Abstract
Previously, we identified proteins released from degenerating cultured cortical neurons as novel cerebrospinal fluid (CSF) markers for acute brain injury in the rat. Here, we investigate relationships between CSF changes in these novel markers and the severity of acute ischemic brain injury. Rats underwent sham surgery or 3,6,8, or 10mins of transient global forebrain ischemia. At 48 h after insult, CSF levels of 14-3-3β, 14-3-3ξ, and calpain cleavage products of α-spectrin and tau were quantified. Regional acute neurodegeneration was assessed by Fluoro-Jade and silver impregnation staining, and confirmed by immunohistochemical detection of the activation of calpain and caspase, cysteine proteases involved in neurodegenerative signaling. Ischemic neurodegeneration and activation of at least one cysteine protease were observed in the hippocampal CA1 sector, dentate hilus, caudate nucleus, parietal cortex, thalamus, and inferior colliculus. As expected, the total number of degenerating cells increased as a function of ischemia duration. Cerebrospinal fluid levels of the four marker proteins increased markedly after ischemia, and rose in proportion with its duration. Irrespective of the length of ischemia, CSF levels of the neuron-enriched proteins 14-3-3β and calpain-cleaved tau correlated significantly with the magnitude of acute ischemic neurodegeneration. Additionally, CSF levels of the two proteins correlated with one another. These results show that certain proteins released from degenerating neurons are CSF markers for brain injury in the rat whose levels reflect the severity of acute ischemic neurodegeneration. Measurement of 14-3-3β and calpain-cleaved tau may be useful for the minimally invasive diagnosis, prognosis, and therapeutic evaluation of acute brain damage.
Introduction
The management of acute ischemic brain injuries resulting from cardiac arrest or stroke would benefit from improvements in diagnosis at the onset of brain damage, rapid identification of patients at risk for mortality, morbidity and long-term neurologic dysfunction, and intervention with neuroprotectant treatments. Toward these ends, numerous studies have identified and characterized candidate biochemical surrogate markers for ischemic brain damage. These studies establish that proteins expressed predominantly in neurons, astroglia, or oligodendroglia can be released into the brain parenchyma and transit to cerebrospinal fluid (CSF) and blood in association with acute brain damage and barrier disruption. After cardiac arrest (Rosen, 1998; Bottiger et al, 2001; Mussack et al, 2002), cardiopulmonary bypass surgery (Basile et al, 2001), stroke (Persson et al, 1987; Wunderlich et al, 1999), or traumatic brain injury (McKeating et al, 1998), the astrocyte-enriched S100β is elevated in CSF and serum, where its levels correlate with measures of neurologic outcome and morbidity. However, S100β can be high in serum even in the absence of acute brain damage (Anderson et al, 2001; Snyder-Ramos et al, 2004; Hasselblatt et al, 2004), and it is unclear whether its alterations relate primarily to astroglial death or induced expression of S100β occurring with the reactive astrogliosis that published online 18 May 2005 accompanies neuronal injury (Miyake et al, 1989). Other well-studied markers include the neuron-enriched proteins neuron-specific enolase (NSE) and tau. After cardiac arrest with resusitation, early alterations in serum NSE are predictive of mortality (Meynaar et al, 2003), and respond to a hypothermia treatment that improves neurologic outcome (Tiainen et al, 2003). After brain trauma, the microtubule-associated protein tau is present in CSF and serum as an ~30–50kDa proteolytic fragment of indeterminate origin, where its levels may have prognostic value (Zemlan et al, 2002). Nevertheless, peripheral measures of neither NSE nor cleaved tau have showed sensitivity sufficient for the detection of mild-to-moderate brain injuries, and a surrogate marker for ischemic brain injury has not emerged yet for broad clinical application. It is possible that this goal may only be achieved with a panel of markers for acute brain injury that have complementary sensitivities, specificities and pharmacokinetics.
Rather than examine brain-enriched proteins on an individual basis, we devised a neurobiological approach to identify candidate surrogate markers for acute brain injury based on the study of protein release from degenerating cultured neurons (Siman et al, 2004). A focus on the most abundant neuron-enriched proteins released specifically with neurodegeneration should identify a set of novel candidate biomarkers with the potential for highest sensitivity to mild insults. The value of this approach was showed by immunodetection of 14-3-3β, 14-3-3ξ, calpain-cleaved α-spectrin and a 17 kDa calpain-derived fragment of tau as abundant proteins released from degenerating neurons that are also markedly increased in CSF of rats after transient global forebrain ischemia or traumatic brain injury. In the adult central nervous system, the two 14-3-3 isoforms and tau are expressed predominantly in neurons (Berg et al, 2003; Lee et al, 1988), suggesting strongly that their release into the CSF is a specific index of neurodegeneration. For a surrogate marker to be useful clinically, it should change in proportion to the extent of irreversible neuronal injury, but the four released proteins have not been studied before in this regard. Here, we use transient global forebrain ischemia of varying durations to investigate the relationships between CSF alterations in these proteins and the severity of acute ischemic histopathology in the rat.
Materials and methods
Rat Model of Transient Forebrain Ischemia
All animal procedures were performed in accordance with NIH guidelines for the care and use of laboratory animals and were approved by our Institutional Animal Care and Use Committee. Male Long Evans rats were subjected to either 3, 6, 8, or 10mins of transient forebrain ischemia using an established model described initially by Smith et al (1984) and modified by Gionet et al (1992) and used routinely in one of our laboratories (Neumar et al, 2001; Zhang et al, 2002). Control (sham) rats were subjected to the surgical procedures but not the ischemia. Rats were kept in animal housing rooms for at least two weeks after shipment and given free access to food and water until the day of experimentation. Rats weighing 400–450g were anesthetized with halothane (3%) in 70% N2O + 30% O2 by insufflation chamber. Rats were orotracheally intubated and mechanically ventilated with 30% O2 + 70% N2O. A surgical plane of anesthesia was maintained with 0.5–1% halothane. PE 50 femoral arterial and venous catheters were placed and temperature monitored with a needle thermocouple probe inserted beneath the temporalis muscle. Temperature was maintained between 37.0°C and 37.9°C using a warming pad and overhead lamp. ECG monitoring was performed with electrocardiographic limb leads. An arterial blood sample was obtained and ventilator adjustments were made, if necessary, to maintain pCO2 at 35 to 45 mm Hg.
Transient forebrain ischemia was initiated by the combination of bilateral carotid occlusion and hypovolemic hypotension to a mean arterial pressure (MAP) of 30 mm Hg. Hypovolemic hypotension was achieved by rapidly withdrawing blood from the femoral arterial catheter and maintained during the ischemic period by withdrawal or infusion of blood. Once a MAP of 30 mm Hg was achieved, both carotid arteries were reversibly occluded with surgical aneurysm clips. After the varying durations of ischemia, the aneurysm clips were removed and shed blood reinfused. Rats were maintained on mechanical ventilation for 1 h. Then the femoral catheters were removed and all surgical wounds closed. Rats were extubated and observed for an additional hour for evidence of respiratory distress, which was treated with oxygen. At 48 h after ischemia, rats were anesthetized using pentobarbital (60mg/kg, intraperitoneally) and CSF samples were obtained by cisterna magna puncture. Samples were centrifuged at 2,000g for 10mins to remove any cells, and the supernatant stored at −80°C. Anesthetized rats were perfused transcardially with ice-cold 0.1mol/L sodium phosphate buffer (PB; pH 7.4) followed by 4% paraformaldehyde in PB. Brains were removed and postfixed 36 h, cryoprotected in 20% sucrose in PB overnight, and frozen in isopentane at −40°C.
Histopathology
Sagittal 40 μm sections were taken starting from the midline with a sliding microtome. Free-floating sections were stained for Fluoro-Jade (Schmued et al, 1997), silver impregnation (Nadler and Evenson, 1983; Roberts-Lewis et al, 1994), or immunostained for calpain- or caspase-derived cleavage products of α-spectrin, using methods and antibodies that have been characterized extensively (Roberts-Lewis et al, 1994; Saatman et al, 1996; Zhang et al, 2002; Oo et al, 2002). Briefly, Fluoro-Jade staining was conducted on slide-mounted sections after oxidation with 0.06% KMnO4 for 20mins. Sections were incubated 30mins at room temperature in a solution of 0.001% Fluoro-Jade (Histochem, Jefferson, AR, USA) in 0.1% acetic acid. Calpain-cleaved α-spectrin was immunolocalized with rabbit antiserum Ab38 (1/30,000), whereas caspase-cleaved α-spectrin was labeled with rabbit antiserum Ab246 (1/5,000). An avidin—biotin—horseradish peroxidase method with diaminobenzidine substrate was used for immunostaining.
Fluoro-Jade-positive cells were counted on every tenth section extending from the midline laterally to the start of the ventral hippocampus. The forebrain ischemia did not change the number of sections between these anatomical landmarks. For cell counting, images were captured at × 200 magnification on a Nikon Eclipse E600 microscope outfitted with Nikon DXM 12-megapixel camera. Stained cells were counted manually on computer screen connected to an image analysis system (MCID/M4 image software; Imaging Research, Brock University, St Catharines, Ontario, Canada). Fluoro-Jade-positive cells were counted in the hippocampal CA1 region, caudate nucleus, parietal cortex and central nucleus of the inferior colliculus. The cortical region analyzed began rostrally with primary motor cortex and extended caudally to the border of primary visual cortex. Counts were made by an investigator masked to the experimental treatment.
Quantitation of Cerebrospinal Fluid Protein Levels
Cerebrospinal fluid levels of 14-3-3β, 14-3-3ξ, α-spectrin, and tau were determined by quantitative Western blotting. Unfractionated CSF samples were separated by SDS-polyacrylamide gel electrophoresis and the polypeptides transferred to PVDF membranes. The membranes were immunostained with the following antibodies: rabbit anti-14-3-3β @ 1/1000 (SC-1657; Santa Cruz Biotechnology, Santa Cruz, CA, USA); rabbit anti-14-3-3ξ @ 1/1000 (SC-1019); mouse anti-α-spectrin @ 1/1000 (MAB1622; Chemicon, Temecula, CA, USA); mouse anti-tau @1/1000 (Tau-1; Zymed, Carlsbad, CA, USA). After incubations with horseradish peroxidase-conjugated secondary antibodies (1/5,000; Santa Cruz), blots were developed with enhanced chemiluminescence substrate (Renaissance Plus, Perkin-Elmer) and used to expose X-ray film (Bio-Max Light, Kodak). To ensure that band density corresponded to polypeptide content, each CSF sample was evaluated at different volumes, and multiple film exposures were analyzed. Band densities were determined from digitized images using ImageQuant software, under conditions where band density varied in linear proportion with sample loading volume (Molecular Dynamics).
Statistical Analyses
The numbers of Fluoro-Jade-positive cells and CSF protein levels across different ischemia durations were compared using a one-way ANOVA followed by post hoc Scheffe test. Pearson's product moment correlation was used to evaluate comparatively CSF proteins levels and Fluoro-Jade-positive cell counts.
Results
Histopathological Assessment of Acute Ischemic Neurodegeneration in the Rat Brain
We used several sensitive and specific histopathological methods to localize cells undergoing acute neurodegeneration after transient global forebrain ischemia in the rat. Previous studies employing two-vessel occlusion with hypovolemic hypotension as a model of transient global forebrain ischemia indicate that acute neurodegeneration first becomes detectable in signature brain regions between 24 and 48 h after the insult. We examined brain sections at 48 h after insult using histopathologic staining methods based on Fluoro-Jade, silver impregnation, and immunohistochemistry for the activation of calpain and caspase, cysteine protease families linked to necrotic and apoptotic neurodegeneration, respectively (Siman et al, 1998; Graham and Chen, 2001).
As shown in Figure 1, the caudate nucleus is a major site of acute neurodegeneration after 10mins of transient global forebrain ischemia. Panels A—D illustrate serial sagittal sections stained for Fluoro-Jade (A), silver degeneration (B), calpain-cleaved α-spectrin (C), and caspase-cleaved α-spectrin (D). Whereas none of these histochemical methods labeled cells in the caudate nucleus of control rats subjected to sham surgical procedures (data not shown), at 48 h after ischemia numerous cells throughout the dorsal and caudal portions of the caudate nucleus were labeled with Fluoro-Jade (A) and silver (B). Immunohistochemical localization of cysteine protease activation with cleavage site-specific antibodies indicative of either calpain or caspase substrate degradation confirmed the presence of acute ischemic neurodegeneration in the caudate nucleus. Postischemic calpain-mediated α-spectrin cleavage, which marks neurons undergoing necrotic ischemic neurodegeneration (Siman et al, 1996), was localized to the neuropil throughout the caudate nucleus. The caspase-mediated α-spectrin degradation product was concentrated in cell bodies located primarily in the dorsal and caudal regions of the caudate nucleus. Double-labeling for caspase activation and cell type-specific markers indicates that postischemic apoptosis in the caudate nucleus occurs in neurons (Zhang et al, 2002).
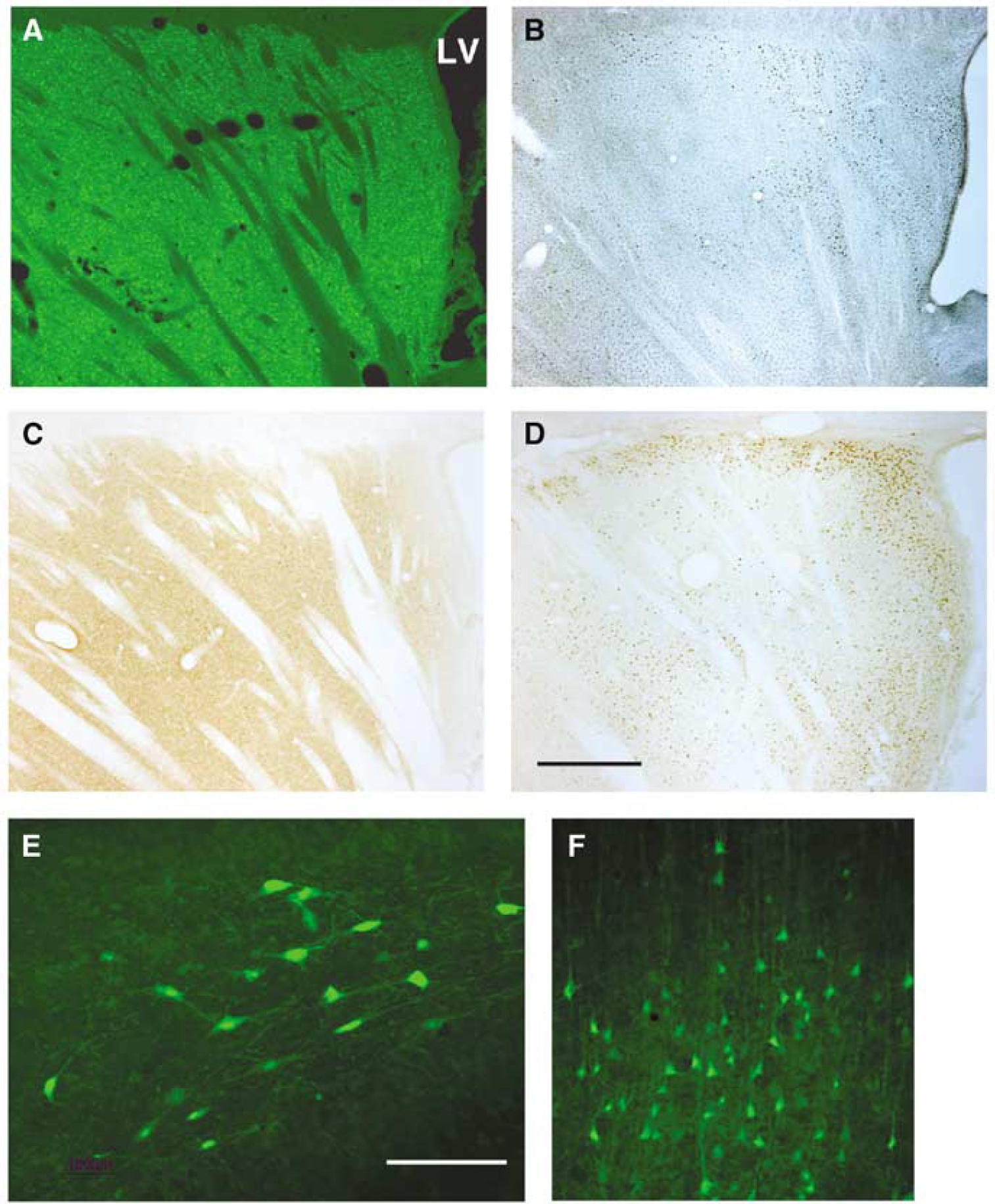
Assessment of acute ischemic histopathology at 48 h after transient global forebrain ischemia in the rat. Representative sagittal sections are shown from a rat subjected to 10 mins of global ischemia, stained either for (
Additional brain regions exhibited acute neurodegeneration consistently after transient forebrain ischemia. At 48 h after a 10-min episode of global forebrain ischemia, the hippocampus, neocortex, thalamus, and inferior colliculus contained cells positive for Fluoro-Jade and silver impregnation staining, and also immunopositive for calpain activation. Acutely degenerating cells included hilar neurons in the hippocampal dentate gyrus (Figure 1E), CA1 pyramidal neurons, neurons in layer V of parietal cortex (Figure 1F), and cells in the central nucleus of the inferior colliculus (Figure 2).
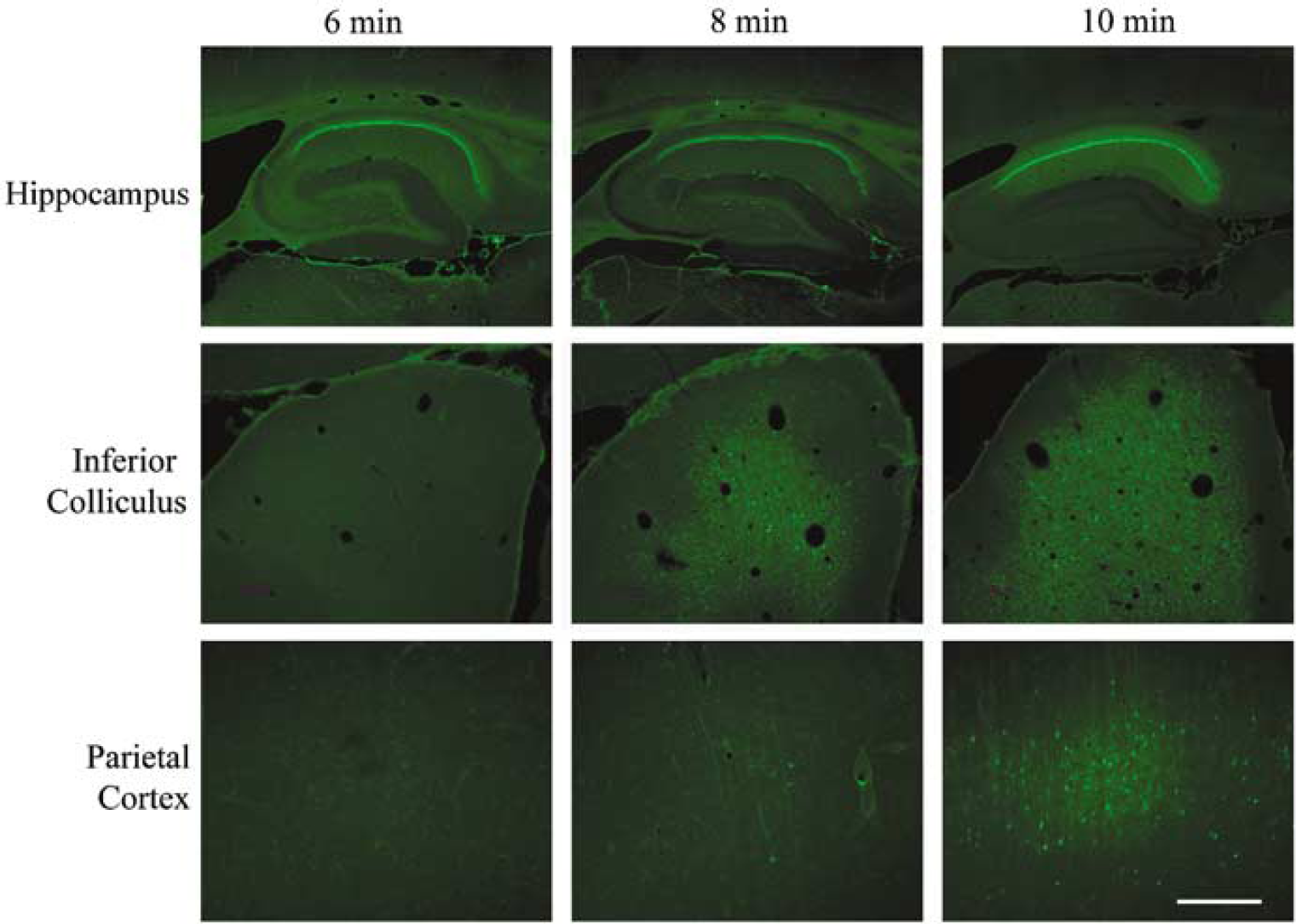
Regional extent of acute neurodegeneration after ischemia episodes of varying durations. Fluoro-Jade staining is depicted for representative cases subjected to either 6, 8, or 10 mins of global forebrain ischemia and analyzed at 48 h after insult. The pyramidal neurons of the hippocampal CA1 region were particularly sensitive to postischemic neurodegeneration, exhibiting consistent and extensive death with all three ischemia durations. The central nucleus of the inferior colliculus, however, exhibited increasing amounts of neurodegeneration detected by Fluoro-Jade staining with escalating ischemia durations. Little neurodegeneration was detected in posterior parietal cortex unless the duration of ischemia reached 10 mins. Scale bar, 1mm (hippocampus), 0.5mm (inferior colliculus), 0.2mm (parietal cortex).
The duration of the ischemic insult influenced the regional distribution and magnitude of acute neurodegeneration. When examined 48 h after insult, 6 mins of ischemia elicited neurodegeneration that was restricted primarily to the pyramidal neurons of the hippocampal CA1 sector (Figure 2), medial portion of the dentate hilus (data not shown), and extreme dorsal and caudal portion of the caudate nucleus (Figure 5). With a shorter 3mins ischemia, acute neurodegeneration was observed only in the CA1 region (data not shown). At 8mins of ischemia, additional acute neurodegeneration occurred in the central nucleus of inferior colliculus, as well as in scattered neurons in the parietal cortex (Figure 2) and dorsal caudate nucleus (Figure 5). The observations reported here using Fluoro-Jade and silver impregnation staining methods as well as immunodetection of calpain and caspase activation are fully consistent with prior studies localizing postischemic cysteine protease activation and neurodegeneration to specific brain regions in a rat transient global forebrain ischemia model (Blomqvist and Wieloch, 1985; Hatakeyama et al, 1988; Crain et al, 1988; Zhang et al, 2002), although the central nucleus of inferior colliculus has not been described before as a locus of postischemic degeneration.
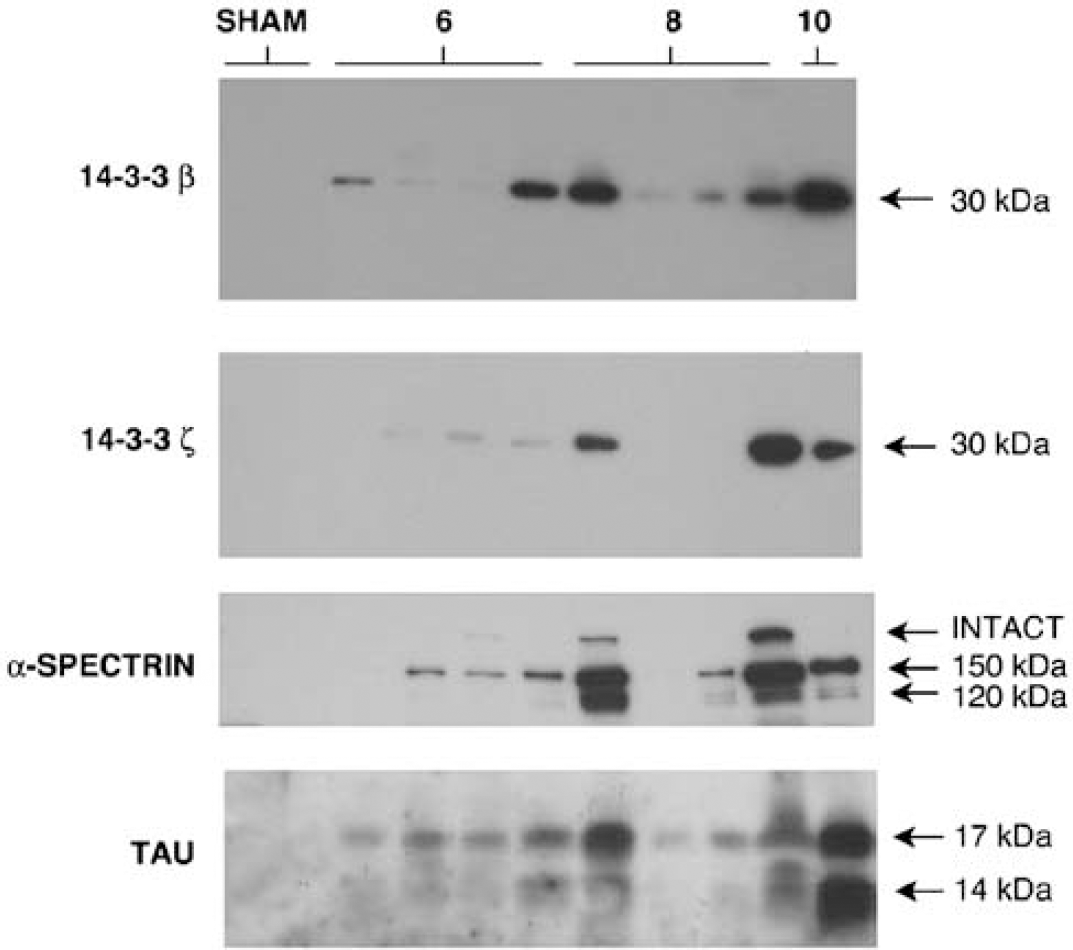
Western blot detection of elevated 14-3-3β, 14-3-3ξ, calpain-cleaved α-spectrin, and 17kDa calpain-cleaved tau in cerebrospinal fluid (CSF) after ischemia. Cerebrospinal fluid samples taken from rats 48 h after sham surgery or the indicated durations of global forebrain ischemia (in minutes) were analyzed by Western blotting. As shown by this representative Western blot, all of the proteins were below the limit of detection in CSF of sham-injured rats, but increased in CSF after ischemia. Note that CSF protein levels varied considerably from case to case for ischemia durations less than 10mins (N = 4/group). In general, cases with relatively high CSF levels of one marker also showed high levels of the other proteins as well.
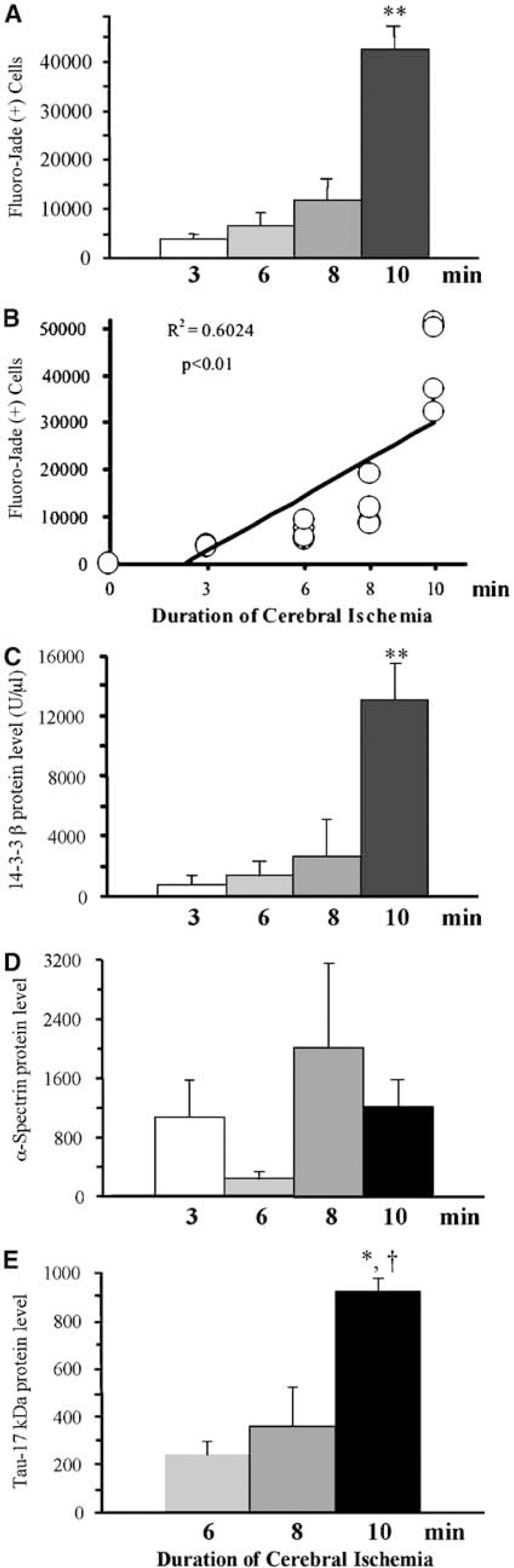
Quantitative analyses of acute neurodegeneration and cerebrospinal fluid (CSF) marker levels at varying ischemia durations. (
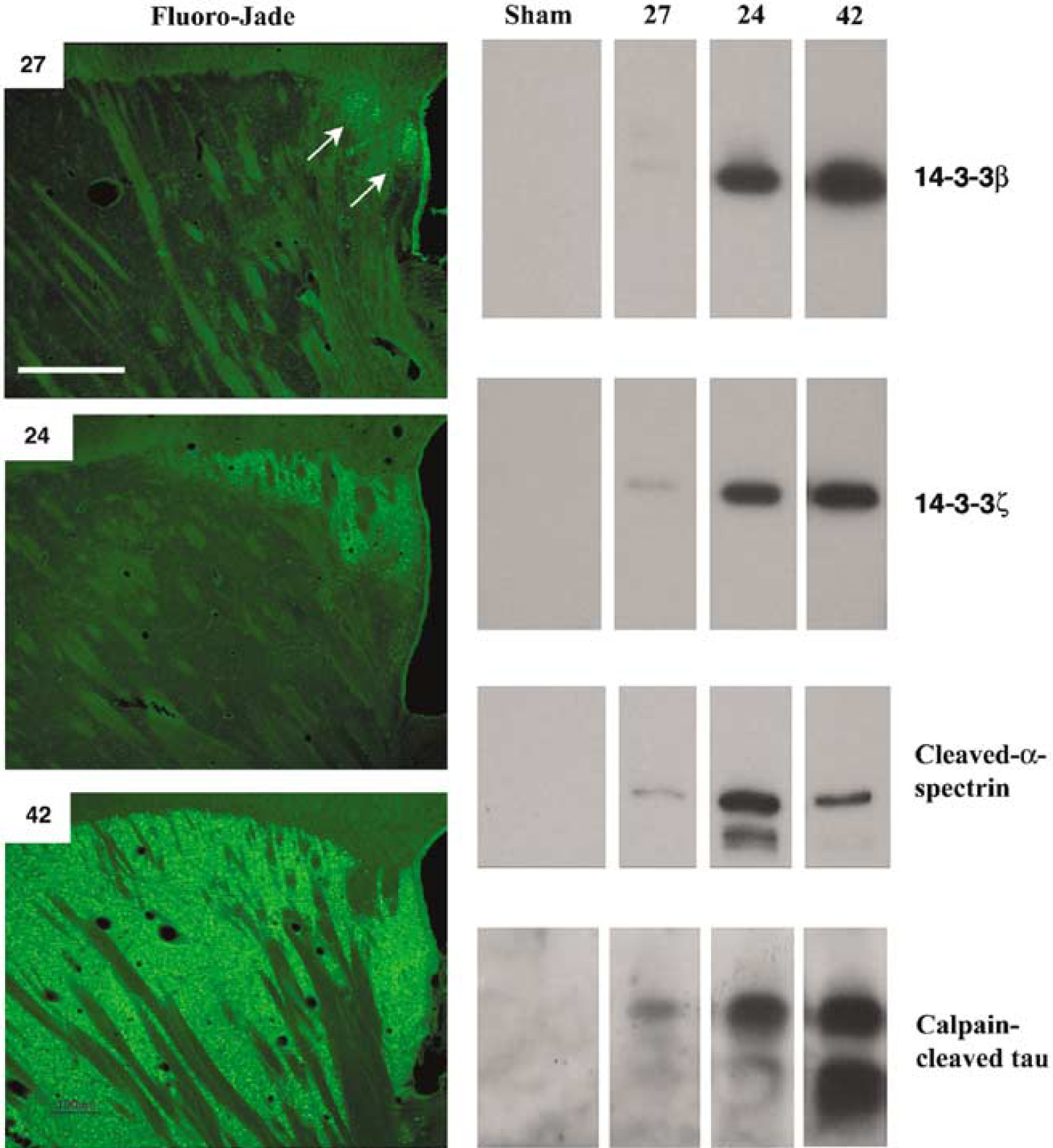
Cerebrospinal fluid (CSF) marker levels under conditions of mild, moderate, and extensive acute ischemic neurodegeneration. The panels on the left are photomicrographs of Fluoro-Jade staining of the caudate nucleus of cases 27, 24, and 42, representative cases with mild, moderate, and extensive acute ischemic neurodegeneration, respectively. The arrows in case 27 denote the ischemic neurodegeneration restricted to the extreme dorsal and caudal region of the caudate adjacent to the lateral ventricle. The panels on the right illustrate Western blot analyses of CSF protein markers from two sham-injured rats, along with cases 27, 24, and 42. Note that CSF levels of 14-3-3β and calpain-cleaved tau (the upper 17kDa derivative) relate well to the magnitude of acute neurodegeneration in these three ischemia cases, whereas CSF levels of 14-3-3ξ or calpain-cleaved α-spectrin do not. Scale bar=500μm.
Novel Cerebrospinal Fluid Markers for Acute Ischemic Neurodegeneration
Previously, we identified 14-3-3β, 14-3-3ξ, a calpain-derived 17kDa tau fragment, and a calpain-derived 150 kDa α-spectrin fragment as proteins released from degenerating cultured neurons that also increased in rat CSF after transient global forebrain ischemia (Siman et al, 2004). The former three proteins are expressed predominantly in neurons, making them excellent candidate surrogate markers for acute brain damage released into the brain parenchyma and measurably increased in CSF specifically in association with neurodegeneration. Important criteria for clinically useful surrogate markers for ischemic brain damage are responses to the severity of the ischemic insult and magnitude of cerebral histopathology. To investigate 14-3-3β, 14-3-3ξ, and calpain-cleavage products of tau and α-spectrin in these regards, we quantified CSF levels of the proteins at 48 h after episodes of forebrain ischemia varying from 3 to 10mins. As shown by the representative Western blot analysis of Figure 3, in sham rats CSF levels of 14-3-3β, 14-3-3ξ, calpain-cleaved tau, and calpain-cleaved α-spectrin were below the limit of detection. At 48 h after episodes of ischemia of 6, 8, or 10mins durations, CSF levels of the four proteins were elevated. Whereas 10mins of ischemia elicited consistently large increases in the four proteins in CSF, the alterations with shorter durations of ischemia were variable (
Quantitative analysis of acute ischemic neurodegeneration from 15 rats based on Fluoro-Jade-positive cell counts from the dorsal hippocampus, caudate nucleus, parietal cortex, and central nucleus of the inferior colliculus showed that lengthening ischemia durations led to increasing numbers of acutely degenerating cells. Whereas hippocampal pyramidal neurons in the CA1 sector underwent acute neurodegeneration after as short as a 3-min episode of forebrain ischemia with hypovolemic hypotension, progressively more cells exhibited acute degeneration with increasing ischemia duration (Figure 4A). In particular, the caudate nucleus and parietal cortex became markedly impacted only after the 10mins ischemia, accounting for the large increase in the total number of acutely degenerating cells with the longest ischemia duration examined. The effect of ischemia duration on acute neurodegeneration is also depicted in Figure 4B, which compares for all 15 cases the ischemia duration with neurodegeneration. There is a significant linear relationship (R2 = 0.60; P < 0.01) between the duration of ischemia and number of acutely degenerating cells.
Evaluation of the four candidate markers showed that CSF levels of 14-3-3β and calpain-derived 17kDa tau varied in accordance with the duration of forebrain ischemia (Figures 4C and 4E). In contrast, there was little relationship between ischemia duration and acute neurodegeneration on the one hand, and CSF levels of 14-3-3ξ (data not shown) or calpain-cleaved α-spectrin (Figure 4D) on the other. These findings suggest that certain proteins released from degenerating neurons are related closely to the duration of the ischemic insult and may reflect the severity of acute ischemic neurodegeneration.
Correlations Between Cerebrospinal Fluid Marker Changes and the Magnitude of Acute Ischemic Neurodegeneration
To investigate further the utility of 14-3-3β, 14-3-3ξ, and calpain cleavage products of tau and α-spectrin as surrogate measures of ischemic neurodegeneration, we compared CSF levels of the four proteins with the numbers of acutely degenerating neurons irrespective of the duration of forebrain ischemia. Examples are shown in Figure 5 of Fluoro-Jade staining in the caudate nucleus and corresponding Western blot analysis of CSF changes for the proteins from three rats, one exhibiting mild ischemic neurodegeneration, a second moderate neurodegeneration, and a third robust neurodegeneration. These cases are representative not only of the varying degrees of acute neurodegeneration in the caudate nucleus, but also the parietal cortex and inferior colliculus as well. Cerebrospinal fluid levels of 14-3-3β and the 17kDa tau fragment corresponded with the severity of ischemic neurodegeneration for these three cases, whereas CSF levels of 14-3-3ξ and calpain-cleaved α-spectrin were higher for the case with moderate injury than the case with extensive injury. Variability in the extrahippocampal neurodegenerative responses to episodes of mild-to-moderate global forebrain ischemia have been noted before (Ginsburg and Busto, 1998).
Quantitative comparisons for all 15 ischemia cases between the numbers of acutely degenerating cells and CSF levels of the four proteins are depicted in Figure 6. Cerebrospinal fluid levels of 14-3-3β (Figure 6A) and calpain-cleaved 17kDa tau (Figure 6C) correlated significantly with the total numbers of Fluoro-Jade-positive cells (P < 0.01). Indeed, CSF levels of the two proteins correlated linearly with acute neurodegeneration (R2 = 0.82 for each) better than the duration of the ischemic insult (R2 = 0.60; Figure 4B). In contrast, there was no significant relationship between the magnitude of acute ischemic brain damage and CSF levels of 14-3-3ξ (Figure 6B) and calpain-cleaved α-spectrin (Figure 6D). We also compared the CSF levels of 14-3-3β with those of the other 3 proteins for all the ischemia cases. As shown in Figure 7, CSF levels of 14-3-3β and calpain-cleaved 17kDa tau correlated significantly with one another. However, CSF levels of 14-3-3β were not significantly related to those of 14-3-3ξ or calpain-cleaved α-spectrin. Collectively, these analyses show CSF levels of 14-3-3β and calpain-cleaved 17kDa tau are surrogate markers for the magnitude of acute ischemic histopathology.
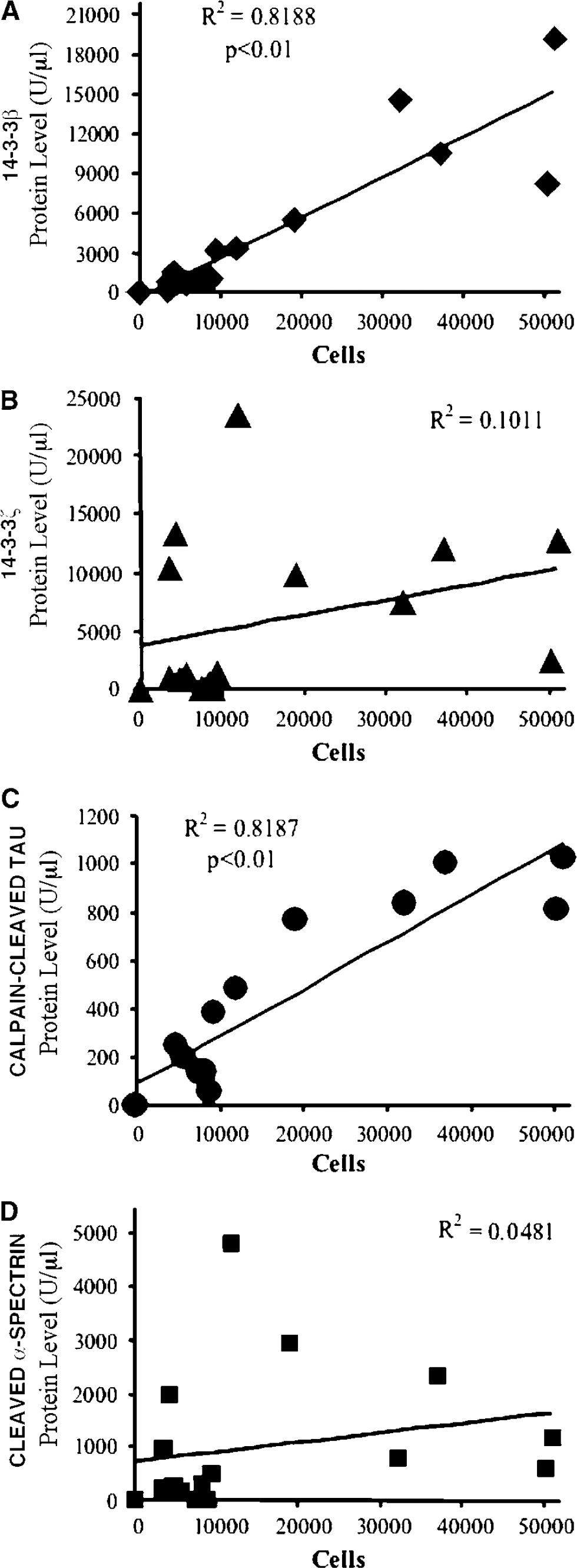
Relationships between cerebrospinal fluid (CSF) marker levels and acute ischemic neurodegeneration, irrespective of ischemia duration. (
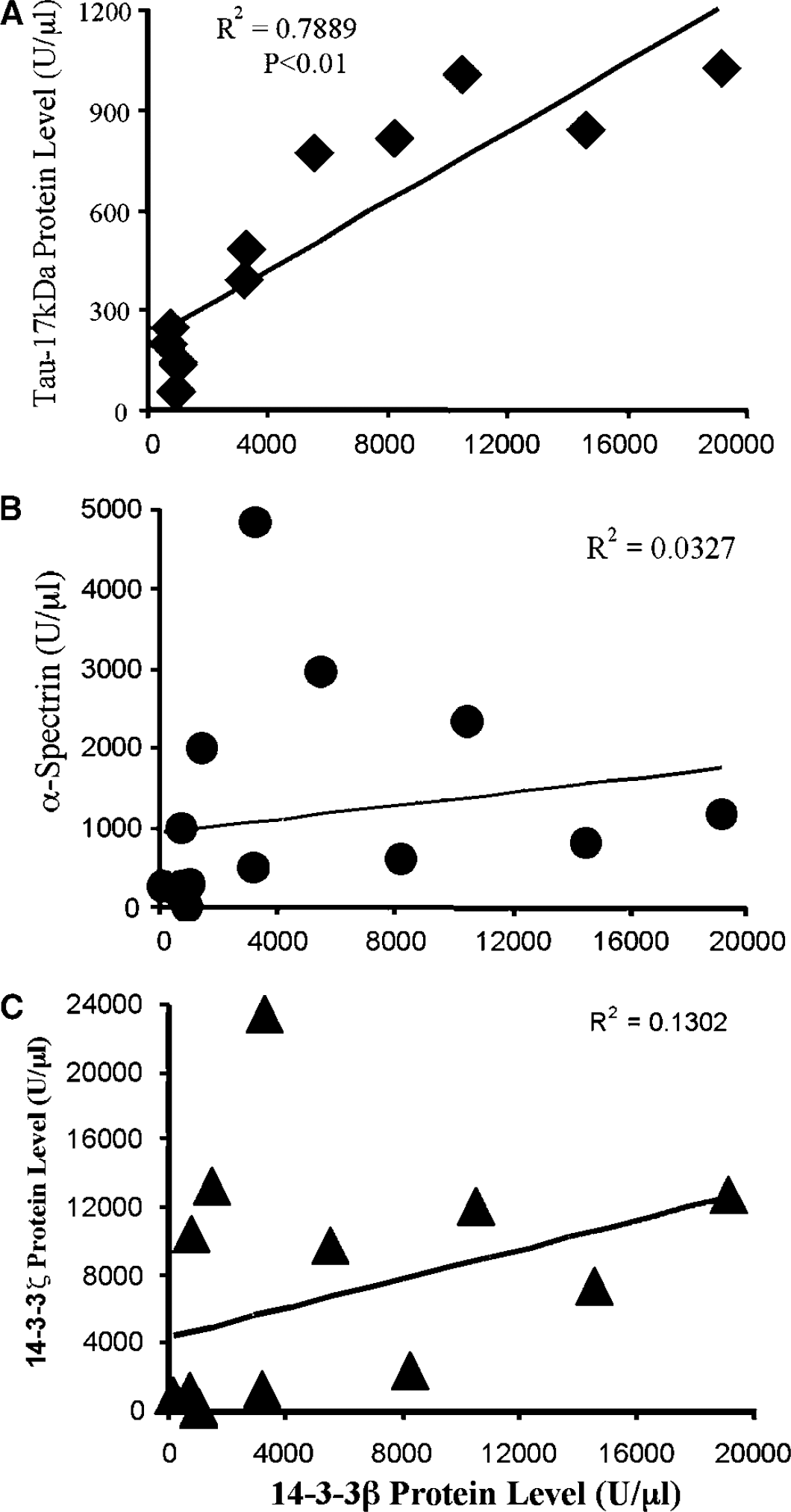
Relationships between cerebrospinal fluid (CSF) levels of marker proteins after transient global forebrain ischemia. Cerebrospinal fluid levels of 14-3-3β at 48 h after insult are plotted in comparison with levels of (
Discussion
The major finding reported here is that certain proteins released from degenerating cultured neurons are CSF markers for ischemic brain damage in the rat whose levels correlate with the magnitude of acute ischemic histopathology, and so predict the extent of neurodegeneration. Prior investigations of candidate surrogate markers for acute brain injury have established the concept that proteins expressed predominantly in the brain can transit to the CSF and serum in association with acute brain injuries, where their levels may provide diagnostic or prognostic information (Marchi et al, 2003) or serve as surrogate measures of the efficacy of experimental neuroprotectant treatments (Shirasaki et al, 2004). Nevertheless, despite the extensive study of selected neuronal-, astroglial-, and oligodendroglial-enriched proteins, limitations in their sensitivity or specificity have precluded the emergence of a biochemical marker with widespread clinical utility (Snyder-Ramos and Bottiger, 2003).
To discover novel candidate surrogate markers for acute brain damage, and as an alternative to the examination of proteins on a one-by-one basis, we devised a neurobiological approach for identifying candidate surrogate markers based on the most abundant proteins released from degenerating cultured neurons. At least four released proteins, 14-3-3β, 14-3-3ξ, and calpain cleavage fragments of α-spectrin and tau, measurably rise in the CSF of rats after transient global forebrain ischemia or traumatic brain injury (Siman et al, 2004). An essential characteristic of a clinically useful surrogate marker for ischemic brain injury is rapid alteration in proportion with the extent of irreversible brain damage, but the four released proteins have not been studied before in this regard. Here we report that, among the four, the neuron-enriched 14-3-3β and a calpain-derived 17kDa proteolytic fragment of tau rise in CSF with increasing duration of global forebrain ischemia and correlate with the numbers of acutely degenerating neurons. These findings provide further validation for the neurobiological approach to identifying novel candidate markers, and indicate that 14-3-3β and the 17kDa tau fragment are deserving of further study as surrogate markers for ischemia-induced brain damage.
To investigate the relationships between CSF protein changes and the extent of acute neurodegeneration, we examined CSF levels of 14-3-3β, 14-3-3ξ, calpain-cleaved α-spectrin, and calpain-cleaved 17kDa tau along with cerebral histopathology 48 h after transient forebrain ischemia of varying durations. In the current study, labeling of acutely degenerating cells by Fluoro-Jade and silver impregnation staining methods identifies signature cell populations in regions susceptible to transient global ischemia, including those in the caudate nucleus, hippocampus, and neocortex (Crain et al, 1988; Hatakeyama et al, 1988; Schmidt-Kastner et al, 1990; Iizuka et al, 1989). In addition, acutely degenerating cells are observed consistently with the two staining methods in the central nucleus of inferior colliculus. This region has not been identified by prior studies as being vulnerable to transient global forebrain ischemia, although, like other susceptible brain regions, it is a preferential site for postischemic calcium uptake (Mies et al, 1993). The localization of acute ischemic neurodegeneration to these signature brain regions (Figure 1) and also the inferior colliculus (data not shown) is substantiated by immunodetection of the activation of calpain and caspase, cysteine protease families implicated in necrotic and apoptotic ischemic neurodegeneration, respectively (Roberts-Lewis et al, 1994; Siman et al, 1998; Graham and Chen, 2001; Zhang et al, 2002). The susceptibility of the central nucleus of inferior colliculus to global ischemic brain injury could contribute to the reported auditory system dysfunction after stroke (Hausler and Levine, 2000) and, along with other auditory brainstem nuclei, is deserving of further study.
The evaluation of 14-3-3β, calpain-cleaved 17kDa tau, 14-3-3ξ, and calpain-cleaved α-spectrin by quantitative Western blotting shows that CSF levels of the former two proteins change in relation to the ischemia duration, and correlate significantly with the magnitude of acute cerebral neurodegeneration measured by Fluoro-Jade-positive cell counts from four vulnerable brain regions. The beta isoform of 14-3-3 and tau are abundant proteins expressed predominantly in neurons (Baxter et al, 2002; Lee et al, 1988), where they play fundamental roles in signal transduction and microtubule dynamics, respectively. They are released either as intact and calpain-cleaved proteins, respectively, in association with necrotic degeneration of cultured cortical neurons (Siman et al, 2004). Prior studies examining ultrastructure and cysteine protease activation provide evidence for abundant neuronal necrosis in the hippocampus, striatum, and neocortex, and also postischemic apoptosis in discrete neuronal populations (Roberts-Lewis et al, 1994; Petito et al, 1997; Lipton, 1999; Zhang et al, 2002). Taken together, these findings suggest that ischemia triggers release of the two proteins into the brain parenchyma and CSF in direct proportion to the extent of acute neurodegeneration. The reason why CSF levels of 14-3-3β and calpain-cleaved tau correlate tightly with the magnitude of acute ischemia neurodegeneration, whereas levels of 14-3-3ξ and calpain-cleaved α-spectrin do not, is unclear presently. One potential factor is the regional and subcellular expression patterns of the proteins. For example, the hippocampal CA1 sector is exquisitely sensitive to neurodegeneration with short episodes of global ischemia in the rat model studied here, whereas the inferior colliculus, caudate nucleus, and parietal cortex require progressively longer ischemia durations to be strongly impacted. Consequently, proteins expressed differentially across vulnerable brain regions may not increase in CSF in proportion to the total magnitude of neurodegeneration. Additionally, the cytoskeletal breakdown occurring with ischemic neurodegeneration develops initially in dendritic processes (Tomimoto and Yanagihara, 1992; Matesic and Lin, 1994), raising the possibility that neuronal proteins with distinct subcellular distributions might exhibit different time courses for release into the CSF. The brain regional and subcellular distributions of the four marker proteins studied here and the kinetics of their CSF alterations currently are under further investigation.
Having showed that CSF levels of two proteins released from degenerating neurons, 14-3-3β and calpain-cleaved 17kDa tau, change in proportion with the magnitude of acute histopathology, and correlate with neurodegeneration better than the duration of ischemic insult, we suggest that the two proteins may be surrogate markers for cardiac arrest-and stroke-induced brain damage with potential clinical utilities. Cerebrospinal fluid levels of 14-3-3β and calpain-cleaved 17kDa tau also correlate with one another, suggesting not only that a common mechanism underlies their injury-induced increases, but also raising the possibility that simultaneous measurement of the two markers might offer improved sensitivity or specificity over either measure alone. Numerous prior studies have evaluated other proteins as potential surrogate markers for ischemic brain damage, including the astrocyte-enriched S100β as well as the neuronal proteins NSE and a cleavage fragment of tau of indeterminate origin. Serum and CSF levels of these proteins have a prognostic relationship with neurologic outcome, morbidity, and mortality (Wunderlich et al, 1999; Zemlan et al, 2002; Meynaar et al, 2003). Unfortunately, high serum concentrations of S100β can arise from extracranial sources (Snyder-Ramos et al, 2004; Hasselblatt et al, 2004), and result indirectly from the reactive astrogliosis and increased S100β expression that accompany neuronal injury (Miyake et al, 1989). Neuron-enriched proteins likely are a more direct index of neuronal degeneration, but serum measures of NSE and cleaved tau reportedly have limited sensitivity, a significant liability for the detection of mild-to-moderate brain injuries that are difficult to diagnose with existing neuroimaging methods (reviewed by McAllister et al, 2001). Consequently, it will be important to evaluate 14-3-3β, calpain-cleaved 17kDa tau, and other proteins released preferentially from degenerating neurons as novel surrogate markers with the potential for improved sensitivity and specificity for ischemic brain damage. Additional studies are warranted to evaluate these novel markers as serum measures of acute brain injuries in experimental animals, as pharmacodynamic markers responsive to neuroprotectant therapies, and as markers of cardiac arrest- and stroke-induced brain damage in humans with potential clinical utilities.
