Abstract
HIV-1 Tat protein plays an important role in inducing monocyte infiltration into the brain and may alter the structure and functions of the blood—brain barrier (BBB). The BBB serves as a frontline defense system, protecting the central nervous system from infected monocytes entering the brain. Therefore, the aim of the present study was to examine the mechanisms of Tat effect on the integrity of the BBB in the mouse brain. Tat was injected into the right hippocampi of C57BL/6 mice and expression of tight junction protein zonula occludens-1 (ZO-1) was determined in control and treated mice. Tat administration resulted in decreased mRNA levels of ZO-1 and marked disruption of ZO-1 continuity. These changes were associated with accumulation of inflammatory cells in brain tissue of Tat-treated mice. Further experiments indicated that Tat-mediated alterations of redox-related signaling may be responsible for decreased ZO-1 expression. Specifically, injections with Tat resulted in activation of extracellular signal-regulated kinase 1/2 (ERK1/2) and pretreatment with U0126, a specific inhibitor of ERK kinase, effectively ameliorated the Tat-induced diminished ZO-1 levels. In addition, administration of
Keywords
Introduction
The impairment of the blood—brain barrier (BBB) and monocyte infiltration are frequent pathological features of HIV-related encephalitis and dementia. Structurally, the BBB is composed of endothelial cells connected by tight junctions, basement membrane, and astrocytes end-feet. Strong evidence indicates that tight junction strands are important in BBB regulation in response to many pathological insults. Tight junctions consist of the integral membrane proteins such as occludin and claudins associated with membrane accessory proteins such as zonula occludens (ZO)-1 or ZO-2 (Mitic and Anderson, 1998), which form a scaffold to link tight junction protein to the cytoskeleton (Stevenson and Keon, 1998). The presence of intact cell junctions is important for maintaining a functional BBB. They seal neighboring cells together to prevent inflammatory cells entering into the brain (Matter and Balda, 1999). Disruption of the BBB is widely believed to be the main route of entry of HIV-1-infected monocytes into the central nervous system (CNS). However, only very limited literature information is available on a possible role for intercellular junctions and/or cell junction proteins in HIV-1 trafficking into the CNS. Disruption of tight junctions demonstrated by fragmentation and decreased immunoreactivity for occludin and ZO-1 was detected in the brains of AIDS patients with HIV-associated dementia and encephalitis (Boven et al, 2000; Dallasta et al, 1999). These changes were associated with accumulation of inflammatory cells, fibrinogen leakage, and cellular apoptosis (Boven et al, 2000; Dallasta et al, 1999).
A growing line of evidence indicates the role of HIV-1 Tat protein in the development of the CNS complications in HIV-1 infection. Tat can be secreted by HIV-infected cells and taken up by the neighboring uninfected cells. These studies were performed using HIV-infected T cell lines such as H9 and Jurkat cells, the epithelial cell line COS-1 (Ensoli et al, 1993; Helland et al, 1991; Marcuzzi et al, 1992; Westendorp et al, 1995) as well as peripheral blood mononuclear cells isolated from HIV-infected patients (Thomas et al, 1994). Tat is released from cells via a leaderless pathway in the absence of cell permeability changes (Chang et al, 1997). Elevated Tat protein and mRNA levels have been detected in the brains and blood of AIDS patients with dementia or progressive multifocal leucoencephalopathy (PML) (Johnston et al, 2001; Valle et al, 2000; Xiao et al, 2000). Regarding CNS localization, Tat-positive immunoreactivity was shown in microglia and astrocytes (Bonwetsch et al, 1999). In addition, Tat-positive monocytes have been found in the perivascular regions of the brain of patients with HIV encephalitis (Hofman et al, 1994).
Strong evidence indicates the HIV-1 infection is associated with long-term oxidative stress, which plays an important role in disease progression (Boven et al, 1999; Gil et al, 2003; Mollace et al, 2001; Perl and Banki, 2000). In particular, the levels of reactive oxygen species (ROS) were found to be significantly increased in demented AIDS patients as compared with nondemented AIDS controls (Boven et al, 1999). Furthermore, free radical product, nitrotyrosine, the footprint of peroxynitrite, was extensively distributed in the perivascular areas of brain tissue in AIDS patients with dementia (Mollace et al, 2001). Free radical products not only may directly contribute to increased permeability of the BBB but also may act as important second messengers in regulating signal transduction pathways and inflammation-related gene expression (Griendling et al, 2000; Kunsch and Medford 1999; Sundaresan et al, 1995). For example, increased oxidative stress can activate specific signal transduction pathways, such as the mitogen-activated protein kinase (MAPK) family (Kevil et al, 2000; Levinthal and DeFranco, 2004).
Our recent data show that cell or tissue exposure to Tat can result in alterations of redox status and increased oxidative stress (Flora et al, 2003; Toborek et al, 2003). However, the involvement of redox-regulated signaling pathways in Tat-induced brain pathology is not known. Therefore, the main goal of the present study was to assess if alterations of redox status can be involved in Tat-mediated dysregulation of the BBB and whether the MAPK pathway can be involved in these effects. Indeed, we found that Tat can activate the MAPK pathways and disrupt the integrity of tight junction protein ZO-1. Antioxidant
Materials and methods
Animals, Tat Injections, and Sample Preparation
The study was performed on male C57BL/6 mice (7- to 8-week-old; weight 25 to 28 g) obtained from the National Cancer Institute (Bethesda, MD, USA). All procedures and handling techniques were approved by the University of Kentucky Institutional Animal Care and Use Committee. Animals were maintained under environmentally controlled conditions and subject to a 12 h light/dark cycle with food and water
HIV-1 Tat protein is encoded by a gene consisting of two exons. The first exon contributes to the initial 72 amino acids and the second exon forms the remaining 14 to 32 amino acids. The functions of transactivation, neurotoxicity, and immune activation are all present within the first 72 amino acids formed by the first exon. Therefore, Tat1–72 was employed in the present study. Recombinant Tat1–72 was produced as described by Ma and Nath (1997). The concentration of Tat used in the present study (25 μg in 1 μL of PBS) was consistent with previously published reports (Flora et al, 2003; Maragos et al, 2003; Toborek et al, 2003). All Tat injections were performed into the right hippocampus using a 2 μL Hamilton syringe. The stereotaxic coordinates were: AP, -1.8; ML, 1.7, and DV, 2.1. After injections, the needle was left in place for 1 min before the scalp was stapled together and animals were returned to their cages. Specificity of Tat1–72-mediated effects was controlled by incubation of Tat1–72 at 100°C for 5 mins, followed by centrifugation of denaturated protein (21,000
To assess the involvement of alterations of redox status and the MAPK pathways in Tat-induced disruption of the BBB integrity, the animals were pretreated 30 mins before Tat injection with NAC (150 mg/kg body weight, intraperitoneally in 100 μL volume) or U0126 (10 mg/kg, intraperitoneally in 100 μL volume), respectively. The controls included animals that were injected with vehicles, such as PBS (vehicle for Tat and NAC) or DMSO (vehicle for U0126).
For immunofluorescence analyses, the brains were removed and serial coronal sections (10 μm each) of samples in OCT compound were cut on a cryostat (Cryostar HM 560 MV). For gene expression and Western blotting studies, the brains were rapidly removed, the hippocampi were dissected, immediately frozen in liquid nitrogen, and stored at -80°C until analysis.
Double-Immunofluorescence Staining and Confocal Laser Microscopy
Brain sections were permeabilized with 100% ethanol for 30 mins at 4°C and fixed with acetone 3 mins at -20°C. They were rinsed with TBS-T (50 mmol/L Tris, 150 mmol/L NaCl, 0.1% Triton X-100, pH 7.4) and nonspecific binding was blocked with 1.5% goat serum in TBS-T for 30 mins at room temperature. The sections were incubated overnight with primary rabbit polyclonal anti-ZO-1 antibody (dilution of 1:50; Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA) at 4°C. Negative controls were prepared by incubation of tissue sections with rabbit IgG. All primary antibodies were diluted in 1.5% goat serum in TBS-T. After removal of primary antibodies, the sections were washed with TBS-T and incubated with biotinylated goat anti-rabbit IgG (diluted 1:100, Santa Cruz Biotechnology) for 1 h at room temperature. After washing, sections were further incubated with streptavidin-conjugated Texas Red (diluted 1:100, Molecular Probes, Eugene, OR, USA) for 1 h at room temperature.
Double immunostaining was performed to determine ZO-1 expression associated with monocytes infiltration in response to Tat treatment. After ZO-1 labeling, the sections were washed with TBS-T, blocked with 1.5% goat serum in TBS-T, and incubated with rabbit anti-macrophage antibody (dilution of 1:100; Accurate Chemical, Westbury, NY, USA) or rat anti-integrin αM antibody (dilution of 1:100; Chemicon, Temecula, CA, USA) at 4°C overnight. Negative controls were prepared by incubation of tissue sections with rabbit IgG or rat IgG. After labeling for macrophage, the slides were washed with TBS-T and incubated with FITC-conjugated goat anti-rabbit IgG or goat anti-rat IgG (dilution of 1:100; Chemicon). After a final washing with TBS-T, slides were mounted and examined with a Leica TCS DM RXE laser scanning confocal microscope, equipped with argon and krypton lasers, and coupled with a Nikon fluorescence microscope (Nikon, Inc., Melville, NY, USA). A plan***neofluor × 40 (numerical aperture, 1.0) oil-immersion objective was used for imaging of Texas red- and FITC-labeled tissues. Images were analyzed with the standard system operating software provided with Leica TCS laser confocal microscopy. For double-labeling studies, separate optical images of Texas red and fluorescence were captured from the same optical section. The captured images were then pseudocolored red or green, a digital overlay was generated, and companion images were superimposed.
The Nomarski differential interface contrast mode was employed for imaging translucent tissues. Images from the same optical sections visualized by fluorescent staining and the Nomarski technique were captured and superimposed.
Western Blotting
The hippocampi were homogenized in ice-cold lysis buffer (20 mmol/L Tris (pH 7.4), 0.5% Triton X-100, 0.5% NP-40, 1 mmol/L Na2VO4, 150 mmol/L NaCl, 1 mmol/L EDTA, 10 μmol/L leupeptin, 1 μmol/L pepstatin, 1 μmol/L aprotinin, 1 mmol/L PMSF, 250 μmol/L 4-2-aminoethyl benzene-sulfonyl fluoride). Forty microgram of protein was loaded onto each well of 7.5% SDS-polyacrylamide gel (SDS-PAGE) for electrophoresis. For detecting phosphorylated extracellular signal-regulated kinase 1/2 (ERK1/2), 10% SDS-PAGE was used. Then, proteins were transferred to a nitrocellulose membrane (BioRad, Hercules, CA, USA) by electroblotting, washed, blocked with 5% milk in Tris-buffered saline with 0.05% Tween 20, and probed with specific primary antibody overnight at 4°C. The following primary antibodies were used in the present study: rabbit anti-ZO-1 (1:500, Santa Cruz Biotechnology), mouse anti-phospho-ERK1/2, mouse anti-ERK (1:500, Santa Cruz Biotechnology), and rabbit anti-actin antibody (1:1000, Sigma, St Louis, MO, USA). As the secondary antibody, HRP-conjugated goat anti-rabbit IgG or HRP conjugated goat anti-mouse IgG antibody (1:1000; Santa Cruz Biotechnology) were used. Membranes were washed and the assessed proteins were detected using a ECL chemiluminescence reagent (Amersham, Biosciences, Piscataway, NJ, USA) and X-ray film. Relative intensity of the bands was quantified using Scion image analysis software (Scion Corporation, Frederick, MD, USA).
Real-Time Reverse Transcriptase-Polymerase Chain Reaction
Real-time reverse transcriptase-polymerase chain reaction (RT-PCR) was employed to determine ZO-1 mRNA in the hippocampi. Briefly, total RNA was extracted from hippocampus using RNeasy Mini Kit (Qiagen, Valencia, CA, USA), and 1 μg of RNA was reverse-transcribed using Reverse Transcription System (Promega, Madison, WI, USA) in a total volume of 20 μL. The following conditions were employed for reverse transcription: 25°C for 10 mins, 48°C for 30 mins, and 95°C for 5 mins. To assess ZO-1 mRNA expression, primer pairs were designed using Primer Express software (Applied Biosystems, Branchburg, NJ, USA). The following primers were used: ZO-1 forward, 5′-TGT GAG TCC TTC AGC TGT GGA A-3′ and ZO-1 reverse, 5′-GGA ACT CAA CAC ACC ACC ATT G-3′. Real-time PCR was conducted on the ABI Prism 7000 system, using 2 μL of RT product, SYBR Green PCR Master Mix (Applied Biosystems), and primers in a total volume of 25 μL. The following thermocycling conditions were employed: 95°C for 10 mins, followed by 95°C for 15 secs and 60°C for 60 secs. Concentration of each sample was calculated from the threshold cycle (Ct) and normalized according to the β-actin (a housekeeping gene) mRNA levels. A standard curve for each gene studied was generated by preparing serial dilutions of the cDNA sample obtained from the hippocampi of vehicle-injected mice.
Statistical Analysis
The data were analyzed by one-way ANOVA and the pairwise comparison was performed using Student—Newman—Keuls
Results
Tat Disrupts ZO-1 Integrity and Induces Brain Infiltration with Inflammatory Cells
Disruption of tight junction proteins was detected in HIV-1 patients with HIV-1 encephalitis and dementia (Boven et al, 2000; Dallasta et al, 1999). Therefore, we assessed whether treatment with Tat can alter expression of tight junction protein ZO-1. The studies were performed on epithelial cells on the edge of the cerebral peduncle near the Tat-injected hippocampus (Figure 1A), in endothelial cells of hippocampal microvessels (Figure 1B) and in choroid plexus of the lateral ventricle (Figures 2A and Figure 2B). In addition, infiltration of the brain tissue with inflammatory cells was evaluated by staining for the presence of integrin αM- (Figures 1A and Figure 2A) or macrophage-immunoreactive cells (Figures 1B and Figure 2B).
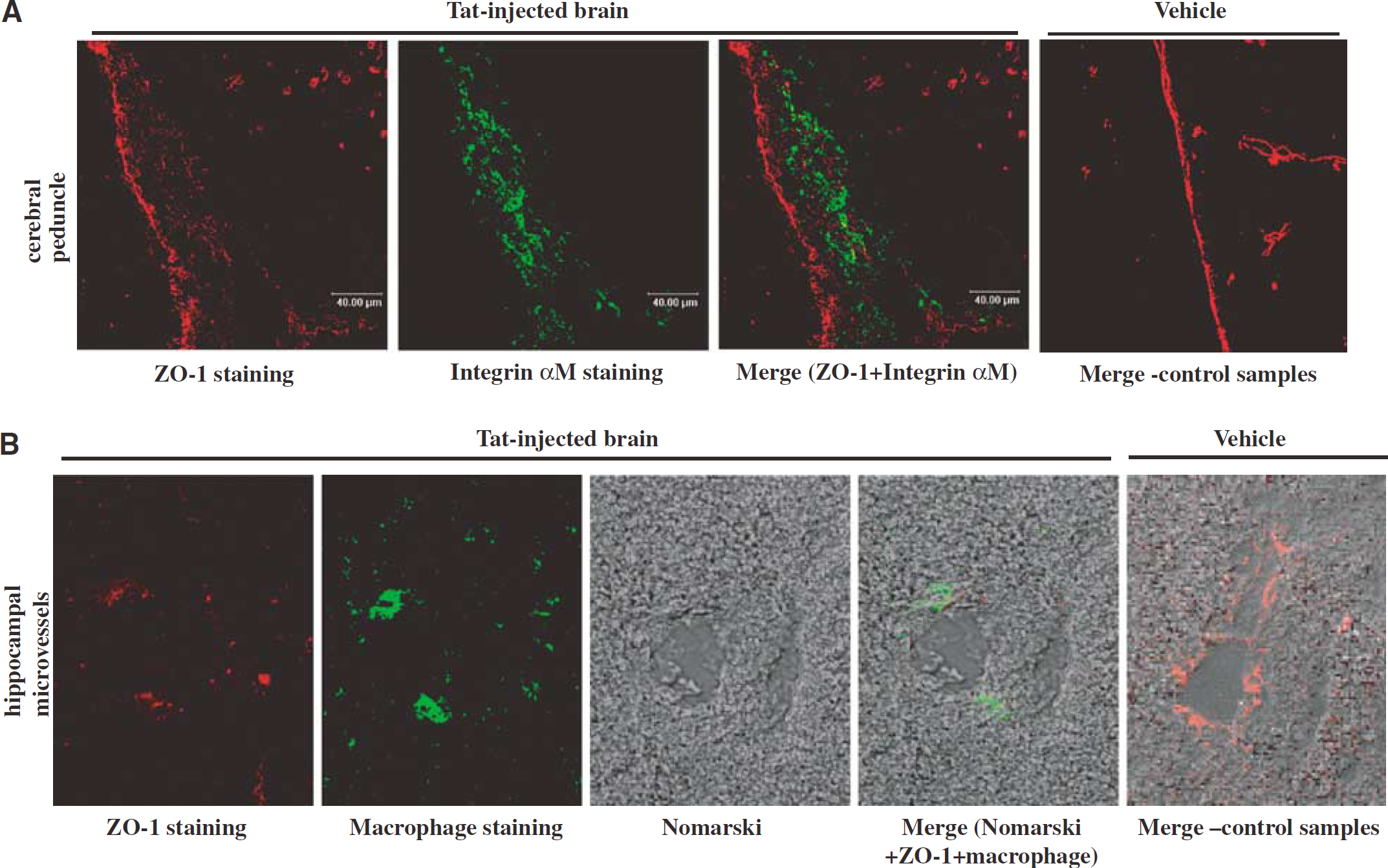
Tat disrupts endothelial and epithelial zonula occludens (ZO)-1 immunoreactivity and stimulates inflammatory infiltration in the hippocampus. Tat1–72 (25 μg/μL) was injected into the right hippocampus, and ZO-1 immunoreactivity and infiltration with inflammatory cells were studied 24 h after the injection. Hippocampal sections were stained for ZO-1 and the same optical sections were stained for the presence of inflammatory cells, such as integrin aM-positive cells (
As indicated in Figure 1, a 24 h exposure to Tat resulted in dramatic disruption of ZO-1 immunoreactivity in and around Tat-injected hippocampus. Specifically, altered continuity of ZO-1 immunoreactivity with the formation of shreds and spot-like structures were displayed on epithelial cells on the edge of the cerebral peduncle near the Tat-injected hippocampus (Figure 1A) as well as on endothelial cells of the microvessels of Tat-injected hippocampus (Figure 1B). Disruption of ZO-1 in Tat-injected animals was associated with infiltration of brain tissue with integrin αM-positive cells, which detects monocyte, macrophages, and activated microglia cells (Figure 1A), as well as accumulation of macrophage-immunoreactive cells in the perivascular space (Figure 1B). In contrast, continuous ZO-1 staining without detectable presence of inflammatory cells was revealed in the corresponding areas of the vehicle-injected brain (Figures 1A and Figure 1B).
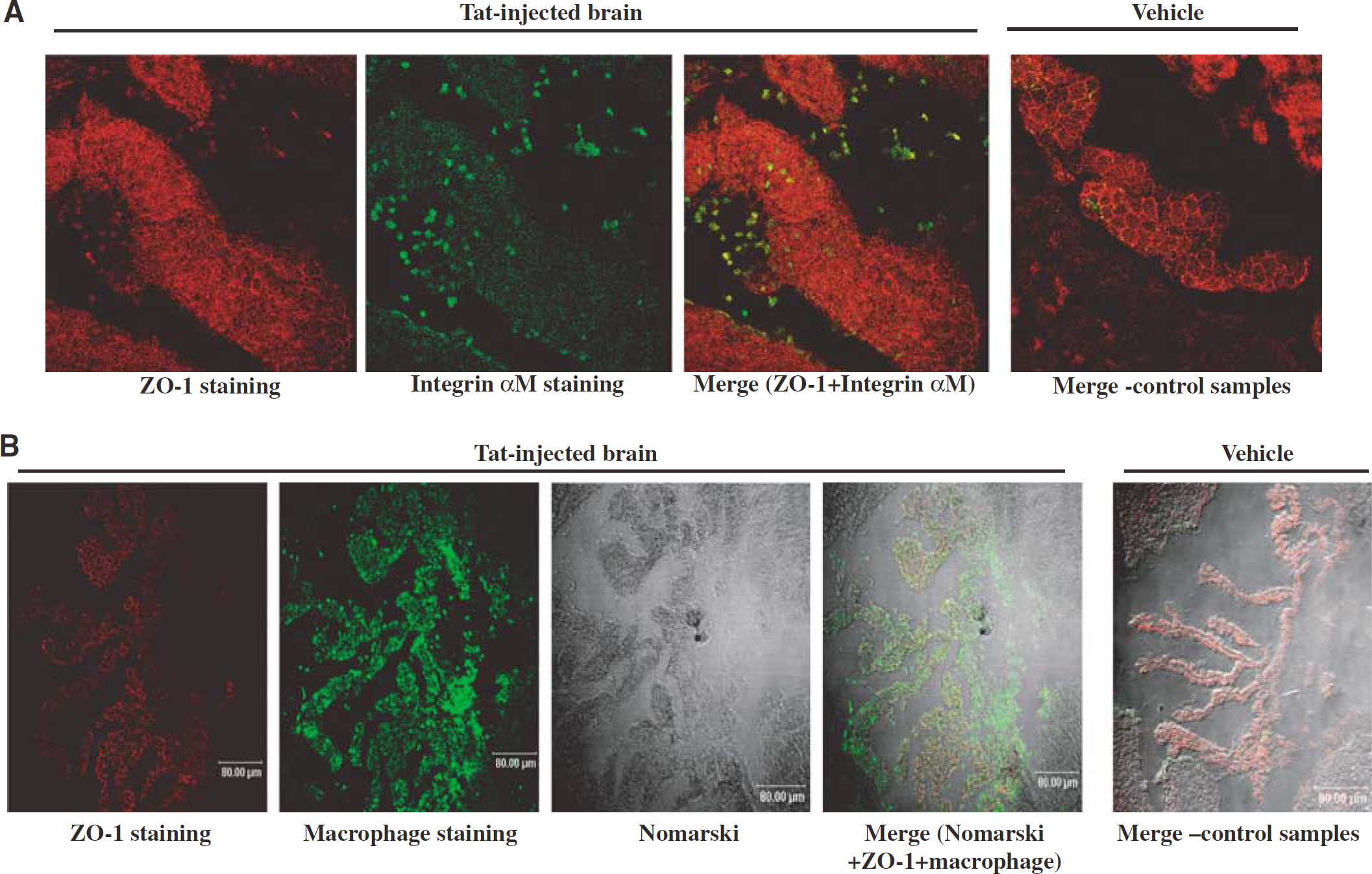
Tat disrupts ZO-1 immunoreactivity and stimulates inflammatory cell accumulation in choroid plexus. Experiments were performed as described in the legend to Figure 1. Brain sections of choroid plexus of the right lateral ventricle were stained for zonula occludens (ZO)-1 and the same optical sections were stained for the presence of inflammatory cells such as integrin αM-positive cells (
Fragmentations and shredding of ZO-1 immunoreactivity associated with prominent infiltration of inflammatory cells were also observed in choroid plexus of the lateral ventricle in animals injected with Tat into the hippocampus (Figure 2). Figure 2A indicates infiltration with integrin αM-positive cells and Figure 2B shows accumulation of macrophage-immunoreactive cells. Intact ZO-1 immunostaining and only very limited number of inflammatory cells were present in choroid plexus of the vehicle-injected animals (Figures 2A and Figure 2B).
Tat Downregulates ZO-1 mRNA and Protein Levels in Dose-Dependent Manners
The effects of different doses of Tat (5, 10, or 25 μg/μL) on the mRNA and protein levels of ZO-1 in mouse hippocampal regions are shown in Figure 3. Real-time RT-PCR indicated that Tat downregulated ZO-1 mRNA in a dose-dependent manner. Decreased ZO-1 mRNA expression was already prominent in mice injected with Tat at a dose as low as 5 μg (Figure 3A).
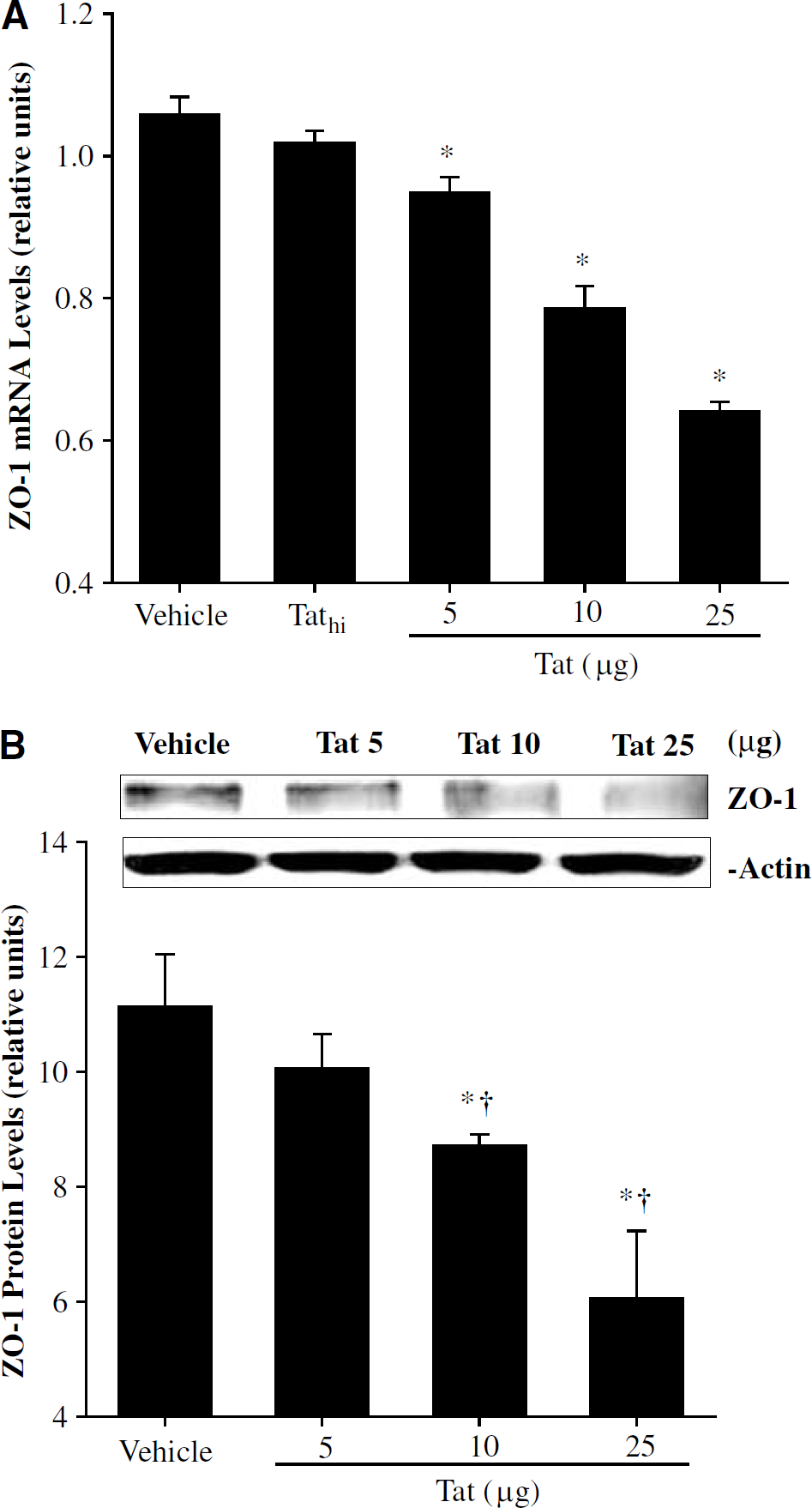
Tat downregulates zonula occludens (ZO)-1 mRNA and protein levels in brain tissue in dose-dependent manners. Tat (5, 10, or 25 μg/μL) was injected into the right hippocampus, and ZO-1 mRNA levels (
The most marked decreased mRNA level of ZO-1 in hippocampus was observed in mice injected with 25 μg Tat (Figure 3A). The supernatant from the heat-inactivated Tat (Tathi) solution did not affect ZO-1 mRNA levels. Therefore, treatment with this negative control was discontinued in the remaining experiments.
Figure 3B indicates the dose-dependent effects of Tat on protein levels of ZO-1 as measured by Western blotting. Tat at the dose of 10 μg significantly decreased ZO-1 protein levels as compared with control values. These effects were even more pronounced in the hippocampi injected with 25 μg f Tat (Figure 3B).
N-acetylcysteine Attenuates Tat-Induced Alterations of Zonula Occludens-1 mRNA and Protein Expression
To quantify Tat-induced alterations of ZO-1 expression, ZO-1 mRNA and protein levels were determined by real time RT-PCR and Western blot, respectively. As indicated in Figure 4A, a 12 h exposure to Tat resulted in reduced ZO-1 mRNA levels as compared with the vehicle-injected animals. However, pretreatment with antioxidant NAC 30 mins before Tat injection markedly attenuated these effects. Indeed, ZO-1 mRNA levels in the Tat plus NAC group were approximately 20% higher than those in the Tat group (Figure 4A).
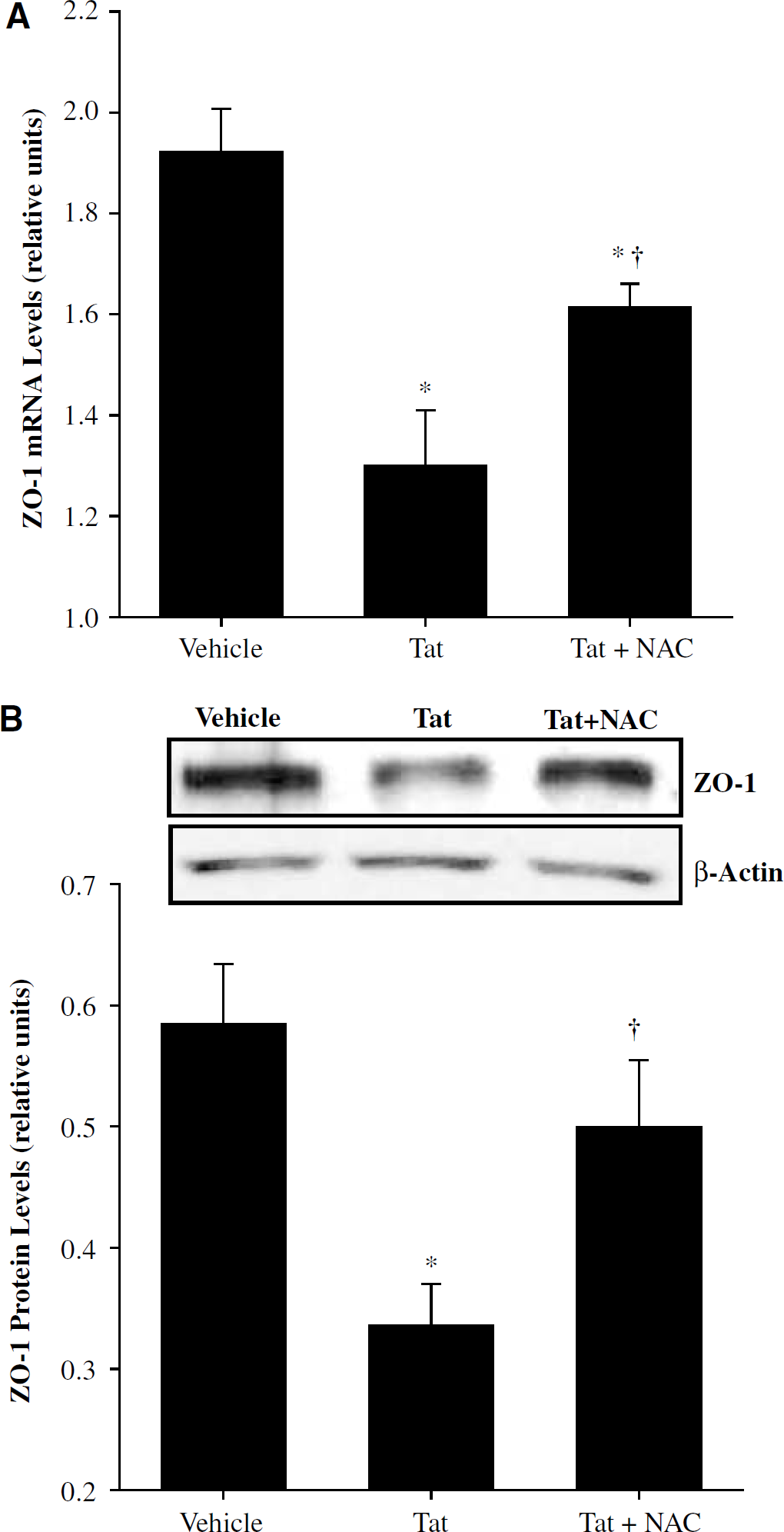
Consistent with the effects on mRNA expression, a 24 h exposure to Tat significantly reduced ZO-1 protein levels as compared with control tissue (Figure 4B). Pretreatment with NAC attenuated Tat-induced downregulation of ZO-1 protein levels, indicating the involvement of oxidative stress and/or alterations of redox status in Tat-induced alterations of ZO-1 expression.
Redox-Sensitive Activation of Extracellular Signal-Regulated Kinase 1/2 is Involved in Tat-Induced Alterations of Zonula Occludens-1 Expression
To determine if Tat treatment can activate ERK1/2 and whether this effect can be mediated by a redox-sensitive pathway, mice were pretreated with NAC before injection with Tat into the right hippocampus. Then, the levels of phosphorylated ERK1/2 (pERK1/2) and total ERK were determined by Western blot. Figure 5 depicts that a 6-h exposure to Tat can significantly increase levels of pERK1/2 without affecting the levels of total ERK. Tat-induced activation of ERK1/2 was completely inhibited by a pretreatment with NAC, indicating that this effect can be mediated by a redox-sensitive mechanism.
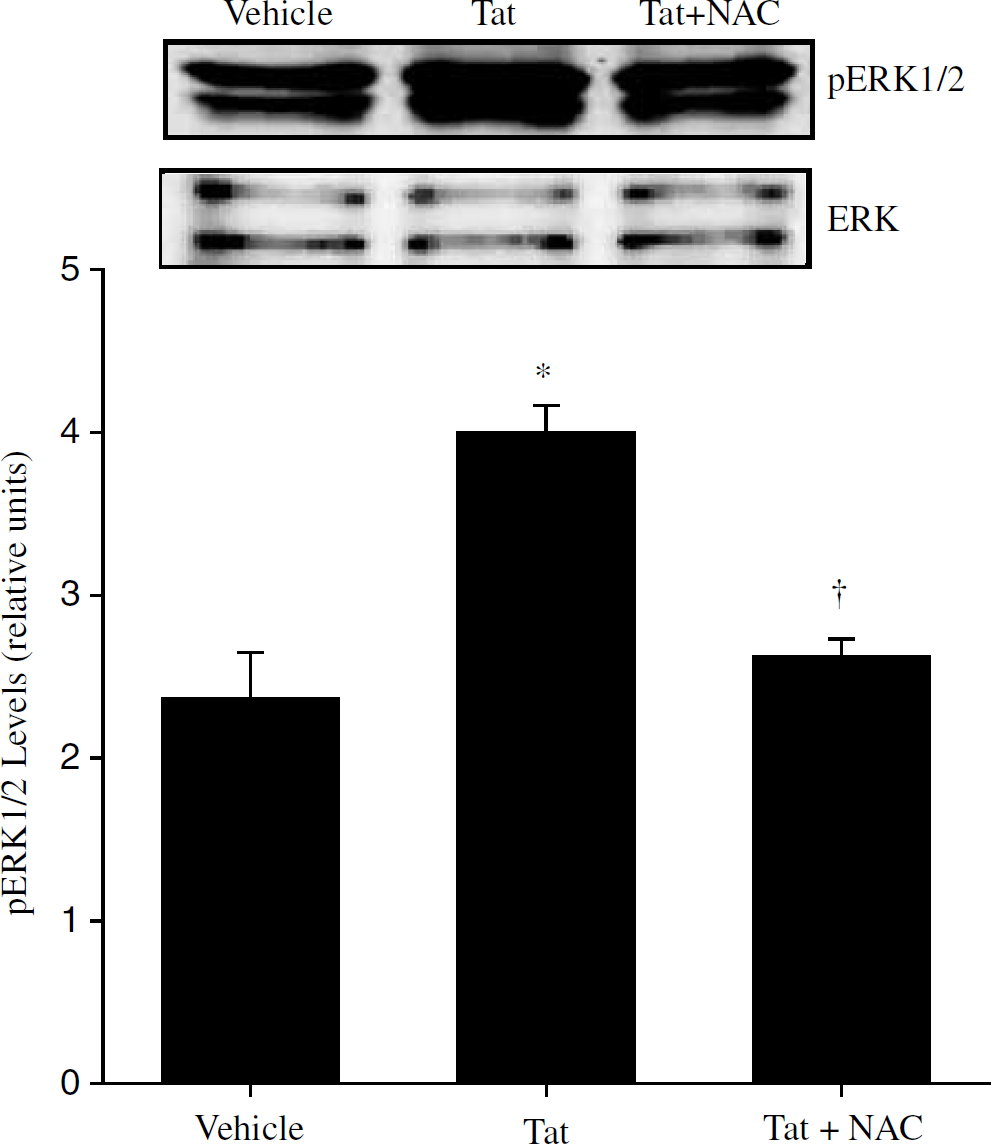
In addition, a series of experiments was performed to determine the involvement of ERK1/2 signaling on Tat-induced alterations of ZO-1 expression. Pretreatment with U0126, a specific inhibitor of ERK1/2 kinase (MEK), attenuated Tat-mediated decrease in ZO-1 mRNA levels (Figure 6A). In addition, U0126 partially protected against Tat-induced alterations of ZO-1 protein level (Figure 6B).
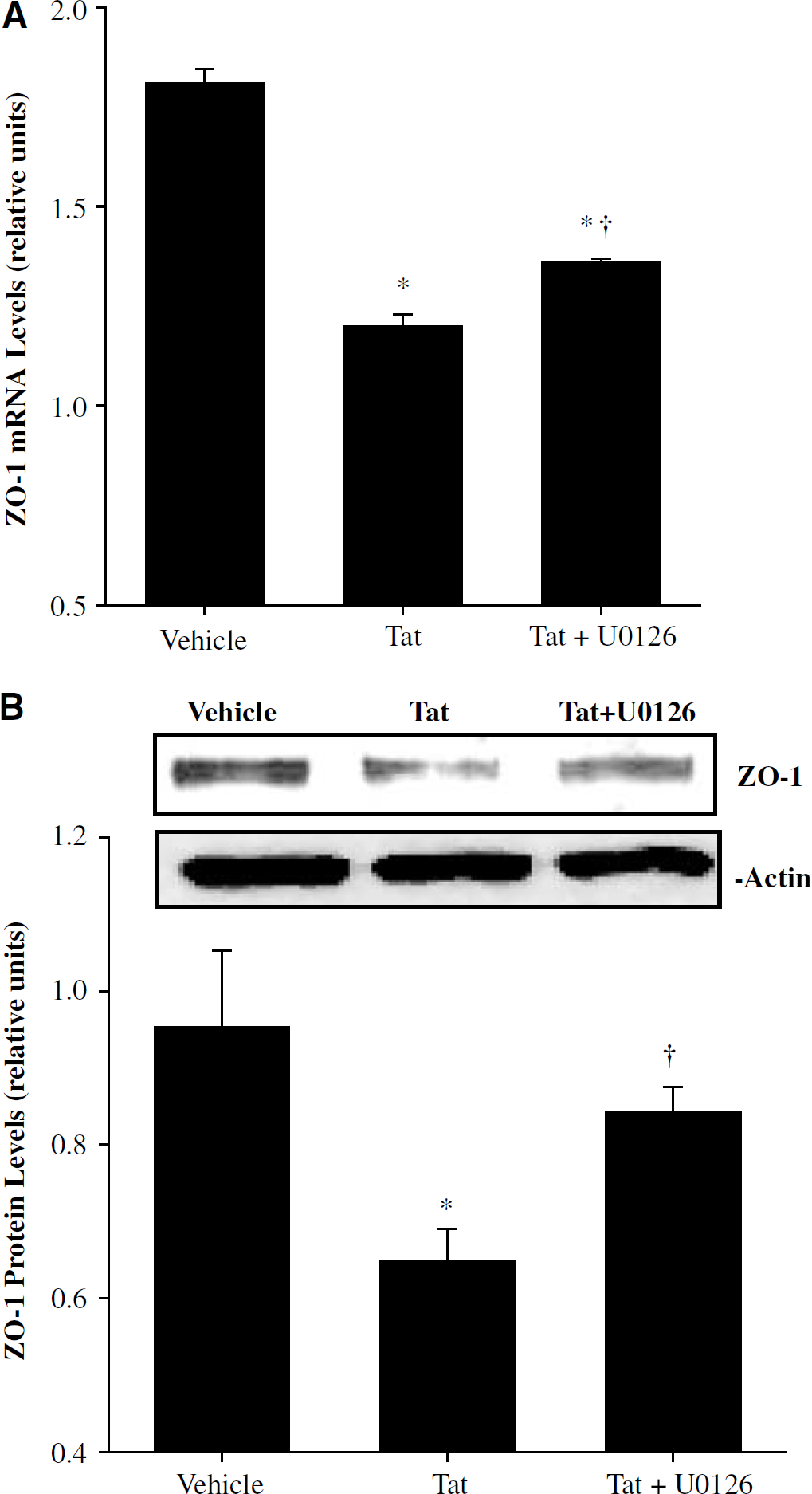
Extracellular signal regulated kinase 1/2 (ERK1/2) is involved in Tat-induced alterations of zonula occludens (ZO)-1 expression. U0126 (10 mg/kg, intraperitoneally) was administrated 30 mins before hippocampal Tat1–72 injection (25 μg/μL). Zonula occludens-1 mRNA levels (
Discussion
Zonula occludens-1 is a member of a family of protein kinases of the membrane-associated guany late kinase homologues (MAGUKs) and is involved in signal transduction at locations of cell-cell contacts. Zonula occludens-1 also acts as the scaffold to organize occludin at the cell junction sites (Mitic and Anderson, 1998) and can link occludin to the actin cytoskeleton (Gumbiner, 1993). Thus, ZO-1 plays a regulatory role in the cellular permeability (Lampugnani et al, 1995; Staddon et al, 1995). Zonula occludens-1 is susceptible to pharmacological regulations, and factors such as glucocorticoids, interferon-γ, epidermal growth factor, or hepatocyte growth factor have all been shown to modulate ZO-1 expression (Gonzalez-Mariscal et al, 2000).
Results of the present paper indicate that administration of Tat into the brain can result in significant disruption of ZO-1 immunoreactivity associated with decreased ZO-1 mRNA and protein levels. These effects resemble those observed in HIV-infected patients. Specifically, fragmentation and loss of immunoreactivity for ZO-1 and occludin were observed in patients who died because of HIV-associated encephalitis (Dallasta et al, 1999). Loss of ZO-1 immunoreactivity also was detected in patients with HIV-associated dementia (Boven et al, 2000). Most importantly, disruption of ZO-1 immunoreactivity was highly correlated with monocyte infiltration and the degree of HIV dementia (Boven et al, 2000). In the present study, the locations of Tat-induced disruptions of ZO-1 also were associated with monocyte infiltration (Figure 1 and Figure 2). This observation is of significant importance, because activated inflammatory cells can initiate cellular release of free radical, cytokines, and growth factors, which can further amplify the initial BBB breakdown. In addition, it is considered that HIV trafficking into the brain is mediated through a ‘Trojan horse’ mechanism, in which HIV-infected circulating monocytes enter the CNS early in the course of infection through breaches of the BBB (Liu et al, 2000). Then, the virus may reside in microglia and macrophages of the perivascular space in the CNS (Gartner, 2000) and induce the development of encephalitis or dementia at the later stages of HIV infection. We also reported
The present study identified for the first time that antioxidant NAC can protect against Tat induced diminished ZO-1 expression (Figure 4). This finding strongly suggests that decreased ZO-1 expression may be mediated by Tat-induced alterations of cellular redox status, implicating also that Tat-induced oxidative stress can be involved in the breakdown of the BBB. To support this hypothesis, our previously published studies indicated that intracerebral administration of Tat can induce oxidative stress and activate redox-stimulated transcription factors and inflammatory genes in brain tissue (Flora et al, 2003; Pu et al, 2003).
Intracellular ROS are recognized as an important second messenger in regulation of signal transduction pathways and inflammation-related gene expression (Griendling et al, 2000; Kunsch and Medford, 1999; Sundaresan et al, 1995). For example: ROS can activate epidermal growth factor receptor, c-Src, protein kinase C (PKC), Ras/MAPK, and Akt/protein kinase B (Finkel, 2003; Griendling et al, 2000; Torres, 2003). In the present study, ERK1/2 activation by Tat was completely inhibited by NAC, a precursor of glutathione and potent antioxidant, indicating that increase of oxidative stress and/or alterations of cellular redox status may be responsible for this effect. To support this notion, literature data also indicated that cellular oxidative stress can lead to stimulation of ERK activity (Genersch et al, 2000; Kevil et al, 2000; Levinthal and DeFranco, 2004). In addition, our results revealed that ERK1/2 activation can be involved in the regulation of ZO-1 expression. Specifically, U0126, an inhibitor of ERK kinase (MEK), attenuated the Tat-induced downregulation of ZO-1 (Figure 6). Thus, Tat-induced oxidative stress can play an important role in affecting the integrity of tight junction system via activation of ERK1/2 pathway.
Our novel finding that activation of the MAPK/ERK1/2 pathway can be involved in Tat-induced alterations of ZO-1 expression are supported by the literature data. For example, Raf-1 (MAPK kinase kinase) was shown to disrupt epithelial tight junctions via downregulation of occludin, another tight junction protein (Li and Mrsny, 2000). The family of Ras-related small GTP-binding proteins, such as RhoA and Rac1, which can initiate MAPK activation, has also been reported to regulate tight junction structure and function (Gopalakrishnan et al, 1998; Jou et al, 1998). Recent data also indicate that activation of the ERK1/2 pathway can induce tight junction disruption in human corneal epithelial cells (Wang et al, 2004). Finally, evidence indicates that tight junction assembly and function can be modulated by a number of signaling molecules, including cAMP, Ca2+, PKC, G proteins, phospholipase C, and diacylglycerol (Balda et al, 1993; Mullin et al, 1998).
The specific mechanisms of decreased ZO-1 expression in response to Tat-induced activation of ERK1/2 are not fully understood. However, it is possible that the ERK pathway can influence ZO-1 mRNA and protein levels through upregulation of redox-regulated transcription factors and inflammatory genes. Specifically, activation of ERK1/2 may directly or indirectly induce phosphorylation of downstream target transcription factors such as NF-κB, Jun/AP-1, and/or Elk (Griendling et al, 2000; Hsu et al, 2000; Whitmarsh and Davis, 1996; Yang et al, 2003), which ultimately alter cellular function. NF-κB and AP-1 have been implicated in transcriptional regulation of a wide range of genes involved in cellular inflammatory responses resulting in producing inflammatory cytokines (Sun and Oberley, 1996). Our previous studies also indicated that Tat activated NF-κB and AP-1 and induced cytokine production in brain tissue (Pu et al, 2003; Toborek et al, 2003). Elevated cytokine levels may contribute to alterations of tight junction integrity and further increase endothelial permeability and leukocyte extravasation (Sharief et al, 1993). Indeed, it was shown that expression of the occludin promoter can be affected by tumor necrosis factor-α (TNF-α) or interferon-γ treatment (Mankertz et al, 2000), and cytokines, nitric oxide, and histamine can reduce ZO-1 mRNA expression (Han et al, 2003; Takeuchi et al, 2001). Elevated production of inflammatory cytokines also can contribute to alterations of cellular redox status and may further increase ERK activation (Elbim et al, 2001; Genersch et al, 2000; Xiong et al, 2000).
In addition to influencing ZO-1 expression at the transcriptional level, the ERK1/2 signaling pathway may be involved in degradation, disruption, and rearrangement of this tight junction protein (Chen et al, 2000; Mori et al, 2002; Wang et al, 2004). For example, activation of ERK1/2 can lead to activation of matrix metalloproteinase-9 (MMP-9), which has been shown to degrade ZO-1 (Mori et al, 2002). Furthermore, it was shown that inhibition of the ERK pathway resulted in the rearrangement of actin filaments from stress fibers into tight junction proteins and E-cadherin (Chen et al, 2000).
In conclusion, our study showed for the first time that intrahippocampal injection with Tat can decrease tight junction protein ZO-1 expression at the gene and protein levels. Pretreatment with NAC partially attenuated Tat-induced downregulation of ZO-1. In addition, inhibition of ERK1/2 signaling either by NAC and U0126 ameliorated Tat-induced alterations of ZO-1 expression. Thus, Tat-mediated disruption of ZO-1 can be mediated by alterations of cellular redox status via activation of the ERK1/2/pathway. These results also indicate that antioxidant treatment may be a supportive therapeutic approach in HIV-1 infected patients to prevent the disruption of the BBB.
Footnotes
Acknowledgements
We would like to thank Dr Avindra Nath (Johns Hopkins University, Baltimore, MD) for providing Tat protein.
