Abstract
To investigate the vasoconstrictor effect of 5-hydroxytryptamine (5-HT1B/1D) receptor agonists for migraine treatment, changes in cerebral blood flow (CBF) and blood volume induced by rizatriptan were assessed by positron emission tomography (PET). Eleven healthy volunteers underwent PET studies before and after rizatriptan administration. Dynamic PET data were acquired after bolus injection of H215O to analyze CBF and arterial-to-capillary blood volume (V0) images using the three-weighted integral method. After a baseline scan, three further acquisitions were performed at 40 to 50, 60 and 70 to 80 mins after drug administration. Global and regional differences in CBF and V0 between conditions were compared using absolute values in the whole brain and cortical regions, as well as statistical parametric mapping (SPM) analysis. The global and regional values for CBF and V0 decreased significantly after rizatriptan administration compared with the baseline condition. However, both values recovered to baseline within 80 mins after treatment. The maximal reduction in global CBF and V0 was approximately 13% of baseline value. The greatest decrease in CBF was observed approximately 60 mins after drug administration, whereas the maximal reduction in V0 was observed approximately 5 mins earlier. Statistical parametric mapping did not highlight any regional differences between conditions. Thus, in brain circulation, rizatriptan caused significant CBF and V0 decreases, which are consistent with the vasoconstrictor effect of triptans on the large cerebral arteries. The gradual recovery in the late phase from the maximal CBF and V0 decrease suggests that rizatriptan does not affect the cerebral autoregulatory response in small arteries induced by CBF reduction.
Keywords
Introduction
Triptans, 5-HT1B/1D receptor agonists used as rapid antimigraine treatment drugs, are known to act by means of the vasoconstrictor effect of 5-HT1B receptors on the cranial arteries and the inhibitory effect of 5-HT1D receptors on the trigeminal terminals (De Vries et al, 1999a; Tepper et al, 2002). The vasoconstrictor effect increases the resistance of carotid arteries and reduces cerebral blood flow (CBF) (Feniuk et al, 1989; den Boer et al, 1991; De Vries et al, 1999b). Cerebral vessels are assumed to dilate during migraine headache and CBF changes during migraine attacks, as reported by Olesen et al (1990). The therapeutic vasoconstrictor effect of triptans presumably acts by counterbalancing the vasodilation of major vessels that might be responsible to induce headache. However, the flow-induced vasodilation in peripheral circulation is not affected by these drugs (Feniuk et al, 1989; Williamson et al, 1997; de Hoon et al, 2000). Although patients with migraine did not show changes in CBF after treatment with sumatriptan (Friberg et al, 1991), triptans may induce small changes in CBF and blood volume as a result of vasoconstriction in the major cerebral arteries, followed by reduction in arterial perfusion pressure. Direct measurement of changes in CBF and intracerebral vascular volume induced by triptans in normal circulation would provide hemodynamic information about changes occurring after administration of triptans.
The purpose of the current study was to investigate the vasoconstrictor effect of triptans by analyzing the time-dependent changes in cerebral hemodynamic parameters in healthy volunteers. Oral dose of rizatriptan, one of the agents known to act as a 5-HT1B/1D receptor agonist, was used to reproduce the effect of triptans. H215O positron emission tomography (PET) was performed to measure CBF and arterial-to-capillary blood volume (V0) using the bolus injection method. Changes in CBF and V0 induced by rizatriptan were compared at different time steps after oral drug administration.
Materials and methods
Subjects
Eleven healthy male volunteers, aged 20 to 31 years (mean = 23.5 ± 3.2), were recruited for the H215O PET study. Two of them had an experience of migraine-like headache, and the rest of them never suffered any migraine. Before the study, they gave their written informed consent to undergo PET scans before and after oral administration of rizatriptan. The study was approved by the Ethical Committee of the University of Fukui, Faculty of Medical Science.
Positron Emission Tomography Procedures
All subjects underwent PET scans with a whole-body tomography scanner (ADVANCE, General Electric Medical System, Milwaukee, WI, USA), which allows simultaneous acquisition of 35 image slices with an interslice spacing of 4.25 mm (DeGrado et al, 1994). Performance tests showed the intrinsic resolution of the scanner to be 4.6 to 5.7 mm in the transaxial direction and 4.0 to 5.3 mm in the axial direction. A transmission scan was performed using 68Ge/68Ga for attenuation correction in each subject before the tracer administration. The PET data were reconstructed using a Hanning filter with a resolution of 6.0-mm full-width at half-maximum (FWHM) in the transaxial direction.
The subjects were positioned on the scanner bed with their heads immobilized using a head-holder. A small cannula was placed in the right brachial artery for blood sampling. Positron emission tomography scans for both CBF and V0 were performed before and 40 to 80 mins after rizatriptan administration using the bolus H215O injection method. Rizatriptan (10 mg, orally disintegrating tablet) was administered after the baseline scan. After baseline scan, all subjects underwent three further scans at 40 or 50 mins, at 60 mins and at 70 or 80 mins after treatment. The interval between each of the scans after treatment was 10 to 20 mins, including scanning time.
As for CBF measurement, a 3-mins dynamic PET scan was started at the time of the bolus H215O injection (555 to 740 MBq) from the left antecubital vein with frame durations of 5 secs × 12, 10 secs × 6 and 20 secs × 3. Radioactivity in the arterial blood was counted continuously using an automatic blood sampling system (ABSS) which includes a positron radioactive counter (Apollomec Co. Ltd, Kobe, Japan) and a bio-minipump (AC-2120; Atto Co., Tokyo, Japan). Arterial blood was sampled and counted continuously with the ABSS at a constant rate of 7mL/mins for the first 2 mins, followed by manual sampling of 0.5 mL of blood every 20 secs during the remaining scan time. Radioactivity counted by the ABSS was calibrated with the blood sampled manually. Decay of the radioactivity from PET and blood data was corrected to the starting point of each scan, and dispersion for the external tube in the arterial curves was corrected with the double-exponential dispersion function described elsewhere (Okazawa et al, 2002). During the PET scanning, the arterial tensions for CO2 (PaCO2) and O2 (PaO2), as well as the hematocrit (Ht), pH and total arterial O2 content (tO2c), were measured from the same blood sample. Blood pressure of each subject was monitored continuously through the arterial line and displayed on a monitor during the PET study.
Calculation of Parametric Images
Cerebral blood flow (mL min−1 100 g−1) image was calculated from the dynamic PET data and arterial blood curves by means of the three-weighted integral (3-WI) method. The calculation procedure for the 3-WI method has been described in detail elsewhere (Ohta et al, 1996; Okazawa and Vafaee, 2001). In brief, the method based on a two-compartment model expressed by the following equation was used:
where K1 (mL min−1 g−1) and k2 (min−1) are rate constants for the tracers, M (Bqg−1) is the radioactivity in brain tissue, and Ca (Bq mL−1) is an arterial input function. The time delay of arterial input was corrected automatically in the program, and a time constant of τ = 4 secs was used for internal dispersion correction (Ohta et al, 1996; Meyer, 1989; Iida et al, 1986). A distribution volume of 0.9 was used in the calculation of CBF (Herscovitch and Raichle, 1985). V0 images (mL 100 g−1) were calculated simultaneously from data of each condition in the 3-WI method.
Coregistration of Positron Emission Tomography and Magnetic Resonance Imaging (MRI)
Subjects underwent a high-resolution MRI scan (124 slices, 1.3 mm thick) using a Sigma Horizon LX scanner (1.5 T, General Electric Medical System, Milwaukee, WI, USA). Coregistration of individual PET and MRI images was performed using an automatic procedure (Woods et al, 1993) that uses the CBF image in each condition. The MRI image of each subject was transformed into Talairach space automatically (Collins et al, 1994; Talairach and Tournoux, 1988). The baseline CBF image coregistered to MRI was then transformed into Talairach space using the individual spatial transformation parameter, which combined the two parameters from coregistration of PET to MRI and transformation of MRI into Talairach space. V0 images were also transformed into Talairach space in accordance with the same individual parameter (Okazawa and Vafaee, 2001).
Statistical Data Analysis
Global values for each parametric image of the 11 subjects were obtained using a template generated from the MRI averaged in Talairach space; that is, the global mean values inside the brain template not including ventricles, after transformation of each CBF and V0 image (Okazawa and Vafaee, 2001). The same template was applied to all parametric images of all subjects. Global values thus obtained were compared between the conditions before and after rizatriptan administration. Analysis of variance (ANOVA) with post hoc Fisher's PLSD was used for the comparison.
Regional values were calculated using multiple regions of interest (ROIs) drawn on cortical territories of the bilateral middle cerebral arteries in Talairach space using individual MRI to avoid including the outside of gray matter. The same ROIs were applied to all parametric images in each subject. Regional values for each parametric image were averaged and compared between different conditions by ANOVA. A probability value of less than 0.05 was considered to indicate a statistically significant difference. Cerebral blood flow and V0 differences at clusters of voxel level were also analyzed between different conditions using statistical parametric mapping (SPM2) (Friston et al, 1990, 1991). Details for the SPM comparison of multiple images were described elsewhere (Okazawa and Vafaee, 2001). In brief, after global normalization of absolute values, the t-test was applied pixel-by-pixel to compare the regional differences in CBF and V0 obtained from the different conditions. The resulting t-value for each pixel was then converted to a normal standard distribution (z-value), independent of the degree of freedom from error, constituting a statistical parametric map, as reported by Friston et al (1991). In SPM analysis, each image was smoothed using a Gaussian filter (10 mm FWHM) and the pixel values were normalized so that the global mean of each parameter should be 50 (mL min−1 100 g−1) for CBF and 2 (mL 100 g−1) for V0. To identify the regions showing significant differences, the height threshold (u) was set at P = 0.001 and the extent threshold (k) was set at k = 100 voxels.
Physiological variables were compared across conditions using paired t-test.
Results
Table 1 shows the average values of the physiological variables measured in the 11 subjects during scanning at baseline and after rizatriptan administration. No physiological parameters were significantly changed after drug administration except for the systolic blood pressure.
Physiological data in 11 healthy volunteers (mean ± s.d.)
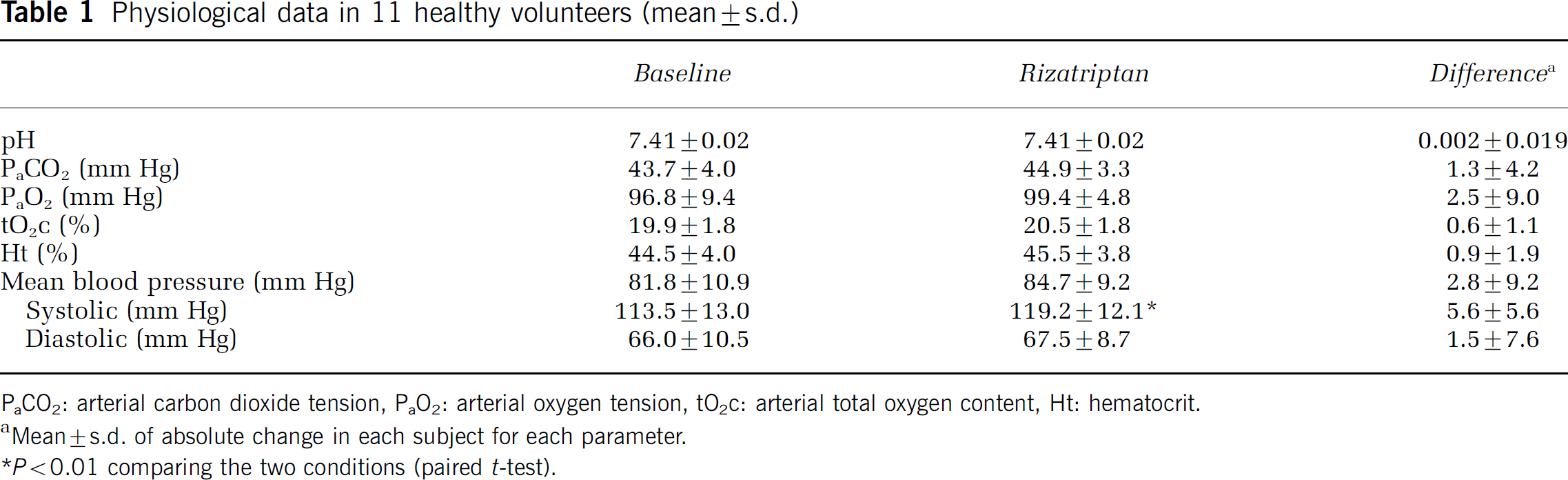
PaCO2: arterial carbon dioxide tension, PaO2: arterial oxygen tension, tO2c: arterial total oxygen content, Ht: hematocrit.
Mean ± s.d. of absolute change in each subject for each parameter.
P > 0.01 comparing the two conditions (paired t-test).
Figure 1 shows the average images of CBF and V0 obtained from the 11 healthy volunteers at baseline and at each time point after drug administration. Both CBF and V0 decreased at 40 to 60 mins after oral administration of rizatriptan. At 70 mins from the baseline scan, V0 almost recovered from the reduction while global CBF was still diminished from baseline level. Both parameters returned to the baseline level 80 mins after treatment.
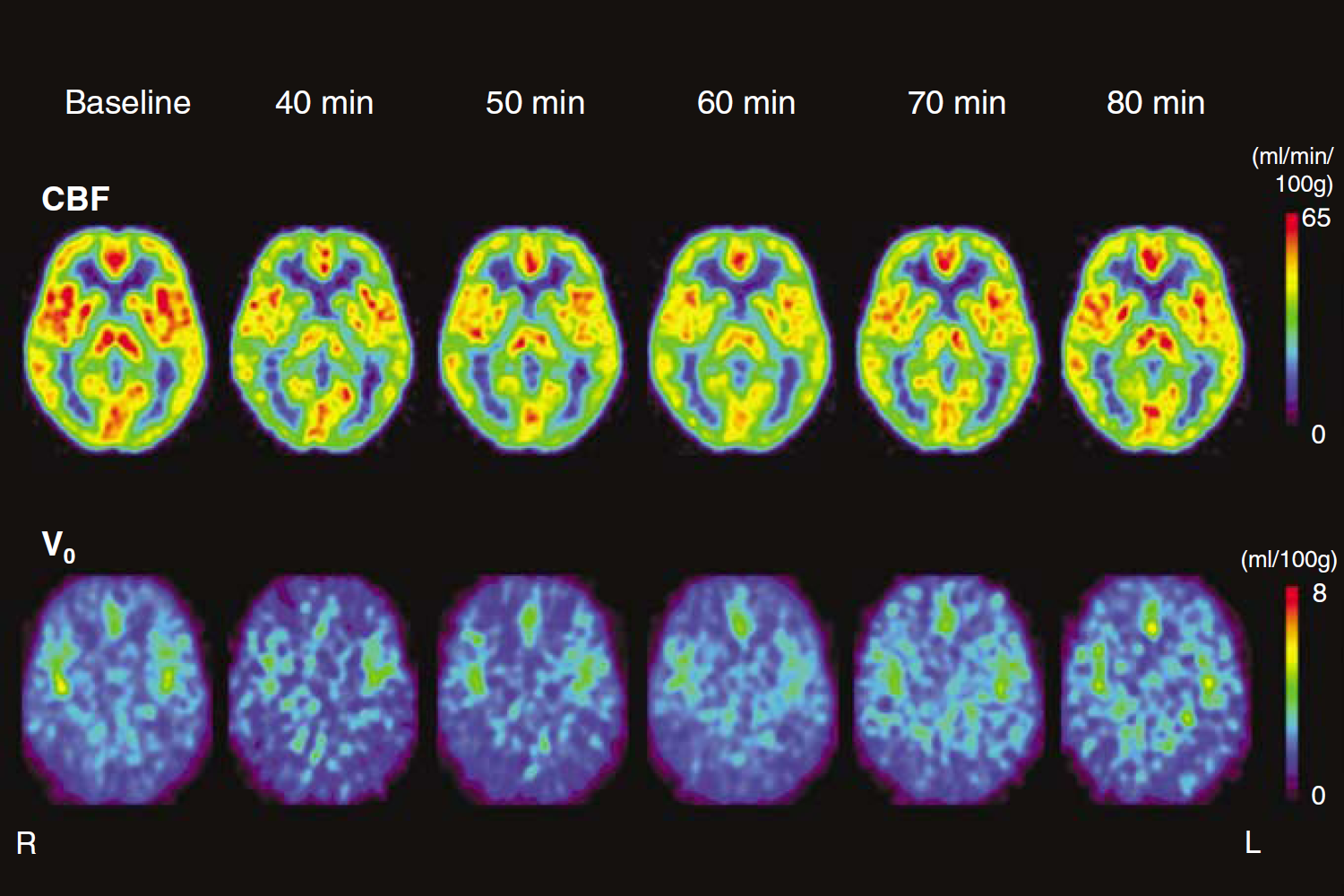
Average images of CBF (upper row) and V0 (bottom row) obtained from 11 healthy volunteers in Talairach space. Images of baseline and at 60 mins after rizatriptan administration were the means of all subjects. Other images were the average of 4 subjects at 40 mins and 6 subjects in the remaining time after treatment. Cerebral blood flow was decreased up to 60 mins after oral administration of rizatriptan and the reduction was gradually recovered in the later phase. V0 also decreased until 50 mins after treatment, and then tended to increase from the minimal reduction, which was earlier recovery than CBF change. Both parameters almost returned to the baseline level 80 mins after treatment. R = right, L = left.
The changes and difference in global CBF and V0 from the baseline values are presented in Figure 2. Significant decreases were observed in CBF from 50 to 70 mins and in V0 from 40 to 50 mins after rizatriptan administration, recovering such reductions later in the experiment. Table 2 shows the changes in global and regional CBF and V0 after medication. Both global and regional CBF showed significant decrease after rizatriptan administration. Approximately 10% reduction of global CBF was observed between 50 and 70 mins, and approximately 8% to 10% reduction of regional CBF was observed between 40 and 70 mins after drug administration. In both analyses, CBF recovered almost to baseline value 80 mins after medication. V0 also showed significant global and regional reductions; however, its recovery was earlier than CBF and no significant difference was observed at 60 mins or later. Maximal changes in CBF and V0 are shown in Table 3. The maximal reduction of CBF was 13.0% in the global and 13.5% in the middle cerebral artery cortices. A similar degree of reduction was observed in V0, although regional reduction tended to be greater. The time at which the maximal change occurred tended to be earlier in V0 than in CBF in both global and regional values, although the difference was not significant.
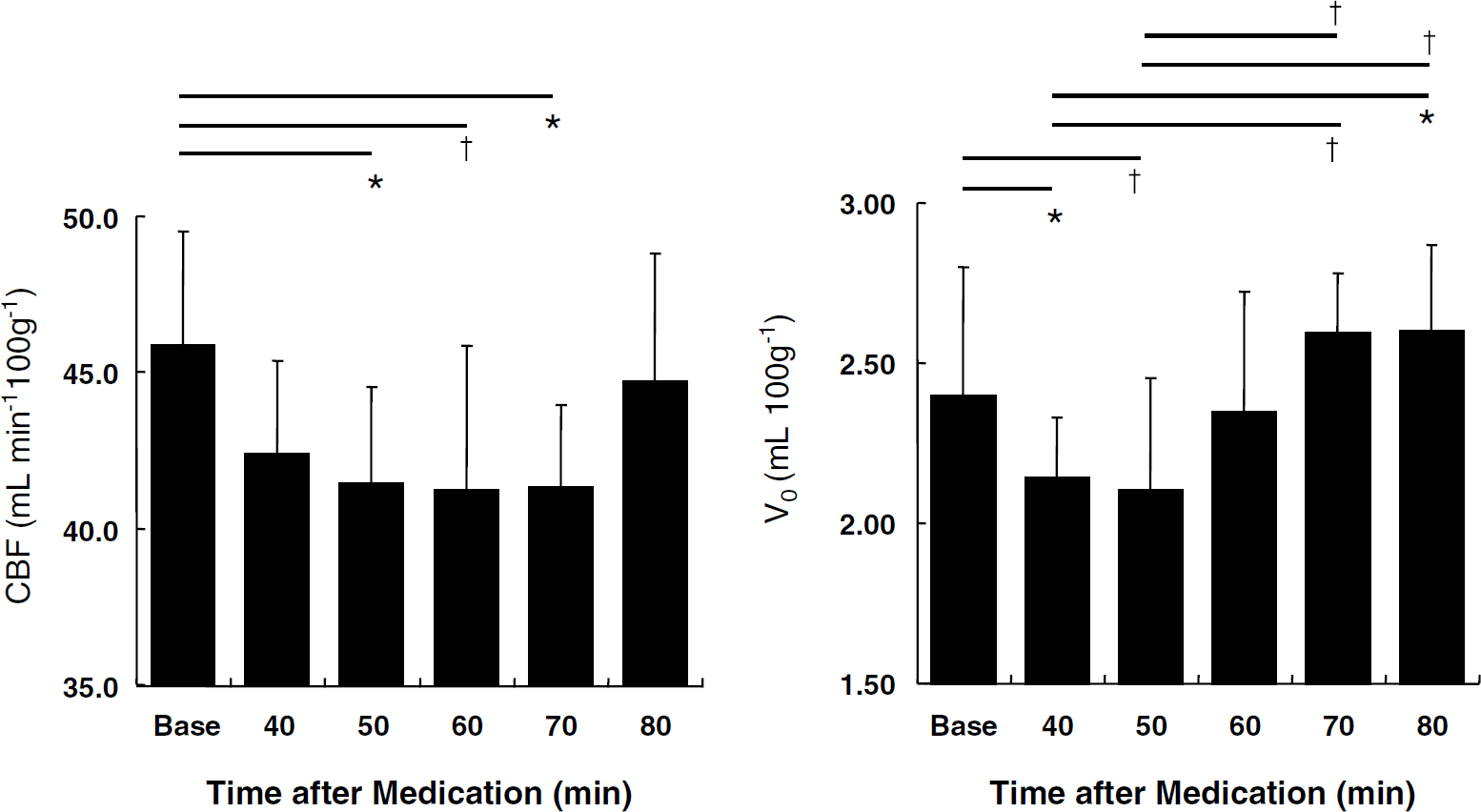
Time-dependent changes and differences in global CBF and V0 before and after rizatriptan administration. Significant decreases were observed during 50 to 70 mins in CBF and during 40 to 50 mins in V0 after treatment as compared with the baseline. The reduction was recovered in the later phase of the experiment. V0 also showed significant difference between 40 to 50 mins and 70 to 80 mins. *P > 0.01, †P > 0.05, compared by ANOVA with post hoc Fisher's PLSD.
Global and regional changes in CBF and V0 before and after rizatriptan administration
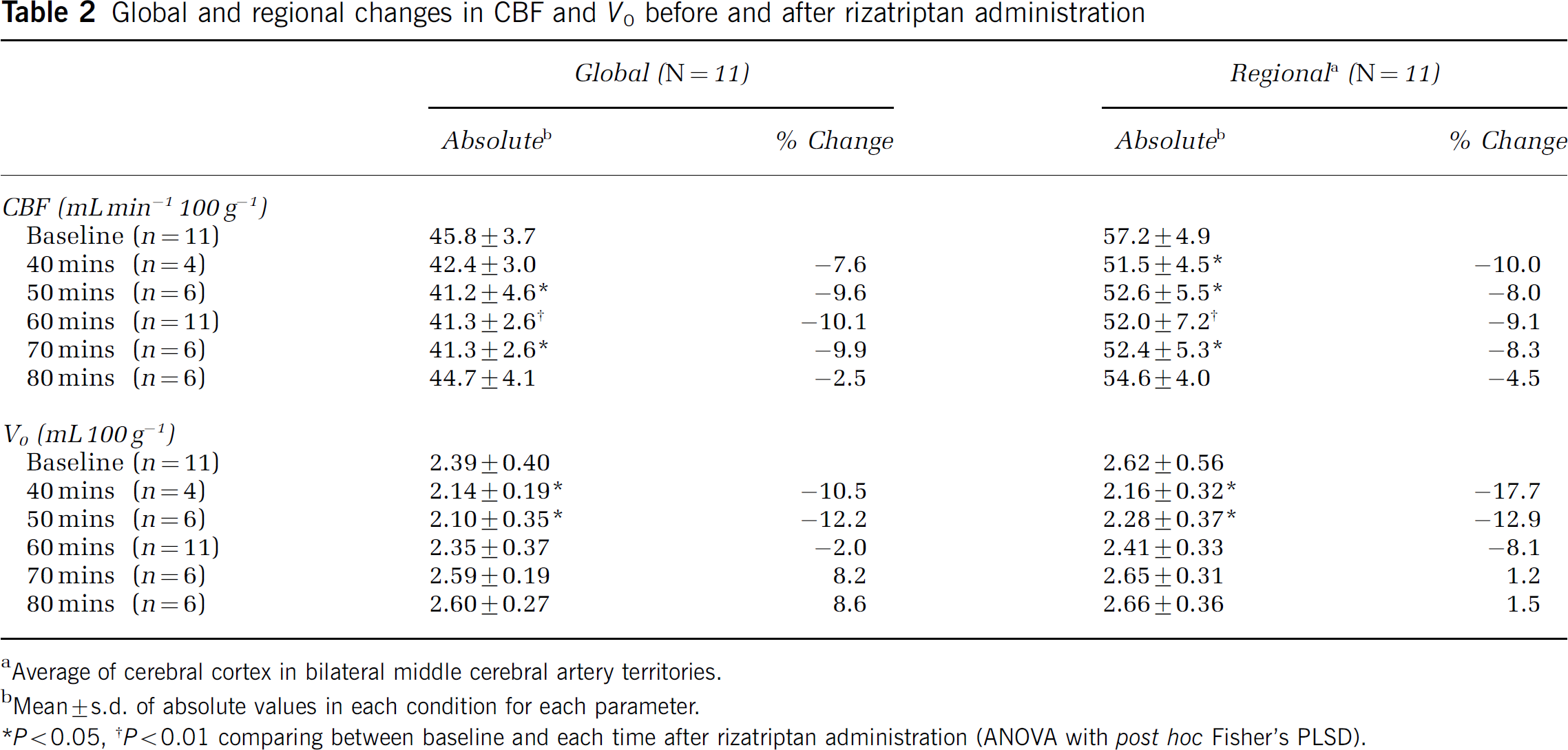
Average of cerebral cortex in bilateral middle cerebral artery territories.
Mean ± s.d. of absolute values in each condition for each parameter.
P > 0.05,
P > 0.01 comparing between baseline and each time after rizatriptan administration (ANOVA with post hoc Fisher's PLSD).
Maximal changes in CBF and V0 (n = 11)a
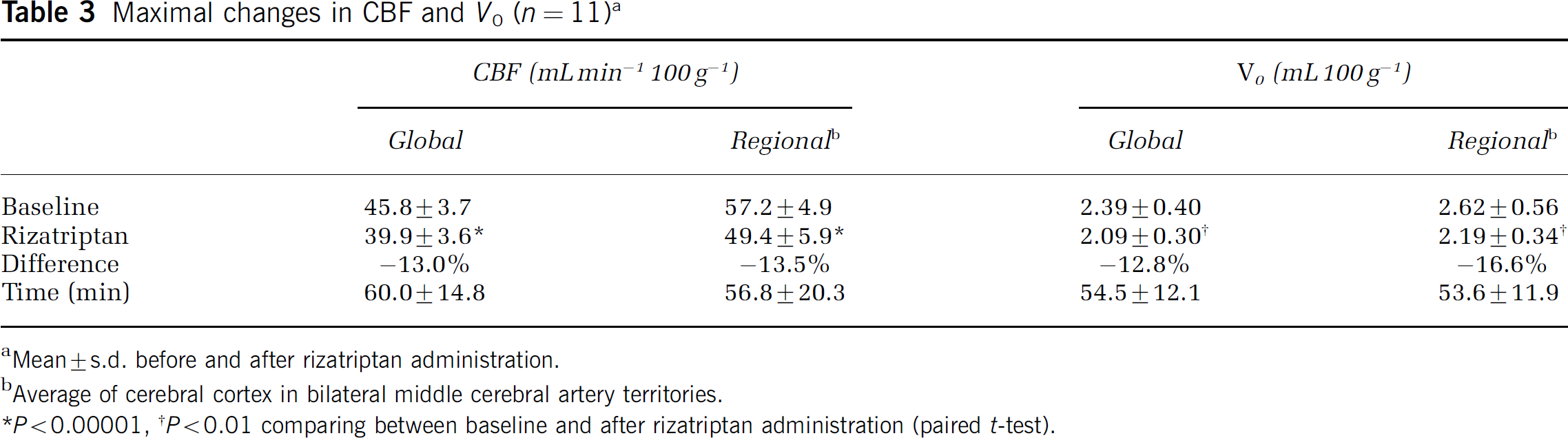
Mean ± s.d. before and after rizatriptan administration.
Average of cerebral cortex in bilateral middle cerebral artery territories.
P > 0.00001,
P > 0.01 comparing between baseline and after rizatriptan administration (paired t-test).
Statistical parametric mapping analysis did not show any significant regional differences in both CBF and V0 between the different conditions after global normalization.
Discussion
5-HT1B/1D receptor agonist, rizatriptan, which is used for migraine treatment, caused a reduction of CBF and V0 for a short period after administration. The absolute values of these parameters recovered almost to baseline 80 mins after rizatriptan challenge. Triptans act on 5-HT1B receptors, eliciting their vasoconstrictor effect on the cephalic arteries and increasing their vascular resistance (Feniuk et al, 1989; Parsons et al, 1989; Longmore et al, 1998; De Vries et al, 1999a). Such response causes changes in blood flow and velocity in the dural and cerebral arteries. During the migraine attack, triptans-driven vasoconstriction acts on the cranial arteries which are expected to be dilated, resulting in a common carotid artery (CCA) blood flow decrease (Feniuk et al, 1989; den Boer et al, 1991). The present study showed a transient but significant decrease in both CBF and V0 observed during a short period of time after oral administration of the drug. This indicates a prompt vasoconstrictor effect of rizatriptan (Tepper et al, 2002), followed by delayed recovery of CBF and V0 in smaller arteries in cerebral tissue including arterioles and precapillaries.
We did not assess directly the vasoconstrictor effect of the drug on the major arteries; however, CBF was measured based on the 2-compartment model, which minimizes influence of the vascular volume on regional CBF (Okazawa and Vafaee, 2001). V0 images calculated from the same dynamic data are an estimate of regional blood volume derived by model analysis. V0 values obtained in gray matter by this method have been shown to represent arterial to capillary blood volume in the brain, which may include a little fraction of venous capillary volume but are not influenced by radioactivity in the large vessels (Ohta et al, 1996; Okazawa and Vafaee, 2001). Thus, reductions of regional CBF and V0 are expected to reflect changes in small arteries in the cerebral cortex not influenced by radioactivity in major vessels. The complete recovery in the late phase of this study could have been induced by the mechanism of hemodynamic autoregulation. Oral administration of rizatriptan might have caused gradual vasoconstriction, and thus the CBF reduction would have occurred sluggishly, which would not cause immediate autoregulatory recovery of CBF via vasodilation of arterioles. Only a significant reduction of CBF or perfusion pressure would cause autoregulatory hemodynamic response to take place and this presumably occurred in the last stage of our experiment. The arterial perfusion pressure, which is expected to be proportionally correlated with CBF to V0 ratio (Okazawa et al, 2003), was mostly reduced at 70 mins from the rizatriptan administration. This suggests that the reduction of arterial perfusion pressure might have continued until cerebral autoregulation-driven vasodilatory reaction in arterioles resulted in CBF recovery. It is also possible that recovery from the significant reduction of CBF and blood volume in the later phase could have been caused by the declining pharmacological effects of the drug. However, the recovery was relatively prompt and the high plasma concentration of rizatriptan is expected to continue during almost 50 to 150 mins after treatment (Vyas et al, 2000), indicating that autoregulatory response seems more likely to explain this reaction.
Triptans have been used for migraine treatment, and several investigations reported that their vasoconstrictor effect counteracted vasodilatory changes in migraine headache (Friberg et al, 1991; Caekebeke et al, 1992). Both animal experiments and isolated human arteries, as well as clinical studies, reported their direct vasoconstrictor effect on various vessels (Feniuk et al, 1989; Parsons et al, 1989; Longmore et al, 1998), and the drug-induced vasoconstriction lasted for 1.5 h after drug administration in patients (de Hoon et al, 2000). In the latter study, the change in diameter of the CCA at 1.5 h from drug administration was significant but relatively small, and the resistance of the CCA was not different compared with placebo treatment. From these results, it can be inferred that CBF, as compared with baseline, might not have been changed at the time points at which those parameters were measured in their study, which is consistent with the present study that showed no change in CBF between baseline and 80 mins after treatment.
The mean maximal reductions in CBF and V0 from baseline were approximately 13% both in global and regional CBF, and approximately 13% and 17% in global and regional V0, respectively. However, focal differences in CBF and V0 were not observed at SPM analysis, suggesting that rizatriptan did not cause any regional effects on specific cortical regions and acted mainly on the large vessels, causing global changes as reported in the previous studies (Feniuk et al, 1989; Williamson et al, 1997; De Vries et al, 1999a, b ). The direct effect of the drugs on 5-HT1B receptors in the smooth muscles of the large vessels is hence confirmed by the absolute changes in CBF and V0. The CBF change was not caused by changes in systemic blood pressure nor blood gas because these parameters did not change after treatment. Although the systolic blood pressure was significantly increased after drug intake, which is caused by vasoconstriction of large vessels, the mean blood pressure did not change significantly as previously reported (Létienne et al, 2003). As flow-mediated vascular dilatation is inversely related to resting arterial diameter (Celermajer et al, 1992), cerebral arterioles would have driven the recovery of CBF even though larger vessels remained constricted.
In the current study, the earlier response of V0 as compared with CBF may reveal a more independent response of cerebral arterioles to the vasoconstriction caused by triptans. This is presumably due to the flow-induced vasodilatation in the smaller arteries known to be not affected by triptans (de Hoon et al, 2000). A small but significant decrease of CBF in earlier phase after treatment might have caused the vasodilatory response in cerebral circulation. Such response might have led the delayed changes in CBF mediated by cerebral autoregulation because of continuous vasoconstrictor effect in the major arteries on which triptans mainly act. This continuous vasoconstrictor effect might have caused slow supply of arterial blood into cerebral circulation and delay of response in CBF.
In conclusion, administration of triptans causes reduction in CBF and arterial-to-capillary blood volume, which is consistent with the therapeutic vasoconstrictor effect of the drug. Such reduction recovers in a short period of time, presumably induced by hemodynamic autoregulation occurring in cerebral circulation.
Footnotes
Acknowledgements
The authors thank staff of the Biomedical Imaging Research Center and the Department of Neurosurgery, University of Fukui, Faculty of Medical Science for technical and clinical support.

