Introduction
Presynaptic cholinergic markers could be used for estimating the integrity of the cholinergic systems in the human brain with neurological diseases such as Alzheimer's disease. Several radiolabeled vesamicol derivatives have been proposed as the probes to detect vesicular acetylcholine transporter (VAChT) by PET and SPECT. Substituted positions and optical isomerization of the derivatives altered their affinities for VAChT and sigma receptors 1 . Here we prepared [11C](−)-o-methylvesamicol ([11C](−)-OMV) and evaluated in vivo its properties as a PET radioligand for mapping VAChT.
Methods
The affinities of vesamicol derivatives for VAChT were investigated by membrane binding assay. [11C](−)-OMV and [11C](+)-p-methylvesamicol ([11C](+)-PMV) were prepared by the reaction of the corresponding trimethylstanylvesamcol and [11C]CH3I in the presence of Pd2(dba)3, (o-tol)3P, CuCl and K2CO3. Biodistribution of [11C]OMV was investigated in male Wistar rats by tissue dissection and ex vivo autoradiography. VAChT-specific uptake of [11C](−)-OMV in the rat brain was evaluated in blocking studies with cold (−)-OMV, (−)-vesamicol, (+)-pentazocine and SA4503 (500 nmol/kg co-injection). A conscious male rhesus monkey underwent 91-min PET scan with [11C](−)-OMV using a model SHR-7700 (Hamamatsu Photonics K.K., Hamamatsu, Japan).
Results
(−)-OMV exhibited in vitro a high affinity for VAChT and low affinity for sigma1 receptor (Table 1). In rats the brain uptake of [11C](−)-OMV was 1.1 %ID/g at 5 min postinjection, and retained a high level for 60 min. The brain uptake was significantly inhibited by co-injection of cold (−)-OMV, (−)-vesamicol and SA4503 (60–70% reduction), but not of (+)-pentazocine. Ex vivo autoradiography showed different regional brain distribution patterns between [11C](−)-OMV and [11C](+)-PMV. In the monkey brain (Fig. 1), the binding of [11C](−)-OMV was reversible and an apparent equilibrium state was found at 20–40 min.
Affinities of vesamicol derivatives for VAChT and sigma receptors.
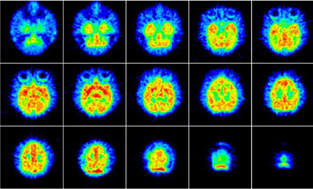
PET image of [11C](−)-OMV in the monkey brain.
Conclusion
These findings show that [11C](−)-OMV is a potential VAChT radioligand that exhibited appropriate brain kinetics during the time frame of 11C-labeled tracers.
