Abstract
Anesthetic exposure during pregnancy is viewed as a relatively routine medical practice. However, recent rodent studies have suggested that common anesthetic agents can damage the developing brain. Here we assessed this claim in a higher order species by exposing previously instrumented near-term pregnant sheep at gestational day 122 (±1) to a combination of midazolam, sodium thiopental, and isoflurane at clinically relevant doses and means of anesthetic delivery (i.e., active ventilation). Four hours of maternal general anesthesia produced an initial increase in fetal systemic oxygenation and a sustained increase in fetal cerebral oxygenation, as determined by in utero near-infrared spectroscopy. Postexposure monitoring failed to identify changes in physiologic status that could be injurious to the fetal brain. Finally, through the histologic assessment of noninstrumented sheep at the same gestational time point, we found no evidence for a direct fetal neuro-toxic effect of our triple-drug regimen. Collectively, these results appear to corroborate the presumed safety of inhalational anesthetic use during pregnancy.
Introduction
The need to administer general anesthesia to a gravid patient can arise at any time before delivery as part of the surgical correction of both obstetric- and nonobstetric-related pathologies (Rosen, 1999). The practice is common enough (50,000 and 75,000 operations per year in the Untied States; Mazze and Kallen, 1989; Beilin, 1998) for maternal and fetal exposure to anesthetic agents to be viewed as a relatively routine occurrence.
In addition to making the patient insensate and amnestic during surgical manipulations, the appeal of general anesthesia (as apposed to regional or local anesthetic techniques) is the control it gives the attending anesthetist over respiratory status. This benefit notwithstanding, at present, it is unclear what if any consequences such drug exposures have on the developing conceptus. Attempts have been made to describe the overt fetal physiologic and pharmacologic effects of anesthetics by noninvasive monitoring during and after completion of the maternal procedure and by retrospective analyses (Levy et al, 1998). However, a thorough delineation of the human fetal responses is complicated by the inability to separate the anesthetic effects from the underlying pathology that necessitated their use.
Recently, a group of investigators reported that exposure to various anesthetic agents causes widespread neuro-degeneration in the developing rat brain (Jevtovic-Todorovic et al, 2003). The report garnered a significant amount of publicity (e.g., Anand and Soriano, 2004; Davidson and Soriano, 2004; Olney et al, 2004; Todd, 2004) because the agents in question (isoflurane, midazolam, and nitrous oxide) are commonly administered to obstetric and pediatric patients. Before eliminating these drugs from clinical practice, one must recognize the limitations associated with this study. The protocol involved passive exposure to the inhalational agents (i.e., rat pups were not intubated) and the period of exposure (6 h) represents between 1% and 2% of the total brain growth spurt period in this species. As a result, the paradigm does not effectively mimic clinical patterns of anesthetic exposure (neither near-term nor neonatal) and thus the report may be overstating the human risk. In this regard, a preliminary study using intubated and actively ventilated neonatal swine found no evidence of increased neuronal cell death after 6 h of exposure to the same drug combination (Loepke et al, 2004).
The present research focused on the other area of concern identified by Jevtovic-Todorovic et al: in utero anesthetic exposure. Experiments were conducted on near-term pregnant sheep at the peak of the brain growth spurt (Dobbing and Sands, 1979). This neurologic developmental stage is roughly equivalent to the full-term human and the 7-day rat pup and it is a time when the developing nervous system can exhibit an increased sensitivity to insult. For the studies, we assessed the fetal systemic (arterial blood gas and cardiovascular changes) and central (cerebral oxygenation monitored with near-infrared spectroscopy, NIRS; Reynolds et al, 2003) effects of maternal general anesthesia. We used a combination of midazolam, thiopental, and isoflurane, a regimen commonly used at our (and other) institution for anesthetizing parturients. The duration of exposure was 4 h, a period that would encompass most, if not all, of the surgical procedures conducted on gravid patients. Fetal brain histologic assessments were conducted on a separate, noninstrumented, cohort after exposure to the same anesthetic combination.
MATERIALS AND METHODS
Subjects
All aspects of the surgical and experimental protocols were approved by the Duke University Institutional Animal Care and Use Committee. Time-dated Q-fever-negative near-term pregnant ewes were obtained from a commercial supplier. On arrival at the Duke University Vivarium, each ewe scheduled to undergo the instrumentation surgery received an intramuscular injection of procaine penicillin G (1,200,000 IU). This penicillin regimen was repeated at 48-h intervals for the duration of the study. The sheep were housed individually and were allowed ad libitum access to food and water except for a 12- to 14-h fasting period before the surgical procedure; food was not withheld before the anesthetic exposure part of the study.
Maternal/Fetal Instrumentation
Instrumentation and catheterization of the ewe and fetus were performed at a mean gestational age of 120±1 days (term is approximately 145 days). The surgery was conducted using sterile technique and all components were cold-gas sterilized before installation. After presedation with midazolam (1 mg/kg, intravenous), surgical anesthesia was induced with sodium thiopental (7 mg/kg, intravenous) and the lungs intubated. Surgical anesthesia was maintained with 1% to 2% isoflurane in oxygen delivered by a Narkomed 2B ventilation system (North American Dräger, Telford, PA, USA). To prevent aortocaval compression, ewes were placed (tilted) in the left lateral position (Eckstein and Marx, 1974). Using standard surgical techniques, catheters were inserted into the maternal left femoral artery and left jugular vein and both the right and left fetal femoral arteries. The fetal head was then exteriorized and the skull was revealed after a midline scalp incision. To install the NIRS fiberoptic bundle, a burr hole was drilled along the midline of the head, approximately 10 mm posterior to the coronal suture. The laser light source was gently inserted through this hole onto the dura and secured with the use of a custom-made metal plate (2 cm diameter) and a series of small self-tapping screws. A second metal plate was used to secure the detector to the skull, 15 mm anterior to the light source. The fetus was then returned to the uterus. As the incisions were closed, a catheter was installed in the amniotic cavity and an electromagnetic flow probe (Transonic Systems Incorporated, Ithaca, NY, USA) was placed around the left uterine artery. The catheters, flow probe, and fiberoptic bundles were tunneled subcutaneously and then exteriorized through a small incision in the left flank of the ewe.
On completion of the surgery (3 to 4 h), bupivacaine (0.25%, subcutaneous) was infused around the incision sites and the animal returned to its pen. Nalbuphine hydrochloride, up to 1.0 mg/kg intramuscular (or similar opioid) was administered to the ewe as needed to control postsurgical pain. In addition to Pen G, prophylactic antibiotic therapy involved two doses of gentamicin (ewe 80 mg intravenous; fetus 40 mg via the amniotic catheter) and daily maternal intravenous infusions of sulfamethoxazole***trimethoprim (800 mg/160 mg intravenous in 5% glucose). All animals were allowed at least 48 h to recover from instrumentation before conducting the anesthetic exposure experiment.
Maternal General Anesthesia
On the day of experimentation, the ewe was placed in a support harness (Munks Livestock Sling Manufacturing Company, Anacortes, WA, USA) within a transportation cart. The arterial catheters were flushed with heparinized saline and then attached to force transducers (Transpac; Abbott Laboratories, North Chicago, IL, USA); maternal and fetal cardiovascular data along with uterine blood flow (UBF) were recorded using a 16-channel PowerLab system (ADInstruments, Colorado Springs, CO, USA). Fetal arterial pressure measurements were corrected for variations in intrauterine pressure by subtracting simultaneously measured amniotic fluid pressure. Fetal cerebral oxygenation, as measured by changes in oxygenated, deoxygenated, and total hemoglobin (oxyHb, deoxyHb, and totalHb, respectively) was continuously recorded using an NIRS monitor (CAS Medical Systems, Branford, CT, USA) adapted for fetal sheep use.
After a baseline recording period (30 to 60 min), general anesthesia was provided by the combination of midazolam, thiopental, and isoflurane. In this setting, isoflurane concentration was kept at exactly 1.5% as measured by an airway gas monitor (Datex Instrumentation Corporation, Helsinki, Finland). The ewe's ventilation was actively managed to keep end-tidal CO2 below 35 mm Hg; other standard operative monitoring devices (e.g., pulse oximetry) were also used. Inhalational anesthesia was delivered for 4 h after which the vaporizer was turned off. Extubation occurred when the swallowing reflex appeared and the ewe resumed a normal respiratory pattern. To monitor for postanesthetic effects, the maternal and fetal physiologic recordings continued overnight. During the study, maternal and fetal arterial blood samples were obtained at regular intervals; blood gas status was quantitated with a Gem Premier 3000 blood gas analyzer (Instrumentation Laboratory, Lexington, MA, USA).
Fetal Brain Histology
To delineate the fetal neurologic anesthetic effects independent of any modifications produced by installation of the catheters and NIRS hardware, this portion of the study was conducted on a separate uninstrumented cohort of near-term pregnant sheep at the same gestational age (120±1 days). Ewes underwent the anesthetic regimen and then 6 days later were reanesthetized. Neural tissues were perfusion-fixed with warmed saline followed by 4% paraformaldehyde in 0.1 mol/L phosphate buffer at pH 7.4. The left hemi-brains were removed and stored in fixative for at least 10 days before processing. (Selection of the left brain was arbitrarily decided before starting the study.) During perfusion, the ewe was euthanized with an intravenous overdose of pentobarbital-phenytoin solution (Euthasol; Delmarva Laboratories, Midlothian, VA, USA). Control fetal tissues were procured in a similar manner from nontreated sheep.
Histologic assessments were conducted on six discrete regions of the left fetal brain: (1) a rostral parasagittal section that included the parasagittal cortex, subjacent subcortical white matter, and periventricular white matter at the dorsolateral tip of the lateral ventricle; (2) a similar but more caudal parasagittal section; (3) the basal ganglia and thalmus; (4) a transverse section of the pons; (5) the hippocampus; and (6) the cerebellum. Coronal blocks (1 cm) of each region were embedded in paraffin from which 8-μm-thick slices were cut. All sections were coded such that the examiners (RJMcC and DJMcC) were unaware of the tissue source (i.e., anesthetized or untreated fetus). Apoptosis in adjacent slices was quantitated by both the TUNEL assay method and after hematoxylin and eosin (H&E) staining. For the former, tissue preparation followed the manufacturer's directions (Intergen, Purchase, NY, USA). Negative controls were created by omitting the TdT enzyme from the end-labeling mixture while positive controls came from sections of adult sheep lymph nodes. TUNEL-positive cells throughout each section (x 10 magnification) were identified by the presence of dark brown nuclear staining. All slides were counted by both examiners. After counting, each slide was photographed and the section area determined (NIH Image, National Institutes of Health, Bethesda, MD, USA). For the H&E-stained sections, apoptotic cells were identified by the presence of cytoplasmic eosinophilia, the appearance of condensed nuclei with dark chromatin inside, and decreased size. Under × 20 magnification, both total and apoptotic cells were counted in three randomly chosen nonoverlapping fields from each section. As with the TUNEL assessment, each slide was viewed and counted by both examiners.
Data Analysis
Arterial blood gas data are presented as group means (±standard deviations, s.d.). For the NIRS data, baseline values of fetal cerebral oxyHb, deoxyHb, and totalHb were calculated for each anesthetized animal by averaging the measurements taken during the preintubation period. Measurements taken during and after anesthesia were divided by these values and expressed as percent of baseline. Similar calculations were performed on the UBF data. With respect to the other cardiovascular parameters, maternal and fetal heart rates and mean arterial pressures (MAP) were averaged at 1-min intervals for each animal and presented as group means. For the histology data, values recorded by the two counters were averaged for each slide and then group means (±s.d.) for the six sections were calculated for the TUNEL and H&E assessments.
The cardiovascular responses to general anesthesia have been well characterized (Bachman et al, 1986). Therefore, to preserve statistical power, we did not test for changes in fetal or maternal hemodynamic parameters. Instead, the statistical analyses focused on the three primary end points, viz., fetal cerebral oxygenation, arterial blood gas status, and fetal neurologic injury. All analyses were conducted using SAS version 9.1 software (SAS Institute Inc., Cary, NC, USA). Changes in oxyHb, deoxyHb, and totalHb during and after anesthesia were assessed by calculating the duration above or below baseline as well as the average distance from baseline (distance was defined as the difference between each minute's measure minus baseline). For each animal, the average of these minute-by-minute distances was taken as a summary measure representing how far above or below baseline the measure was and for how long. Mean distances during and after anesthesia (along with 95% confidence intervals) were calculated and compared with baseline using t-tests. To assess for changes in maternal and fetal blood gas status, a mixed-model repeated-measures analysis of variance was employed. Measurement times were treated categorically to allow post hoc comparisons to baseline with Dunnett's test adjusting as needed for multiple comparisons. A follow-up analysis treating minutes numerically in a test for time effect was also conducted to confirm the initial results. Student's t-test (unpaired) was used to determine if general anesthesia produced any evidence of neurologic injury. In addition, power analysis calculations were conducted to determine that the group sizes were sufficient to detect the large effects reported in the rodent study (Jevtovic-Todorovic et al, 2003). For all end points, P-values of <0.05 were considered significant.
RESULTS
A total of 13 near-term sheep were instrumented for the anesthetic physiologic monitoring portion of the study. Within this group, three fetuses died during the surgical recovery period and one animal was excluded because the fetal arterial catheters became blocked. As a result, nine instrumented fetuses underwent the exposure paradigm. All of these animals tolerated the procedure well with no fetal or maternal intra- or postanesthesia complications. There was some variation in the time required to stabilize inspired isoflurane concentration at 1.5% after intubation of the lungs (11±7 mins) and in the time to extubation after termination of isoflurane exposure (19±5 mins). To account for these differences, changes in the continuously monitored parameters (i.e., maternal and fetal cardiovascular status and fetal brain NIRS signals) during intubation and extubation have been omitted. The 4-h period of general anesthesia started when inspired/expired isoflurane concentration was constant at 1.5% while the postanesthetic time points mentioned later in the text refer to the time after extubation.
Values for maternal and fetal arterial blood gas status during and after the 4 h of general anesthesia are presented in Table 1. The ewes were well-ventilated throughout the procedure with average end-tidal CO2 staying below the preset limit of 35 mm Hg. As expected, maternal SaO2 increased to 100% during anesthesia and then returned to the preintubation level of 97% after extubation; anesthesia produced a minor increase in pCO2 and nonsignificant changes in arterial pH. Mean standard base excess (SBE) changed little during or after isoflurane exposure although, as indicated by the large s.d. values, there was some interanimal variation. On the fetal side, there was an initial rise in SaO2 followed by a gradual decline such that by the 4-h time point, systemic oxygenation returned to baseline; fetal SaO2 remained at this level during the postanesthetic monitoring period. Modest alterations in fetal pH and pCO2 were observed during anesthesia. These changes resolved after extubation. Fetal SBE values were essentially constant during the study.
The maternal and fetal physiologic responses to 4 h of isoflurane exposure are presented in Figures 1 and 2, respectively. General anesthesia produced an initial period of maternal bradycardia (nadir of 87±15 bpm) and hypotension (nadir of 65.4±19 mm Hg) along with a decrease in UBF (nadir of 75%±27% of basal). These effects started to resolve after 30 mins and by the 120-min time point, all three cardiovascular parameters were within 10% of their baseline values. As noted in Table 1, the initial decline in UBF did not have an adverse effect on fetal systemic oxygenation. Extubation was accompanied by marked increases in maternal heart rate (peak of 142±31 bpm) and MAP (120.3±7.7 mm Hg); the former gradually resolved during the overnight monitoring period while MAP remained elevated. Uterine blood flow reflected these apposing responses by varying between 80% and 135% of baseline. Isoflurane produced fetal hypotension (nadir of 37.8±11.2 mm Hg) and bradycardia (nadir of 140±17 bpm) with the exception of an initial spike in heart rate (peak of 196±39 bpm). While initially similar to the maternal responses, fetal cardiovascular depression persisted for the duration of anesthetic exposure. Extubation produced a sharp rise in both fetal heart rate (peak of 199±40 bpm) and MAP (peak of 54.3±8.2 mm Hg). Again, similar to the ewe, the changes in heart rate gradually resolved while fetal MAP remained above baseline during the postanesthesia monitoring period.
Changes in fetal cerebral oxygenation (viz., time curves for deoxyHb, oxyHb, and totalHb) are presented in Figure 3. During the anesthesia exposure period, there was little change in deoxyHb level (<10%) but there was an increase in oxyHb (peak of 130% of baseline) and totalHb (peak of 122% of baseline). These latter two observations are indicative of an increase in the amount and supply (i.e., an increase in blood flow) of oxygen to the fetal brain. As a group, oxyHb was above baseline for 85.1% of the 4-h exposure while totalHb was above baseline for 86.5% of the time. After extubation, deoxyHb rose (peak of 120%) before returning to baseline. Both oxyHb and totalHb returned to baseline and then fluctuated between 90% and 120% for the next several hours. During this period, deoxyHb was above baseline for 70.2% of the time while oxyHb and totalHb were above baseline for 54.3% and 59.5%, respectively. Distance from baseline was used as a summary measure of these changes (Table 2). During anesthesia, both oxyHb (P = 0.03) and totalHb (P = 0.04) were significantly elevated above baseline whereas deoxyHb was not (P = 0.59). For the postanesthetic period, the distance from baseline was nonsignificant for all three parameters.
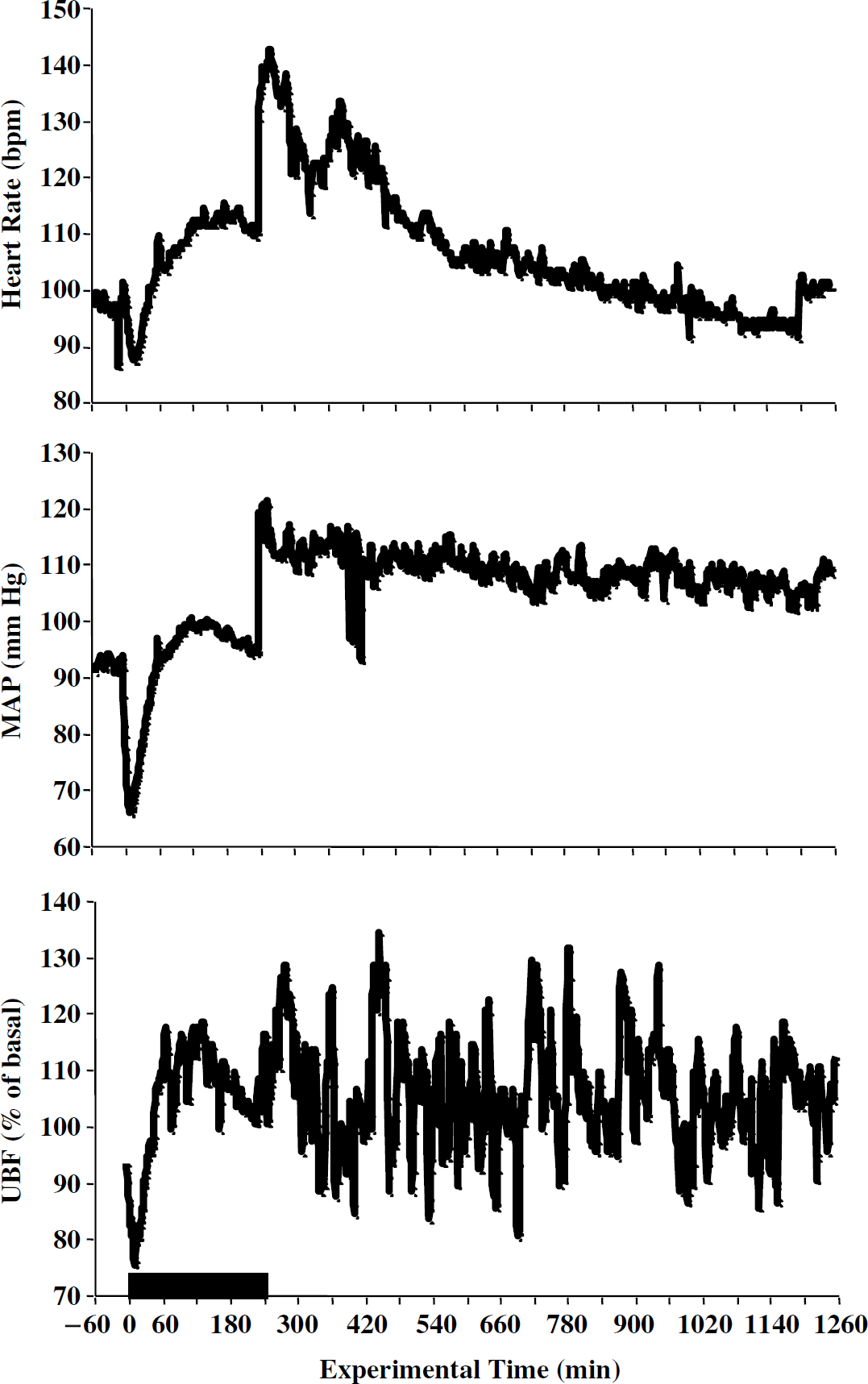
Maternal cardiovascular responses to general anesthesia. Maternal heart rate, mean arterial pressure (MAP), and uterine blood flow (UBF) time curves during and after 4 h of exposure to 1.5% isoflurane (demarcated by the box). The data are presented as group means with heart rate expressed as beats per minute (bpm) and UBF expressed as percent of baseline. The s.d. values did not exceed 25% of the means and have been omitted for clarity. As noted in the text, the observed fluctuations were expected and were not tested for significance.
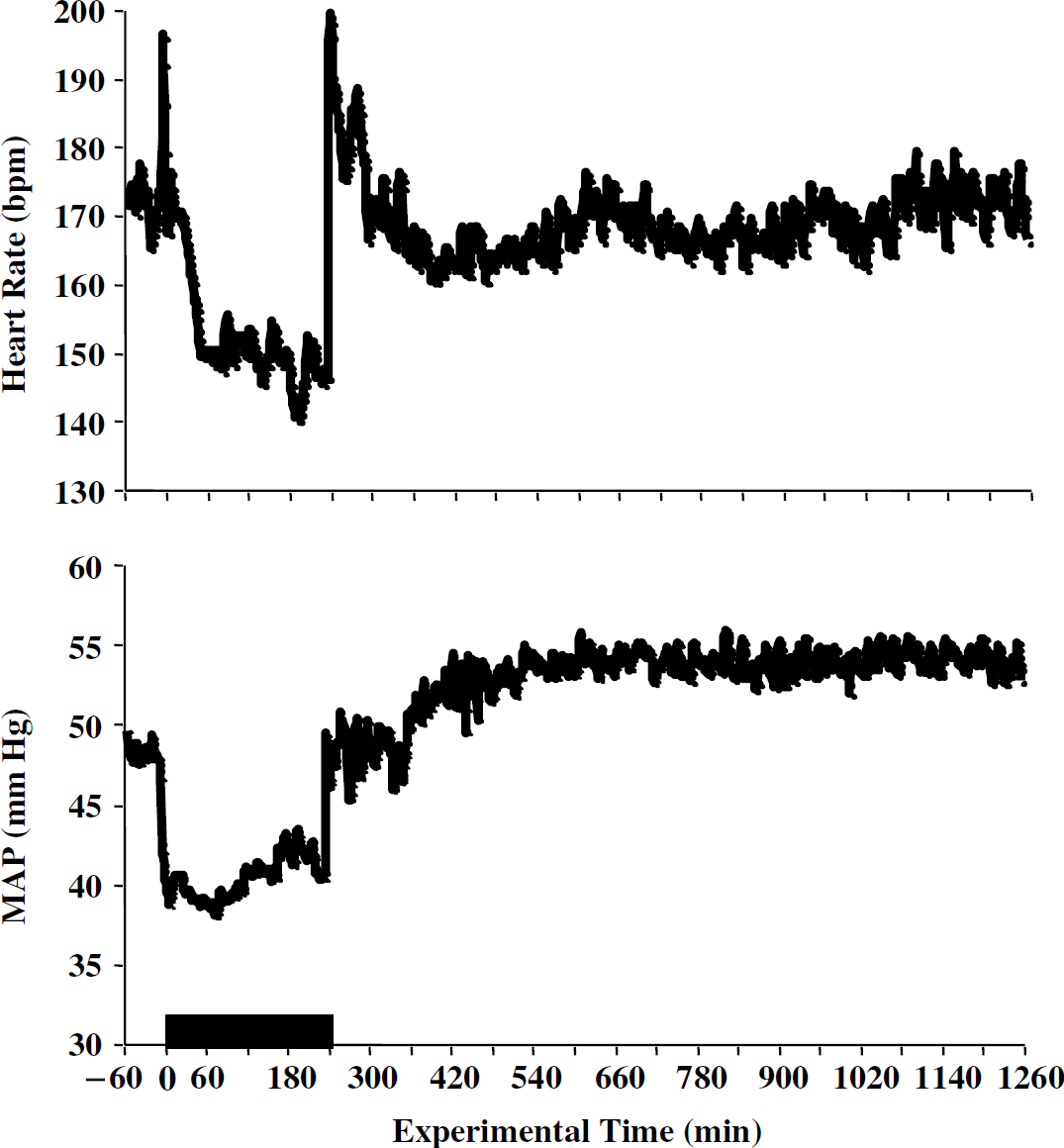
Fetal cardiovascular responses to maternal general anesthesia. Fetal heart rate and mean arterial pressure (MAP) time curves during and after 4 h of maternal exposure to 1.5% isoflurane (demarcated by the box). The data are presented as group means with heart rate expressed as beats per minute (bpm). The s.d. values did not exceed 25% of the means and have been omitted for clarity. The heart rate and MAP changes were not tested for significance.
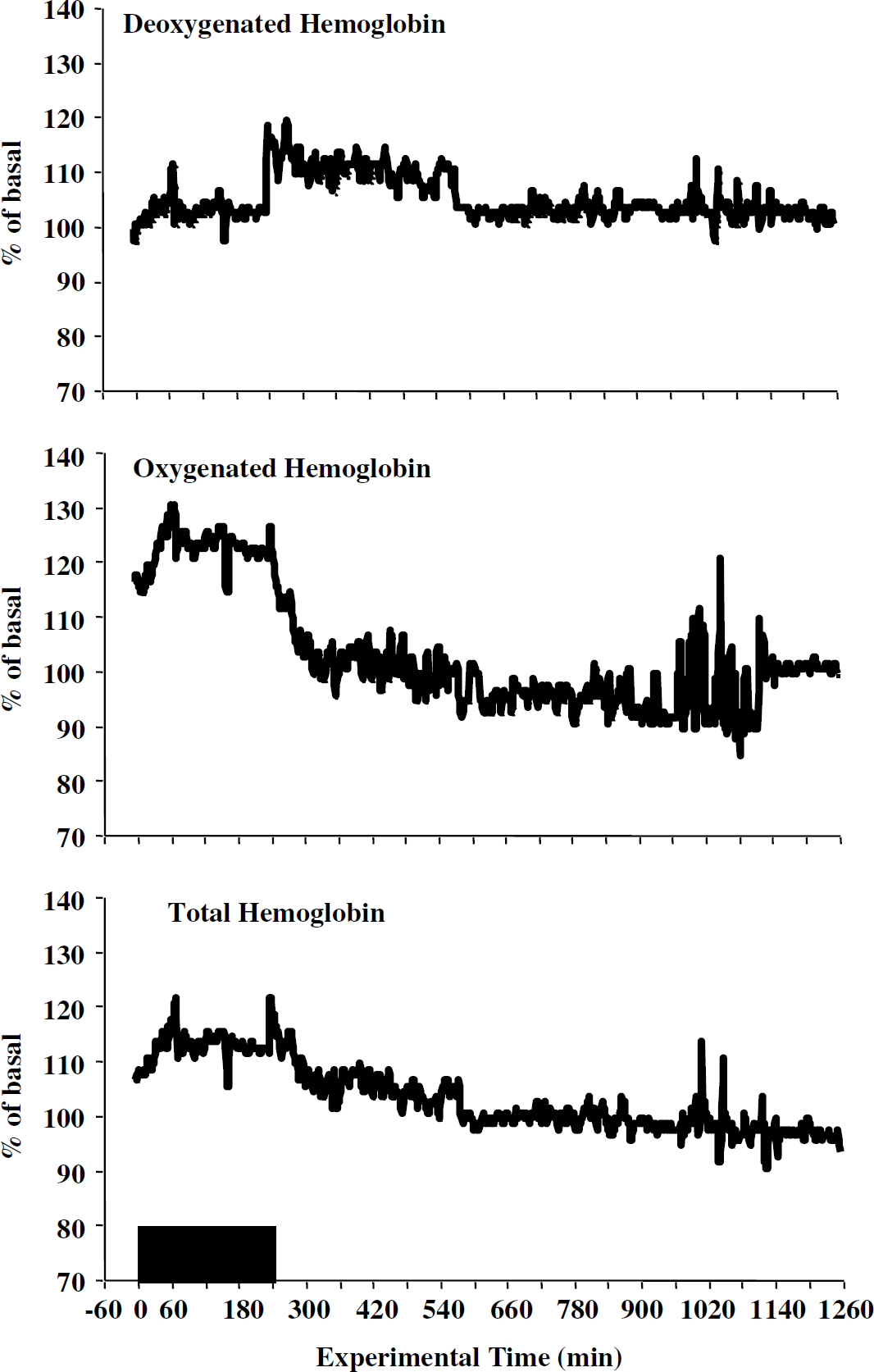
Fetal cerebral oxygenation. Time curves for fetal cerebral deoxygenated (deoxyHb), oxygenated (oxyHb), and total hemoglobin (totalHb) during and after 4 h of maternal exposure to 1.5% isoflurane (demarcated by the box). All three parameters are expressed as percent of baseline; the s.d. values have been omitted for clarity. During general anesthesia, both oxygenated and total hemoglobin were significantly elevated above baseline. Extubation produced a nonsignificant rise in deoxyHb while oxyHb and totalHb returned to baseline.
Arterial blood gas status
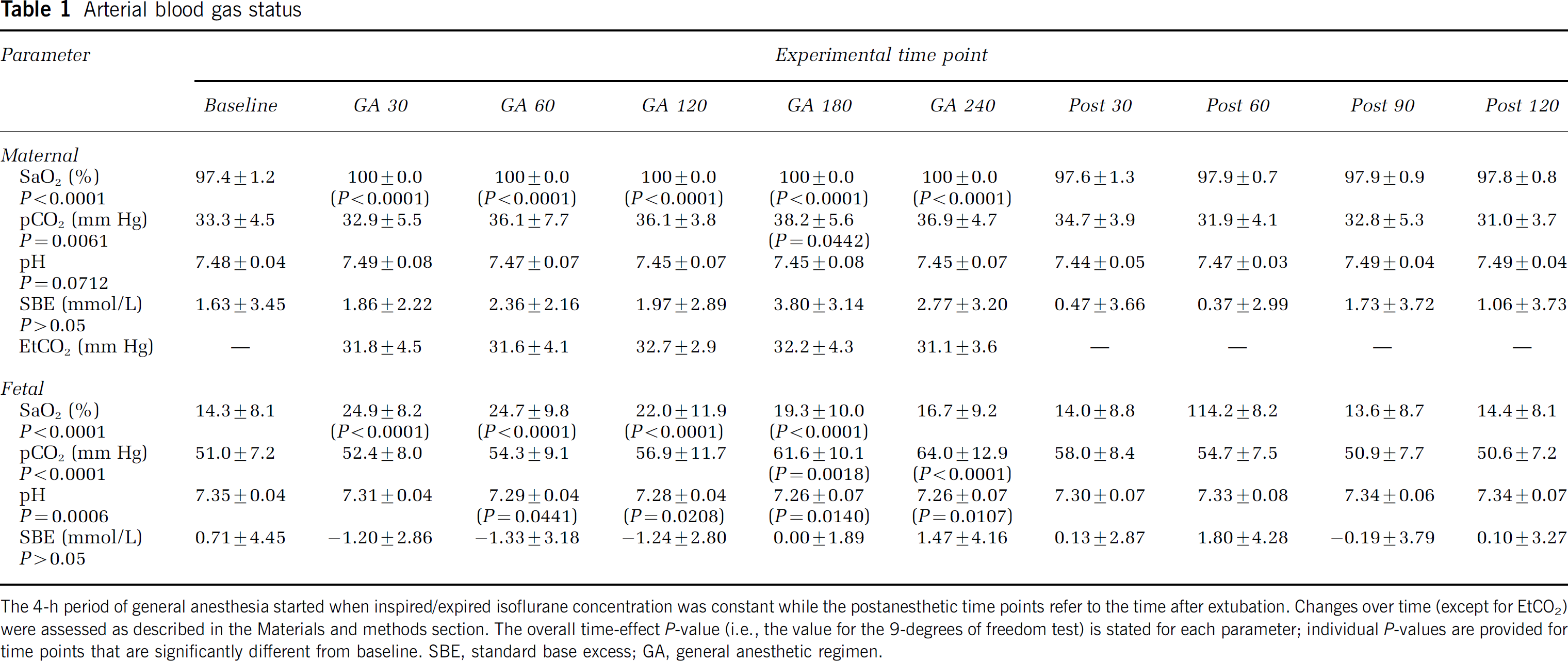
The 4-h period of general anesthesia started when inspired/expired isoflurane concentration was constant while the postanesthetic time points refer to the time after extubation. Changes over time (except for EtCO2) were assessed as described in the Materials and methods section. The overall time-effect P-value (i.e., the value for the 9-degrees of freedom test) is stated for each parameter; individual P-values are provided for time points that are significantly different from baseline. SBE, standard base excess; GA, general anesthetic regimen.
Summary measures of the NIRS data
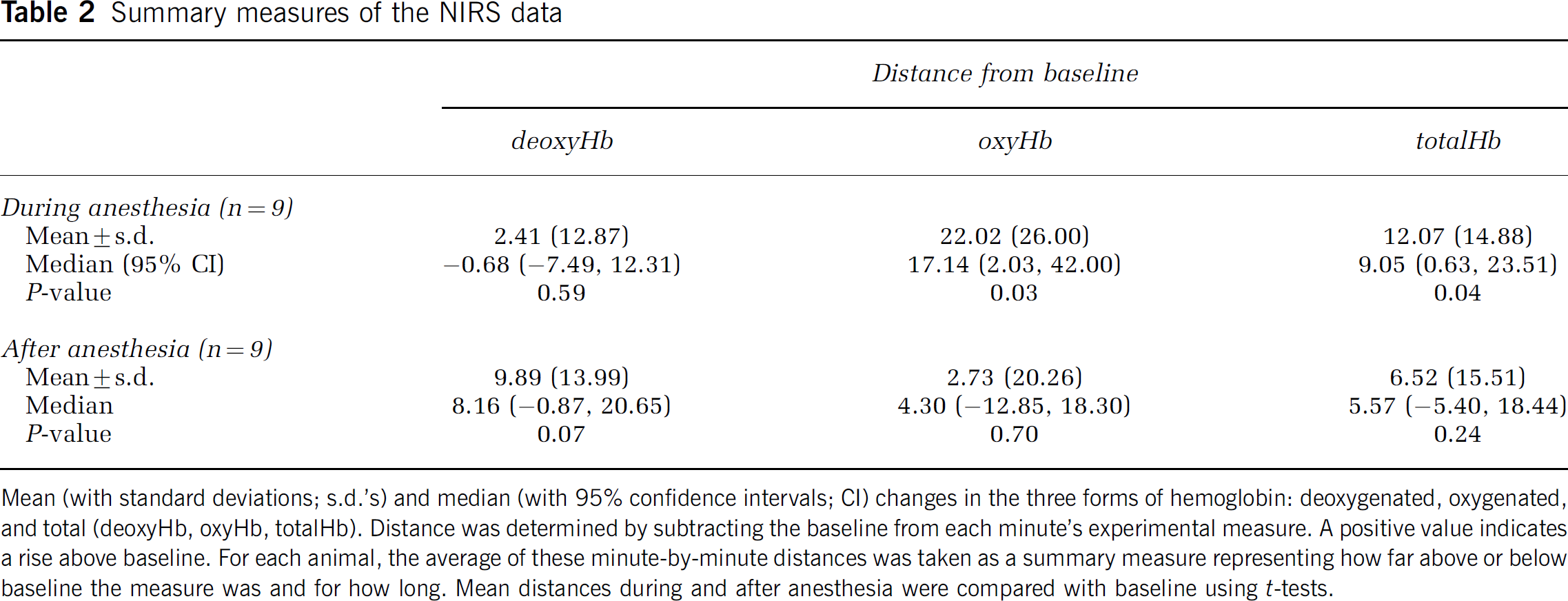
Mean (with standard deviations; s.d.'s) and median (with 95% confidence intervals; CI) changes in the three forms of hemoglobin: deoxygenated, oxygenated, and total (deoxyHb, oxyHb, totalHb). Distance was determined by subtracting the baseline from each minute's experimental measure. A positive value indicates a rise above baseline. For each animal, the average of these minute-by-minute distances was taken as a summary measure representing how far above or below baseline the measure was and for how long. Mean distances during and after anesthesia were compared with baseline using t-tests.
Nine near-term ewes were used for the histology component broken down into six experimental (i.e., anesthesia exposure) and three controls (i.e., no manipulation before fetal brain procurement); no adverse anesthesia effects were observed in this cohort. Results from the histologic assessments are presented in Table 3. There was no difference between the anesthesia and control groups in the number of TUNEL-positive cells or in the number of H&E-stained apoptotic cells in any of the brain regions that were scored. Likewise, there was no difference in the total number of neurons.
DISCUSSION
This study was prompted in part by the publication of a manuscript that reported passive exposure of neonatal rat pups to the inhalational agent isoflurane (with and without concomitant administration of injectable drugs) damages the developing brain (Jevtovic-Todorovic et al, 2003). Using a clinically relevant method of anesthetic delivery, our present findings do not support this assertion. Maternal general anesthesia produced an initial increase in fetal systemic oxygenation and a sustained increase in fetal cerebral oxygenation even in the face of the expected maternal and fetal cardiovascular depression (Bachman et al, 1986). Towards the end of the exposure period, there was some modest maternal and fetal respiratory acidosis, although this level of fetal hypercarbia has previously been reported to have minimal physiologic consequences (Walker et al, 1976). All changes resolved within 30 mins of extubation. There was no evidence of metabolic acidosis as both maternal and fetal SBE values stayed within normal levels (Ross and Gala, 2002). Additional monitoring during the postanesthesia period failed to identify changes in physiologic status that could be injurious to the fetal brain. Finally, through the histologic assessment of noninstrumented sheep, we found no evidence for a direct fetal neuro-toxic effect of our triple drug regimen of midazolam, sodium thiopental, and isoflurane.
One of our principal end points, fetal cerebral oxygenation, was assessed by monitoring changes in hemoglobin oxidation and concentration. To accomplish this, we used an NIRS device of our own design optimized for long-term in utero applications (Reynolds et al, 2003). The spectrophotometric method is based on the principle that light in the near-infrared range (700 to 1000 nm) can pass easily through the skin, bone, and other tissues but, within these frequencies, hemoglobin has specific absorption spectra dependent on its oxidation state (Jobsis, 1977). By using fiberoptic probes that transmit (and other fiberoptic bundles that receive) near-infrared light at specific wavelengths, changes in attenuation can be converted to changes in oxyHb, deoxyHb, and totalHb. The limitation of this technology is that it provides a global measure of tissue or organ hemoglobin status; changes in oxygenation within discrete brain regions cannot be derived with our machine. Nonetheless, the ability to record hemoglobin changes continuously within the fetal brain is a significant advance over other blood flow ‘type’ monitoring methods (such as microspheres) that only allow for a limited number of measurements at discrete time intervals.
Using our NIRS device, we determined that maternal general anesthesia produces sustained increases in oxyHb and totalHb within the near-term fetal brain. These effects most probably result from an isoflurane-mediated reduction in cerebral oxygen metabolism coupled with a rise in cerebral blood flow. The ability of isoflurane to augment cerebral oxygenation by these actions has been well documented in adult humans (Newman et al, 1986; Oshima et al, 2003) and a variety of experimental animal preparations (Scheller et al, 1990; Hoffman and Edelman, 2000; Sicard et al, 2003) but this is the first time such effects have been observed in the fetus. Cerebral oxygenation remained elevated for the duration of isoflurane exposure despite the prolonged fetal bradycardia and hypotension and even as fetal systemic oxygenation returned to baseline by the 3 h time point. Overall, the findings suggest a direct positive action of isoflurane on the fetal cerebro-vasculature.
Fetal brain histologic assessment
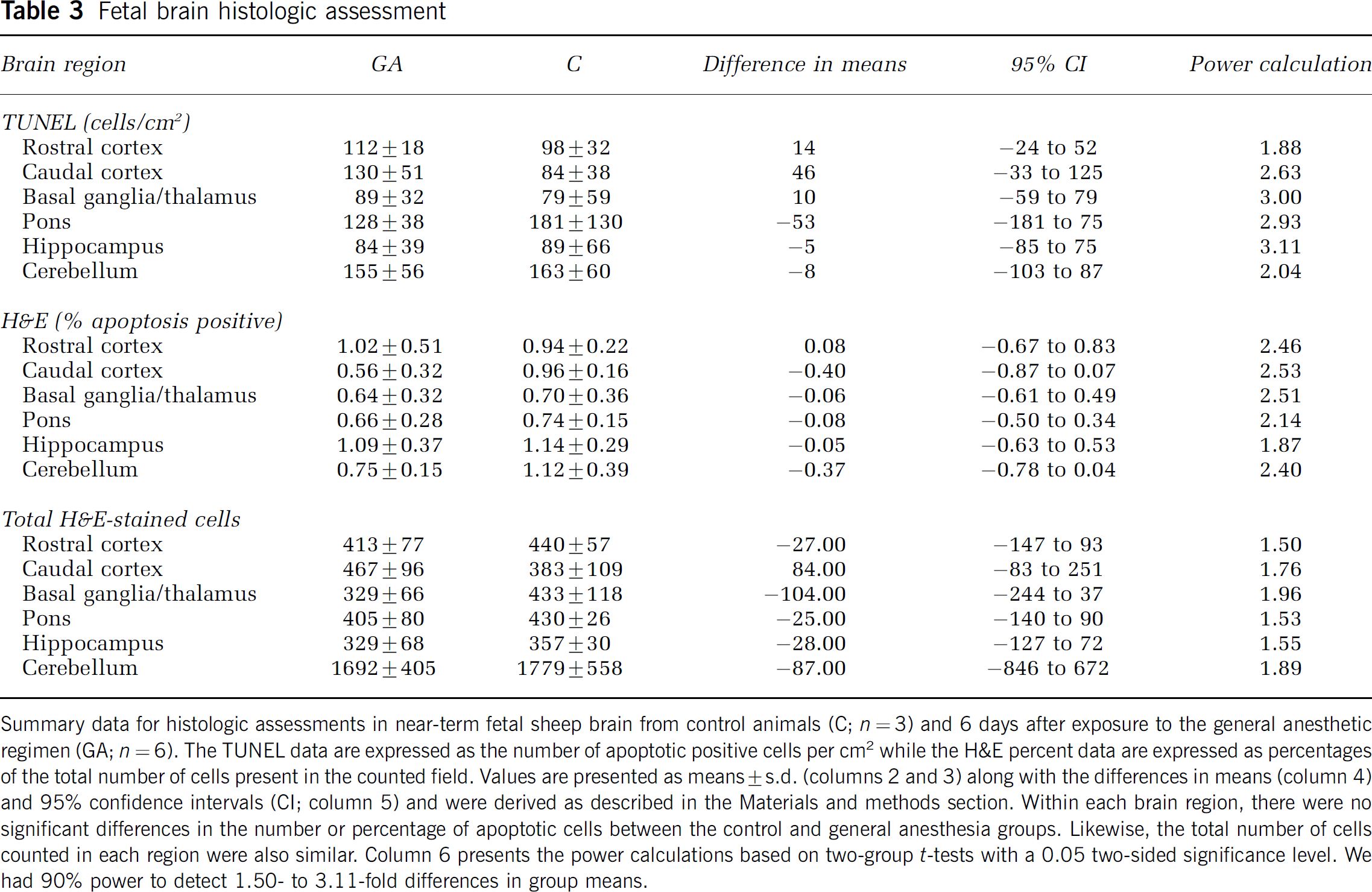
Summary data for histologic assessments in near-term fetal sheep brain from control animals (C; n = 3) and 6 days after exposure to the general anesthetic regimen (GA; n = 6). The TUNEL data are expressed as the number of apoptotic positive cells per cm2 while the H&E percent data are expressed as percentages of the total number of cells present in the counted field. Values are presented as means ± s.d. (columns 2 and 3) along with the differences in means (column 4) and 95% confidence intervals (CI; column 5) and were derived as described in the Materials and methods section. Within each brain region, there were no significant differences in the number or percentage of apoptotic cells between the control and general anesthesia groups. Likewise, the total number of cells counted in each region were also similar. Column 6 presents the power calculations based on two-group t-tests with a 0.05 two-sided significance level. We had 90% power to detect 1.50- to 3.11-fold differences in group means.
After the termination of anesthesia and extubation of the ewes' lungs, fetal cerebral oxygenation decreased as reflected by the decline in oxyHb, and totalHb, and the modest rise in deoxyHb. After the initial recovery phase (2 h), all 3 parameters remained within 10% to 15% of their preanesthetic levels for the remainder of the monitoring period. The observed fluctuations suggest that, at this stage in development, the fetal cerebral-vasculature is independently regulated as none of the observed changes temporally correlated with the bi-phasic changes in heart rate or the prolonged elevation in fetal MAP. Most importantly, at no time did deoxyHb increase or oxyHb decrease to a level that would indicate a significant reduction in fetal brain oxygen availability (Reynolds et al, 2003).
Consistent with the physiologic data, histologic assessment revealed no evidence of fetal neurologic damage after maternal general anesthesia. While our histology data are in contrast to Jevtovic-Todorovic et al's neonatal rat report, they are in agreement with the preliminary results of Loepke et al, who found no neuronal damage in ventilated neonatal swine after a 6-h exposure to midazolam, isoflurane, and nitrous oxide. From these findings one could infer that administration of clinically relevant doses of anesthesia via a clinically relevant mode of delivery are not injurious to the developing central nervous system (CNS) of high-order species (i.e., gyrencephalic as apposed to lissencephalic brains). However, the small sample size for the histologic component of the present study precludes us from making an unequivocal statement on the safety of general anesthesia. Brain tissues were prepared from near-term fetal sheep 6 days after drug exposure, a sufficient period of time to allow for the appearance of permanent injury (Kusaka et al, 2002) and yet still determine changes in apoptosis markers such as caspase activity (Cao et al, 2003). Nonetheless, it is possible we missed an earlier surge in apoptosis after general anesthesia, albeit one that did not result in a readily distinguishable change in neuronal population. Along these lines, it is worth noting that the developmental pattern of apoptosis in the ovine brain has not been well characterized (for a thorough review see Falkowski et al, 2002). As a result, additional serial assessments in a higher-order species after general anesthesia need to be conducted to delineate fully any early, and more importantly permanent, drug-induced neurologic changes.
The anesthetic regimen we used would be administered to pregnant patients, but it does differ from that used by Jevtovic-Todorovic et al. We included sodium thiopental for induction (a requirement for lung intubation), which has some neuroprotectant activity (Cheng et al, 1997). The period of exposure was reduced to 4 h as we felt this duration would encompass most, if not all, surgical procedures performed under general anesthesia during pregnancy. Isoflurane was delivered in 100% oxygen, which improved fetal systemic oxygenation consistent with human data (Ngan Kee et al, 2002) yet was well below levels that might be injurious to the developing brain (Felderhoff-Mueser et al, 2004). Finally, we omitted the use of a second volatile anesthetic agent noting the infrequent administration of nitrous oxide to parturients in North America (Littleford, 2004) and because Jevtovic-Todorovic et al had reported significant apoptosis after isoflurane exposure alone. In addition, we had concerns (at least hypothetical) that nitrous oxide-induced diffusion hypoxia could impact our observations (Rosen, 2002). Of these differences, it may well turn out that the duration of anesthetic exposure is the most important factor especially when contrasted to the rate of brain development. Neural activity is a key component of CNS maturation (Corner et al, 2002) and at some point continued depression of this activity would be expected to have a material impact on brain development; studies describing the time-dependent developmental neurotoxic potential of other anesthetic agents such as ketamine would support this contention (Hayashi et al, 2002). By extension, it is reasonable to conclude that additional research must be conducted before attempting to delineate human fetal or neonatal anesthesia exposure limits.
Our fetal sheep study is not without its limitations. The anesthetic exposure occurred at a fixed period of gestation so we cannot discount the possibility that developmental changes in the cerebro-vasculature (or other organ systems) may produce periods of vulnerability during which the fetal CNS could exhibit a greater sensitivity to maternal general anesthesia. However, it is worth noting that Jevtovic-Todorovic et al's mechanism for anesthetic toxicity focussed on N-methyl-d-aspartate (NMDA) receptor blockade; their rat study was conducted at the time when the NMDA receptor population peaks in this species (Tremblay et al, 1988). A surge in NMDA receptor population is an integral part of normal CNS development but it has long been recognized that this event confers an enhanced susceptibility to neurologic insult (McDonald and Johnston, 1990). The NMDA receptor populations in the sheep CNS peak during the late prenatal period (Anderson et al, 1999) so any anesthetic/NMDA-mediated injury should have been observed at the gestational time point we studied, notwithstanding the histologic limitations noted above. The second limitation is that we have no long-term outcome data. While the physiologic and histologic findings are suggestive of a positive outcome, we have previously detected behavioral differences in lambs after prenatal ethanol exposure in the absence of neurologic injury (Spear-Smith et al, 2000). As already noted, useful information would be obtained from experimental animal outcome studies along with behavioral assessments of humans after prenatal and/or early neonatal anesthetic exposure. The final limitation is that our study was conducted on healthy fetuses. In humans, anesthetics are administered to gravid patients in response to some type of maternal or fetal pathology. While it is tempting to speculate that the anesthetic-induced increase in cerebral (and systemic) oxygenation would be beneficial to the distressed fetus, this hypothesis has yet to be tested. Likewise, it is unknown how or if the fetal systemic and central actions of general anesthesia may be modified by surgical manipulation.
In conclusion, a 4-h episode of anesthesia during the third trimester equivalent does not appear to produce conditions during or after exposure that would be injurious to the fetus. Instead, maternal general anesthesia causes a prolonged increase in fetal cerebral oxygenation along with a transient increase in fetal systemic oxygenation. The absence of a detrimental effect was supported by the negative histology findings. While much additional work is needed to explain the dichotomy between these results and the study by Jevtovic-Todorovic et al and ultimately to define exposure limits, the present study does corroborate the presumed safety of moderate inhalational anesthetic use during pregnancy.
Footnotes
Acknowledgements
The authors thank Tom Cummings, Cheryl Miller (Duke University Medical Center), and Marjorie Grafe (Oregon Health Sciences University) for their assistance with the histology component of this study and David S Warner (Duke University Medical Center) for his editorial suggestions.
