical role in the processing of fear reactions (Sah et al, 2003; LeDoux, 2003). Thus, it is consistent with this role that the ischaemia-sensitive behavioural differences seen between the neonatally salinetreated and neonatally LPS-treated animals, reflecting more fearful behaviour in the latter group, were associated with differences in surviving cell numbers in the CeA.
Other investigations have provided indications of altered amygdala excitability after cerebral ischaemia. For instance, Fos expression is elevated in the CeA after a coronary artery occlusion (Xie et al, 2000). Focal cerebral ischaemia has been associated with acutely elevated corticotrophin-releasing hormone (CRH) release and mRNA in the amygdala (Wong et al, 1995; Khan et al, 2004) with a corresponding decrease in intracellular CRH (Khan et al, 2004), as well as changes in other neurotransmitters (Allen et al, 1995). Also, expression of voltage-gated K+ channels is increased in the rat amygdala after a transient focal ischaemia (Chung et al, 2001), providing a possible molecular basis for altered neuronal excitability in the region. The amygdala has also previously been shown to undergo long-term changes in response to a neonatal challenge. For instance, changes in amygdala synaptic regulatory proteins have been seen after a neonatal infection with influenza A virus (Beraki et al, 2004). While it is unclear as to how the neonatal LPS treatment might alter the brain such that regions like the CeA are permanently affected, it is possible that such selective action may occur via activation of the hypothalamic–pituitary–adrenal axis, which would alter glucocorticoid actions on various brain regions. Alternatively, cytokines such as interleukin-1β are activated in the brain after LPS exposure (van Dam et al, 1998) and could lead to long-term alterations in N-methyl-
It is possible that a more sensitive CeA, such as that which may arise as a result of selective synaptic remodelling after a neonatal immune challenge, would be more easily excitable after an ischaemic insult affecting the region. This hyperexcitability may result in enhanced release of potentially neurotoxic neurotransmitters such as glutamate or CRH (Lyons et al, 1991; Strijbos et al, 1994; Khan et al, 2004) that may account for the enhanced CeA cell death seen in the neonatally LPS-treated animals after ischaemia. The enhanced exploration of the novel objects seen 1 day after the bilateral carotid artery occlusion would therefore occur as a result of a subtle decrease in anxiety because of a decreased excitatory output.
Conclusion
The present investigation has shown that exposure to an inflammatory challenge as a neonate can alter neuronal survival, at least in the CeA, as well as body temperatures and behavioural responses after cerebral ischaemia in adult rats. These results may have relevance to other neurologic disorders that have been associated with exposure to inflammation early in development (Eklind et al, 2001). More investigation will be necessary to determine in more detail how the amygdala and other relevant brain regions are affected by challenges to the young animal, and the implications this has for the adult.
Footnotes
Acknowledgements
We thank Mio Tsutsui and Svetlana Farkas for technical assistance.
