Abstract
Developing cerebral infarction obscures the relationship of neurons to their local supply microvessels. We tested the notion that in the basal ganglia (i) an ordered relationship between neurons and their nearest neighboring microvessel exists, and (ii) focal ischemia predictably affects neuron integrity based on microvessel–neuron proximity. Distances between individual microvessels and their nearest neurons ([m–n distance]s) were measured in normal primates and ischemic subjects undergoing middle cerebral artery occlusion for 2 hours. An ordered microvessel–neuron relationship exists in the normal nonischemic basal ganglia within the early hours of focal ischemia. During ischemia normal (n) and sensitive (n∗) neurons are interspersed. On average, neurons more distant from their nearest microvessel are most sensitive ([m–n distance]=16.2±11.2 μm versus [m–n∗ distance]=22.2±13.0 μm, 2P≪0.00000001). Neurons not expressing glutamic acid decarboxylase were more likely to be sensitive than those with a normal microvessel–neuron relationship. In contrast, the [m–n distance] distribution of injured tyrosine hydroxylase-containing neurons was similar to those without tyrosine hydroxylase. Hence, the [m–n distance] relationship in the normal and ischemic basal ganglia is highly ordered, and distant neurons are consistently perturbed by ischemia, although this is not uniformly dependent on neurotransmitter type.
Introduction
In stroke patients and experimental models, tissue infarction is taken as the measure of neuron injury after focal ischemia (Garcia et al, 1993; Barid et al, 1997; Mabuchi et al, 2000). This tenet assumes that neuron injury and necrosis are due directly to hypoxia and its consequences (Siesjo, 1985). Strategies which alter neuron responses to hypoxia, including antagonists to neuron NMDA or AMPA ion channel receptors, decrease infarction volume in experimental models of middle cerebral artery occlusion (MCAO) without altering the ischemic stimulus (Dirnagl et al, 1990; Minger et al, 1998). But, the decrease in regions of neuron injury should also reflect a dependence on vascular supply. In this formulation, the greatest relevance to the individual neuron is its immediate supply microvessel. But, there is little information about the local direct and indirect interactions between individual neurons and their neighboring microvessels.
To date, no cerebral vascular territory has been mapped for the microvascular supply of individual neurons, although the microvessel array of the rodent cortex has been described (Bär and Wolff, 1972; Bär, 1978). Carpenter and others have suggested that the distributions of neurons within the primate basal ganglia are relatively homogeneous (Parent, 1996). This should allow a quantitative assessment of the organization and fate of neurons and their proximate microvessel supply after ischemia. However, late-following MCAO, secondary inflammation and edema obscure these relationships.
Microvessel responses to ischemia depend in part on their structure. Cerebral capillaries, the identified site of O2 exchange, consist of endothelial cells and astrocyte end-feet resting on the intervening basal lamina matrix (ECM) (del Zoppo, 1994). Pericytes and histiocytes also contribute to this complex (Ookawara et al, 1996). Krogh and others proposed a limit to O2 diffusion for a hypothetical capillary in the central nervous system (CNS) (Quistorff et al, 1977). Hence, neurons might be positioned less than a fixed critical distance from the nearest capillary (microvessel) if diffusion distance were the major factor in tissue organization.
A second consideration is whether neurons exchange information with their proximate microvessels. Astrocytes provide a link between endothelial cells and neurons. The recent report of Zonta et al (2003) supports this concept. If an ordered relationship between viable neurons and their proximate microvessels exists, then it should be possible to observe alterations within the target territory in the early moments of focal ischemia before the onset of the secondary injury.
Based on these considerations, we tested the hypotheses that within the nonhuman primate basal ganglia (i) an ordered relationship exists between neurons and their nearest neighboring microvessels, and (ii) focal ischemia affects neuron responses in a consistent manner based on proximity. Here, neuron ‘sensitivity’ is defined by evidence of nuclear DNA scission (dUTP incorporation) at 2 hours MCAO in the absence of large artery reperfusion, as shown by Tagaya et al (1997). These studies are the first to show a consistent and coordinate response of cerebral microvessel–neuron pairs to controlled focal cerebral ischemic injury in a single arterial territory.
Materials and methods
The experimental procedures used here were approved by the institutional Animal Research Committee and were performed according to standards published by the National Research Council (The Guide for the Care and Use of Laboratory Animals) and the United States Department of Agriculture Animal Welfare Act. Every effort was made to ensure that the subjects were free of pain and discomfort. All procedures were attended by the veterinarians, the primate handling staff, research personnel, and the principal investigator.
Experimental Stroke Model in the Nonhuman Primate
Because of their very close homology to humans, cerebral tissues from 12 adolescent male baboons (Papio anubis/cynocephalus) were used for these studies (normal, n=6; 2 hours MCAO without reperfusion, n=6). The detailed surgical and experimental approaches to the awake nonhuman primate stroke model have been described previously (Fukuda and del Zoppo, 2003).
Tissue Processing
Each experiment was terminated by left ventricular transcardiac perfusion of the cranial structures at 180 to 220 Torr with chilled isosmotic perfusate containing heparin (200 IU/L), nitroprusside (1 mg/L), and bovine serum albumin (BSA, 50 g/L) (Sigma, St Louis, MO, USA), under thiopental Na+ anesthesia. Tissue blocks (1.0 × 1.0 × 0.5 cm3) from symmetrically located sites of both left and right basal ganglia were excised, and immediately immersed in 2% paraformaldehyde (PFA) for 24 hours before to paraffin embedding. Sections from one subject were prepared in the coronal, sagittal, and horizontal planes. All remaining blocks were cut in the coronal plane.
Antibodies
Specific components of the microvessel wall were identified with well-characterized antibodies against the human antigens. These included a rabbit polyclonal antibody against human laminin (applied in 1:30 dilution; DAKO, Carpentaria, CA, USA) and a murine monoclonal antibody (MoAb) against collagen (clone COL-94, collagen type IV, 1:100; Sigma) (Hamann et al, 1995). Neurotransmitters or their processing enzymes were identified with a rabbit gamma-aminobutyric acid (GABA) polyclonal antibody (1:800; Sigma), a goat choline acetyltransferase (ChAT) polyclonal antibody (1:30; Chemicon, Temecula, CA, USA), and murine tyrosine hydroxylase (TH) MoAb (1:100; Chemicon) and anti-MAP-2 MoAb (1:50; Sternberger Monoclonals Incorporated, Lutherville, MD, USA). Glutamic acid decarboxylase (GAD) was identified with a rabbit polyclonal antibody (1:1000; Sigma).
Determination of the Region of Cellular dUTP Incorporation
Evidence of nuclear DNA scission/repair was taken as an indication of significant cellular injury as described by (Tagaya et al, 1997; Abumiya et al, 1999). The DNA polymerase I-based procedure for incorporation of digoxigenin dUTP was performed on deparaffined sections employed for the antigen studies. Hereafter, neurons that incorporated dUTP (dUTP+) are designated n∗, in contrast to dUTP− neurons (n).
Immunohistochemistry
Immunoperoxidase studies were performed on serial 3 μm paraffin sections after dUTP incorporation, as described previously (Abumiya et al, 1999). Sections were incubated with proteinase K (20 μg/mL) (Sigma) for 5 mins, washed, then incubated overnight with the primary antibody at 4°C. The sections were incubated with biotinylated horse anti-mouse, goat anti-rabbit (Vector Laboratories, Burlingame, CA, USA), or biotinylated donkey anti-goat IgG (Jackson Immunoresearch Laboratories, West Grove, PA, USA), where appropriate. Avidin–biotin complexes were detected with NovaRED, SG, or DAB substrate kits (Vector Laboratories), and counterstained for Nissl substance (Polysciences, Inc., Warrington, PA, USA). In all sections, microvessels (laminin) and neurons (MAP-2, or Nissl counterstain) were identified.
Quantitative Analysis
The minimum linear distances between the nearest facing outer boundaries of each neuron and its nearest microvessel (m–n distances) were measured with computerized digital video-imaging microscopy (Carl Zeiss, Oberkochen, Germany) at × 400. Each data set was obtained separately by two investigators each masked to the section assignments and sample origins, and to each other's experience. The quantitative observations between the observers were indistinguishable statistically. All sections were scanned under the mode of 8 bits per pixel digital resolution and 0.49 μm2 pixel size.
The absolute number of m–n pairs and their respective distances were realized in coregistered 1.5 mm2 regions of interest (ROI). In the ischemic basal ganglia, two identical ROI were chosen within the ischemic core (Ic) and peripheral (Ip) regions for each serial section from each block (Tagaya et al, 1997). One ROI was chosen from the contralateral nonischemic region and from the matched basal ganglia of normal subjects at the levels of the anterior commissure (AC) and the anterior thalamic (AT) structures in view of known regional differences in neuron density and identity.
Statistical Analysis
In a preliminary study, m–n distances were calculated within four noncontiguous ROI each in three coronal sections of normal basal ganglia (Figure 1). The m–n distance distributions ([m–n distance]) were right-skewed (Figure 1C); no statistically significant differences among the grouped individual [m–n distance]s were seen via multi-sample Kolmogorov–Smirnov goodness-of-fit statistic. Square root transformation of the pooled data induced approximate normality of the [m–n distance] (Figure 1D), as demonstrated by the linearity of the expected order statistics of the standard normal distribution to the order statistics of the sample. The Shapiro–Francia statistic (w) for assessing normality with the transformed [m–n distance]s is 0.99. Normality is achieved when w=1.00. Variations at the extremes of the plot are indicative of possible outliers. With (approximate) normality, assumptions for the validity of subsequent parametric analyses of variance are satisfied.
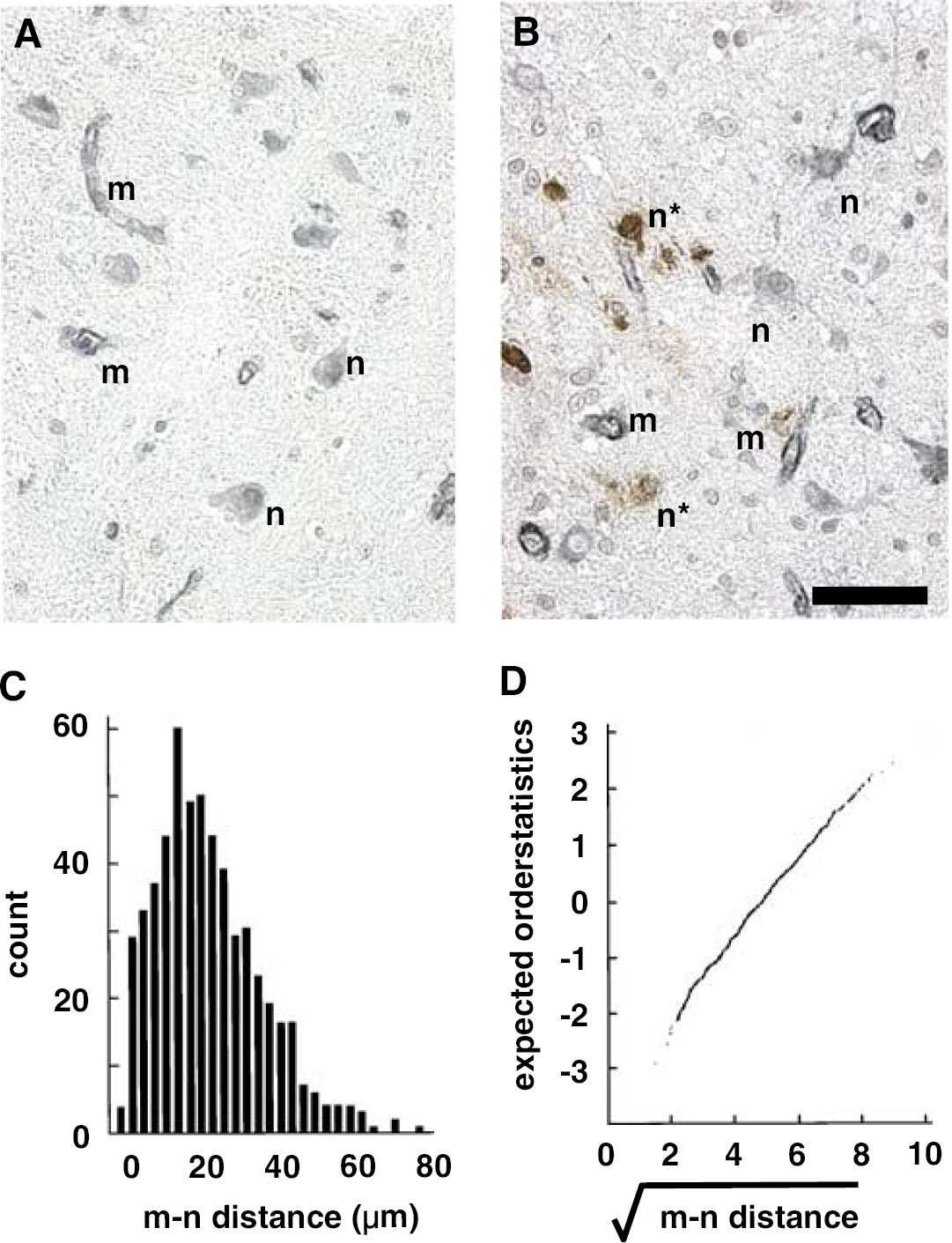
Photomicrograph showing distribution of microvessels and their neighboring neurons in the normal primate striatum (
All data are presented as the mean±standard deviation (s.d.). For each antigen, differences between the ischemic and matched nonischemic samples were assessed by two-way analysis of variance (ANOVA), or Student's t-test with Bonferroni corrections for multiple comparisons, where appropriate. Comparisons of distance measurements among sample blocks and serial planes were made by ANOVA. Significance was set at 2P<0.05.
Results
The Microvessel–Neuron Relationship in the Normal Basal Ganglia
Based on the preliminary data, [m–n distance]s were tabulated in five ROI each from five consecutive sections of normal basal ganglia in three planes at the levels of AC and AT in a single subject. Significant small differences in the transformed [m–n distance] at both levels were seen among the three planes only in the caudate and putamen (Table 1). Differences in the mean [m–n distance]s among the ROI were at most 1.8 μm in any dimension. Hence, data in subsequent experiments were taken from the coronal plane of the basal ganglia (caudate nucleus and putamen).
Comparison of [m–n distance] in the striatal regions of orthogonal planes
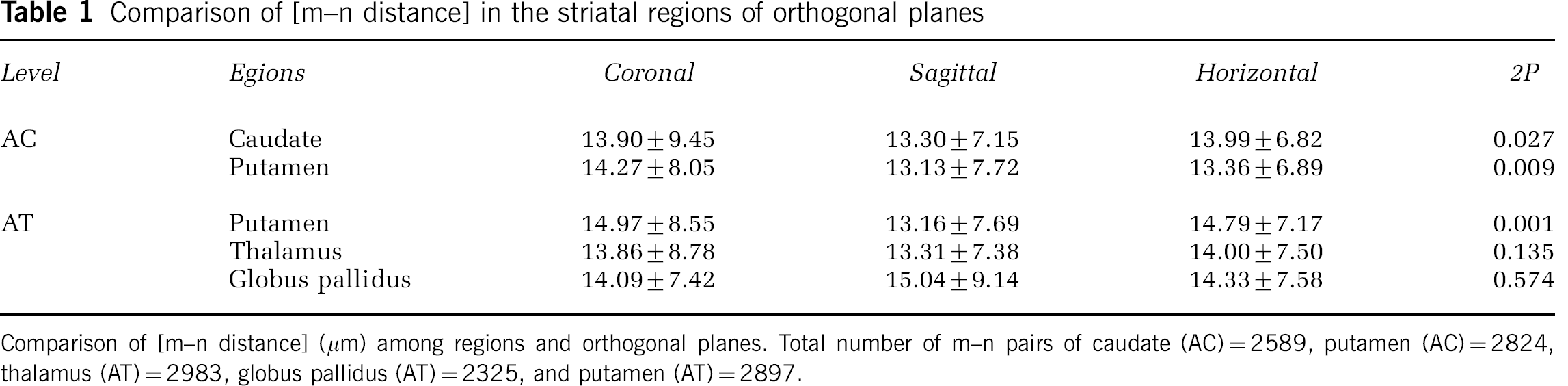
Comparison of [m–n distance] (μm) among regions and orthogonal planes. Total number of m–n pairs of caudate (AC)=2589, putamen (AC)=2824, thalamus (AT)=2983, globus pallidus (AT)=2325, and putamen (AT)=2897.
The consistency of the microvessel–neuron relationships was demonstrated from [m–n distance] within coregistered ROI of five serial coronal sections of three separate normal subjects (Figure 2). No differences in the transformed [m–n distance] were observed within any subject (P=0.92), or among the [m–n distance] distribution between either basal ganglia (P=0.249).
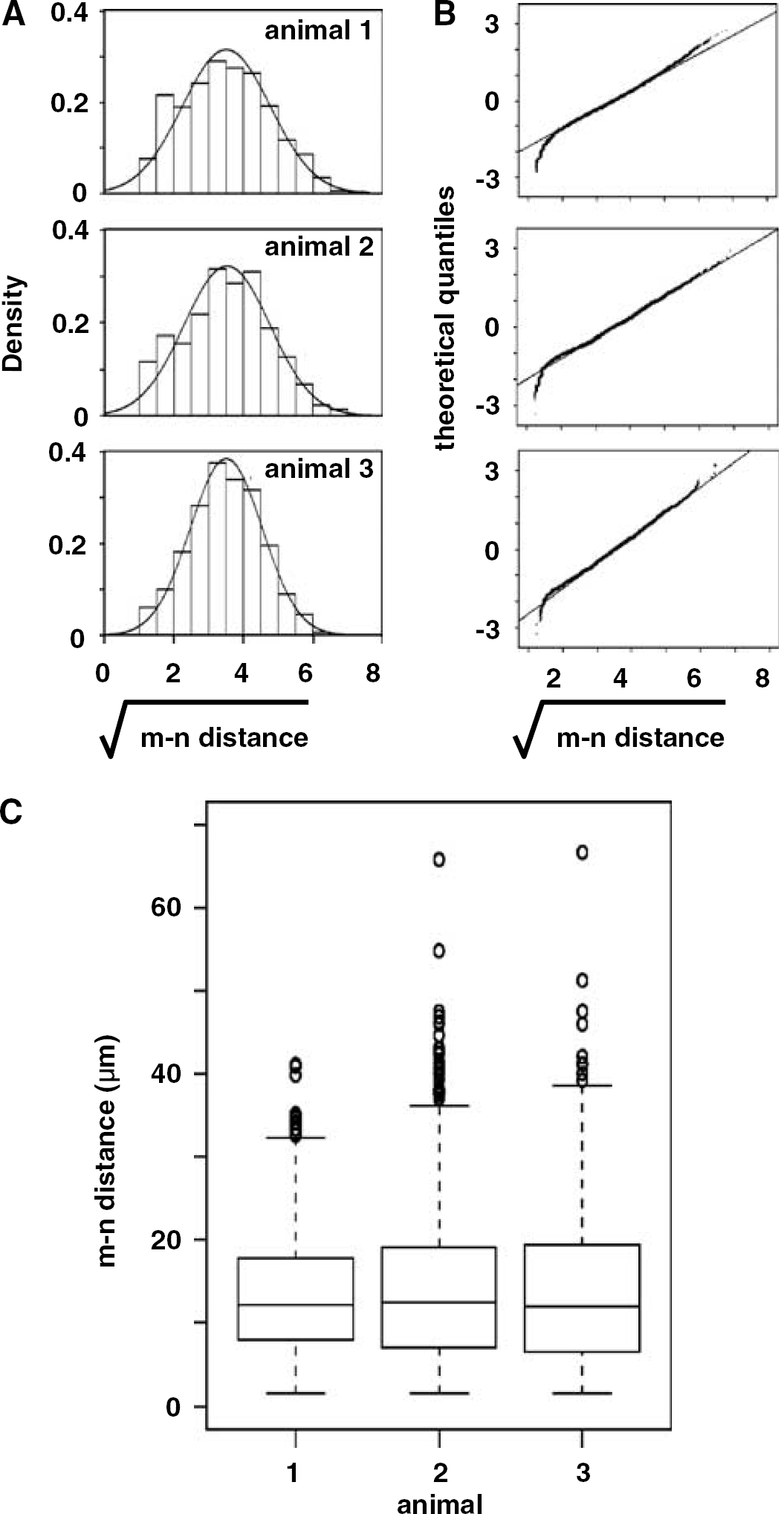
Identity of [m–n distance] among three normal animals. Normal distribution of transformed [m–n] data in each animal (
Neuron Subtype Identity
Next, [m–n distance]s for neurons displaying the individual neurotransmitter processing enzymes TH or ChAT, or the neurotransmitter GABA, were tabulated against MAP-2. Each transformed distribution also approximated normality, with [m–n distance]s characteristic for each neuron subtype (Figure 3). The [m–n distance] pattern of GABA-immunoreactive neurons was identical to that of MAP-2+ Nissl+ neurons. These medium-sized round-shaped neurons were chiefly distributed in the caudate-putamen at the level of the AC. Although fewer in absolute number, the [m–n distance] of TH-immunoreactive neurons were not different from those identified by GABA, Nissl, or MAP-2. TH+ neurons were characteristically pyramidal, larger than GABA+ cells, and distributed in the caudal portion of the basal ganglia. Large ChAT+ neurons, distributed chiefly at the AT level, were much fewer in number, and displayed a significantly longer mean [m–n distance] compared with the other neurotransmitter types.
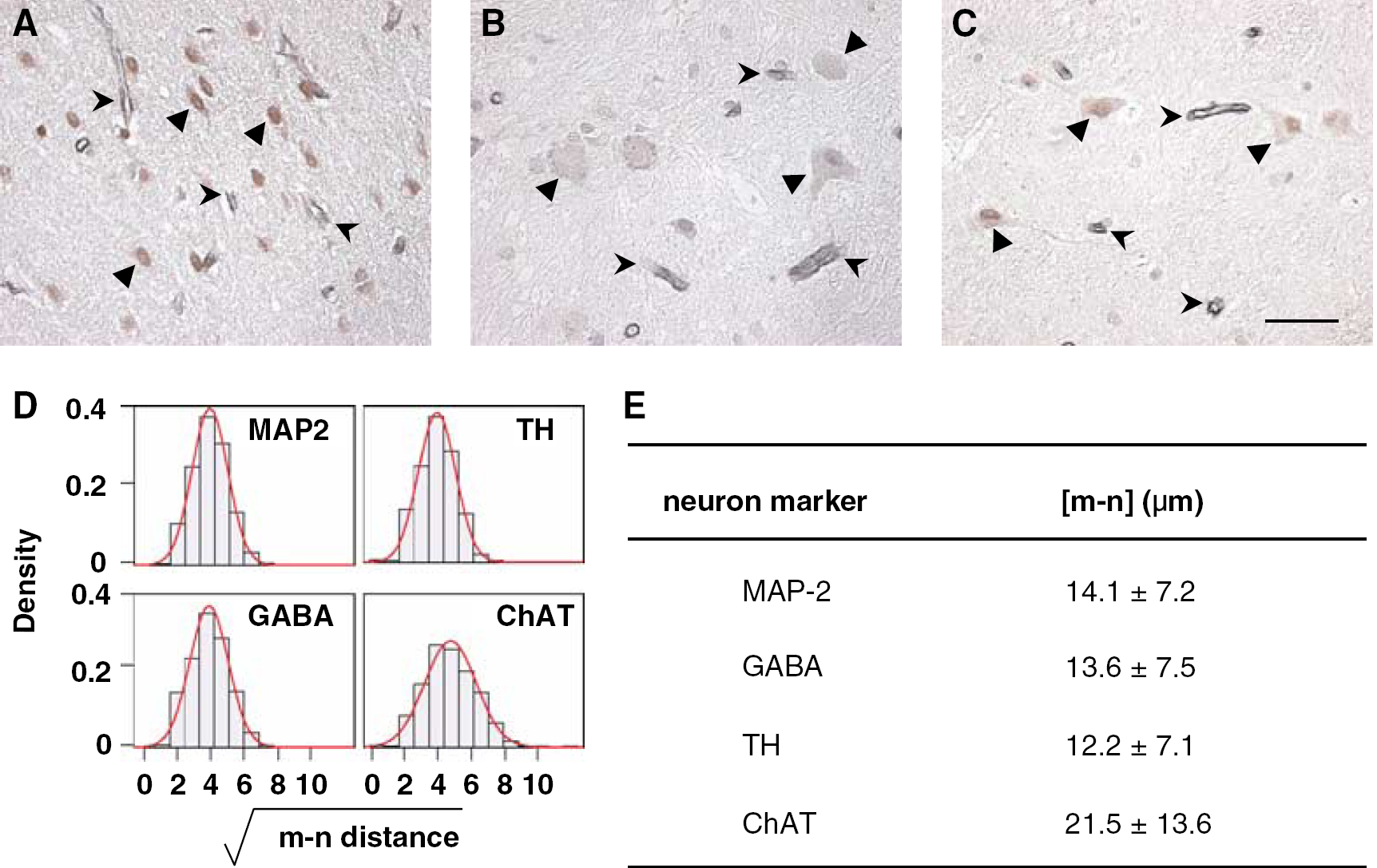
Distribution and profiles of neuron subtypes. GABA+ neurons (arrows) (
Focal Ischemia and the Microvessel–Neuron Relationship
After MCAO, Nissl+ neurons were identified by evidence of nuclear DNA scission (n∗), no DNA scission (n), or were indeterminate by dUTP incorporation techniques (Tagaya et al, 1997). At 2 hours after MCAO, injured neurons were heterogeneously distributed within the ischemic core of the basal ganglia. Prespecified ROI within the Ic region contained 49.2±11.3% dUTP+, 35.8±11.5% dUTP−, and 14.9±2.2% dUTP indeterminate neurons. The m–n∗ and m–n pairs were scattered within the Ic regions, without subregional preponderance of injury (‘hot spots’ of m–n∗ pairs). Furthermore, the distance between dUTP+ neurons and their nearest neighboring microvessel significantly exceeded that of dUTP− neurons within the same ROI (mean [m–n∗ distance]=22.2±13.0 μm versus mean [m–n distance]=16.2±11.2 μm; 2P≪0.00000001 overall) (Table 2). This observation was reproduced serially through the ischemic core, and was consistent among the three animal subjects (Figure 4).
Differences in [m–n distance] between dUTP− and dUTP+ neurons
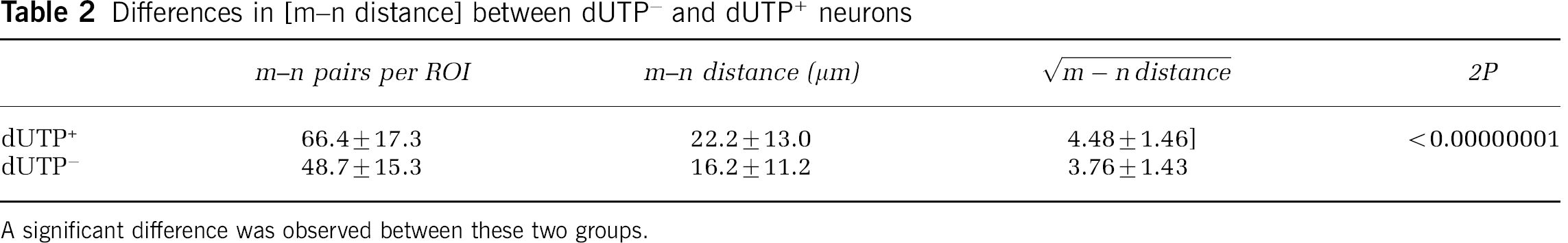
A significant difference was observed between these two groups.
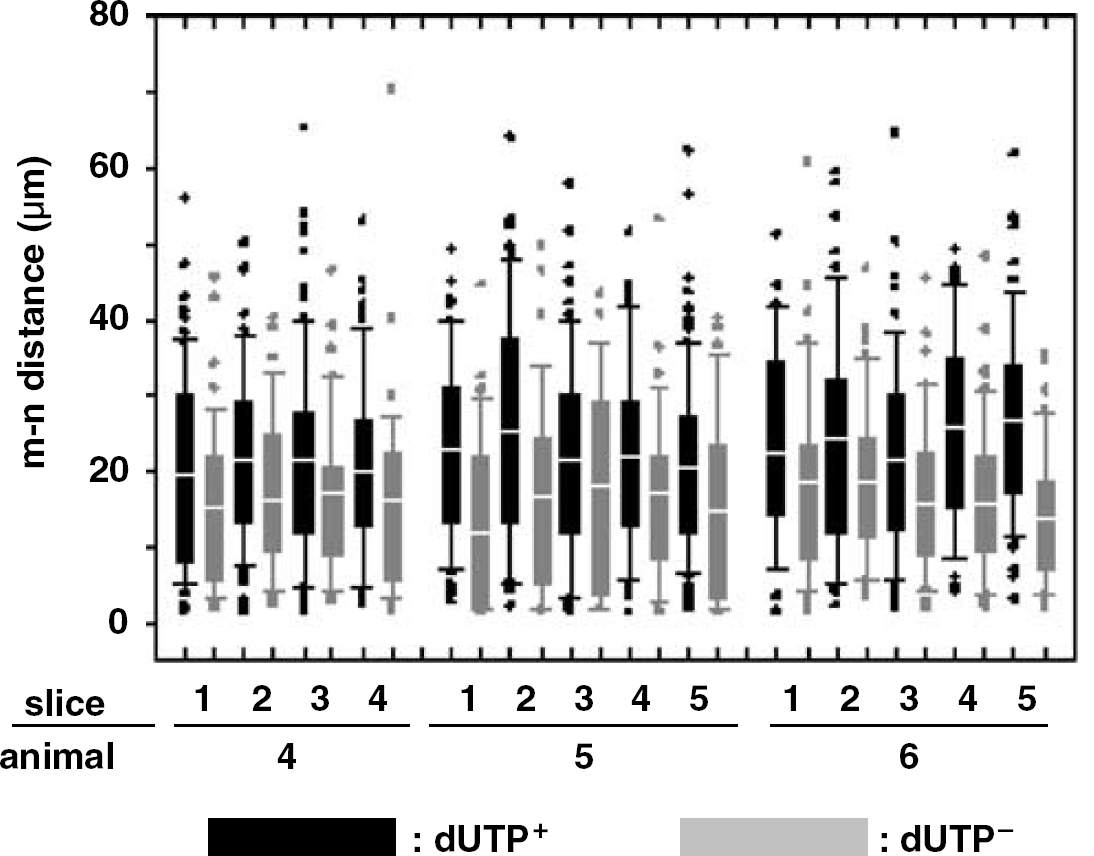
Comparison of [m–n distance] between dUTP− and dUTP+ neurons within the same ROI at 2 hours MCAO, presented as boxplots. Black bars display m–n∗ data, and gray bars display m–n data from ROI in respective slices.
Distortion of the Ischemic Regions During Ischemia
If local swelling (e.g. edema) lengthened the m–n distances of dUTP+ neurons, then the density of neurons or microvessels would substantially decrease in ROI throughout the Ic regions. However, no difference between the neuron densities in normal and ischemic tissues (309.3±23.8 versus 296.8±10.1 neurons/1.5 mm2, respectively; n=12; 2P=0.106) or in neuron cross-sectional diameter was observed (10.8±1.9 versus 10.5±1.8 μm, 2P=0.307) in the coronal plane.
Range of Effect of Ischemia on the Neuron–Microvessel Relationship
To determine the range of this phenomenon, distances from the target neurons to the nearest (primary), second, and third closest microvessels were determined for all neurons in three subjects (Figure 5). At 2 hours MCAO, significant increases in the [n∗–m distance] over [n–m distance] were observed in the prespecified ROI up to the tertiary microvessel. The majority (99±1%) of dUTP− neurons were detected within 30 to 40 μm of n∗ neurons. A decreasing proportion of dUTP− neurons were detected beyond this threshold in association with secondary (73±14%) and tertiary (24±10%) microvessels.
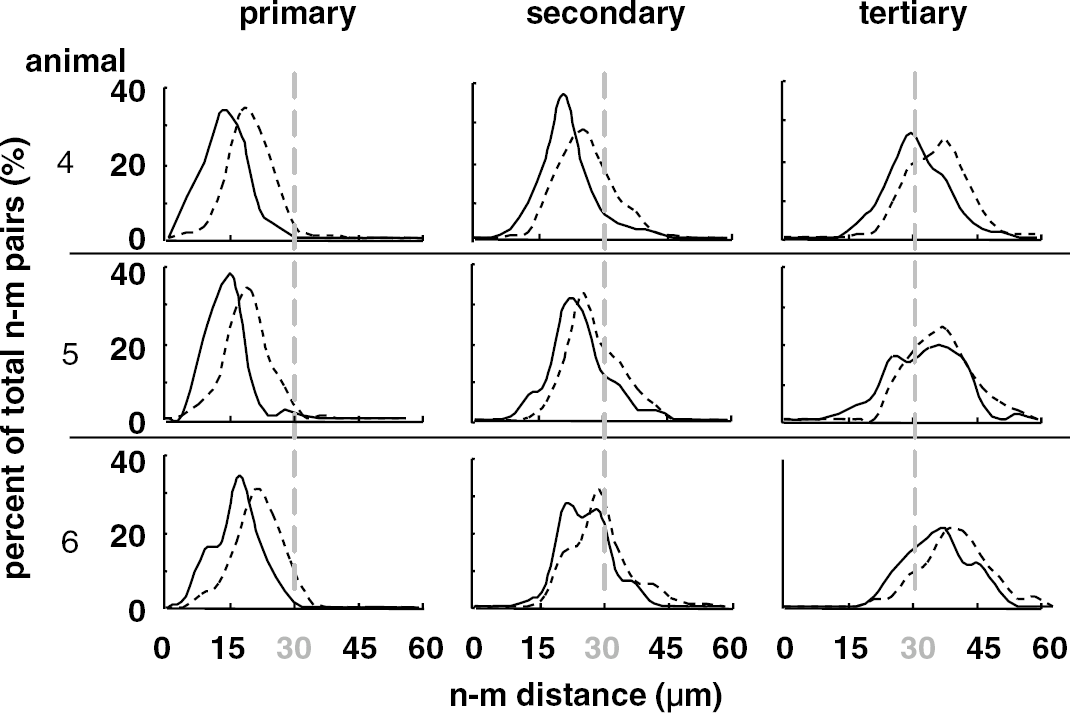
Range of effect of ischemia on [n–m distance] at 2 hours MCAO. Solid and broken curves represent the [n–m distance] of dUTP− (n) and dUTP+ neurons (n∗), respectively. The difference is distinct within 30–40 μm from a target neuron.
The Microvessel–Neuron Relationship and Neuron Subtype
Because neurotransmitter content changes in response to ischemia, the enzyme GAD was chosen to define a neuron subgroup. GAD+ neurons accounted for 61.7±0.4% of neurons in normal tissues and 55.0±4.2% of all neurons in ROI at the AC level at 2 hours MCAO. There was a highly significant relationship between neuron GAD content and the absence of dUTP incorporation (2P<0.00001). Only 5.1±2.0% of GAD+ neurons were dUTP+ at 2 hours MCAO. Furthermore, among dUTP+ neurons, 14.3±4.4% were GAD+ and 85.7±4.4% were GAD−. This implies that GAD-immunoreactive neurons were relatively resistant to ischemic injury at 2 hours.
Within normal tissue, GAD− neurons were distributed on average significantly further from their proximate microvessel than GAD+ neurons (mean GAD− [m–n distance]=19.5±8.3 μm versus mean GAD+ [m–n distance]=11.9±6.2 μm, respectively; n=3, 2P≪0.00000001) (Figure 6). This relationship was not substantially altered at 2 hours MCAO (GAD−, 16.3±7.6 μm versus GAD+, 11.7±6.5 μm; n=3, 2P≪0.00000001) (Figure 6), indicating that for this neuron subpopulation the [m–n distance] profiles in normal tissue were preserved, and that tissue distortion did not play a substantial role.
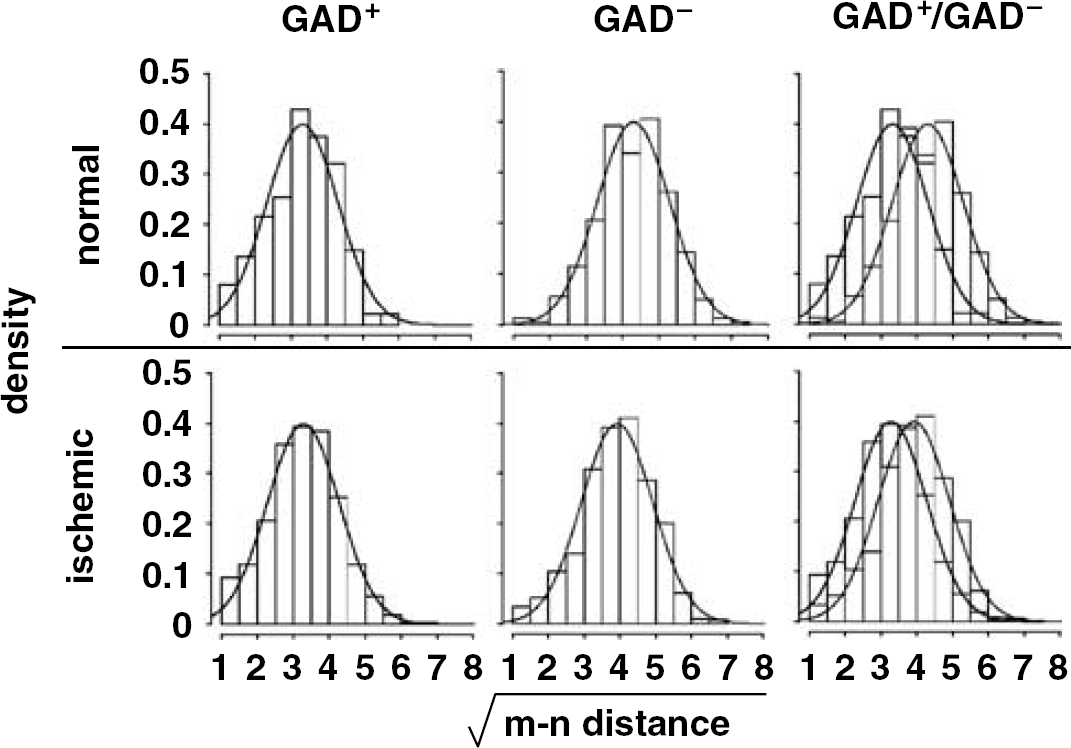
Comparison of transformed [m–n distance] between GAD− and GAD+ neurons in both normal (n=3) and ischemic (n=3) animals. Black and gray curves represent transformed [m–n distance] distribution of GAD+ and GAD−, respectively. The difference between the distributions before and after ischemia was consistent.
To test the generality of this observation, the effect of MCAO on TH+ neuron responses was tabulated. No difference in the [m–n∗ distance] and the [m–n distance] for the TH+ and TH− neurons was observed (2P=0.643). Here, 34.6±5.3% of GAD+ neurons were TH+ in the basal ganglia ROI, or of the TH+ neurons, 70.4±6.6% were GAD+ (Table 3).
Relative neuron distribution (in percent) by neurotransmitter subtype
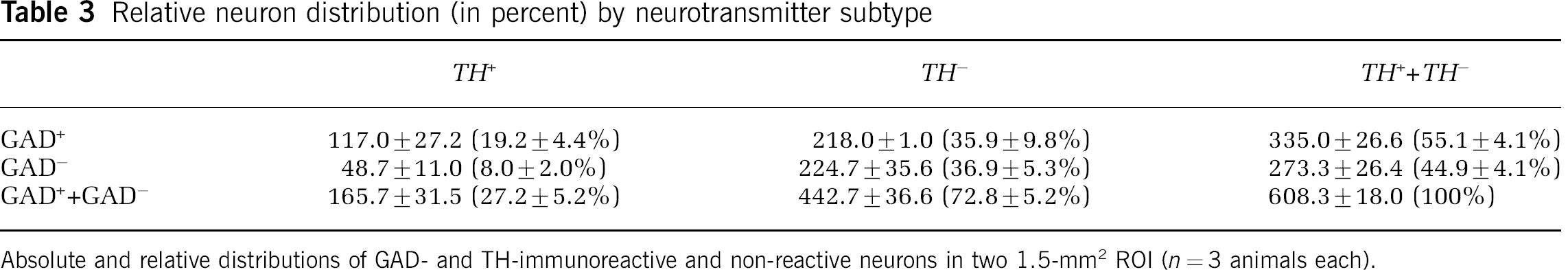
Absolute and relative distributions of GAD- and TH-immunoreactive and non-reactive neurons in two 1.5-mm2 ROI (n=3 animals each).
Discussion
The dependence of cerebral tissue on local blood flow implies that communication between the microvessel and its nearby neurons may play a unitary role (Iadecola et al, 1997; Zonta et al, 2003). Here, we show for the first time that a highly ordered reproducible spatial relationship exists between individual neurons and their nearest neighboring microvessels throughout the normal primate basal ganglia. In the ischemic core, within 2 hours after MCAO in the absence of arterial reperfusion, neurons positioned most distant from their nearest detectable microvessel are more likely to display evidence of DNA injury/repair. This observation is strengthened by: (i) the absence of substantial tissue distortion that could account for a shift in [m–n distance], (ii) the interspersal of neurons displaying evidence of DNA scission by dUTP incorporation (n∗) among normal appearing neurons (n) in the Ic region, and (iii) the correlation of both resistance to ischemia and short [m–n distance] for a distinct subpopulation of neurons that express the enzyme GAD. Hence, when flow ceases, neurons (mostly GAD−) on average furthest from their primary neighboring microvessel are more likely to suffer injury than those in close proximity.
While a microvessel–neuron relationship during focal ischemia has been presumed, it has not been shown. The primate basal ganglia is of particular importance to these studies because these tissues are highly sensitive to occlusion of their arterial supply during thromboembolic stroke.
The basis for the ordered [m–n distance] relationship is not known. One hypothesis is that it represents the limits of nutrient diffusion or support. In an experimental model of the Krogh tissue cylinder, the width of the oxygen-diffusion area from the primary capillary is proportional to the square of oxygen tension within the capillary (Quistorff et al, 1977). Moreover, the threshold for the [m–n∗ distance] and the primary [n–m distances] in the ischemic basal ganglia appears consistent with a 30- to 40-μm diffusion boundary (Quistorff et al, 1977). Interestingly, branch points for capillaries within the microvascular network of the primate basal ganglia occur at 30- to 40-μm intervals (del Zoppo et al, 1991). Furthermore, in normal subcutaneous tissues, nearly all intravascular points are at most 30 μm from the nearest microvessel (Baish et al, 1996).
A second contributor to the ordered microvessel–neuron relationship observed here is undoubtedly genetic. Significant differences in cerebral vascular arrangements and in the complexity of leptomeningeal anastomoses depend on phylogeny (Edvinsson et al, 1993), which implies that collateral protection and perhaps the capillary arrays can differ among species. Among rodents, strain differences in infarction volume after focal cerebral ischemia (Maeda et al, 1999) and in related outcomes (Barone et al, 1993; Maeda et al, 1999) have been observed. In part, these differences have been associated with arterial supply (Maeda et al, 1999) and the completeness of the circle of Willis (Barone et al, 1993). Even within a single strain, atypical branching of the arterial supply or hypervascularization of the capillary bed has been observed (Fox et al, 1993). Those reports suggest that multiple genetic factors might be involved in cerebral vascular architecture and physiology, and in the pathophysiological responses to ischemic stroke.
For these experiments, several technical considerations are also relevant. Early selective injury to basal ganglia neurons most distant from their supply microvessels is likely to occur in all dimensions. Differences in normal mean [m–n distance] among the three dimensions were small (<1.8 μm) and not believed to be biologically significant for two reasons: (i) the exceptionally large numbers of m–n distance measurements, and (ii) within the primate, basal ganglia gray matter normally contains few scattered large white matter tracts (in contrast to rodents). Therefore, depending on the ratio of the fiber tracts to gray matter in the ROI, the [m–n distance]s could vary within a small range.
Local tissue distortion leading to longer m–n∗ distances is unlikely. Distortion of the m–n∗ distance in the neighborhood of the injured neuron could occur through: (i) tissue expansion due to microvessel permeability, (ii) loss of tissue scaffolding, and (iii) the effects of tissue ‘warp’ introduced during histologic preparation of CNS tissues. Tissue swelling and volume expansion of astroglial cells is an early consequence of MCAO in primates and other species (Watanabe et al, 1977; Garcia et al, 1993; Kimelberg et al, 1995). Although glial cell swelling occurs within minutes or hours after ischemia onset, which encompasses the period studied here, an ∼40% increase in the linear m–n distance and a compensatory change elsewhere would be required to explain these results. No such tissue distortion was observed in the coronal plane. We cannot rule out some distortion in the rostral–caudal direction, however. Alternatively, loss of the tissue scaffold after MCAO, perhaps through degeneration of the matrix around primary microvessels and their neurons, could distort local m–n relationships without tissue expansion. ‘Warping’ of the tissues during histologic processing is a concern with samples from the CNS (Uylings et al, 1986; Capowski, 1989). But, such tissue processing-related changes are likely to be stochastic. The consistency of the increased m–n∗ distances, the heterogeneity of neuron and microvessel injury, and the lack of a detectable tissue volume change imply that ‘warping’ did not play a major role in the fundamental observation.
The interspersal of n∗ neurons among those without injury indicates that very early after MCAO neuron sensitivity and injury development in the basal ganglia might not be uniform. This is paralleled by changes in microvessel integrity, which are also heterogeneously distributed within the Ic region (Tagaya et al, 1997; Abumiya et al, 1999). Furthermore, within selected regions of ischemic cortex in the rat, neuron injury may also be heterogeneously distributed under specific conditions (Koh and Choi, 1988; Dawson and Hallenbeck, 1996). This raises the possibility that neurons in the basal ganglia are heterogeneously affected. In time, secondary injury processes would be expected to affect all microvessels and neurons in the ischemic core (del Zoppo, 1994).
An important concern is that heterogeneous neuron injury could result from the differential sensitivity of selected neuron subtypes. This would require that vulnerable neurons could be on average further from their primary microvessel than those that are resistant. But, it is not known whether and how the clear relationship between early neuron injury (sensitivity) and long [m–n distance] may relate to ‘selective neuron vulnerability’ or ‘delayed neuronal necrosis’ observed late (i.e. days) after global ischemia. Selective neuron vulnerability has been attributed to neuron subtype and differential glutamate sensitivity (Pulsinelli, 1985; Calabresi et al, 1999), depression of intrinsic excitability (Gajendiran et al, 2001), ion channel density (Centonze et al, 2001), and other attributes. On this basis, we examined neuron sensitivity relative to neurotransmitter subtype.
Subtle differences in neuron resistance to focal and to global ischemia have been reported. Among neurons identified by specific enzymes of neurotransmitter processing, thalamic and striatal cholinergic neurons appear more resistant to ischemia than GAD+ (GABAergic) neurons in rodents (Nyberg and Waller, 1989; Gonzales et al, 1992). In the present experiments, all ChAT neurons were confined to the thalamus, and did not display evidence of injury (data not shown). To transient focal ischemia, TH immunoreactivity in rat substantia nigra interneurons was preserved up to 3 days after MCAO, and then decreased (Yamada et al, 2003; Huh et al, 2003). Little is known about the responses of GAD+ neurons to focal ischemia, but GAD+ hippocampal neurons appear relatively resistant in the setting of global ischemia in the rat (Schlander et al, 1988).
In the primate basal ganglia, nonischemic GAD− neurons are situated on average farther from the primary microvessel than GAD+ neurons, and are significantly more likely to display injury (incorporate dUTP). Nitsch et al (1989) and Gonzales et al (1992) reported little or no change of hippocampal or striatal GAD-immunoreactive neurons to global ischemia, suggesting that those GAD+ neurons might not be sensitive. Francis and Pulsinelli (1982) concluded that GABAergic neurons were not equally sensitive to ischemia throughout the brain. In these studies, the proportion of basal ganglia GAD+ neurons decreased from ∼62% to 55% by 2 hours MCAO, not inconsistent with reports of decreased GAD activity late (1 to 21 days) in the ischemic territory (Bowen et al, 1976; Francis and Pulsinelli, 1982).
Betarbet et al (1997) showed that TH-immunoreactive neurons in the primate striatum are a subset of GAD 67-immunoreactive neurons. The anti-GAD polyclonal antibody used here identified both GAD 65 and GAD 67 antigens. It cannot be excluded that the two GAD subpopulations do exhibit differential sensitivity to focal ischemia. TH− and TH+ neurons were equally affected by focal ischemia and there was no clear relationship between TH immunoreactivity and [m–n distance] as seen for GAD. Finally, preservation of normal m–n relationships among basal ganglia GAD neurons, after-ischemia (Figure 6), suggests that neuron–microvessel proximity and neuron identity might be relevant to early neuron sensitivity. However, based on the TH neuron responses, neurotransmitter subtype is not an inviolable determinate of neuron sensitivity, or neuron vulnerability (Pulsinelli, 1985).
Alternatively, because neurons communicate to supply microvessels via intervening astrocytes, or indirectly through supportive cells (Nedergaard, 1994; Bernaudin et al, 1998; Aguado et al, 2002), the more distant communications might be more sensitive to ischemia or to injury (e.g. occlusion) within the microvessel.
Finally, if the diffusion boundary is limited to approximately 30 to 40 μm from the capillary, then a 12% to 14% loss of O2 tension would be expected over 6 μm in this range, in the absence of active O2 consumption (Quistorff et al, 1977). During focal ischemia, the increase in O2 extraction fraction (OEF) would be expected to steepen the O2 gradient such that the mean 6-μm difference in [m–n distance] observed may make the most distant neurons more susceptible. In addition, neurons most distant from the supply microvessels would be expected to be exposed to neurotoxic products (e.g. glutamate) that might not be readily cleared from the locale of such neurons. However, without experimental evidence to support either hypothesis, we hesitate to impute any precise biological significance to the 6-μm difference in nearest neighbor distances between the dUTP+ or dUTP− neurons and their respective but adjoining microvessels. A current research endeavor is to devise a mathematical model that would be concordant with the two distance distributions, taking into account their overlap.
So, basal ganglia neurons more distant from their neighboring microvessel appear most sensitive to injury early after MCAO. Neuron subtype experiments confirm that the relationship is not a result of tissue distortion. However, there is no intrinsic reason that enzymes in neurotransmitter processing should confer sensitivity to focal ischemia on selected neurons, or convey vulnerability. In contrast, [m–n distance] could be one of the contributors to sensitivity of neuron subpopulations in the basal ganglia, although it does not explain all of the phenomena. Ongoing studies will examine the proximity of microvessel injury to neuron sensitivity early after MCAO to address this question.
Footnotes
Acknowledgements
This work was supported in part by grants NS 26945 and NS 38710 of the National Institutes of Health. Dr. Mabuchi was supported in part by a fellowship award from the Sumitomo Pharmaceuticals Co., Ltd (Tokyo). We wish to dedicate this manuscript to the contributions of Julio H Garcia whose input and friendship were invaluable to this project.
