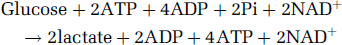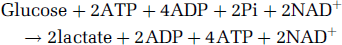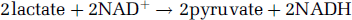Abstract
Research over the past two decades has renewed the interest in lactate, no longer as a useless end product of anaerobic glycolysis in brain (and other tissues), but as an oxidative substrate for energy metabolism. While this topic would be considered blasphemy only three decades ago, much recent evidence indicates that lactate does play a major role in aerobic energy metabolism in the brain, the heart, skeletal muscle, and possibly in any other tissue and organ. Nevertheless, this concept has challenged the old dogma and ignited a fierce debate, especially among neuroscientists, pitting the supporters of glucose as the major oxidative energy substrate against those who support lactate as a possible alternative to glucose under certain conditions. Meanwhile, researchers working on energy metabolism in skeletal muscle have taken great strides toward bridging between these two extreme positions, while avoiding the high decibels of an emotional debate. Employing their findings along with the existing old and new data on cerebral energy metabolism, it is postulated here that lactate is the only major product of cerebral (and other tissues) glycolysis, whether aerobic or anaerobic, neuronal or astrocytic, under rest or during activation. Consequently, this postulate entails that lactate is a major, if not the only, substrate for the mitochondrial tricarboxylic acid cycle. If proven true, this hypothesis could provide better understanding of the biochemistry and physiology of (cerebral) energy metabolism, while holding important implications in the field of neuroimaging. Concomitantly, it could satisfy both ‘glucoseniks’ and ‘lactatians’ in the ongoing debate.
Keywords
Introduction
The controversy surrounding the role lactate plays, if any, in the adult brain is as ferocious as any other past or present scientific controversy. For many years, most scientists agreed that lactate is a useless end product of anaerobic energy metabolism, which at times can become harmful. These notions have manifested, among others, the concept that lactate accumulation in muscle tissue is responsible for muscle fatigue, a concept that has recently been refuted (Pedersen et al, 2004). In the brain, lactate has been promoted as a major exacerbating factor of cerebral ischemic damage. Lactate's bad reputation could explain why an important body of research on the oxidation of this monocarboxylate in the brain has all but been ignored, and especially why recent evidence that indicates lactate to be an important cerebral oxidative energy substrate has met with great skepticism. A hypothesis proposed by Magistretti and colleagues (Pellerin and Magistretti, 1994), known as the astrocyte—neuron lactate shuttle hypothesis (ANLSH), has added much fuel to the ongoing debate (Chih et al, 2001; Chih and Roberts, 2003; Pellerin and Magistretti, 2003; Hertz, 2004). Recently, this very hypothesis awakened another old controversy regarding the role of hypotheses in scientific research (Kimelberg, 2004; Pellerin and Magistretti, 2004b).
Nonetheless, the present paper advances a new hypothesis on the role of lactate in cerebral (and other tissues) energy metabolism. In constructing the hypothesis, I have relied on studies, old and new, that were conducted both in the brain and other tissues. It seems that, ‘de facto,’ there is greater agreement among scientists today on the role of lactate in energy metabolism than meets the eye. The division, at least among neuroscientists, seems to focus mainly on whether lactate plays as important a role in cerebral oxidative energy metabolism as does glucose. If one believes that it is not, any hypothesis that promotes lactate as a cerebral oxidative energy substrate, such as the ANLSH, would be objectionable. It is my belief that the debate over ‘which is more important’ has distracted many from realizing a more significant possibility, namely, that both glucose and lactate are integral and important entities of energy metabolism.
Background Considerations
Much of our knowledge on the formation and fate of lactate originates from early studies on muscular exercise (Hill et al, 1924). The investigators involved in those studies have also coined most of the terminology associated with tissue lactate and are responsible in large part for the conceptions and misconceptions regarding lactate, some of which continue to persist today. As late as 1958, one can find statements such as ‘…the end-product of reduction in the lactate dehydrogenase (LDH) system is lactate, which has no other function in metabolism’ (Huckabee, 1958a) or ‘The LDH system, however, is unique in being ‘dead-end;’ endogenous lactate does not participate in any other reaction which would be affected by its accumulation in the cell’ (Huckabee, 1958a). ‘This assertion,’ noted Huckabee, ‘is not dependent solely on the failure to discover any other lactate reaction in mammalian tissues up to the present time, but is confirmed by the close parallelism of specific activities of lactate and pyruvate from living tissue following exhibition of isotope-tagged pyruvate.’
However, a large body of work on brain energy metabolism, published in the late 1920s and the early 1930s, had been completely ignored over the years. As one reviews it, one should remember that the glycolytic pathway had not been completely elucidated until 1940 and that the mitochondrial tricarboxylic acid (TCA) cycle had not been proposed by Krebs and Johnson until 1937. Nevertheless, the fact that the TCA cycle was formulated 3 years before the formulation of the glycolytic pathway could contribute, a priori, to the prevailing concepts regarding the roles of certain substrates and products of the glycolytic pathway (see below). The most active investigators in the area of nervous system metabolism during that early period were Holmes and Ashford. These two researchers published several studies on the oxidation of glucose and especially lactate in brain tissue. In one of their earlier studies (Ashford and Holmes, 1929), they concluded that brain tissue possesses two mechanisms of lactate formation: ‘one, involving glucose, is quantitatively the more important, and is independent of phosphate; the other is much smaller, involves glycogen, and depends on the availability of phosphate.’ Approximately a year later, these two pioneers published detailed studies on the oxidation of lactic acid in the brain (Holmes and Ashford, 1930; Ashford and Holmes, 1931). In these studies, they related to what was known then as the ‘Meyerhof quotient,’ the ratio [Total lactic acid disappeared]/[Lactic acid oxidized], which normally has a value greater than unity. Meyerhof theorized that, in muscle tissue, any lactic acid disappearance above the amount equivalent to oxygen used is directed toward carbohydrate synthesis (see refs. to Meyerhof's work in Holmes and Ashford, 1930). However, Holmes and Ashford were able to show ‘that, although the ’Meyerhof quotient’ is readily obtainable under their experimental conditions, there is no synthesis of carbohydrate from that portion of the lactic acid which disappears but is not accounted for by O2 uptake.’ Moreover, they found that oxygen uptake in the presence of glucose was inhibited by NaF, while such uptake in the presence of lactic acid was insensitive to fluoride (Holmes, 1930). Thus, they were the first to show aerobic utilization of lactate in brain tissue, that is, a correlation between lactic acid disappearance and oxygen consumption. It is even more telling how advanced in their studies these two investigators were by their demonstration that O2uptake by lactate-supplemented brain tissue is greater in the presence of a CO2-bicarbonate buffer than in the presence of phosphate buffer and that this uptake increases with increased oxygen tension in both cases (Ashford and Holmes, 1931). They also showed that the respiratory quotients, both of brain tissue with and without lactate supplementation, are close to unity, even when animals from which the brain tissue was prepared were rendered hypoglycemic by insulin injection (Holmes, 1930; Ashford and Holmes, 1931). Moreover, they found that glucose, lactate, succinate and glyceropho-sphate all act as hydrogen donors to methylene blue, while glycerol and dihydroxyacetone were without effect on the reduction time, while formate, acetate and mandelate prolonged it. They also concluded that lactate oxidation is unlikely to spare the utilization of another substrate (Ashford and Holmes, 1931). One may find this conclusion somewhat bewildering, if one is to assume that lactate utilization replaces that of glucose. However, in vivo, lactate originates from glucose and the oxidation of the former should drive the utilization of the latter, rather than sparing it. Sparing of glucose may occur only when lactate (or pyruvate) is supplied exogenously in sufficiently high levels directly into the brain, bypassing the liver. In addition, these investigators were not equipped with the luxury of experimenting with two different substrates (glucose and lactate) concomitantly to directly assess whether the addition of lactate to their preparation could spare the utilization of glucose or vice versa. Thus, their conclusion should be looked upon mainly as an estimate.
Obviously, Holmes and Ashford were not aware of the, yet nonexistent, notion that under aerobic conditions glucose is supposed to be reduced to pyruvate, not to lactate. The concept of pyruvate as the end product of aerobic glycolysis was formulated with the introduction of the glycolytic pathway. The most plausible explanation for the decision of the pathway formulators to place pyruvate as its aerobic end product had to do with the introduction of the TCA cycle three years earlier, where pyruvate was proposed to play a central role in the cycling formation of citrate from oxaloacetate and acetyl-SCoA (Krebs and Johnson, 1937). Clearly, many biochemists of the period were already familiar with the TCA-cycle requirement for pyruvate as its key substrate. Glycolysis was, and still is, the best source of pyruvate supplies in sufficient quantities. While there were isolated reports regarding lactate conversion to pyruvate (Peters, 1936), most studies had shown the opposite reaction, namely, the production of lactate from glucose, fructose, hexosediphosphate or pyruvate (Flock et al, 1938; Bueding and Goldfarb, 1943; Friedemann et al, 1945; Meyerhof and Geliazkowa, 1947; Meyerhof and Wilson, 1947, 1948).
Thus, more than six decades after the introduction of the first enzymatic pathway, an event that launched the era of modern biochemistry, glycolysis is now being taught in grade school. As it is described in numerous text books, the glycolytic pathway begins with the phosphorylation of glucose and ends with the formation of lactate (reaction I),
unless oxygen is present, in which case glycolysis stops short of its last enzymatic step, namely, LDH, ending with pyruvate, the substrate that enters the mitochondrial TCA cycle (reaction II):
Although exceptions to the aerobic sequence have been documented, where aerobic glycolysis proceeds to form lactate, such as in red blood cells, heart muscle and retina, the prevailing notion even today is that, under ample oxygen supplies, pyruvate, not lactate, is the final product of glycolysis. Notwithstanding, evidence has been accumulated over the past half-century, which indicates that lactate is a suitable aerobic energy substrate for brain tissue (McIlwain, 1953; Ghosh and Quastel, 1954; Ide et al, 1969; Korinkova and Lodin, 1976; Schurr et al, 1997a, b , 1999; Schurr and Rigor, 1998; Fellows et al, 1993; Izumi et al, 1994, 1997; Larrabee, 1995, 1996; Tabernero et al, 1996; Wada et al, 1997; Hu and Wilson, 1997; Qu et al, 2000; Bliss and Sapolsky, 2001; Takata et al, 2001; Smith et al, 2003; Bouzier-Sore et al, 2003). This evidence should at least raise some doubt regarding the notion that lactate is a dead-end product. Moreover, a recent human study by Dalsgaard et al (2004) has indicated that lactate, produced during exercise, is taken up and metabolized by the brain.
Nevertheless, the widely accepted dogma that, aerobically, pyruvate, not lactate, is the final product of glycolysis (reaction II) is both chemically and thermodynamically flawed. Thermodynamically, glycolysis should proceed to completion, that is, lactate formation, since pyruvate conversion to lactate yields free energy (ΔG0‘ = −6kcal/mol). Chemically, glycolysis should proceed to form lactate, since the LDH reaction regenerates nicotenamide adenine dinucleotide (NAD +) by oxidizing its reduced form (NADH) formed during the glyceraldehyde phosphate (GAP) dehydrogenase reaction and thus assuring the cyclical nature of glycolysis (reaction III).
Furthermore, the assumption that under aerobic conditions pyruvate conversion to acetyl-SCoA, a reaction that takes place in the mitochondrion and requires NAD +, is somehow thermodynamically preferable over pyruvate conversion to lactate is unsubstantiated. This assumption would have some basis if the glycolytic enzymes, substrates and products were, as was believed for many years, floating freely in a cytosol that behaves like an aqueous medium.
In his famous book, Bioenergetics, Lehninger (1971) generalized on aerobic glycolysis by stating that either lactate or pyruvate is oxidized to CO2 and H2O. However, in dealing with the intracellular organization of the glycolytic system (p. 69), he stated: ‘the eleven enzymes which catalyze the glycolytic sequence exist free in solution in the soluble portion of the cytoplasm, at least in most cells. They are apparently not grouped or arranged in an intracellular structure, as is the case for the much more complex enzyme systems responsible for respiration and photosynthesis.’ Clearly, it did not occur to many great biochemists that the assumption regarding the glycolytic sequence of reactions and enzymes as existing in free solution might not necessarily reflect the in situ situation. Thus, the prevailing notion, even today, is that the glycolytic reactants and their enzymes flow freely in the cytosol. Only such notion would explain the classic representation of a branched glycolytic chain, where one substrate/product, pyruvate, does not behave according to the rules of thermodynamics in the presence of oxygen, and is changing direction in search of a mitochondrion rather than to proceed to its natural last step in the pathway, the LDH reaction.
Nonetheless, Lehninger's statement, as cited above, is somewhat surprising, considering that 6 years before the publication of his book questions had been raised regarding the validity of the concept that the glycolytic pathway exists free in solution. Green et al (1965) concluded from their studies of red blood cells and yeast ‘that in the intact cell the complete glycolytic complex of enzymes is associated with the plasma membrane and is not in free solution.’ Schrier (1966), concomitantly working on the erythrocyte, arrived at the same conclusion. Knull (1978), working on rat brain tissue, showed that the glycolytic enzymes are all associated with particulate fractions of lysed nerve endings. Clegg (1984), in his excellent opinion paper, concluded that there is ‘an intimate connection between cellular architecture and most, and possibly all, of the metabolic machinery. These relations are dynamic and under tight control, and their disruption leads to malfunction and loss of regulation.’ Consequently, lactate formation from pyruvate takes place within an aggregate of glycolytic enzymes, while pyruvate conversion to acetyl-SCoA, the initial step in the TCA cycle, takes place in the mitochondrion, a conversion that first requires the transport of the monocarboxylate into this organelle via its inner membrane. Moreover, the existence of mitochondrial LDH is well established, and lactate has been shown to be as good as, if not better than, pyruvate as a substrate for the mitochondrial TCA cycle (Kline et al, 1986; Brandt et al, 1987; Brooks et al, 1999).
Interestingly, in their study of mitochondrial preparation from different tissues, Brandt et al (1987) placed a large part of the mitochondrial LDH inside the mitochondria. They distinguished it from cytosolic LDH, characterized by them as LDH5 (see also below), which preferentially adheres to the outer mitochondrial membrane. Of all the different tissues from which these investigators isolated mitochondria, including the heart, kidney, liver lymphocytes and brain, only the latter's mitochondrial preparations were contaminated to a certain extent with three glycolytic enzymes, phosphoglucomutase, phosphoglucose isomerase and 3-phos-phoglycerate kinase (Brandt et al, 1987).
Hence, aerobic glycolysis, when coupled to the mitochondrial TCA cycle, would be better described by reactions IV and V as follows:
In other words, glycolysis proceeds to produce lactate because it is the most feasible path thermodynamically, chemically and spatially.
There is also the issue of tissue levels of lactate and pyruvate. In the early years, after the formulation of the glycolytic pathway, many studies dealt with this issue, in which both monocarboxylates and their ratio were measured in human blood at rest, during exercise, after a meal, after administration of pyruvate, lactate or glucose and in various pathological states (Bueding et al, 1942; Bueding and Goldfarb, 1943; Friedemann et al, 1945; Goldsmith, 1948; Huckabee, 1958a, b ). First, pyruvate levels are always significantly lower than those of lactate and thus the measured ratio [lactate]/[pyruvate] is anywhere from 7.0 to 25.0, depending on the conditions under which measurements are made. The low end of this ratio usually associates with pathological situations.
Flock et al (1938) showed the conversion of perfused pyruvate to lactate in the dog. Huckabee (1958aa), in his attempt to understand the relationships of pyruvate, lactate and oxygen, suggested that these relationships can be written as follows:
The above equation illustrates the dependence of lactate formation on pyruvate and the constant K × [NADH]/[NAD +;]. Thus, under fully oxygenated conditions, when the ratio [NADH]/[NAD +] is held, more or less, constant, any change in pyruvate concentration should bring about a corresponding change in lactate concentration, that is, an increase in [pyruvate] would follow immediately by a similar increase in [lactate] without any change in oxygen consumption. Of course, these relationships hold true as long as blood pH remains unchanged. Hence, an increase in glycolytic flux under fully oxygenated conditions should result in increased pyruvate and a concomitant increase in lactate concentration, keeping the ratio [lactate]/[pyruvate] constant. According to the above equation and the high ratio [lactate]/[pyruvate] that is normally maintained, it is clear that even under fully aerobic conditions, lactate is the end-product of glycolysis.
Curiously, Huckabee (1958a) did not consider in his calculations two important points: (a) localized changes in oxygen concentration, which could lead to changes in the ratio [NADH/[NAD +] and (b) a decrease in the concentration of lactate that would drive a decrease in [NADH]. These points are elaborated on later in this monograph.
Several of the above issues, as they relate to energy metabolism in skeletal and heart muscle and the putative role of lactate in this process, are elegantly detailed in a recent review by Gladden (2004).
The Hypothesis
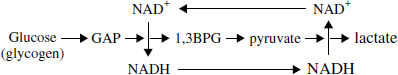
Based on the preceding considerations, it is hypothesized that in the brain, and most likely in many other tissues, whether at rest (does the brain ever rest‘) or during activation, both under aerobic and anaerobic conditions, glycolysis always proceeds to its final step, the LDH reaction and the formation of lactate. Consequently, this hypothesis entails that lactate, not pyruvate, is the oxidative substrate for the mitochondrial TCA cycle in situ (Figure 1).
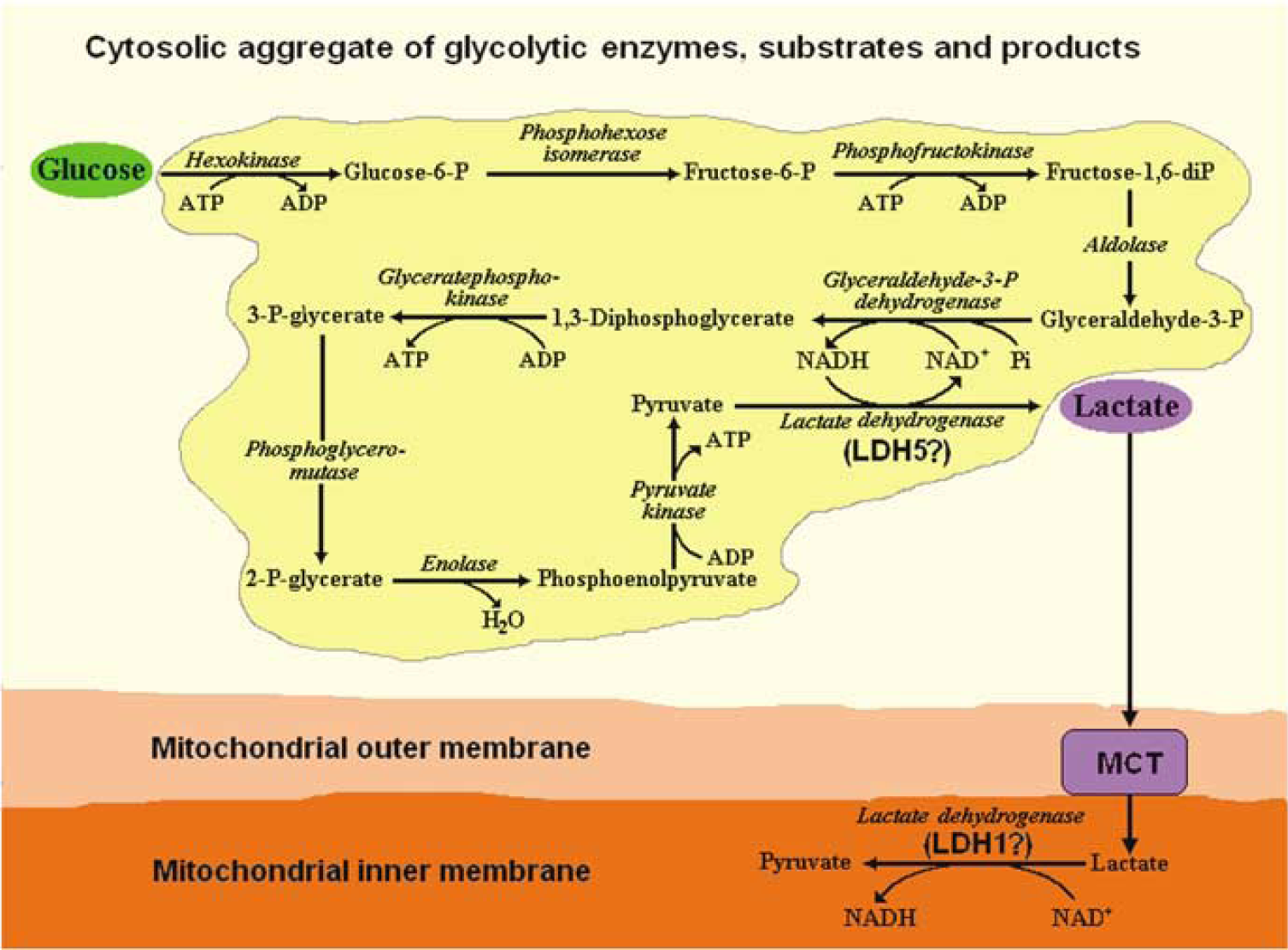
A schematic depiction of a hypothetical arrangement of the glycolytic pathway aggregate with all its enzymes, substrates and products. According to the hypothesis put forward in this paper, glycolysis, whether aerobic or anaerobic, always proceeds to its last step, the formation of lactate, the real in situ substrate for mitochondrial respiration. The last enzymatic step in the cytosolic glycolysis is LDH composed generally of the isoform that catalyzes the conversion of pyruvate to lactate (LDH5?). In contrast, the isoform that converts lactate to pyruvate (LDH1?) is the first enzyme of the mitochondrial oxidative pathway, supplying pyruvate to the tricarboxylic acid cycle.
It is important to clarify that investigators working on energy metabolism of skeletal and heart muscles have toyed with the idea that lactate is the product of aerobic glycolysis, which, on its formation and after its shuttling either intracellularly or extracellularly from its place of formation, is being oxidized in the mitochondrion (Brooks, 1985, 1998; Brooks et al, 1999; Van Hall, 2000; Chatham et al, 2001; Gladden, 2004). Nonetheless, the hypothesis, as presented here, namely, that cerebral glycolysis always ends with lactate formation has not been presented before.
Testing the Hypothesis
In his review, Gladden (2004) lists some of the key concerns/questions regarding the ANLSH. While these questions relate mainly to the validity of the ANLSH per se, many of them are dealing in essence with the issue of lactate as a major oxidizable energy substrate. The hypothesis forwarded here is not aimed to validate or discredit the ANLSH, but rather to provide a feasible explanation to the overwhelming amount of data accumulated over the past two decades, indicating that lactate is an important oxidizable energy substrate in the brain.
Since the ANLSH has become a central issue in the debate over the role of lactate in energy metabolism, it would be appropriate to briefly describe the ANLSH for those who are not immersed in the ongoing debate. According to the ANLSH, on neuronal activation by glutamate, astrocytes take up presynaptically released glutamate from the synaptic cleft through specific Na+-dependent glutamate transporters. The resulting intracellular increase in astrocytic [Na+] induces Na—K–ATPase activity aimed at pumping out the extra Na+. The increased demand for ATP by the pumping activity activates astrocytic glycolysis and thus the production of lactate. The mounting astrocytic lactate levels are then transported via MCTs, found in both astrocytic and neuronal membranes, first to the extracellular space and then into neighboring neurons, where it is oxidized in the mitochondria. Moreover, it has been suggested that neurons exclusively contain the LDH1 isoform, while astrocytes contain both LDH1 and LDH5 (Pellerin and Magistretti, 1994; Tsacopoulos and Magistretti, 1996; Bittar et al, 1996; Magistretti, 1999, 2000; Magistretti and Pellerin, 1999; Bouzier-Sore et al, 2002).
Recently, the ANLSH has been revised (Pellerin and Magistretti, 2004a) and thus not all the details above are updated. Nevertheless, it should be reemphasized that much of the criticism of the ANLSH, whether in its original or revised format, is directed toward the notion of lactate as a major aerobic energy substrate. As such, any hypothesis that promotes that notion would not be spared from such criticism.
The relevancy of the distribution of the two LDH isoforms, LDH1 and LDH5, between neurons and astrocytes, respectively, has been questioned by the ANLSH critics, since the LDH reaction is near equilibrium and thus the isoform that carries out this reaction might have only a small effect on the reaction's flux in vivo (Newsholme, 2003). Although the relevancy of the distribution of LDH isoforms between neurons and astrocytes is yet to be determined, an intracellular distribution, where LDH1 is the mitochondrial isoform and LDH5 is the cytoplasmic one, could be an important part of the premise of the present hypothesis and thus relevant. In their review, Chih and Roberts (2003) clarify that ‘glycolysis refers to the conversion of glucose to pyruvate under aerobic conditions and anaerobic glycolysis refers to the conversion of glucose to lactate,’ the classical definition of glycolysis. These authors reject lactate as a significant oxidative energy substrate in situ due to what they have termed ‘thermodynamic feasibility for conversion of lactate to pyruvate’ (Chih and Roberts, 2003). Accordingly, in the initial stages of neural activation, as the glycolytic flux increases, pyruvate levels rise, and the cytosolic NADH/NAD+ ratio increases. These changes arguably drive the LDH-catalyzed reaction toward lactate production rather than lactate use, making lactate oxidative utilization unlikely (Chih and Roberts, 2003). However, this argument contradicts their own ‘classical’ description of aerobic glycolysis and is actually in full agreement with the present hypothesis that the increased glycolytic flux due to higher energy demands would result in increased aerobic lactate production. More importantly, if one accepts the premise of the present hypothesis that the glycolytic (cytosolic) LDH(5?) always catalyzes the hydrogenation of pyruvate to lactate, while mitochondrial LDH(1?) is the enzyme that dehydrogenates lactate to pyruvate, then, there should be no conflict between glycolytic lactate formation and concomitant mitochondrial lactate utilization, whether the lactate originates intracellularly or extracellularly. Moreover, there should not be any problem with the NADH/NAD+ ratio, since the cytosolic LDH-cata-lyzed formation of lactate also assures the oxidation of NADH and the cyclical nature of glycolysis. NAD + reduction to NADH during lactate oxidation takes place in the mitochondrion and should have little or no effect on cytosolic glycolysis. The work of Brooks and his colleagues (Brooks, 1985, 1998, 2000, 2002a, b ; Brooks et al, 1999), using isotopic tracers, strongly implies that glycolytically produced pyruvate is predominantly converted to lactate, not to acetyl-CoA, while lactate, supplied exogenously, is preferentially metabolized to acetyl-CoA. Brooks (1985) was the first to propose the existence of an intracellular lactate shuttle, which by no means supports or refutes the ANLSH.
It has been pointed out that the ANLSH applies only to glutamatergic neurons and does not address how activity-related energy demands are met in nonglutamatergic neurons (Chih and Roberts, 2003). According to the hypothesis proposed here, an increase in energy demands, regardless of the type of the excitatory neurotransmitter, would result in significant increases in lactate production, due to the glycolytic pathway's tight regulatory control, and thus the rapid response to an increase in demand for ATP by subsequent increased glycolytic flux. However, there is no reason that increases in lactate production should result in an observable lactate accumulation, given the postulate that at the onset of activation every molecule of lactate produced is being transported to and oxidized by the mitochondria. Nevertheless, examination of the elegant work of Hu and Wilson (1997) clearly indicates that immediately on neuronal stimulation there is a dip in all the three cerebral energy substrates, glucose, oxygen and lactate. If astrocytes are the cells responsible for clearing the excitatory neurotransmitter from the synaptic cleft, then, this clearing activity is the signal that induces the increased flux of the glycolytic pathway in astrocytes. As has been shown by Hu and Wilson (1997), the increase in extracellular lactate levels on stimulation is significant (160% to 200%) and is accompanied by a measurable decrease in extracellular glucose levels, without a significant change in tissue oxygen concentration (Hu and Wilson, 1997). Since glutamate is the most prominent excitatory neurotransmitter in the brain, the role of astrocytes in its uptake most probably plays a central role in the above-mentioned signaling. Studies using hippocampal slices have clearly shown that any increase in tissue lactate levels due to activation by glutamate is hardly detectable, unless the transport of lactate is inhibited (Schurr et al, 1999). Lactate transport inhibition, either from astrocytes or the extracellular space, into neurons should cause its accumulation either in the astrocytes themselves or extracellularly. In addition, N-methyl-
The report by Kasischke et al (2004) drove Pellerin and Magistretti to revise the Pellerin and Magistretti (2004aa). Accordingly, the increase in energy demands on neural activation is supported by neuronal oxidative lactate utilization, indicated by dips in both dendritic mitochondrial NADH (Kasischke et al, 2004) and extracellular lactate levels (Hu and Wilson, 1997), followed by a longer-lived overshoot in astrocytic NADH and lactate concentrations. Hence, it is now clear that the original notion of the ANLSH, postulating that astrocytic glutamate uptake is the signal coupling neural activity to glucose consumption and increased energy metabolism, was wrong. It is more likely that neuronal ionic movement is the signal that induces the increase in dendritic mitochondrial substrate oxidation (oxygen, lactate and NADH dips) that follows an elevation in astrocytic glucose consumption (lactate and NADH overshoot).
In his critique of the ANLSH, Hertz (2004) rejects the notion that lactate is predominantly a neuronal oxidative energy substrate. To support his stance, he uses several studies in which either 14C- or 13C-labeled glucose or lactate was employed. For instance, Itoh et al (2003) found that unlabeled glucose did not inhibit 14CO2 production from labeled lactate in cortical neurons. In contrast, both Itoh et al (2003) and Bouzier-Sore et al (2003) have shown that unlabeled lactate strongly reduced the production of 14CO2 from labeled glucose. Itoh et al (2003) explained their results by postulating that lactate conversion to pyruvate also reduces NAD+ to NADH, minimizing the availability of the former for the oxidation of glyceraldehyde-3-phosphate during glycolysis. Hertz (2004) argues that if this were the mechanism by which lactate inhibits glycolytic glucose utilization, then pyruvate, which is not involved in oxidation—reduction reactions of NAD+/NADH, should be less efficient an inhibitor of 14CO2 production from labeled glucose than lactate. At this point in his argument, Hertz (2004) reveals the results of experiments from his laboratory that indicate pyruvate to be as good an inhibitor of 14CO2 production from labeled glucose as is lactate. Although these unpublished results may refute the explanation given by Itoh et al (2003) for the possible mechanism of inhibition of 14CO2 production from labeled glucose, Hertz's very experiments strongly support the present hypothesis, namely, that lactate (and pyruvate), when supplied exogenously, is an oxidative substrate for the mitochondrial TCA cycle. Consequently, abundant exogenous supplies of either lactate or pyruvate would be expected to inhibit the glycolytic utilization of glucose, since the oxidative utilization of any of these monocarboxylates results in the production of ∼15 mol of ATP per mol of monocarboxylate, without the need for ATP investment when glucose is the substrate. In addition, no shortage of NAD+ would be expected when lactate is the substrate, since the enzyme that oxidizes it to pyruvate is not the glycolytic LDH; rather it is the mitochondrial LDH.
A ‘white elephant’ thus emerges from the arguments of those who discredit lactate as a cerebral oxidative substrate of importance, while simultaneously arguing that glycolytic pyruvate is the important one. Interestingly, those who dismiss lactate as an oxidative energy substrate on the grounds that its levels do not rise to the occasion of activation do not hold pyruvate to that very standard: none of the critics of the ANLSH expect pyruvate levels to rise significantly during activation although, according to their predominant notion, pyruvate is the aerobic glycolytic product and thus its levels must also rise if it is to answer the call for higher energy demands.
Nonetheless, it should be emphasized that ‘lactate as the ultimate oxidative energy substrate’ hypothesis has not been constructed merely to support or refute the ANLSH. This hypothesis aims at furnishing a framework to explain the results of numerous studies of the past two decades on cerebral (and other tissues) energy metabolism that have challenged one of the most enduring dogmas in biochemistry.
Conclusion
Undoubtedly, the research on brain energy metabolism lags behind that of muscle. Yet, there are many parallels between the two tissues. In both, there are at least two types of neighboring cells that influence each other, especially during heightened activity: neurons and astrocytes in the brain and type I and II fibers in the skeletal muscle. Many of the features of skeletal muscle fibers, so elegantly elaborated on by Brooks (1985, 1998) and Brooks et al (1999), the first investigator to coin the term ‘lactate shuttle,’ and Van Hall (2000), are features that could easily be applied to brain tissue. For now, the ‘muscular’ investigators are ahead of the ‘brainy’ ones, at least where the clarity of the glycolytic picture is concerned. Recently, members of the first group shot down another ‘white elephant,’ showing that lactate is not the cause of muscle fatigue (Pedersen et al, 2004). The hypothesis put forward here postulates that lactate is the major product of cerebral (and other tissues) glycolysis, whether aerobic or anaerobic, neuronal or astrocytic. It provides the necessary bridge between the two factions in the debate on ‘who's more important, glucose or lactate.’ Accordingly, both are: glucose as the substrate of the glycolytic pathway and lactate as the substrate of the mitochondrial TCA cycle.
Footnotes
Acknowledgements
The author thanks MM Schurr and E Gozal for their invaluable comments and critique during the preparation of this manuscript.