Abstract
Despite caffeine's wide consumption and well-documented psychoactive effects, little is known regarding the effects of caffeine on neurovascular coupling. In the present study, we evaluated the effects of caffeine, an adenosine receptor antagonist, on intracerebral arterioles in vitro and subsequently, on the pial circulation in vivo during cortical activation induced by contralateral sciatic nerve stimulation (SNS). In our in vitro studies, we utilized isolated intracerebral arterioles to determine the effects of caffeine (10 or 50 μmol/L) on adenosine-induced vasodilatation. At the lower concentration, caffeine was without effect, but at the higher concentration, caffeine produced significant attenuation. In our in vivo studies, we determined the cerebrospinal fluid (CSF) caffeine concentrations at 15, 30, and 60 mins after intravenous administration of 5, 10 and 40 mg/kg. At the latter two concentrations, CSF levels exceeded 10 μmol/L. We then evaluated the pial arteriolar response during cortical activation caused by contralateral SNS after administering caffeine intravenously (0, 5, 10, 20 30, and 40 mg/kg). The pial circulation was observed through a closed cranial window in chloralose-anesthetized Sprague—Dawley rats. The contralateral sciatic nerve was isolated, positioned on silver electrodes and stimulated for 20 secs (0.20 V, 0.5 ms, and 5 Hz). Arteriolar diameter was quantified using an automated video dimension analyzer. Contralateral SNS resulted in a 23.8%±3.9% increase in pial arteriolar diameter in the hindlimb sensory cortex under control conditions. Intravenous administration of caffeine at the lowest dose studied (5 mg/kg) had no effect on either resting arteriolar diameter or SNS-induced vasodilatation. However, at higher doses (10, 20, 30, and 40 mg/kg, intravenously), caffeine significantly (P<0.05; n=6) attenuated both resting diameter and cerebral blood flow (CBF) responses to somatosensory stimulation. Intravenous administration of theophylline (10, 20, and 40 mg/kg), another adenosine receptor antagonist, also significantly reduced SNS-induced vasodilatation in a dose-dependent manner. Hypercarbic vasodilatation was unaffected by either caffeine or theophylline. The results of the present study show that caffeine significantly reduces cerebrovascular responses to both adenosine and to somatosensory stimulation and supports a role of adenosine in the regulation of CBF during functional neuronal activity.
Introduction
Metabolic regulation of cerebral blood flow (CBF), first proposed by Roy and Sherrington (1890) is a widely accepted hypothesis. Indeed, several studies have since showed a clear coupling between neuronal activation and increases in cerebral energy metabolism and CBF. However, the metabolic factor(s) involved in CBF regulation during somatosensory stimulation remain unclear. Among the list of proposed metabolic regulators is adenosine, a purine nucleoside and a product of ATP metabolism (Dirnagl et al, 1994; Ko et al, 1990; Li and Iadecola, 1994; Meno et al, 2001; Winn et al, 1981). Adenosine has many of the prerequisites necessary to participate in the coupling between local CBF and brain metabolism. For example, adenosine is a potent dilator of cerebral arterioles (Ibayashi et al, 1991; Ngai et al, 2001; Ngai and Winn, 1993). Moreover, brain tissue is capable of producing significant concentrations of adenosine and in a time course that parallels metabolically induced increases in CBF (Winn et al, 1980). Adenosine is also a potent inhibitor of neuronal transmission (Fredholm and Dunwiddie, 1988; Schubert, 1988). Thus, in addition to increasing blood flow, adenosine is capable of decreasing metabolic demand in situations where there is a mismatch between brain activity and CBF.
A number of studies (Dirnagl et al, 1994; Ko et al, 1990; Li and Iadecola, 1994; Meno et al, 2001) have showed that CBF increases during neuronal activation are significantly attenuated by adenosine receptor antagonists such as theophylline and ZM241385 (an A2a receptor blocker). Surprisingly, the effect of caffeine, a widely consumed psychoactive compound (Nehlig, 1999; O'Brien and Kinnaird, 2003) and a nonselective adenosine receptor antagonist, has received relatively little attention about its effects on CBF changes during somatosensory stimulation. With the exception of Gotoh et al (2001), most previous studies have focused on the effects of caffeine on resting CBF (Laurienti et al, 2002; Laurienti et al, 2003; Mathew and Wilson, 1985; Mulderink et al, 2002). The experiments by Gotoh et al (2001) were performed on unanesthetized rats and used autoradiographic methods to quantify changes in cerebral energy metabolism and CBF during whisker stimulation. These investigators showed that caffeine attenuated increases in CBF during neuronal activation in subcortical regions but was without effect in the cortical barrel fields. In contrast to the investigations by Gotoh et al (2001), earlier studies, as we noted above, have showed that theophylline and ZM241385 did attenuate the increases in CBF during neuronal activation (Dirnagl et al, 1994; Ko et al, 1990; Meno et al, 2001).
Given the high daily consumption of caffeine and the relative lack of data regarding its effects on neurovascular coupling, the purpose of the present study was to evaluate the effects of caffeine on the cerebrovascular response to somatosensory stimulation. In addition, we sought to compare the effects of caffeine with that of theophylline. We used both in vitro and in vivo techniques to evaluate the effects of caffeine on the cerebral circulation. First, using isolated perfused penetrating intracerebral arterioles in vitro (Ngai and Winn, 1993), we evaluated the ability of caffeine to affect adenosine-induced vasodilatation. In our in vivo studies, we quantified cerebrospinal fluid (CSF) concentrations of caffeine after intravenous administration. Then, we used a closed cranial window preparation (Morii et al, 1987; Ngai et al, 1988) to visualize the cerebral circulation and to document pial arteriolar changes during contralateral sciatic nerve stimulation after administration of caffeine intravenously (5, 10, 20, 30, and 40 mg/kg). The lower dose range (5 and 10 mg/kg) of caffeine represents typical human consumption of coffee (2 to 4 cups). The higher dose range of caffeine was evaluated for comparisons with the experimental findings (Gotoh et al, 2001; Horinaka et al, 1997) on the effects of caffeine in the cerebral circulation. Lastly, we evaluated the cerebrovascular effects of caffeine as contrasted to those of theophylline during cortical activation.
Methods
Isolation and Cannulation of Cerebral Microvessels
This study received prior approval from Mt Sinai School of Medicine and the University of Washington Animal Welfare Committee. Detail methods for isolation and cannulation of intracerebral arterioles have been described previously (Dacey and Duling, 1982; Ngai et al, 2001). Briefly, male Sprague—Dawley rats (350 g) were anesthetized with pentobarbital (50 mg/kg intraperitoneally) and decapitated. The brain was rapidly removed from the skull, immersed in cold (4°C) physiological salt saline (PSS) containing (in mmol/L) 144 NaCl, 3 KCl, 2.5 CaCl2, 1.5 MgSO4, 5.0 glucose, 2.0 pyruvate, 0.02 EDTA, 2.0–(N-morpholino)propanesulfonic acid (MOPS), and 1.21 NaH2PO4, plus 1% dialyzed bovine serum albumin. A thick (2 mm) section of the cerebral cortex containing the first portion of the middle cerebral artery was dissected from the brain. The pia matter and its attached penetrating intracerebral arterioles were then separated from the parenchyma, and an unbranched distal segment of a vessel, ∼0.5 to 1 mm in length, was severed from the pia. The vessel segment was then transferred to a temperature-controlled tissue chamber (3.0 mL volume), mounted on the stage of an inverted microscope (Nikon Inc.).
The isolated vessel was cannulated using a system of concentric glass pipettes filled with buffered saline-albumin. The cannulated vessel was observed on a video monitor connected to a video camera, and lumen diameter was measured with a video dimension analyzer. The bath solution was then changed to one without albumin, and the bath temperature was raised to 37°C. The vessel was perfused at 4 μL/min; this rate of intraluminal perfusion was chosen to avoid flow-mediated effects on vessel diameter (Ngai and Winn, 1995). After an equilibration period of ∼40 mins, viable arterioles developed vasomotion and spontaneously contracted; control diameter was measured after the development of steady tone. Reactivity of the vessels was assessed by changing the extraluminal pH from 7.3 to 6.8 and from 7.3 to 7.6. Vessels with weak pH response (<15% dilation or constriction) were excluded from data collection. All drugs for this study were applied extraluminally at a rate of 1 mL per minute.
Experimental Protocol for In Vitro Studies on Isolated Intracerebral Arterioles
These experiments were designed to determine the effects of caffeine on adenosine dose-dependent dilations of intraparenchymal arterioles, in vitro. Adenosine, in increasing concentrations from 10−3 to 10−9 mol/L, was perfused extraluminally. Each concentration of adenosine was applied for 5 mins before proceeding to the next higher concentration. After establishing a control dose—response to adenosine, the arterioles were pretreated extraluminally with caffeine (10 or 50 μmol/L) for 5 mins. A dose—response to adenosine was then repeated in the presence of caffeine (either 10 or 50 μmol/L). After the second dose—response in which both adenosine and caffeine were perfused, washout of the bath chamber with control buffer was performed for 10 mins. The arteriolar responses to acidic and basic pH were then assessed.
Determination of Cerebrospinal Fluid Concentrations of Caffeine
Animal preparation
Male Sprague—Dawley rats weighing 350 to 400 g were anesthetized with pentobarbital (50 mg/kg intraperitoneally) and their right femoral vein was cannulated for administration of caffeine (5, 10, or 40 mg/kg). Animals were maintained on a warming pad to stabilze body temperature. At 15, 30, or 60 mins, CSF samples (100 to 150 μL) were drawn through suboccipital puncture of the cisterna magna. A minimum of two animals were studied at each dose and at each time period. Control CSF samples were obtained after saline injection.
In a separate set of experiments, we also analyzed CSF concentrations of caffeine in animals (n=2) undergoing the experimental protocol for cranial window/sciatic nerve stimulation (except craniotomy) to determine if there is any difference in CSF caffeine levels between the two anesthesia methods (pentobarbital versus halothane, α-chloralose and urethane, see below). Cerebrospinal fluid was obtained at 60 mins after 40 mg/kg caffeine injection and compared with a similar time period and dose in animals treated with pentobarbital.
High-performance liquid chromatographic analysis
We used the method and protocol of Mei et al (1996) to measure caffeine by high-performance liquid chromatography (HPLC). In all, 50 μL of CSF samples was added to 150 μL of acetonitrile and centrifuged at 3500 g for 15 mins to precipitate proteins (Walker, 1989). Of the sample supernate or caffeine standard 5 μL was injected onto an 1100 series liquid chromatograph (Agilent, Palo Alto, CA, USA) using an autosampler. A microbore column 1.0 × 150 mm2 3.5 μm (Zorbax Eclipse XDB-C18, Agilent, Palo Alto, CA, USA) with a linear gradient of 10 mmol/L KH2PO4 (A) and 10 mmol/L KH2PO4 in 50% (v/v) methoanol/water (B) (both adjusted to pH 3.5 with phosphoric acid) was used. The gradient started at 4% to 10% B in 10 mins, 10% to 70% B in 5 mins, 70% to 100% B in 12 mins, held at 100% B for 2 mins, 100% to 4% B in 1 min, and a 20-min post run at 4% B. The flow rate was 50 μL/min. A photo diode array detector was used to record chromatograms at 270 nm. Peak identity was confirmed by comparison of the retention time of peaks from samples and their absorption spectra with those of caffeine standard. Caffeine standards were dissolved in water at 50 × 10−5, 50 × 10−6, 10 × 10−6, and 50 × 10−7 mol/L. Linear calibration curves were obtained with each batch of samples by plotting the peak areas as a function of their known concentration. Calculations of sample caffeine concentrations were performed using the software ChemStation for LC 3D (Agilent, Palo Alto, CA). For intravenous injection, caffeine powder was dissolved in physiological saline at 1 g/100 mL.
In Vivo Cranial Window and Sciatic Nerve Stimulation Studies
A detailed description of the closed cranial window preparation and the sciatic nerve stimulation (SNS) paradigm have been published previously (Morii et al, 1986; Ngai et al, 1988). Briefly, male Sprague—Dawley rats weighing 350 to 400 g were initially anesthetized with 1.5% to 2.0% halothane. The right femoral artery was cannulated for monitoring blood pressure and blood gas sampling. The right femoral vein was also cannulated for intravenous drug administration. The rats were tracheotomized, immobilized with tubocurarine chloride (1 mg/kg, intravenously), and mechanically ventilated. The ventilated gases were adjusted to maintain physiologic blood gas tensions and rectal temperature was maintained at 37°C. Halothane anesthesia was discontinued and replaced with α-chloralose and urethane (50 and 500 mg/kg, respectively, intraperitoneally). The animals were positioned onto a stereotaxic frame and a closed cranial window created over the sensory cortex allowing visualization of the pial vasculature over the right somatosensory cortex. The pial arterioles were observed by means of a microscope and changes in diameter evaluated (see Morii et al (1986) and Ngai et al (1988) for details).
The contralateral sciatic nerve was exposed, cut proximal to the bifurcation into tibial and peroneal nerves and placed on stimulating electrodes immersed in a pool of warm mineral oil. An automatic video dimension analyzer (IPM Inc., model 303, San Diego, CA, USA), which tracks the width of red blood cell columns, allowed continuous on-line monitoring of pial vessel diameter. The contralateral sciatic nerve was stimulated for 20 secs (0.20 V, 5 Hz, 0.5 ms rectangular pulses) and vascular response to SNS was calculated as percent increase over resting diameter. Our previous studies reveal that these stimulation parameters optimize the vascular response without affecting systemic blood pressure (Ngai et al, 1988).
Caffeine and theophylline were obtained from Research Biochemicals International (Natick, MA, USA). Caffeine or theophylline (40 mmol/L) were directly dissolved in water and subsequently diluted in artificial CSF, phosphate-buffered saline or physiologic saline to reach desired final concentrations. Artificial CSF had the following composition: Na+ 156.5 mEq/L, K+ 2.95 mEq/L, Ca2+ 2.50 mEq/L, Mg2+ 1.33 mEq/L, HCO3− 24.6 mEq/L, dextrose 66.5 mEq/L, and urea 40.2 mEq/L.
Experimental Protocol for Cranial Window/Sciatic Nerve Stimulation Studies
The contralateral sciatic nerve was stimulated for 20 secs and changes in pial arteriolar diameter were observed and recorded. A minimum of three consistent responses to SNS under control conditions were obtained before administering the test compounds. Only one compound was tested per animal. The effects of caffeine or theophylline on the CBF response to SNS were evaluated at 15 mins postintravenous administration of the test compounds. Mean arterial blood pressure (MABP) and arterial blood gases were monitored and remained stable throughout the entire duration of the experiment. In addition, reactivity of pial arterioles to CO2 inhalation was assessed before and after exposure to the test compounds, as previously described (Morii et al, 1986). Briefly, the respirator was switched to a special gas mixture containing 6% CO2, 10% O2 and 84% N2 for 2 mins. Changes in pial arteriolar diameter and arterial PCO2 were determined for calculation of CO2 reactivity. CO2 reactivity is expressed as percent increase in diameter per mm Hg increase in PaCO2. The pial arteriolar response to CO2 inhalation was not affected by any of the compounds tested in this study.
Statistical Analysis
All values are expressed as means±s.d. Analysis of variance and paired t-tests were used to make statistical comparisons. When multiple simultaneous comparisons were made, a Bonferonni correction was applied. A P-value of less than 0.05 was used to indicate statistically significant differences.
Results
In Vitro Studies
Effect of caffeine on adenosine-induced vasodilatation of cerebral arterioles in vitro
In our initial experiments, we evaluated the effects of caffeine on vasodilatation of intracerebral arterioles to extraluminally applied adenosine. Intracerebral arterioles, observed in this study, spontaneously constricted to 68.4%±6.6% of their maximally dilated (passive) diameter when bath temperature was increased to 37°C (Table 1). Average arteriolar diameter was 66.1±9.7 μm (range: 51 to 77 μm) after developing spontaneous tone. A well-documented characteristic of cerebral arterioles is their sensitivity to extraluminal pH changes. In this study, intracerebral arterioles constricted 15.7%±1.2% when the bath pH was increased from 7.3 to 7.6. Predictably, we observed significant vasodilatation (21.6%±9.0%) when the bath pH was lowered to 6.8 from 7.3 (Table 1).
Diameters of intracerebral arterioles in vitro
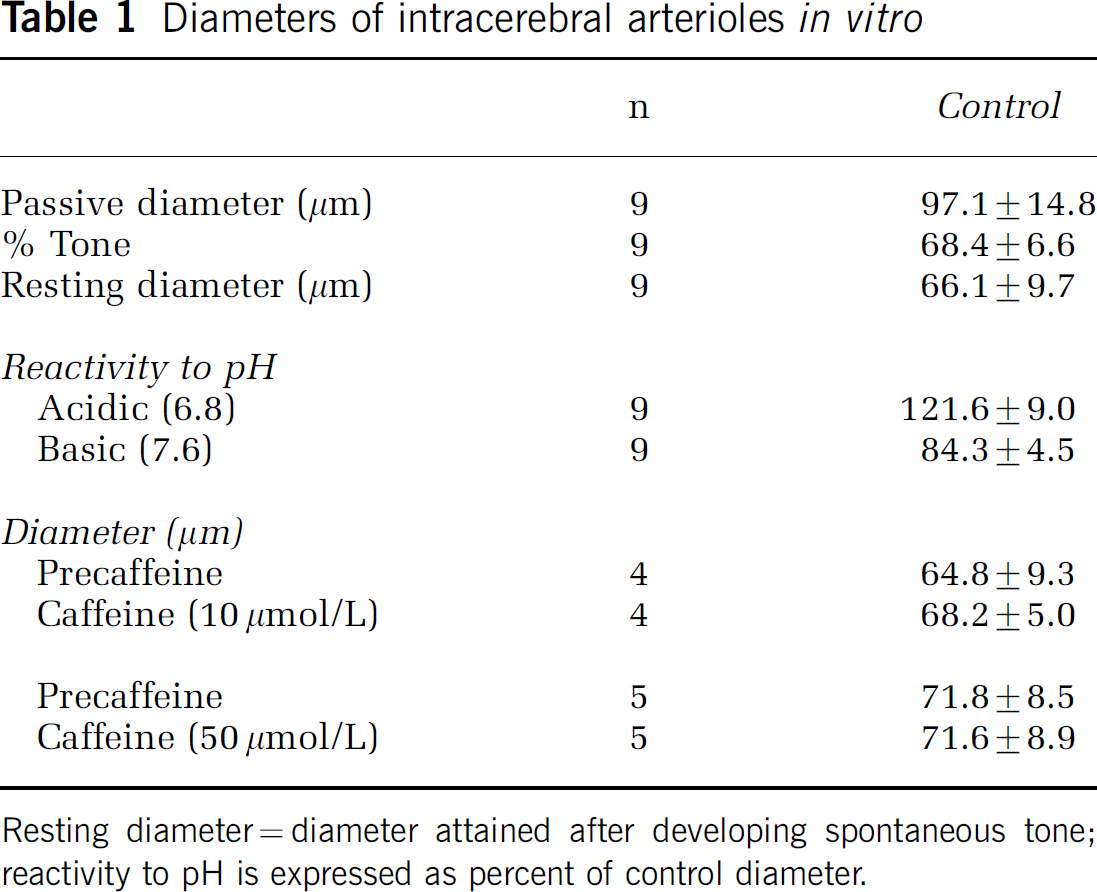
Resting diameter=diameter attained after developing spontaneous tone; reactivity to pH is expressed as percent of control diameter.
Extraluminal application of adenosine caused dose-dependent dilations similar to previously reported values. A second dose response to adenosine was performed in the presence of caffeine, either 10 or 50 μmol/L. Pretreatment with caffeine alone (10 or 50 μmol/L), had no effect on resting arteriolar diameter (Table 1). In the presence of the lower dose of caffeine (10 μmol/L), dose-dependent vasodilatation to extraluminally applied adenosine was unaffected (data not shown). However, pretreatment with the higher concentration of caffeine (50 μmol/L) caused significant attenuation of adenosine-induced vasodilatation (Figure 1). The −logEC50 value for adenosine alone was 5.5±0.2 and 4.9±0.5 for adenosine plus caffeine (50 μmol/L).
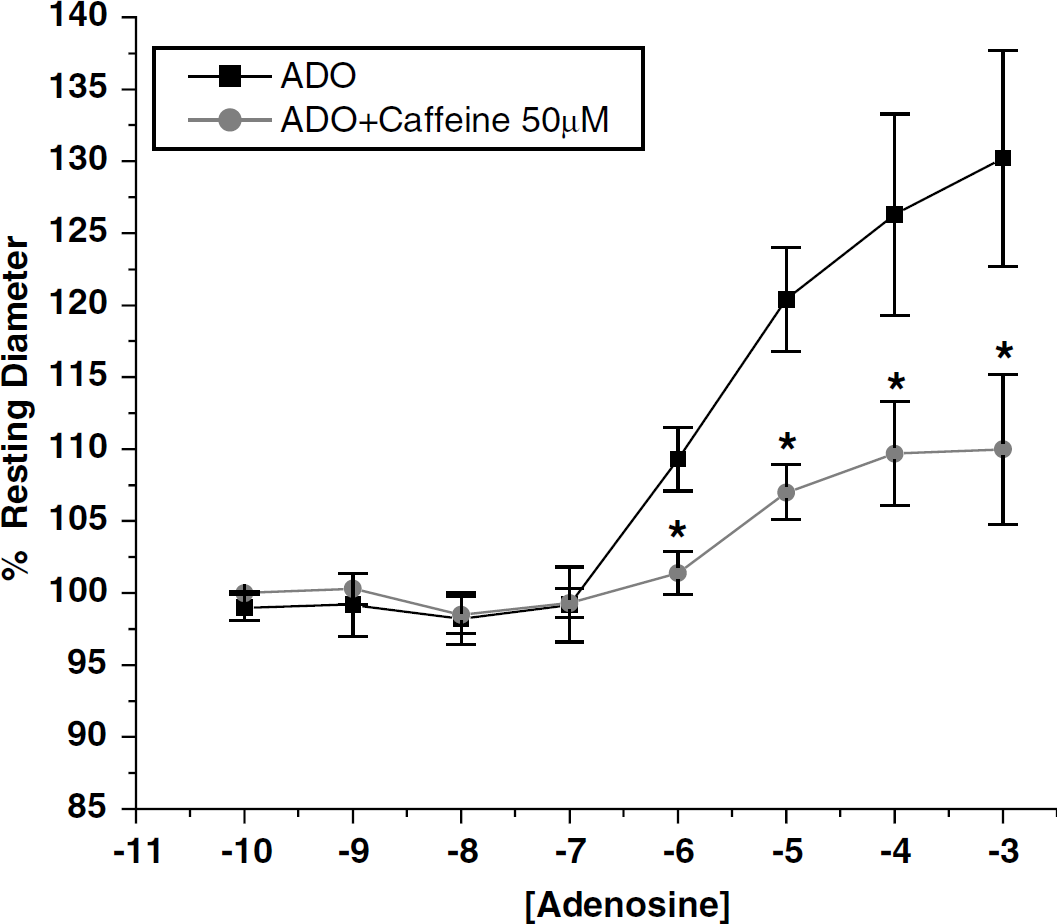
Effect of caffeine on dose-dependent adenosine-induced vasodilatation of intraparenchymal arterioles, in vitro. Dilation responses are expressed as percentage of baseline diameter. Data are expressed as means±s.d. (n=5). *P<0.05 versus corresponding adenosine alone values.
In Vivo Studies
Determination of cerebrospinal fluid caffeine concentrations
As illustrated in Figure 2, CSF caffeine levels were measured at 15, 30, and 60 mins after intravenous injection of 5, 10, and 40 mg/kg. Only the higher two doses (i.e., 10 and 40 mg/kg) reached CSF concentrations greater than 10 μmol/L. At the higher doses, CSF concentrations appeared to peak after 30 mins. There were no significant differences in the CSF concentrations between the animals anesthetized with pentobarbital and those anesthetized with halothane, α-chloralose and urethane and prepared for cranial window studies (data not shown).
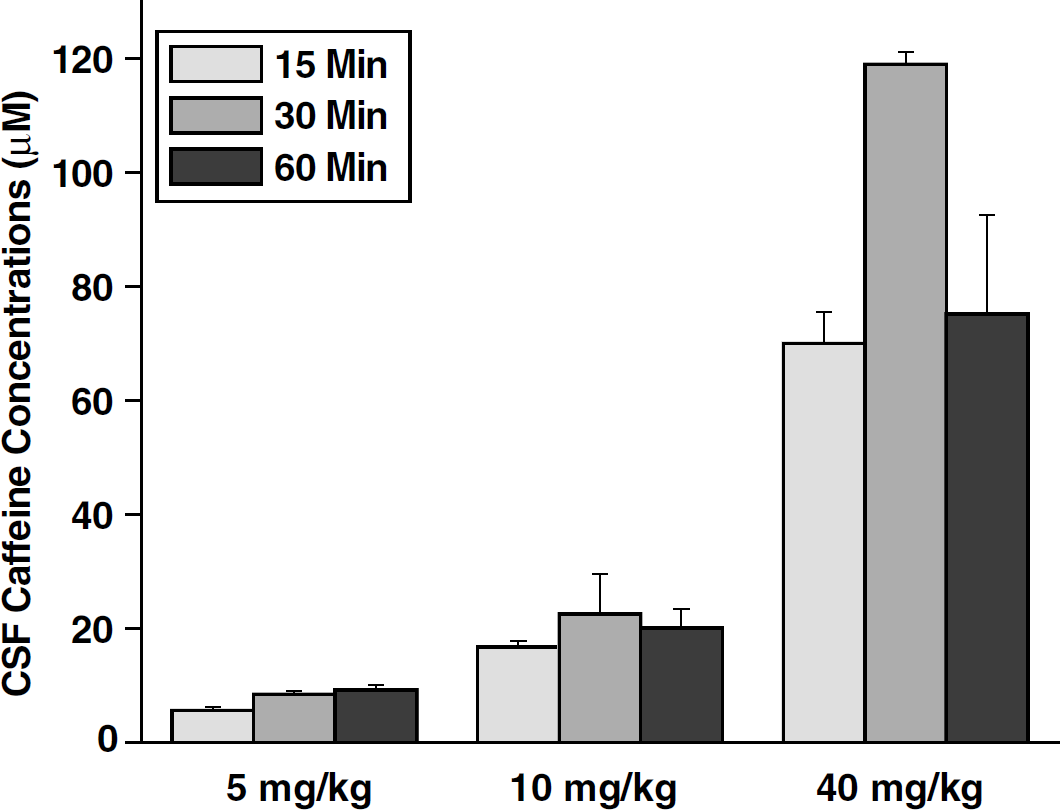
Cerebrospinal fluid (CSF) Caffeine concentrations. Cisternal CSF was sampled at 15, 30, or 60 mins after intravenous injection of caffeine at 5, 10, or 40 mg/kg. Data are expressed as mean±s.d. (n=18, with 2 animals in each group).
Cortical Activation Studies
Physiologic parameters
The effects of intravenous administration of caffeine or theophylline on physiologic parameters are presented in Tables 2 and 3, respectively. To summarize, systemic administration of caffeine had no effect on MABP, either under control conditions (just before SNS) or during SNS. However, caffeine tended to increase heart rate, particularly at the highest dose (40 mg/kg) where the effect was statistically significant from control (P<0.05).
Effect of intravenous caffeine on physiologic parameters during sciatic nerve stimulation
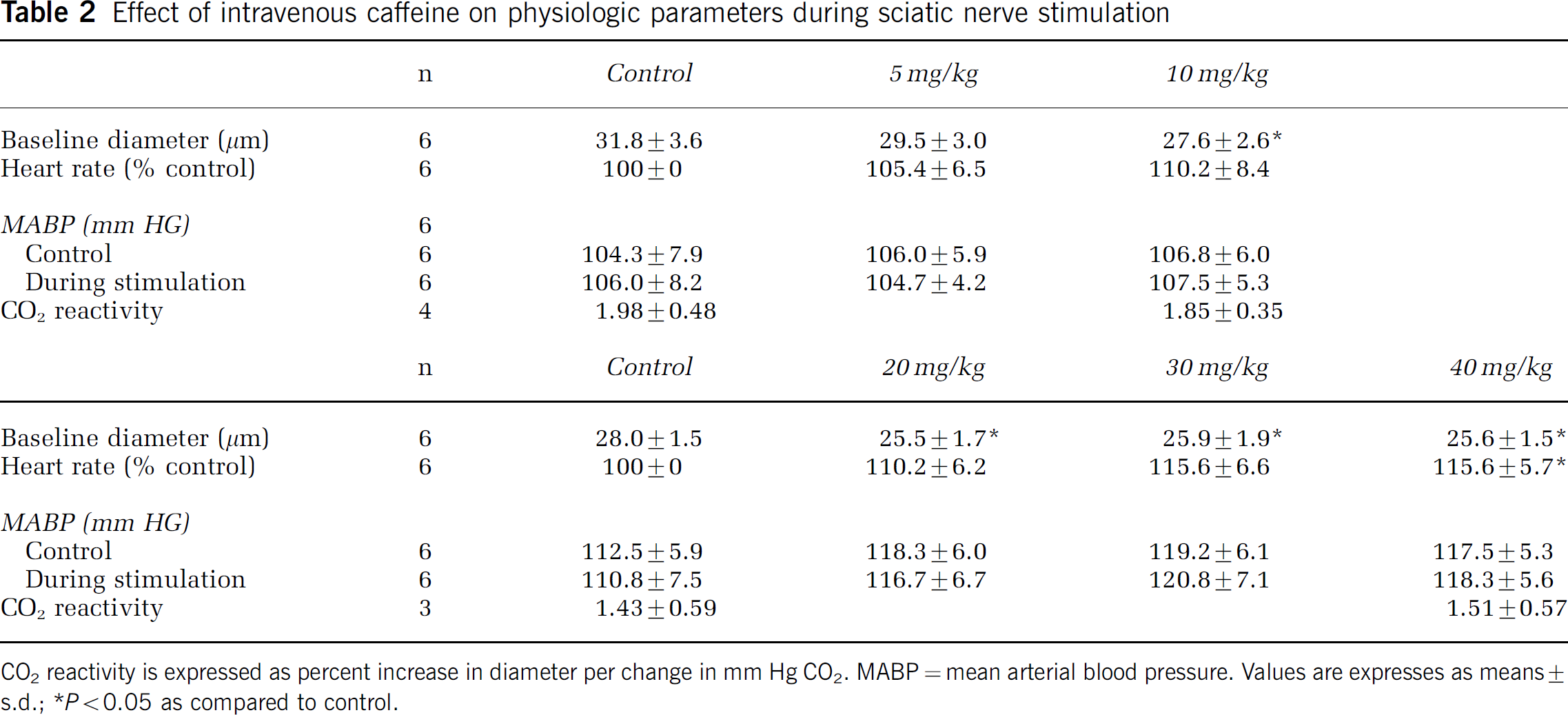
CO2 reactivity is expressed as percent increase in diameter per change in mm Hg CO2. MABP=mean arterial blood pressure. Values are expresses as means±s.d.;
P<0.05 as compared to control.
The effects of theophylline on physiologic parameters are presented in Table 3. Like caffeine, theophylline had no effect on MABP, either before or during SNS. Similar to caffeine, theophylline also tended to increase heart rate, however, none of the doses tested resulted in a statistically significant effect.
Effect of intravenous theophylline on physiologic parameters during sciatic nerve stimulation
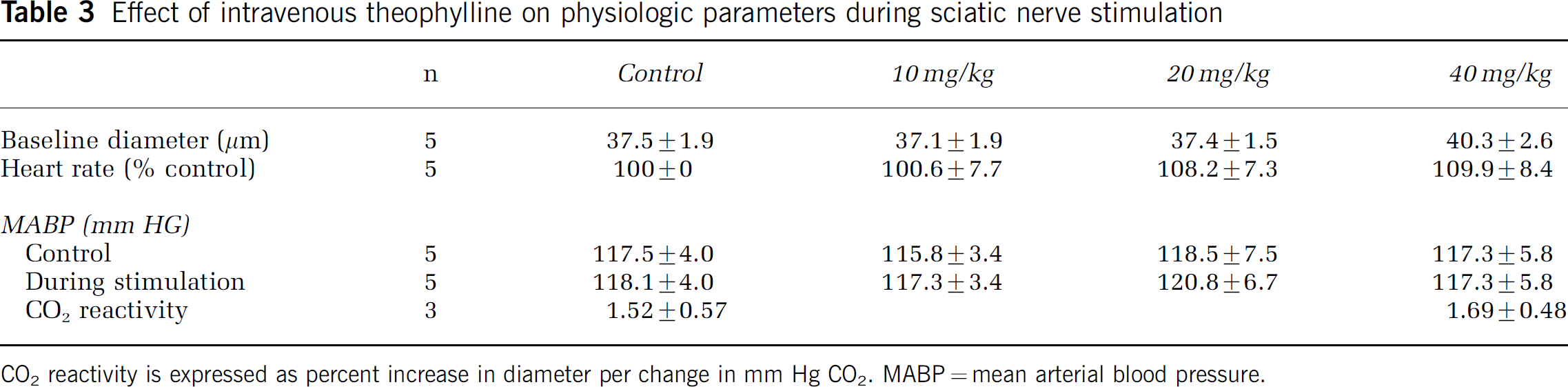
CO2 reactivity is expressed as percent increase in diameter per change in mm Hg CO2. MABP=mean arterial blood pressure.
Effect of intravenous caffeine and theophylline on resting pial arteriolar diameter
The effects of caffeine on resting arteriolar diameter are presented in Table 2. In one group of animals where lower doses of caffeine was evaluated, the lowest dose of caffeine (5 mg/kg) had no effect on resting diameter. Doubling the dose of caffeine (10 mg/kg) caused a significant reduction in baseline arteriolar diameter (P<0.05; n=6). Moreover, caffeine caused small but statistically significant decreases (P<0.05; n=6) in resting pial arteriolar diameter at all of the higher doses tested (20, 30, and 40 mg/kg; Table 2). For example, resting arteriolar diameter before caffeine administration was 28.0±1.5 μm, which was significantly (P<0.05, n=6) reduced to 25.5±1.7 μm 15 mins after the first dose of caffeine (20 mg/kg, intravenous).
In contrast to caffeine, theophylline (Table 3) was without effect on resting pial arteriolar diameter at all concentrations. The highest dose (40 mg/kg) theophylline tended to increase baseline diameter. However, this trend failed to reach statistical significance.
Effect of intravenous administration of caffeine on sciatic nerve stimulation-induced pial arteriolar dilation
Stimulation of the contralateral sciatic nerve caused significant (P<0.001; n=12) vasodilatation of pial arterioles in the hindlimb somatosensory cortex. Before intravenous administration of caffeine, pial arterioles dilated an average of 23.8%±3.9% in response to contralateral SNS (n=12). The effects of low doses of caffeine (5 and 10 mg/kg) on SNS-induced vasodilatation are illustrated in Figure 3. The lowest dose of caffeine (5 mg/kg) resulted in a downward trend that did not reach statistical significance. In the same group of animals, 10 mg/kg of caffeine significantly attenuated arteriolar dilatation to SNS from 26.1%±2.4% to 19.1%±4.9% (P<0.05; n=6).
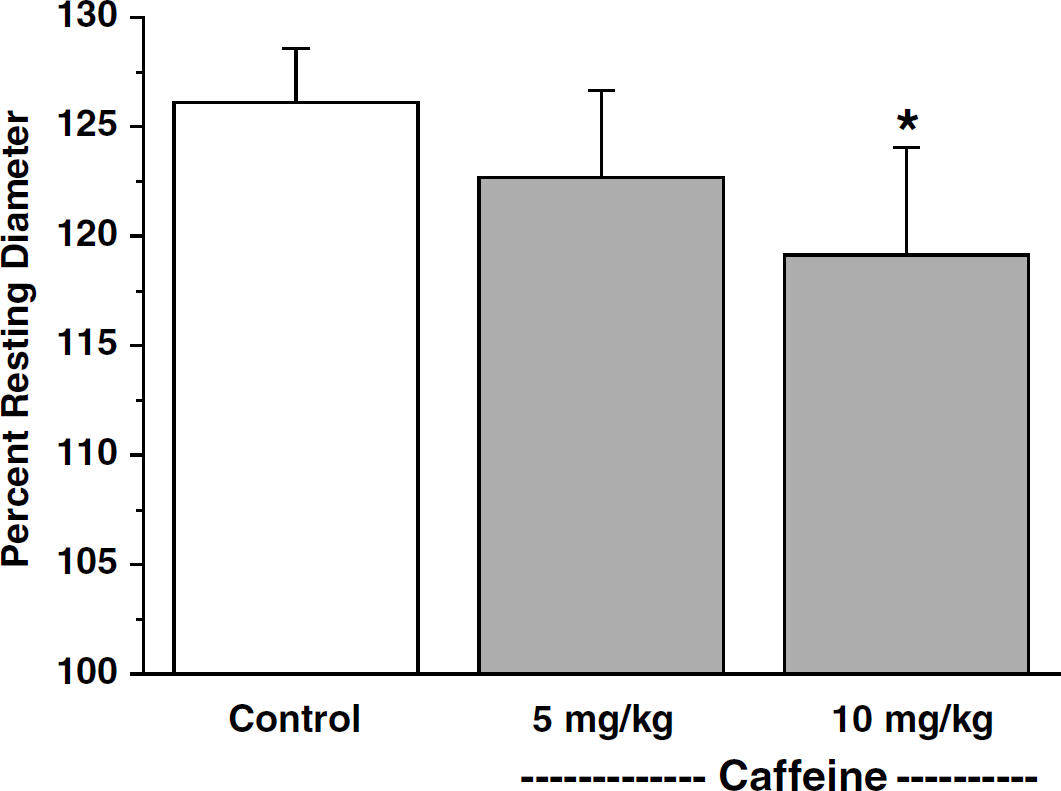
Effects of intravenous administration of low doses of caffeine on pial arteriolar dilation to contralateral sciatic nerve stimulation (SNS). Dilation responses are expressed as percentage of resting baseline diameter. Data are expressed as means±s.d. (n=6). *P<0.05 versus control.
Higher doses of caffeine (20, 30, and 40 mg/kg), similar to amounts utilized in the study by Gotoh et al (2001), were evaluated in another group of animals (Figure 4). All of the doses resulted in significant (P<0.05, n=6) and dose-dependent reduction of SNS-induced vasodilatation of pial arterioles. For example, 15 mins after caffeine administration of 20, 30 and 40 mg/kg, SNS-induced vasodilatation was significantly reduced by 39.2%, 59.2%, and 70%, respectively.
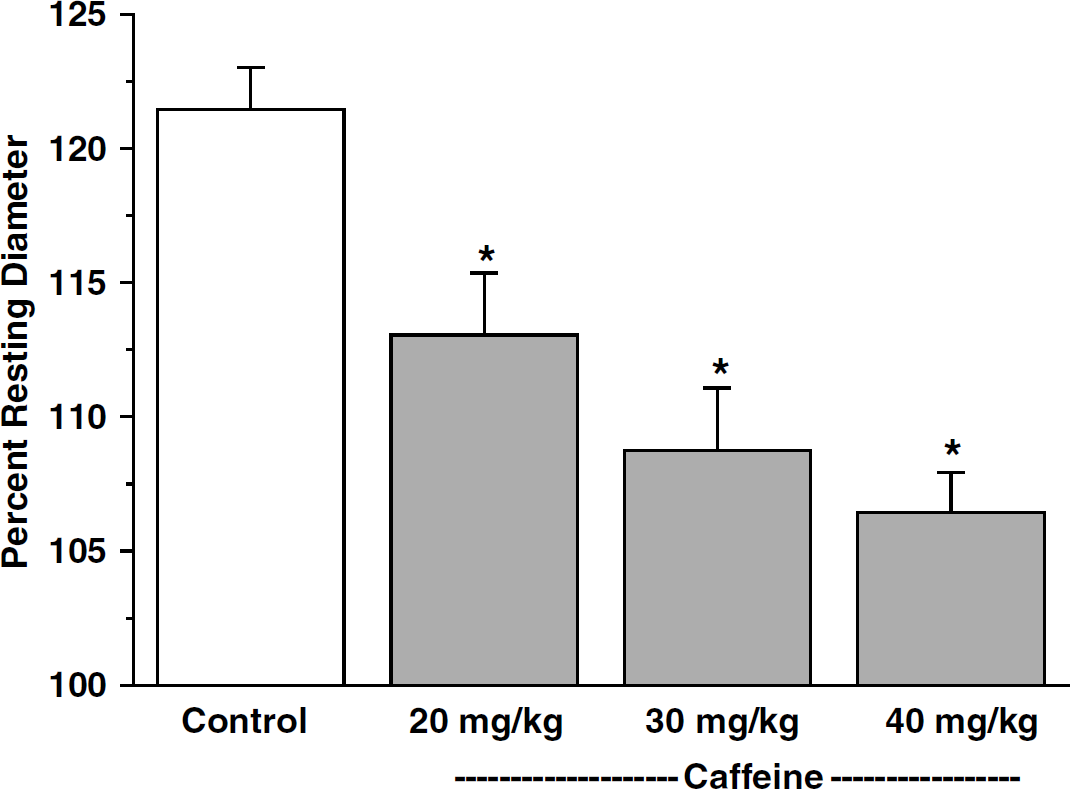
Effects of intravenous administration of high doses of caffeine on pial arteriolar dilation to contralateral sciatic nerve stimulation (SNS). Dilation responses are expressed as percentage of resting baseline diameter. Data are expressed as means±s.d. (n=6). *P<0.05 versus control.
To rule out potential nonspecific effects that caffeine may be causing, we also evaluated the effect of caffeine on pial arteriolar dilation to CO2 inhalation. Intravenous administration of caffeine had no effect on reactivity of pial arterioles to CO2 inhalation (control=1.98%±0.48%/mm Hg; caffeine (10 mg/kg)=1.85%±0.35 %/mm Hg; see Table 2).
Effect of intravenous administration of theophylline on sciatic nerve stimulation-induced pial arteriolar dilation
Similar to the control group in the caffeine experiments, stimulation of the contralateral sciatic nerve caused an 18.5%±1.7% increase in pial arteriolar diameter in the hindlimb sensory cortex (n=5; Figure 5) under control conditions. In contrast, 15 mins after intravenous administration of theophylline (10, 20, and 40 mg/kg), SNS-induced vasodilatation was significantly (P<0.05) reduced by 48.4%, 60.4%, and 70.3%, respectively. In addition, as in the case of caffeine, there was no effect of theophylline on the reactivity of pial arterioles to hypercarbia (Table 3).
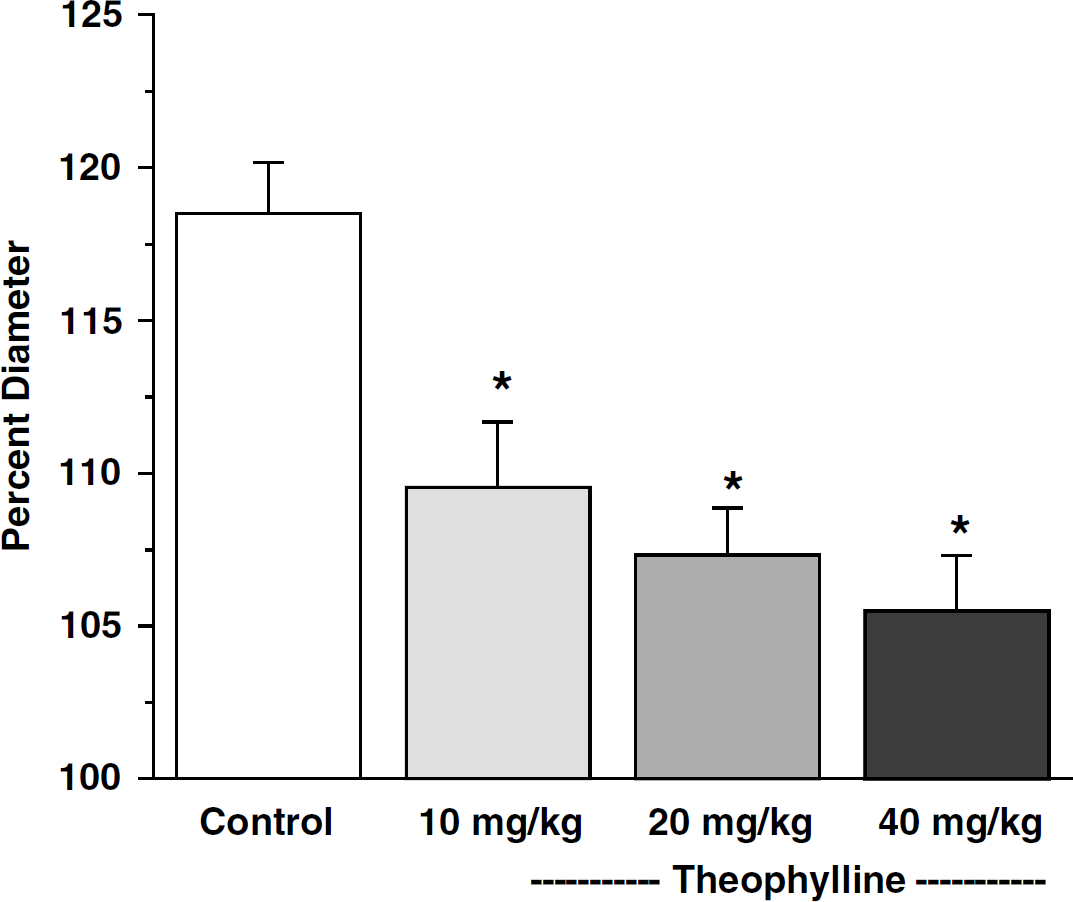
Effects of intravenous administration of theophylline on pial arteriolar dilation to contralateral sciatic nerve stimulation (SNS). Dilation responses are expressed as percentage of resting baseline diameter. Data are expressed as means±s.d. (n=5). *P<0.05 versus control.
Discussion
The primary goal of the present study was to examine the effects of caffeine on the cerebrovascular responses to functional neuronal activation. Caffeine is among the most widely consumed CNS stimulants (Nehlig, 1999; O'Brien and Kinnaird, 2003) and can be found in a variety of common food and beverages including coffee, tea, chocolate, and cola drinks (Gilbert and Scott, 2000). Despite this, experimental data regarding the effects of caffeine on CBF response to neuronal activation are sparse. In the present study, intravenous administration of caffeine at four different doses (10, 20, 30, or 40 mg/kg) led to significant and dose-dependent attenuation of the pial arteriolar dilation in response to contralateral SNS. After intravenous administration of 5 mg/kg (comparable to 2 cups of brewed coffee; Nehlig, 1999), we noted only a downward trend that did not reach statistical significance. Our observations parallel the report by Horinaka et al, 1997 in the conscious rat where increases in CBF during insulin-induced hypoglycemia were nearly abolished by caffeine. In the present study, theophylline, another widely consumed methylxanthine, had comparable effects to caffeine on attenuating pial vasodilatation during SNS-induced cortical activation. Moreover, the effects of intravenous theophylline were similar to our previous observations with intraperitoneal administration (Meno et al, 2001).
Caffeine and theophylline are well-known potent and nonselective adenosine receptor antagonists affecting A1 and A2 receptors (but without effect on A3 receptors). Methylxanthines have other well-documented actions, including inhibition of phosphodiesterase as well as mobilization of catecholamines and calcium (Morii et al, 1987). However, the primary CNS actions of caffeine and theophylline resulting from daily food/beverage consumption are related to adenosine receptor blockade (Nehlig, 1999; O'Brien and Kinnaird, 2003). The doses of caffeine and theophylline used in the present study are in the range that predominately affect adenosine receptors (Hirsh, 1984; Morii et al, 1987).
The effects of caffeine on resting levels of CBF have been reported in a number of previous studies (Field et al, 2003; Mathew and Wilson, 1985; Mulderink et al, 2002). For example, Mathew and Wilson (1985) using 133 Xenon technology and Field et al (2003) using inversion-recovery perfusion MR imaging both showed reduction in resting CBF values after caffeine administration in humans. The discovery that caffeine significantly reduces baseline cerebral perfusion has benefited recent efforts to quantify functional CBF changes using BOLD fMRI (Laurienti et al, 2002, 2003; Mulderink et al, 2002). By reducing resting CBF levels, caffeine decreases deoxyhemoglobin and BOLD baseline levels that afford enhanced BOLD contrast during activity-evoked CBF changes. More recent BOLD studies have begun to evaluate caffeine-induced regulation of adenosine receptors (Laurienti et al, 2003). Gotoh et al (2001) using awake rats also documented significant reductions in resting rCBF in multiple brain regions, using [14C] IAP method.
In the present in vitro studies, caffeine was without effect on the resting diameter of intracerebral arterioles. In contrast, in our in vivo studies, systemic administration of caffeine caused significant constriction of pial arterioles as visualized through a cranial window (Table 2). Taken together, the decrease in the resting CBF noted by earlier investigators, the reduction in pial arteriolar diameter observed in our in vivo studies, and the lack of effect on resting diameter of our in vitro arterioles strongly suggest that the observed decrease in CBF after caffeine administration is related to a parenchymal effect of caffeine rather than a direct vascular effect.
In our in vitro arterioles, 50 μmol/L (but not 10 μmol/L) caffeine significantly attenuated adenosine-induced vasodilatation. This information suggests that the higher doses of caffeine we used in our subsequent in vivo SNS experiments resulted in CSF caffeine concentrations that were greater than 10 μmol/L. Indeed, our in vivo dose/CSF concentration studies confirmed that CSF concentrations greater than 10 μmol/L were achieved after intravenous administration at all doses except 5 mg/kg. Previous studies have shown that after oral administration of 10 mg/kg caffeine to rat, plasma levels were 20 to 50 μmol/L (Abdi et al, 1993; Aldridge et al, 1977).
Our observation that caffeine significantly attenuated SNS-induced vasodilatation is in agreement with several previous studies aimed at determining the role of adenosine in the regulation of cerebrovascular responses to increased cortical activity (Dirnagl et al, 1994; Ko et al, 1990; Li and Iadecola, 1994; Meno et al, 2001). For example, Ko et al (1990) and Meno et al (2001) showed respectively, that topical or intraperitoneal administration of theophylline significantly attenuated pial arteriolar dilation to contralateral SNS. Moreover, Dirnagl et al (1994) reported that both theophylline or adenosine deaminase significantly blunted increases in laser Doppler flow during whisker stimulation. Lastly, Li and Iadecola (1994) showed that increases in CBF in the cerebellar cortex are significantly attenuated by theophylline. In the present study, both caffeine and theophylline dose-dependently reduced arteriolar dilations during SNS. At the highest dose of either of these compounds, SNS-induced vasodilatation was significantly reduced by greater than 70%.
The attenuation in CBF during neuronal activation after administration of caffeine and theophylline could be related to depression of electrical excitation and/or brain metabolism, which in turn could lead to a secondary reduction of the vascular response. However, in the present study, depression of electrical activity or metabolism is not likely since we have previously shown that SSEP measurements remain robust after theophylline administration and Gotoh et al (2001) noted that ICMRglu is not depressed after caffeine administration.
In contrast to the present and previous studies, Gotoh et al (2001) reported the caffeine was without effect on cortical blood flow during neuronal activation (whisker barrel stimulation); but caffeine did attenuate CBF in subcortical areas (trigemenal nuclei in the brain stem) and led to an insignificant downward trend in the thalamic relay nucleus (VPM) and a nonsignificant enhanced percentage increase in barrel field cortex. The observations by Gotoh et al (2001) represents the only study aimed specifically at determining the effects of caffeine on CBF during somatosensory stimulation. Moreover, the lack of effect of caffeine in the barrel field cortex is in variance with observations of the present study with caffeine and to earlier studies using theophylline (Dirnagl et al, 1994; Ko et al, 1990; Meno et al, 2001).
There are a number of considerations that may account for the differences in the effects of caffeine between the present and the study of Gotoh et al (2001). One potentially significant difference is that we used anesthetized animals whereas Gotoh et al (2001) performed their experiments on awake animals. Anesthesia and various anesthetic agents have well-known effects on both resting CBF and CBF reactivity. For example, α-chloralose anesthesia reportedly decreases baseline CBF and blunts increases in CBF in the barrel field cortex during whisker stimulation (Nakao et al, 2001). In the present study, the animals were anesthetized with α-chloralose and urethane (50 and 500 mg/kg, respectively) as previously described (Ngai et al, 1988) and in accordance with IACUC guidelines. Despite the anesthesia, we were able to evoke significant increases in pial arteriolar diameter during SNS, averaging 23.8%±3.9% (n=12) in the absence of caffeine or theophylline. Such changes in pial arteriolar diameter correspond to a 70% increase in CBF, based on Poiseuille's law and our previous measurements using laser Doppler flowmetry (Ngai et al, 1995). Our observed increases in pial arteriolar diameter and derived CBF increases are comparable to the 51% increase in CBF reported by Gotoh et al (2001) in the barrel cortex during whisker stimulation. Thus, we would conclude that depression of the CBF response in anesthetized animals may not account for the differences between the results of the present study compared with that of Gotoh et al (2001).
The different methodologies (i.e., autoradiographic quantification of CBF used by Gotoh et al versus in situ observation of cerebral arterioles used in the present study) may provide additional explanation. For example, the temporal pattern of CBF response to forepaw (Ances et al, 2000) and whisker (Gerrits et al, 1998) is such that there is an initial peak followed by a plateau of lesser magnitude that persists throughout the stimulus duration. We have observed a similar pattern in pial arterioles during SNS (Ngai et al, 1988) and subsequently showed a strong correlation between arteriolar measurements and laser Doppler flowmetry (Ngai et al, 1995). Gotoh et al (2001) used autoradiographic quantification of CBF, which is based on the Fick principle and requires a stable CBF. Thus, temporal changes in CBF that occur during somatoasensory stimulation are averaged over the entire stimulation period, making valid comparisons between the two studies difficult. However, Gotoh et al (2001) did document a difference in the CBF changes in subcortical areas after caffeine administration. Although the pattern of dynamic changes in CBF during neuronal activation (initial short duration peak and subsequent longer lower plateau) have been documented in activated cortex, whether a similar pattern of response occurs in subcortical regions during neuronal activation is unknown. Perhaps a more robust and sustained response occurs in subcortical regions, which would be more accurately quantified using the autoradiographic technique used by Gotoh et al (2001).
In summary, we report that caffeine dose-dependently attenuates CBF increases during neuronal activation. Moreover, caffeine does not have a primary vascular effect on in vitro arterioles, but does attenuate vasodilatation induced by exogenous application of adenosine. Thus, the present study demonstrates that caffeine and theophylline, both widely consumed compounds, have significant effects on neurovascular coupling and further supports a role for adenosine in the regulation of CBF during somatosensory stimulation.
